Abstract
ABSTRACT
Introduction
Myelodysplastic syndrome (MDS) comprises a group of clonal hematological disorders, characterized by ineffective hematopoiesis and progressive bone marrow failure. It increases the risk of transformation to acute myeloid leukemia (AML). Therapeutic benefit should include overall survival increase (OS), hematological improvement, transfusion dependence and time to progression to AML decrease.
Objective
Assess, from a Mexican health-care perspective, the cost-effectiveness of azacitidine compared with low-doses of chemotherapy (LDC) plus best supportive care (BSC) for the treatment of adult patients with intermediate-2 and high-risk MDS, who are not eligible for hematopoietic stem-cell transplantation. We developed a cost-effectiveness survival analysis model of three stages: MDS, AML, and death. OS and costs are extrapolated beyond three-year time horizon. Discount rate of 5% was applied. To estimate the model cycle probability transition to mortality state, survival curves were constructed for each treatment arm using individual patient-level data from Study AZA-001. Unitary costs are from public price list, and profiles for the management of MDS and AML were collected separately using a structured questionnaire. Probabilistic sensitivity analyses (PSA) were conducted by simultaneously sampling from estimated probability distributions of model parameters.
Results
Overall survival was projected to increase by 72.26 weeks with azacitidine. Incremental expected total costs for azacitidine compared to LDC was MXN$68,045. However, the cost of the drug therapy was lower with azacitidine. The incremental cost-effectiveness ratio (ICER) for azacitidine compared to LDC was MXN$48,932 per life-year gained (LYG). PSA showed that azacitidine was a highly cost-effective option in 96.49% of the simulated cases in MXN$180,000/LYG willingness-to-pay.
Conclusions
Compared with LDC, azacitidine represents a cost-effective treatment alternative in patients with MDS from a Mexican perspective.
Keywords
Introduction
Myelodysplastic syndrome (MDS) comprises a heterogeneous group of clonal hematological disorders that are characterized by ineffective hematopoiesis leading to one or more peripheral blood cytopenias and progressive bone marrow (BM) failure. This results in an increased risk of malignant transformation to acute myeloid leukemia (AML) (1, 2).
The pathogenesis of MDS is complex and has not been fully characterized. Models have been proposed whereby MDS develops along a multistep process, during which a hematopoietic stem cell is mutated and attains a growth advantage. The resulting mutated cell clone causes morphological dysplasia, impaired cell differentiation and hematopoiesis, and genomic instability. The immune system is impaired as a result of altered cytokine secretion and apoptotic pathways: in early MDS, excessive apoptosis is thought to contribute to cytopenias and a hyper-cellular BM, whereas in later stages of MDS, decreased apoptosis and subsequent clonal expansion is thought to promote progression to AML; BM failure results (3). Overall, an estimated 20%-30% of MDS patients will eventually progress to develop AML. However, even in the absence of progression to AML, the prognosis for MDS patients is poor: frequent complications include infections as a result of neutropenia, which may be fatal, and life-threatening hemorrhages as a result of thrombocytopenia (4).
Given the rarity of MDS and its poor prognosis, it is difficult to ascertain incidence and prevalence accurately. MDS can affect all ages; however, it is predominantly a condition of the elderly, with the highest occurrence seen in patients >65 years of age (1, 4). Most epidemiological knowledge to date has been obtained from statistical studies of selected regional populations, making a true reflection of the figure across a wider population difficult to obtain (5). An increase in the incidence of MDS has been noted in recent years, but this is predominantly attributed to wider recognition and diagnosis of the condition and the introduction of an improved classification system. Incidence rate per 100,000 habitants/year was estimated by 4-12 in Mexico (6).
MDS management is complicated due to the advanced age of the patient population and their inability to tolerate standard-dose chemotherapy, as well as the coexistence of non-hematological morbidities (4). Treatment of all MDS patients should consider both management of symptoms and prolongation of survival. Aside from clinical response, benefit may also be obtained from hematological improvement, a decrease in transfusion dependence and a decrease in time to progression to AML (7, 8). Patients need to be managed on an individual basis, with age, performance status, major comorbid conditions, psychosocial status and availability of a caregiver being taken into consideration when deciding treatment choice, dose and length. Management decisions should be taken with the full involvement of the patient (9).
