Abstract
Background
Thanks to immense improvements in technology over the past few decades, we have witnessed a major shift towards the idea that breast cancer results from a combined effect of multiple common alleles conferring low risk. This study investigates the role of 3 nonsynonymous SNPs in the DNA repair genes XRCC1 (R399Q), RAD51 (G135C) and TP53 (Arg72Pro) in breast cancer in Serbian women.
Patients and Methods
Cases of BRCA1/2-negative hereditary breast cancer (n = 52), sporadic breast cancer (n = 106) and age-matched cancer-free female controls (n = 104) were obtained from the Institute for Oncology and Radiology of Serbia's blood bank. Restriction fragment length polymorphism analysis was used for genotyping. Descriptive analyses included genotype and allelic frequencies; the odds ratio and 95% confidence interval were calculated as an estimate of the relative risk.
Results
A significant difference in QQ+RQ versus RR genotype distribution of XRCC1 was observed between hereditary breast cancer patients and cancer-free controls. The association was confirmed among young breast cancer patients from these high-risk families. The existence of 3 recessive alleles in the RAD51 and XRCC1 genotype combination showed an association with hereditary breast cancer. Odds ratio analysis indicated a strong protective role of the RAD51 GG + TP53 ArgArg + XRCC1 RR combined genotype against hereditary breast cancer negative for BRCA1/2 mutations.
Conclusions
The XRCC1 R399Q polymorphism showed an association with increased breast cancer risk in Serbia, especially in the hereditary form of the disease and in young breast cancer patients. Dominant alleles of RAD51, TP53 and XRCC1 combined genotypes indicated a strong protective role against hereditary breast cancer.
Introduction
Breast cancer is the most common cancer in women and the second cause of death from all malignant diseases (1). The majority of breast cancers are sporadic (70%-75%), with usually only 1 family member affected and with disease onset later in life. However, 5%-10% of all breast cancers have a strong inherited component and some of these cases arise as a consequence of deleterious mutations in the high-penetrance genes BRCA1 and BRCA2 (2-3-4). Apart from BRCA1/2, a number of other genes associated with increased breast cancer risk have been identified (5, 6). Delicate functional alterations in highly conserved DNA repair or growth regulatory processes resulting from germline genetic variations are among the key topics in current breast cancer research. These polymorphic genetic variants have been proposed as a possible cause of increased susceptibility to breast cancer.
The tumor suppressor gene TP53 encodes a protein which acts as a transcription factor that activates a number of processes resulting in prevention of neoplastic transformation (7). Codon 72 of TP53 is in a region of the gene that encodes the polyproline domain, which lies between the trans-activating and the DNA-binding domains of the protein (8). This codon in exon 4 either has the sequence CCC, which encodes proline (Pro), or the sequence CGC, which encodes arginine (Arg) (Arg72Pro; rs1042522). Although the current consensus is that the Arg72 variant is more effective in apoptosis induction than Pro72, their role in cancer development is still unclear.
One of the most important ways to repair double strand breaks is by homologous recombination, an error-free mechanism consisting of a well-orchestrated protein network with BRCAs as the leading ones. BRCA2 binds directly to RAD51, promoting its assembly onto a single-stranded DNA to stimulate DNA strand exchange (9). TP53 also binds directly to RAD51 and can downregulate RAD51-mediated homologous recombination events (10). Although truncating mutations in RAD51 are hard to identify since they are rare and population specific, a polymorphic variant in the 5’ untranslated region G135C (rs1801320) has been intensively studied as a potential risk factor for breast cancer because it lies in the important regulatory region of the gene.
X-ray repair cross-complementing group 1 (XRCC1) plays a role in highly conserved base excision repair and acts as a scaffold protein that brings together proteins of the DNA repair complex (11, 12). More than 30 SNPs have been identified in the coding and regulatory regions of XRCC1. A nucleotide change G to A in exon 10 (codon 399) leads to amino acid change Arg to Gln (R399Q; rs25487) and is located in the BRCT domain within a poly (ADP-ribose) polymerase binding region of the gene (13).
