Abstract
Background
Genotoxic factors, including ionizing radiation and oxidative stress, are associated with genomic instability and development of breast cancer (BC). The homologous recombination DNA repair (HRR) pathway, base excision repair (BER) mechanism, and antioxidative enzymes are required as defense mechanisms against these DNA damaging agents. GSTP1, XRCC1, XRCC3 and RAD51 proteins are essential components of antioxidation, BER and HRR of DNA, respectively. Deficiencies in BER, HRR and antioxidation pathways are involved in the progression of cancer.
Methods
Genomic DNA was extracted from formalin-fixed, paraffin-embedded tissue and blood samples of BC patients of an Italian population. Genomic DNA was also extracted from blood specimens of a control group. DNA sequencing was performed for six single-nucleotide polymorphisms (SNPs) in the GSTP1, RAD51, XRCC1 and XRCC3 genes in BC patients and the control group.
Results
Two variants in the 5′-UTR of the XRCC3 (rs1799794 A/G) and RAD51 (rs1801321) genes showed a significant association with susceptibility to BC (OR = 4.125; 95% CI 1.057-16.102; p = 0.03 and OR = 2.04; 95% CI 0.4925-8.449; p = 0.007, respectively). Additionally, we reported 2 mutations in intron 7 of the XRCC3 gene, CTdel (rs543072564) and A/G (rs369703243).
Conclusions
Our results underscored the existence of an association between XRCC3-5′-UTR-A/G (rs1799794) and RAD51-5′-UTR G172T (rs1801321) genotypes and BC risk in an Italian population. The presence of mutations in the intronic region of the XRCC3 gene highlights the importance of more sequence screening of DNA repair genes for possible genetic penetrance in BC.
Introduction
Breast cancer (BC) has been estimated to be the second leading cause of death among cancer patients, and to account for 25% of all cancers globally (1). In Italy, around 48,000 new cases of BC were diagnosed in 2013 (2). The contribution of a genetic predisposition to BC is limited to mutations in the BRCA1 and BRCA2 genes, which are rare in the general population. Therefore, other candidate genes have been proposed to be associated with BC development, including DNA repair complex proteins. However, only rare mutations have been identified in these genes. Thus, there has been extensive research to elucidate the possible role of particular genotypes in the development of many cancers including BC (3).
Genotoxic agents such as ionizing radiation can modify biological molecules including DNA by a direct or indirect mechanism of the generation of reactive oxygen species (ROS) (4). This may subsequently cause harmful mutations more readily than cell damage. Eukaryotic cells have been reported to respond to ionizing radiation by several DNA damage repair pathways and detoxification of ROS by certain antioxidant mechanisms (5-7). For instance, base excision repair (BER) requires action of XRCC1. Additionally, XRCC3 and RAD51 recombinases are required for homologous recombination DNA repair (HRR) and DNA double-strand break (DNA-DSB) repair pathways (8, 9). Furthermore, glutathiones-transferases (GSTs) are involved in detoxification of oxidative stress damage (7).
Genetic polymorphisms and mutations in the GSTP1, RAD51, XRCC1 and XRCC3 genes have been investigated in various cancer types including BC (10-19). Controversially, in some studies, XRCC1-399Gln (rs25487), XRCC3-241M (rs861539) and XRCC3 5' untranslated region (UTR) (rs1799794) genotypic variants have been associated with susceptibility to BC. Other studies did not find an association between these variants and the risk of BC (20).
Also 5′-UTR polymorphisms of the RAD51 gene, namely G135C (rs1801320) and G172T (rs1801321), have been evaluated in BC. In particular, extensive studies of the G135C variant and few studies of the G172T variant have shown a significant association between C and T alleles and BC development in different populations (21, 22). Additionally, BRCA2 carriers have shown a significant association between G135C variants and the risk of developing BC (23).
The glutathione-S transferase-pi (GST-π) isozyme is coded by the GSTP1 gene to detoxify xenobiotic oxidative stress (24). The GSTP1 polymorphism (rsl695; A→G) changes Ile to Val at position 105 (Me105Val). This alteration has been associated with lower activity of GST-π, which may influence mutations in DNA by genotoxic factors (25-27). Previous studies have shown an inconsistent association between the GSTP1 (Me105Val) polymorphism and susceptibility to BC (28-31).
