Abstract
BACKGROUND:
Reduced efficiency of DNA repair systems has long been a suspected factor in increasing the risk of cancer.
OBJECTIVE:
In this work we investigate influence of three selected polymorphisms of DNA repair gene
METHODS:
In group of 359 patients with HNSCC (diagnosed with OSCC) the occurrence of polymorphic variants in Arg399Gln, Arg280His and Arg194Trp of
RESULTS:
Arg399Gln polymorphism and Arg194Trp polymorphism of
CONCLUSION:
Patients with HNSCC not only have an increased level of 8-oxoguanine and the Arg399Gln and Arg/Trp of
Introduction
Head and neck cancer is a heterogeneous group of tumors located in the head and neck region. About 90% of these tumors are squamous cell carcinomas (head and neck squamous cell carcinomas, HNSCC). HNSCC is the sixth most common cancer worldwide, with 700,000 cases diagnosed per year [15]. Despite intensive research and advances in surgical techniques, chemo- and radiotherapy, it is estimated that overall 5-year survival rate remains at a level of 40% to 50% [10, 47]. Among HNSCC the most common subtypes of cancer are oropharyngeal cancer, laryngeal and oral cavity cancers [12]. When located in mouth area, the most frequent one is oral squamous cell carcinoma (OSCC) [37]. The definitive etiology of the HNSCC remains unknown, and the primary risk factors are tobacco and alcohol use, involved in 75% of the cases, and human papilloma virus (HPV) infection, present in 40–60% of the head and neck cancers [26, 42]. OSCC is primarily associated with smoking and can be referred to as HPV-negative HNSCC [22]. No screening strategy has proved to be effective yet, and careful physical examination remains the primary approach for early detection, which increases the need for an effective method of early diagnosis. As with other types of malignant tumors, the most important role in oral cavity cancer development seems to be the combination of genetic predispositions with the influence of environmental factors. Critical damage to DNA, which occurs under the influence of both endogenous and exogenous factors, if not repaired, as a result of accumulation can lead to the cancer transformation. To defend itself against the risks associated with this phenomenon, the human organism on cellular level has developed a number of DNA repair mechanisms that effectively remove damage within DNA, significantly reducing the risk of cancer. However, when DNA repair mechanisms are diminished, the risk of cancer increases significantly as confirmed by abnormalities in various repair pathways observed and identified as a risk factor for many types of cancer, e.g. colorectal cancer [27], lung cancer [9, 48], bladder cancer [36], breast cancer [13, 46] or HNSCC [6, 14, 20]. One of the functions of DNA repair mechanisms is the removal of oxidative damage which is one of the most common types of DNA damage with an established role in modulating cancer risk [30, 33], in particular in the case of one of the oxidized bases: 8-oxo-guanine [24, 32]. The key DNA repair protein XRCC1 (X-ray repair cross-complementing protein 1) is involved in this repair. XRCC1 is part of Base Excision Repair (BER) pathway where it is associated with DNA ligase III as a complex protein, and while it does not have enzymatic activity, it plays a crucial role in DNA repair by acting as a scaffolding protein that interacts with multiple repair enzymes [28]. Polymorphisms within the XRCC1 gene are the subject of intense research as factors that may affect the effectiveness of the protein and, consequently, the efficiency of the entire DNA repair system, which translates into modulation of cancer risk [18, 34, 35]. The aim of this study was to investigate the effect of Arg399Gln, Arg280His and Arg194Trp polymorphisms of
Materials and methods
DNA isolation
DNA for genotyping was isolated from lymphocytes of the peripheral blood. The blood samples were taken from 359 unrelated patients hospitalized in Provincial Multispecialist Center of Oncology and Traumatology M. Kopernika in Łódź. Each patient had histopathologically confirmed OSCC, which was the inclusion criterion for the study. Patients with incomplete or inconclusive diagnosis and/or another type of neoplastic disease were excluded from the study group. All samples were collected from patients after admission to the hospital and confirmation of the diagnosis, before the start of the therapeutic process. The studied group included 207 men and 152 women (average age 63 years
DNA isolation was carried out with a commercial kit QIAamp DNA Blood Mini Kit for isolation of high-molecular-weight DNA (Qiagen).