Since the effects of different SNPs can be subtle and may vary according to genetic background, there are serious challenges to determining their effects on cancer development in different populations (14). We have previously shown the importance of RAD51 G135C and TP53 Arg72Pro polymorphisms as single markers in breast cancer in Serbian patients, especially in the hereditary form of the disease (15). In the present study we analyzed the association of the XRCC1 R399Q polymorphism with BRCA1/2-negative hereditary breast cancer as well as with sporadic breast cancer in Serbia. Our second aim was to investigate the contribution of XRCC1, RAD51 and TP53 combined genotypes to the breast cancer risk. We also analyzed the impact of these polymorphisms on early-onset breast cancer in both hereditary and sporadic forms of the disease.
Methods
Study Participants
The investigated women were enrolled as 3 groups. The first, “hereditary” group consisted of 52 patients from Serbian high-risk breast/ovarian cancer families (BRCAPRO: CP>10%) (age range 25-69 years, median 49). All of them had previously tested negative for sequence alterations and large exon deletions and duplications in the BRCA1/2 genes. These individuals were selected from a larger group of BRCA1/2-negative high-risk patients (n = 132) tested in our laboratory from 2010 to 2015. Together with the BRCAPRO result, the inclusion criteria for women with a family history of breast/ovarian cancer and for classifying them into the hereditary group were: breast cancer patients with at least 1 breast or ovarian cancer case diagnosed in first- or second-degree relatives, patients with both breast and ovarian cancers, patients with bilateral breast carcinoma, and young breast cancer patients (less than 40 years of age). The subgroup of patients from the hereditary group with the disease diagnosed before the age of 40 was also analyzed for genotype associations with early-onset hereditary breast cancer. The second, “sporadic” group consisted of 106 breast cancer patients without a family history of breast/ovarian cancer, all treated at the Clinic for Medical Oncology at the Institute for Oncology and Radiology of Serbia (age range 32-77 years, median 52). The third, “control” group consisted of 104 age-matched (by 10-year age intervals) cancer-free women (age range 31-69 years, median 53). All tested individuals were of Caucasian descent.
The study was approved by the Ethics Committee of the Institute and each patient gave written informed consent.
Dna Isolation
DNA was isolated from peripheral blood on an ABI Prism™ 6100 Nucleic Acid PrepStation (Applied Biosystems) using a BloodPrep™ chemistry according to the manufacturer's instructions.
Genotype Determination
The XRCC1 R399Q polymorphism was determined using the following primers: sense: 5’-CAAGTACAGCCAGGTCCTAG-3’, antisense: 5’-CCTTCCCTCATCTGGAGTAC-3’, resulting in a 268-bp PCR product. Thermal cycling conditions were as follows: initial denaturation step at 94°C for 5 minutes, 32 cycles of 94°C for 60 seconds, 58°C for 30 seconds, 72°C for 40 seconds and final elongation for 10 minutes at 72°C. PCR products were visualized by electrophoresis on 2% agarose gel and digested with the BcnI fast digest restriction enzyme (Thermo Scientific) according to the manufacturer's instructions. Digestion resulted in 91-bp and 177-bp fragments corresponding to the Arg (R) allele, or a 268-bp fragment representing the Gln (Q) allele. Fragments were detected using a 2100 Bioanalyzer (Agilent Technologies) according to the manufacturer's instructions. The RAD51 G135C and TP53 Arg72Pro polymorphisms were genotyped with restriction fragment length polymorphism polymerase chain reaction (RFLP-PCR) as described previously (15). A sample with each genotype was sequenced for confirmation and was further used as positive control. To exclude potential contamination, negative controls consisting of dH2O instead of DNA were used in both PCR and RFLP reactions after each batch of samples.