In this pilot study, we aimed to compare 6 polymorphisms in 4 genes, XRCC1 Gln399Arg, XRCC3 Met241Thr, XRCC3-5′-UTR A/G, RAD51-5′-UTR-G135C, RAD51-5′-UTR-G172T and GSTP1 Ile105Val, in tumor and normal tissue is the blood sample of BC patients. Secondly, we aimed to investigate the possible association of these polymorphisms and mutations with the susceptibility to BC in an Italian population.
Materials and Methods
Ethics statement
For sample collection from archived storage, approval was obtained from the ethics committee of Santa Chiara Hospital, Pisa, Italy.
Study subjects
The study population included 23 BC patients with no family history and negative results for BRCA1 and BRCA2 mutations. Formalin-fixed, paraffin-embedded (FFPE) tissue blocks and blood specimens were collected from female patients diagnosed with BC according to the medical records of Santa Chiara University Hospital. The control group included 16 normal individuals with no previous diagnosis of cancer matched for age with the patient group. Blood specimens were collected from the control group during their visits to the hospital, after informed consent had been obtained.
DNA extraction
DNA extraction from the FFPE samples was performed as described before (32). Briefly, 5 sections of 10-μm thickness were deparaffinized in xylene (Sigma-Aldrich) followed by absolute ethanol clearance (Sigma-Aldrich); evaporation of ethanol was done in a safety cabinet for 45 minutes. A Qiagen DNeasy kit (Qiagen GmbH) was used to complete genomic DNA extraction from the FFPE samples by following the manufacturer's instructions.
DNA extraction from blood specimens was performed according to the manufacturer's instructions by a commercial genomic extraction kit (QIAamp DNA Blood Mini Kit by Qiagen).
Primer design, PCR and sequencing
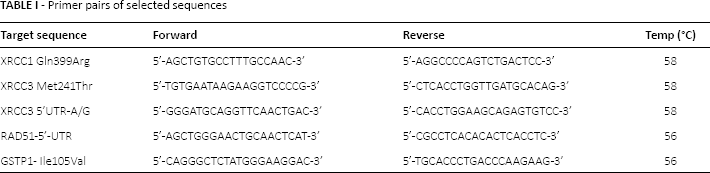
Specific primers were designed for the selected genotypes based on the gene sequences available from the National Center for Biotechnology Information (NCBI) (Tab. I). The PCR reactions targeting the specified genotypes were performed in 30 μL reaction volume containing 1.5 mM MgCI, 75 mM Tris-HCI, 20 mM (NH4) 2S04, 50 mM KCI, 0.2 mM of primers, and 1 U of TAKARA DNA polymerase. Cycling steps were as follows: after 7 minutes at 95°C, 45 cycles of 45 seconds at 94°C, 45 seconds at annealing temperatures from 56°C to 59°C, and 1 minute at 72°C, then a final extension at 72°C for 7 minutes. After proper cleanup of the PCR products, it was subjected to sequencing PCR according to the manufacturer's instructions by means of a BigDye Terminator version 3.1 kit (Applied Biosystems); this was followed by the cleanup protocol as described in the kit. Finally, all amplicons were applied to an automated DNA sequencer (ABI Prism Genetic Analyzer 3130×1 System, Applied Biosystems).
Primer pairs of selected sequences
Statistical analysis
The GraphPad Prism 6 software was used to perform the chi-square test, Fisher's exact test and Hardy-Weinberg equilibrium test. An association was considered significant when the p value was less than 0.05.
Results
DNA sequences were analyzed by ABI software in the FFPE and blood samples of 23 BC patients and the DNA extracted from 16 samples of the control group. The genotypes of the target sites of the GSTP1, RAD51, XRCC1 and XRCC3 genes were confirmed by Sanger sequencing (Supplementary Figure, available online at www.biological-markers.com). The genotypes and variants of the 6 positions of the studied genes are illustrated in Table II. The XRCC3-5'UTR (rs1799794 A/G) genotypic and allelic distribution exhibited a significant association with the susceptibility to BC (OR = 4.125; 95% confidence interval [CI] 1.057-16.102; p = 0.0316; χ 2 for trend = 4.621). Moreover, the G172T genotype of RAD51-5′-UTR exhibited a more significant association with the risk of BC development in the selected Italian population (OR = 2.04; 95% CI 0.4925-8.449; p = 0.007; χ 2 for trend = 7.165).