Genotyping
The occurrence of polymorphic variants in Arg399Gln (rs25487), Arg280His (rs25489) and Arg194Trp (rs1799782) of
The refSNP’s and thermal conditions used in the PCR reaction
The refSNP’s and thermal conditions used in the PCR reaction
Level of 8-oxo-guanine was determined in isolated DNA using HT 8-oxo-dG ELISA II Kit (R&D Systems). Final DNA concentration used in measurement was 500
Statistical analysis
Number of genotypes received was compared with the expected value under Hardy-Weinberg law. The significance of differences between allele frequencies and genotypes for individual groups was assessed using the
Results:
Genotyping:
The genotyping results indicate that Arg399Gln polymorphism of
The distribution of genotypes, allele frequencies and the analysis of the odds ratio (OR) for Arg399Gln polymorphism of XRCC1 gene in patients with HNSCC and the control group
The distribution of genotypes, allele frequencies and the analysis of the odds ratio (OR) for Arg399Gln polymorphism of XRCC1 gene in patients with HNSCC and the control group
*genotype distribution in Hardy-Weinberg equilibrium,
The distribution of genotypes, allele frequencies and the analysis of the odds ratio (OR) for Arg280His polymorphism of XRCC1 gene in patients with HNSCC and the control group
*genotype distribution in Hardy-Weinberg equilibrium,
The distribution of genotypes, allele frequencies and the analysis of the odds ratio (OR) for Arg194Trp polymorphism of XRCC1 gene in patients with HNSCC and the control group
*genotype distribution in Hardy-Weinberg equilibrium,
In order to investigate the mutual effect of the occurrence of two studied polymorphisms simultaneously we performed intra-gene analysis for all three polymorphisms. Results show that simultaneous occurrence of Arg399Gln – His280His increases the risk of cancer (Table 5, OR 3.079 (1.395–6.798);