Statistical Analysis
Allele and genotype distribution between groups was tested by Pearson's chi-square test (χ2) with Bonferroni's correction or by Fisher's exact test. A p value <0.05 was considered statistically significant. The odds ratio (OR) and 95% confidence interval (95% CI) were calculated to estimate the strength of associations of allele and genotype variants with breast cancer.
Results
An example of the RFLP analysis results of XRCC1 polymorphic variants is presented in Figure 1.

Detection of XRCC1 R399Q by PCR-RFLP. Column L: High-sensitivity DNA ladder (Agilent Technologies). Column 1: 268-bp-long PCR product. Columns 2, 4 and 6: both alleles contain restriction sites cleaved by the BcnI enzyme; 2 bands on the gel (91 bp and 177 bp) represent the RR genotype. Column 3: one allele contains restriction sites (91 bp and 177 bp) while the other does not (268 bp); 3 bands on the gel represent the heterozygote RQ genotype. Column 5: the QQ genotype is characterized by the absence of a BcnI restriction site on both alleles which showed a single gel band (268 bp).
The distribution of genotypes in all tested groups was as follows: 26.34% for the dominant homozygote (R399R), 33.21% for the heterozygote (R399Q), and 40.46% for the recessive homozygote (Q399Q). The frequency and distribution of the XRCC1 399R and 399Q alleles among all tested groups are shown in Table I.
Allele and genotype frequencies, odds ratios and 95% confidence intervals of the XRCC1 R399Q polymorphism in hereditary breast cancer, sporadic breast cancer and control samples
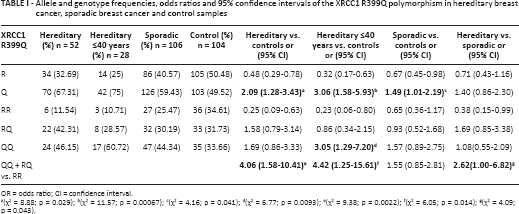
OR = odds ratio; CI = confidence interval.
(χ2 = 8.88; p = 0.029);
(χ2 = 11.57; p = 0.00067);
(χ2 = 4.16; p = 0.041);
(χ2 = 6.77; p = 0.0093);
(χ2 = 9.38; p = 0.0022);
(χ2 = 6.05; p = 0.014);
(χ2 = 4.09; p = 0.043).
Analysis of the XRCC1 polymorphic variants showed statistically significant differences in distribution between the 3 investigated groups (χ2 = 10.718; df = 4; p = 0.0299). Pearson's chi-square test with Bonferroni's correction showed a significant difference in the XRCC1 genotype distribution between the hereditary group and the control group (χ2 = 9.389; df = 2; p = 0.00914). Further analysis revealed the Q allele to be associated with both hereditary and sporadic breast cancer. The OR (and 95% CI) for the Q allele in relation to the R allele in the hereditary group was 2.09 (1.28-3.43) and for carriers of the Q allele in the dominant genetic model (QQ + RQ vs. RR) it was 4.06 (1.58-10.41) in the same group (Tab. I). The importance of the XRCC1 399Q allele was confirmed in patients aged younger than 40 years of the hereditary group. The OR (and 95% CI) for the Q allele in relation to the R allele in this group was 3.06 (1.58-5.93). The significance of the Q allele in both recessive (3.05 [1.29-7.20]) and dominant (4.42 [1.25-15.61]) genetic models was shown in women younger than 40 years of the hereditary group (Tab. I). The Q allele showed a statistically significant association with sporadic breast cancer with an OR (and 95% CI) for the Q allele in relation to the R allele of 1.49 (1.01-2.19). We found no statistically significant difference regarding the variant XRCC1 allele and genotype frequencies between the sporadic group of patients younger than 40 years and the control group (data not shown). We also analyzed the differences between patients from the hereditary group and patients from the sporadic group. In dominant genetic model, data showed statistical significance with OR of 2.62 and 95% CI of 1.00-6.82 (Tab. I). Results about individual RAD51 G135C and TP53 Arg72Pro polymorphisms have been published previously (15).