Genotype distribution of the selected genes
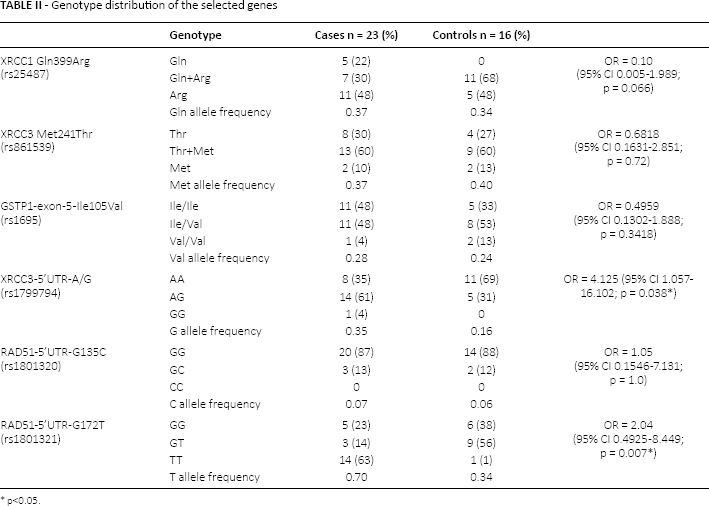
p<0.05.
On the other hand, the XRCC1-Arg399Gln genotypic distribution showed no significant correlation with the risk of BC in the selected population (OR = 0.10; 95% CI 0.005225-1.989). Furthermore, the frequencies of the M241T genotypes of the XRCC3 gene did not show a significant difference between BC patients and controls (OR = 0.6818; 95% CI 0.1631-2.851). Moreover, the distribution of the He105Val (rs1695) genotype of the GSTP1 gene was not associated with the risk of BC in the selected Italian population (OR = 0.4959; 95% CI 0.1302-1.888). Also the genotypic distribution of G135C of the RAD51-5′-UTR was not associated with BC (OR = 1.05; 95% CI 0.1546-7.131).
Interestingly, sequence analysis of the selected genes revealed a germline frameshift mutation and somatic mutation in intron 7 of the XRCC3 gene in 2 different samples. Specifically, CTdel (rs543072564) in intron 7 of the XRCC3 gene was found in the tumor tissue as well as blood sample of the patient (Fig. 1), while A/G (rs369703243) was found in tumor tissue only (Fig. 2).
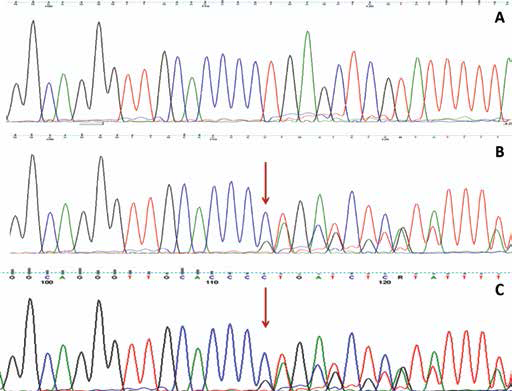
DNA sequencing electropherogram of the deletion mutation CTdel (rs543072564) in intron 7 of the XRCC3 gene. Wild-type (
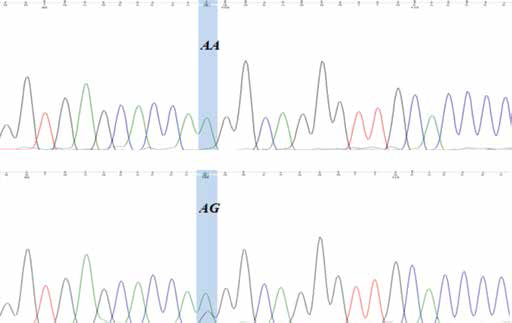
DNA sequencing electropherogram of A/G (rs369703243) in intron 7 of the XRCC3 gene. Wild-type (AA); mutant in cancer tissue (AG).
Discussion
Genetic instability is an eventual event in cancer. Therefore, an inadequate repair mechanism has been suggested to be associated with cancer development and progression (33). This hypothesis is supported by the limited genetic predisposition of BRCA1/2 mutations in BC (34). Consequently, there could be other genetic polymorphisms associated with the risk of BC. Our results supported this model by studying BER (XRCC1), HRR and DNA-DSB repair complex proteins such as RAD51 and XRCC3, and the antioxidant enzyme GSTP1.