The distribution of genotypes and the analysis of the odds ratio (OR) for XRCC1 intra-gene interactions: Arg399Gln – Arg280His in patients with HNSCC and the control group
The distribution of genotypes and the analysis of the odds ratio (OR) for XRCC1 intra-gene interactions: Arg194Trp – Arg280His in patients with HNSCC and the control group
The distribution of genotypes and the analysis of the odds ratio (OR) for XRCC1 intra-gene interactions: Arg399Gln – Arg194Trp in patients with HNSCC and the control group
First, the level of 8-oxo-guanine was compared between the healthy subjects and the patient group, showing that the mean level for patients is higher (Table 8, 56,392 vs 155,418 (nM) / DNA (
Statistical analysis (
-Test: Two-Sample Assuming Unequal Variances) of the mean values of 8-oxo-guanine levels (8-oxoG (nM) / DNA (
g/
l)) in the groups of patients with HNSCC and control. Results for the entire study groups and by genotype
Statistical analysis (
Statistical analysis (
8-oxo-guanine levels in HNSCC patients and control group for genotypes of Arg399Gln polymorphism of 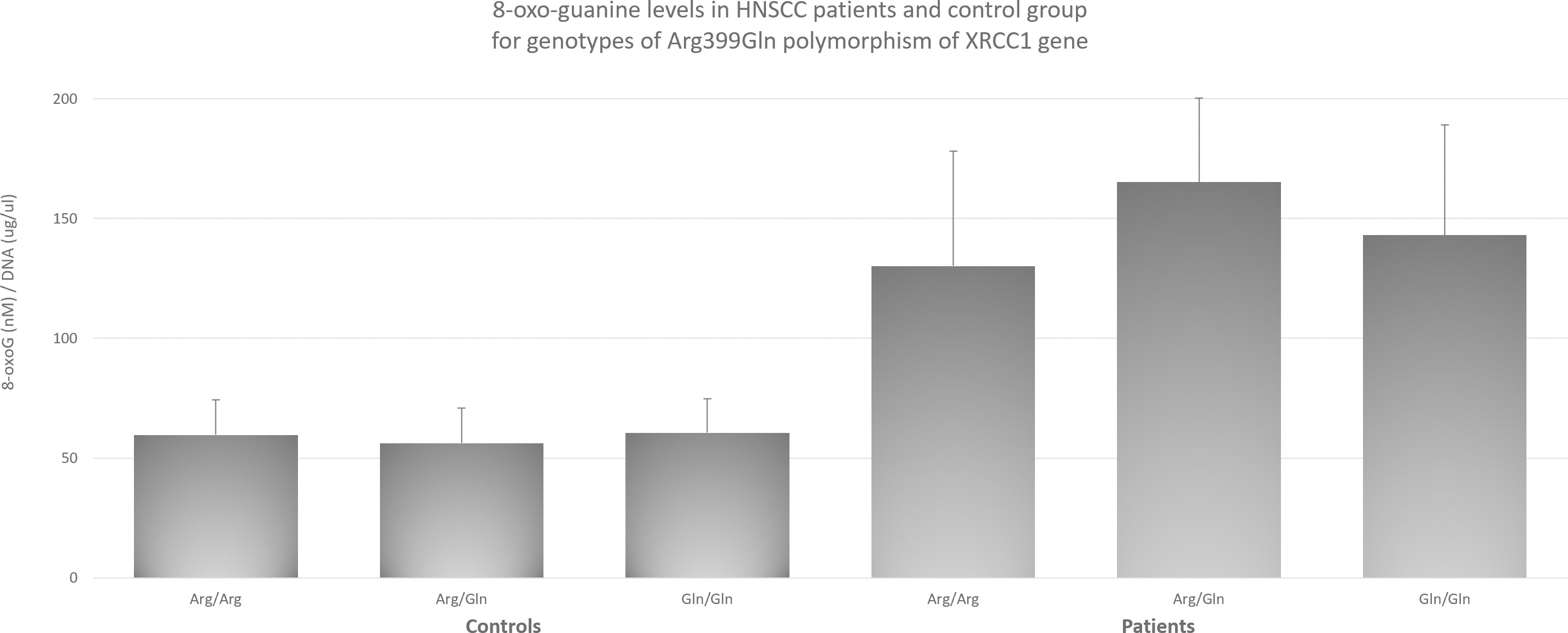
8-oxo-guanine levels in HNSCC patients and control group for genotypes of Arg280His polymorphism of 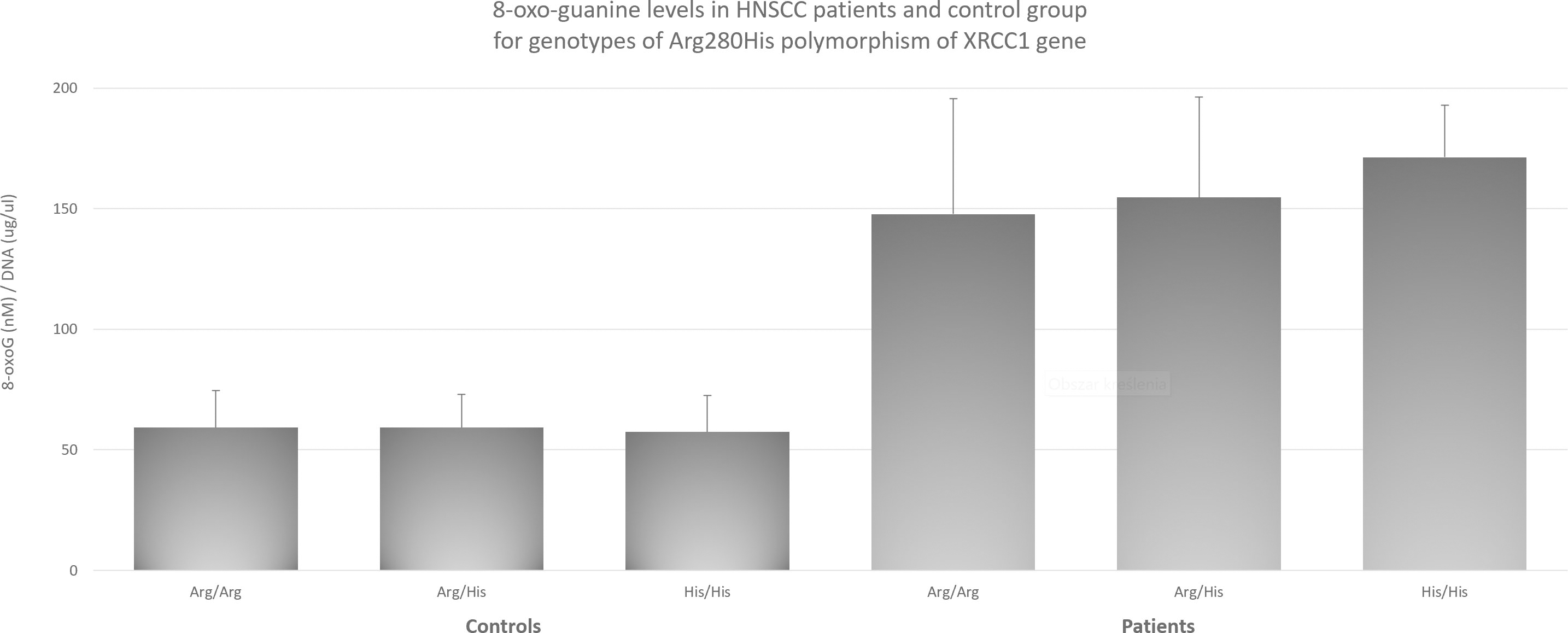
8-oxo-guanine levels in HNSCC patients and control group for genotypes of Arg194Trp polymorphism of 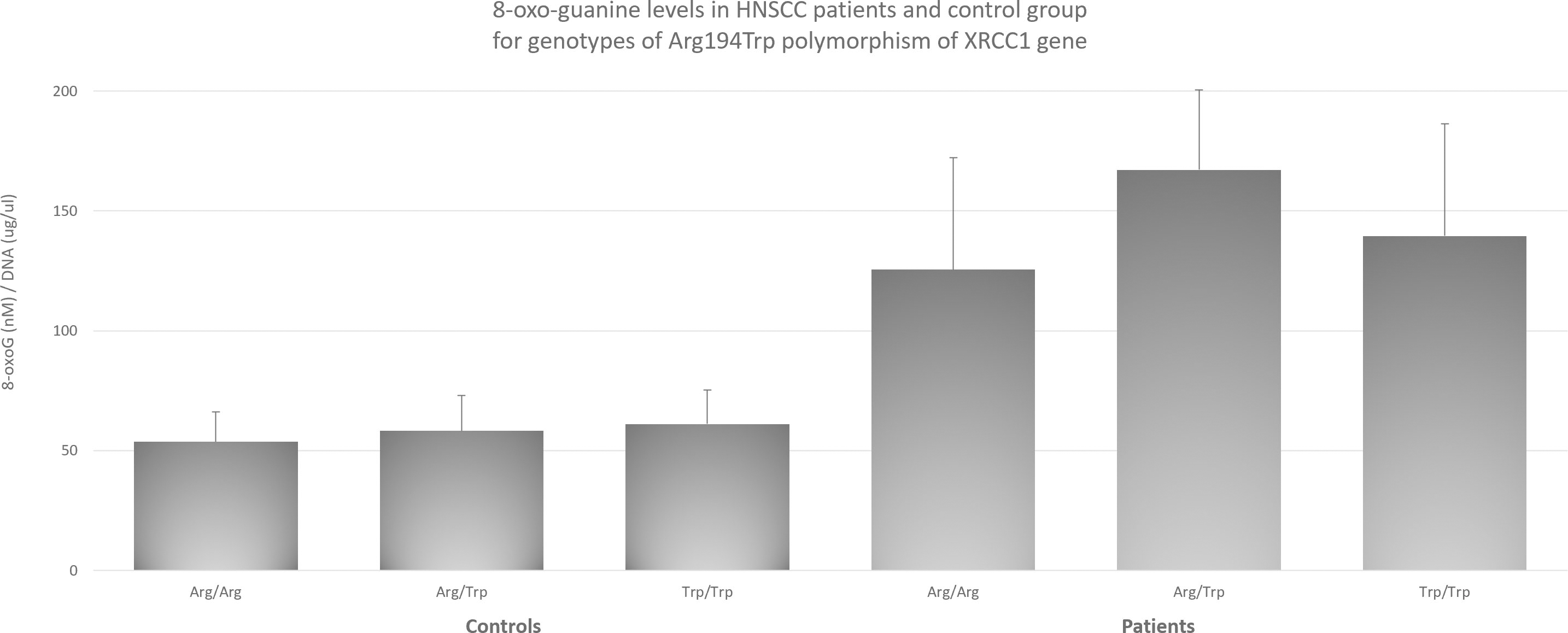