There is a considerable unmet treatment need in higher-risk MDS patients, owing to the high morbidity, greater transfusion burden, rapid progression to AML and high mortality in this population. UK treatment guidelines for MDS suggest that, from a patient quality of life (QoL) perspective, a stable augmented hemoglobin concentration may be preferable to the cyclical fluctuations of red blood cell (RBC) transfusions. The Cancer and Leukemia Group B (CALGB) conducted studies of single-agent low-dose azacitidine, which demonstrated clinical benefit (9). Thus, there is a need for novel forms of treatment that can improve hematological parameters, and azacitidine fulfils this criterion. The aim of Study AZA-001 was to compare the effect of azacitidine plus BSC with conventional care regimens (CCRs) plus best supportive care (BSC) on overall survival (OS) in higher-risk MDS patients (10). The primary efficacy endpoint in Study AZA-001 was time to death from any cause. Secondary efficacy endpoints were time to transformation to AML, hematological response and improvement according to International Working Group criteria for MDS, independence from RBC transfusions for 56 consecutive days or more, number of infections requiring intravenous antimicrobials, and occurrence of adverse events (AEs).
The reduction in risk of death on azacitidine compared with CCR was 42% (p = 0.0002). At two years, the proportion of patients surviving was approximately twice in the azacitidine group than in the CCR group (50.8% vs. 26.2%; p<0.0001). The median time to transformation to AML was also greater in the azacitidine group (17.8 vs. 11.5 months; p<0.0001). In summary, azacitidine significantly lengthens overall survival in patients with higher-risk disease (International Prognostic Scoring System [IPSS] categories intermediate-2 and high-risk MDS) (10). The objective of the present evaluation was to assess, from a Mexican health-care system perspective, the cost-effectiveness of azacitidine compared with low-dose chemotherapy plus BSC for the treatment of adult patients who are not eligible for hematopoietic stem-cell transplantation with intermediate-2 and high-risk MDS according to the IPSS.
In Mexico, the Diagnostic and Treatment Guide for Myelodysplastic Syndrome (6), recommends the use of low-dose cytarabine in patients with MDS. Based on this and having few alternatives within the institutions and being the more appropriate clinical comparator for the treatment of these patients it was decided to take low-dose chemotherapy (LDC) as the alternative to be compared in this model.
Methods
A partitioned survival analysis model was developed to estimate the expected life-time outcomes and costs of treatment of MDS patients with intermediate-2 and high-risk MDS according to the international prognostic scoring system who were alternatively assumed to receive azacitidine 75 mg/m2 daily for seven days, followed by a rest period of 21 days (28-day treatment cycle) or LDC regimen of cytarabine 20 mg/m2/day administered subcutaneously for 14 days every 28 days. BSC, as described above, is provided in addition to LDC (10).
Clinical effectiveness (progression-free survival [PFS], OS, and AEs) for azacitidine and LDC were based on the results of the AZA-001 trial. Other model parameters were based on data from secondary sources identified by a review of the literature and from a structured questionnaire. To the possible extent, the methods used in the evaluation are consistent with guidelines for the economic evaluation from the Mexican General Health Council (GHC) (11).
The partitioned survival analysis model used in this study is similar to the q-twist approach, a well-established analytic framework for evaluating oncology therapies (12), and also to the models used in numerous earlier economic assessments of treatments for MDS, including recent evaluations of the cost-effectiveness of azacitidine for the same disease (13, 14).
In this approach, survival is partitioned into three mutually exclusive health states:
MDS (using the PFS estimated from the trial AZA-001).
AML (blasts >30%).
Dead (using the OS estimated from the trial AZA-001).
OS and the associated costs are extrapolated beyond the three-year time horizon of the pivotal clinical trial. To estimate the model cycle transition probability to the mortality state and the life-time extrapolation, survival curves were constructed for each treatment arm using individual patient-level data from Study AZA-001.
The patients treated with azacitidine were stratified according to their pre-randomization arm, so that like-for-like patient groups are compared (for example, only patients who had been preselected by the investigating physician as suitable for treatment with LDC before randomization to azacitidine are compared with patients randomized to receive LDC).
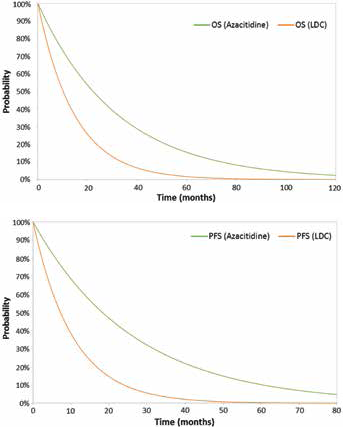
Expected MDS and expected OS are calculated as the area under their respective survival curves. PFS and OS (Fig. 1A, 1B).