This study supported the presence of an association between polymorphisms at the 5′-UTR of the XRCC3 and RAD51 genes and the susceptibility to BC (21, 32). The rs1799794 A/G genotype of XRCC3-5′-UTR showed a significant association with susceptibility to BC. Moreover, the G172T (rs1801321) genotype of RAD51-5′-UTR exhibited a more highly significant association with the risk of developing BC in the selected Italian population. In addition, we reported the presence of a frameshift mutation in intron 7 of the XRCC3 gene in about 4% of the selected cases.
The role of RAD51 in carcinogenesis is not completely understood. Hasselbach et al (35) in a functional study showed a positive impact of alterations of G135C and G172T on the expression of RAD51. Theoretically, expression of homologous recombination proteins, namely XRCC3 and RAD51, is protective against DNA damage generated by genotoxic agents. However, our results contradict this assumption. Overexpression of RAD51 may therefore have an impact on DNA instability and carcinogenesis without a completely understood mechanism of action. Nevertheless, RAD51 overexpression has been demonstrated in BC cells (36). Moreover, clinicopathological factors and metastasis have been found to be associated with overexpression of RAD51 and XRCC3 (37, 38). Therefore, the balanced expression of RAD51 and other repair complex proteins is hypothesized (39). Further molecular analyses will be helpful in understanding the impact of 5′-UTR on the expression of RAD51 and XRCC3 and the role of the expression of these genes in genomic instability and carcinogenesis.
In the present study, we extracted DNA from tumor tissues and blood samples of BC patients to investigate a possible variation in the selected genotypes between tumor and normal tissues. None of the analyzed samples showed any genotypic difference between FFPE and blood samples. Therefore, the current results of the G172T genotype support our previous finding of the association between rs1801321 and the risk of BC (21).
The other genotypes (XRCC1-Arg399Gln, XRCC3-M241T, GSTP1-Ile105Val and RAD51-G135C) did not show any association with the risk of BC, which is consistent with previous studies (40, 41). The lack of any association between the XRCC3-M241T polymorphism and cancer risk is supported by the in vitro finding of no significant differences in DNA repair responses between wild-type and normal genotypes (42). Consistently, Thyagarajan et al (43) did not find an association between the XRCC1-Arg399Gln and XRCC3-M241T polymorphisms and susceptibility to BC. In contrast, an association between the GSTP1 polymorphisms and BC has been found to be exacerbated by alcohol consumption (44). Many factors can be considered for the inconsistent findings, such as population differences, ethnic origin, lifestyle habits, smoking, and alcohol consumption (45). Furthermore, other genetic variations might be associated with the risk of BC; for instance, hypermethylation of the GSTP1 promoter has been associated with BC risk (46, 47). In another study, smoking and age were found to influence the association of the GSTP1 polymorphism with lung cancer (48). Moreover, BC survival but not BC risk has been found to be associated with the GSTP-Ile105Val polymorphism (49). The G135C genotype of the RAD51 gene was not associated with susceptibility to BC, which is consistent with previous reports (50, 51). In other studies the association of the G135C polymorphism with the development of BC was found to be influenced by BRCA status and age (52-54).
Our study has a limitation of sample size, due to the investigation of 6 polymorphisms in 4 genes. Besides, the primary aim was to compare tumor tissues with blood samples.
Conclusion
Our findings supported the association between the 5′-UTR polymorphisms of HRR genes (RAD51 and XRCC3) and the susceptibility to sporadic BC in an Italian population. However, more studies are required to clarify the precise role of these polymorphisms in the development of BC and analyze the 5′-UTR polymorphisms and expression levels of the RAD51 and XRCC3 genes. Moreover, the finding of 2 mutations in intron 7 of the XRCC3 gene (32) suggests the presence of other unknown mutations in DNA repair complex proteins. Since the study population was relatively small, future studies with larger sample sizes are required to confirm our findings.
Footnotes
Acknowledgment
We appreciate the valuable effort of Dr. Riyadh Muhaidat in helping us to finish this work. We sincerely thank Dr. Greg Eaton of Rowan University (USA) for his editing suggestions.
List of Abbreviations
Financial support: We are very thankful to the University of Pisa and Yarmouk University for the financial support to complete this work.
Conflict of interest: The authors declare they have no conflict of interest.