Stability of the genome, and consequently protection against cancer transformation, is largely provided by DNA repair systems. When the ability to repair DNA is reduced, damages are accumulated which can consequently lead to consolidation of the mutations and malignant transformation. Among DNA damages, such as a break, a base missing, or a chemically changed base, very widespread are those caused by reactive oxygen species, including the most frequent one: 8-oxo-guanine. It is estimated that every day, in each cell about 2400 8-oxoG molecules are reported [43], and thanks to effective DNA repair systems, their removal is carried out on an ongoing basis. This process is extremely important because any unrepaired damage can be a potential cause of cancer due to the fact that 8-oxo-guanine interferes with the transcription [23] and DNA replication [38]. Oxidative damages are removed by DNA Repair Mechanisms, primarily with Base Excision Repair (BER). Key elements of BER are enzymes involved in removal of damaged base and replacing it with correct one: glycosylase 1 (OGG1) producing an apurinic site (AP-site) by base removal, which is then eliminated through backbone incision by AP-lyase (APEX1), and end processing through flap-endonuclease 1 (FEN1) which removes 5’ overhanging DNA fragment left after damaged base removal. In the last step, the correct, undamaged base is built in [23]. An essential component of this mechanism is the XRCC1 protein, which interacts not only with the BER mechanism (in particular with the proteins mentioned above), but also with single-strand break repair and Nucleotide Excision Repair (NER), acting as a scaffold with which enzymatic systems of DNA repair mechanisms interact [28]. Due to its key role in DNA repair mechanisms, XRCC1 is the subject of extensive research in the field of modulating the risk of various types of cancer.