In this study, we assessed the distribution of RAD51, TP53 and XRCC1 genotype combinations among the groups (Tab. II). The combination of the RAD51/XRCC1 genotypes with 1 recessive homozygote and 1 heterozygote (CC/RQ or GC/QQ), which means the existence of 3 recessive alleles in this genotype combination, showed a significant difference between the hereditary group and the control group and an association with hereditary breast cancer with an OR (and 95% CI) of 3.6 (1.11-11.63) (Tab. II). The frequency of a combined RAD51CC+TP53ArgPro+XRCC1RQ genotype was significantly higher in the hereditary group than in the sporadic group (χ2 = 6.233; p = 0.013) with an OR (and 95% CI) of 4.08 (1.26-13.22). The combination of the 3 dominant genotypes, RAD51GG+TP53ArgArg+XRCC1RR, was significantly more frequent among healthy controls than in the hereditary cancer group (χ2 = 10.8; p = 0.001) with an OR (and 95% CI) of 0.05 (0.003-0.85). All other combinations of the RAD51, TP53 and XRCC1 genotypes were tested but the differences between their frequencies were not significant and no association was confirmed. There were no subjects with a recessive homozygote status for all 3 investigated genes.
Distribution of combined genotypes of RAD51 G135C, TP53 Arg72Pro, and XRCC1 R399Q in breast cancer
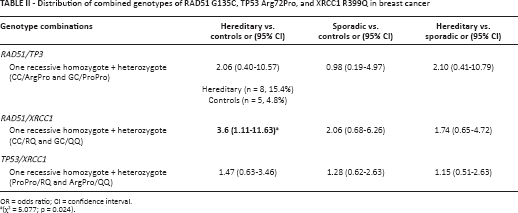
OR = odds ratio; CI = confidence interval.
(χ2 = 5.077; p = 0.024).
Discussion
Breast cancer remains the second most common cause of cancer mortality in the Western world despite ongoing efforts to understand its etiology and improve its outcomes (16). In Serbia, breast cancer is diagnosed in approximately 4,600 women annually, of whom more than a third are diagnosed at the Institute for Oncology and Radiology of Serbia. Although important, mutations in the BRCA1/2 genes account for only a small part of the inherited susceptibility to breast cancer. In principle, the remaining susceptibility could be explained by single genes with rare, strongly predisposing alleles similar to BRCA1/2. However, despite significant efforts invested so far, such a high-penetrance gene in breast cancer (BRCA3) has not been identified. Another explanation for the hereditary predisposition is that it could be polygenic – the result of combined effects of moderately or weakly predisposing alleles in a number of genes. The contribution to the overall genetic effect thus depends on the frequency of each allele and its strength in terms of the risk. In the focus of current research are common alleles conferring a low risk identified by genome-wide association studies (GWAS) (4). Differences in allele frequencies observed between populations today have been gradually shaped by various historical events (e.g., genetic drift, admixture, founder effects) (14). Thus, some genetic variants are frequent in specific populations, while in others they would be difficult to find. Similarly, some variants are likely to be subgroup specific and require studies focused on specific populations.
Common small alterations in low-penetrance genes (SNPs) can have low individual impact, but it is also believed that combinations of low-risk alleles may have a greater impact and can act additively or multiplicatively, contributing significantly to the cancer risk (17). The effect of a particular SNP may be reflected in the earlier onset of the disease, as the SNP causes or increases the risk of its development (8). Even though they are very powerful means to identify risk alleles for common diseases (18, 19), GWAS still have considerable limitations. Thus, understanding predisposition across populations will require discovery efforts in diverse populations.