Influence of Arg399Gln and Arg194Trp polymorphisms of 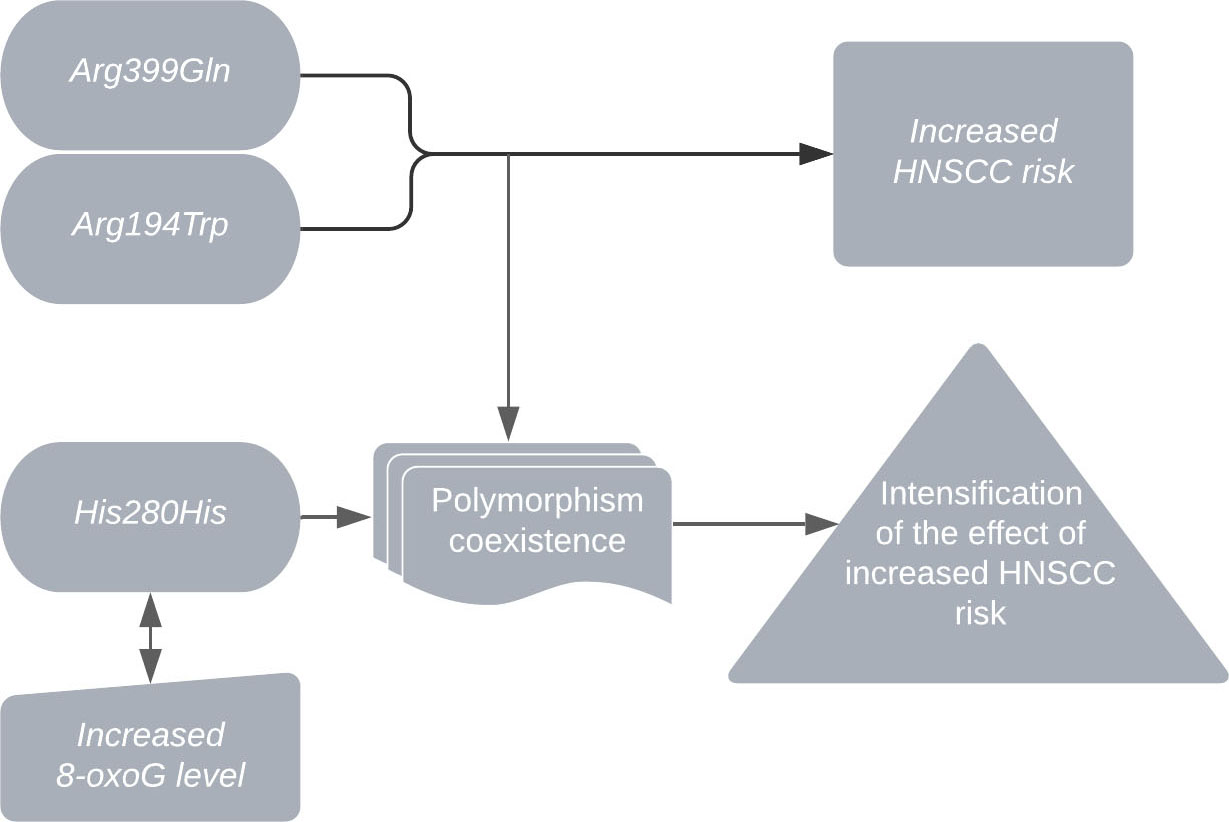
with Arg194Trp and Arg399Gln it increases their effect. This supports the theory, that the most responsible for data differences is the degree of complexity and multiplicity of factors responsible for malignant transformation, which can lead to different results and conclusions for the same genotype of a given gene. Since, as mentioned earlier, XRCC1 plays a key role in DNA repair mechanisms, and the main DNA damages are oxidative damages that may lie at the root of the cancer process, we decided to check whether there is a relationship between the polymorphisms studied and the level of 8-oxo-guanine and try this way potentially elucidate the mechanism behind HNSCC risk modulation by XRCC1. Regarding the influence of 8-oxo-guanine on HNSCC, the research to date has focused primarily on the role of hOGG1 as a factor directly responsible for cutting out the damaged base [7, 39]. In case of OSCC available data suggests influence of hOGG1 polymorphisms on cancer risk [5]. However, despite the fact that oxidative damage is a recognized factor in the pathogenesis of HNSCC [2], there are no reports of its level being associated with changes in XRCC1 due to polymorphisms. The obtained results indicate that the level of 8-oxo-guanine is significantly higher in patients with HNSCC than in the control group. At the same time, to check whether these differences are also observed at the level of individual genotypes, measurements were made for each of the polymorphisms. In all three cases, polymorphisms with the highest level of 8-oxo-guanine showed statistically significantly higher levels than the other studied ones. In the case of the Arg399Gln polymorphism, the highest level of 8-oxo-guanine was observed (Figure 1), which has previously been shown to increase the risk of HNSCC. Similar results were obtained for the Arg194Trp polymorphism (Figure 3), which, according to our results, is also associated with an increased risk. However, in the case of the His280His, which showed highest level of 8-oxo-guanine (Figure 2), no effect on the increased risk of HNSCC was observed. It is particularly worth emphasizing that this genotype enhances the increased risk effect through the interaction with Arg399Gln and Arg194Trp, as described earlier. We postulate that although His280His does not have a direct impact on the increased risk of HNSCC, through protein dysfunction leading to impaired 8-oxoG removal function, its coexistence with the polymorphisms increasing the risk of HNSCC intensifies this effect. The fact that His280His has the highest average level of 8-oxo-guanine of all tested groups allows, in our opinion, to conclude that this is a potential explanation of the mechanism behind the increased risk of HNSCC in the case of coexistence of His280His with Arg399Gln or Arg194Trp. Summary of proposed mechanism has been presented on flowchart on Fig. 4. At the same time, we do not usurp the right to fully explain the process underlying at malignant transformation in case of studied polymorphisms, but we believe that linking specific genotypes with 8-oxo-guanine levels will help to better understand the mechanisms involved in that complex process. The co-occurrence of the discussed polymorphisms may prove to be a promising diagnostic factor. Further research should focus on the unquestionable link between the coexistence of polymorphisms and an increased risk of HNSCC, as well as the inclusion of further polymorphisms in this set. Ultimately, this may lead to the creation of a diagnostic panel that will allow the patient to be quickly and conveniently classified as at increased risk and to be included in a special preventive program. Due to the complexity of the carcinogenesis process and the large role that oxidative stress may play in it, the need for an effective processing of DNA repair mechanisms is critical. DNA repair mechanisms, one of the last lines of cell defense against cancer transformation, have been shown to play a key role in maintaining cellular homeostasis and genome stability. Impaired repair mechanisms, as we have demonstrated in this article, are directly related to an increased level of HNSCC risk and clearly linked to levels of 8-oxo-guanine. Further, in our opinion elevated level of 8-oxo-guanine associated with
Arg399Gln and Arg194Trp polymorphisms of
Conflicts of interest
The authors declare no conflict of interest.
Footnotes
Acknowledgments
This work has been supported by Statutory financial resources of the Department of Clinical Chemistry and Biochemistry, Medical University of Lodz.
Author contributions
Conception: Jacek Kabzinski, Ireneusz Majsterek.
Interpretation or analysis of data: Jacek Kabzinski, Monika Maczynska, Dariusz Kaczmarczyk.
Preparation of the manuscript: Jacek Kabzinski, Monika Maczynska, Dariusz Kaczmarczyk.
Revision for important intellectual content: Ireneusz Majsterek.
Supervision: Ireneusz Majsterek.