The association between the TP53 Arg72Pro polymorphism and breast cancer has been examined in many populations, with conflicting results. Besides the sample size and differences in defining investigated groups, the main reasons for these discrepancies are evolutionary factors and population structure. While some publications report the Pro allele and ProPro genotype as a risk factor for breast cancer and early breast cancer (20-21-22-23), others emphasize the importance of the ArgArg genotype (24, 25). Proline homozygosity was also associated with decreased breast cancer risk (26), but there were also studies that found no correlation of the Arg72Pro polymorphism with breast cancer risk (27, 28). In our previous study (15) we found that the Pro allele was associated with the hereditary form of breast cancer, but it was investigated only as a single marker.
Given that many regulatory elements are linked to the 5’ untranslated region, it is believed that RAD51 G135C can influence mRNA processing, transcriptional regulation, translational efficiency and mRNA stability. Although this SNP has been extensively studied as a modifier of BRCA1/2 penetrance (29), the results about its individual effect on breast cancer are conflicting. It has been reported that the 135C allele in the homozygote form is associated with breast cancer in European populations (30, 31), but the heterozygous genotype GC was also associated with an increased risk of breast cancer (32). The 135C genotypes (GC and CC) were associated with an increased disease risk In South Americans only among women with a) a family history of breast cancer, b) a BRCA1/2-negative genotype, and c) age at onset <50 years (33). Our previous results showed an association of the CC genotype with an elevated breast cancer risk among women with a positive family history of the disease (15). However, RAD51 G135C was investigated only as a single marker.
The XRCC1 protein has a leading role in base excision repair and its polymorphic variants R194W (rs1799782), R280H (rs25489) and R399Q (rs25487
To our knowledge, the specific role of combined TP53 Arg72Pro, RAD51 G135C and XRCC1 R399Q genotypes in breast cancer has not yet been reported on. Since the protein products of these genes form a complex array of mutual interactions and may contribute to interindividual variation of DNA repair capacity and affect cancer susceptibility, we investigated the distribution of their combined genotypes and its role in breast cancer in Serbian women. We showed that carriers of 3 recessive alleles of the RAD51 and XRCC1 variants in their genome simultaneously (CC/RQ or GC/QQ) have an increased risk of hereditary breast cancer. Analysis of combined genotypes indicated that the RAD51 CC + TP53 ArgPro + XRCC1 RQ combined genotype is more frequent in hereditary than sporadic breast cancer. OR analysis confirmed its importance, which means that those women who have this exact genotype combination and come from high-risk families have an increased risk of developing the disease compared to women who have the exact same genotype but come from families without the burden of the disease. OR analysis indicated a strong protective role of the RAD51 GG + TP53 ArgArg + XRCC1 RR genotype against hereditary breast cancer negative for BRCA1/2 mutations. This means that women who have all dominant alleles of these variants in their genome are less likely to develop the disease than women who have at least 1 recessive allele. A strong protective role against hereditary breast cancer of 3 dominant genotypes in combination has been indicated through our results, but larger studies are needed to confirm this observation due to the fact that this exact genotype combination is rare. Further studies will also be needed in order to evaluate the distribution of this genotype combination in different populations.
In conclusion, our findings suggest that the XRCC1 R399Q polymorphism has a significant association with breast cancer risk in the Serbian population. Its role is emphasized in the BRCA1/2-negative hereditary form of the disease and in young breast cancer patients from breast/ovarian cancer families. Dominant alleles of the RAD51, TP53 and XRCC1 combined genotypes showed a strong protective effect against BRCA1/2-negative hereditary breast cancer compared to women who have at least 1 recessive allele in their genome. Considering their great importance in double strand break repair and the potential to modify the penetrance of BRCA1 and BRCA2 mutations, the problem of the genetic variability of these polymorphisms in BRCA1/2-mutated breast cancer also needs to be further investigated.
Footnotes
Financial support: This study was supported by a grant from the Ministry of Education and Science of the Republic of Serbia (grant number 41026).
Conflict of interest: The authors declare that they have no conflict of interest.