ICER = incremental cost effectiveness ratio; AEs = adverse events.
Expected AML survival is the area between the MDS and OS curves. Costs were assumed to be conditioned on treatment and expected time in the given disease states. This approach is similar to a traditional Markov model (15), except that it does not require explicit calculation of transition probabilities between states. Weekly cycles and a half-cycle correction were used in the analysis.
Outcomes calculated by the model for each treatment included expected progression-free life-years, expected post-progression life-years, expected overall life-years and expected life-time costs of this disease. The incremental cost-effectiveness ratio (ICER) was calculated as the ratio of the difference in expected life-time cost of MDS care to the expected difference in life-years (“cost per life-years gained [LYG]”) between azacitidine and LDC. All outcomes were evaluated over a 10-year time-frame, beginning with treatment start. This time-frame approximates a life-time projection, consistent with GHC recommendations (11). The analysis was conducted from the perspective of the Mexican publicly funded health-care system and is focused specifically on the costs of MDS-related care. Expected outcomes and costs were calculated on a discounted basis using an annual discount rate of 5% (16). The model was programed using Microsoft Excel (Microsoft Corporation, Redmond, WA, USA).
Parameters used in the model are reported in Table I and described below.
Model parameters
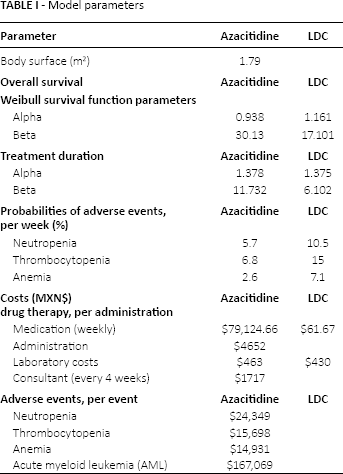
Because Kaplan-Meier estimates of PFS and OS were greater than zero when analyses of PFS and OS were conducted, it was necessary to project them beyond the end of the trial to obtain life-time projections. Consistent with GHC guidelines (11), estimates of PFS and OS were based on extrapolation.
Disease progression occurs when the patient progresses to AML. While this is recorded in the clinical trial, there are a large number of censored data points, which makes estimation of the time to transformation to AML difficult in each treatment arm. Rather than forward estimating the time to AML progression from randomization, the model calculates the pooled time in AML across all treatment arms and back calculates the time to AML progression from the survival curves. The pooled median time spent in AML across all treatment arms is 3.65 five-week cycles. This average time is used to recalculate the progression to AML curves based on the survival curves. It is assumed that the progression to AML curves maintain the same shape as the survival curve for each treatment arm, but the curve median is adjusted so that:
AML curve median = survival curve median – 3.65.
The curve fits to the trial survival data were analyzed using the Akaike Information Criteria (AIC). AIC values are based on model fit (how well the observed data fit the model) and the principle of parsimony (simple models are better, so there is a penalty for added parameters). The AIC statistics showed that there is very little to choose between the Weibull and log-logistic curve fits to the observed overall survival data. There is, however, a difference when the tail of the curve fits is examined. It appeared that the log-logistic curve overestimates the PFS and OS; therefore, the Weibull survival function was used.
The Weibull is a flexible survival function that allows for increasing or decreasing risk of events over time and takes the general form:
S(t) = exp (-αtβ)
where S[t] is the probability of not having experienced the event (for example, progression or death) at time t (17). Alpha (α) is often called the “event rate parameter,” and beta (β), the “shape parameter.” For β = 1.0, the hazard rate is constant over time and the inverse of α is the mean failure time. For β<1, the hazard rate for the event is a decreasing function of time. For β>1, the hazard rate is an increasing function of time.
The modelled median survival times are slightly different from the trial medians, due to the curve fits, which extrapolate the long-term survival. This is mainly caused by the numerous censoring points around the median survival in the observed data and in the tail of the curve, which drive the curve shape. Figure 1 shows empirical and fitted survival functions for PFS and OS for azacitidine and LDC.
Adverse events
Estimates of the incidence of AEs for azacitidine and LDC were obtained from the AZA-001 trial. AEs considered in the model included neutropenia, thrombocytopenia and anemia. Those events were included either because their incidence was higher among patients receiving azacitidine or because the event is of particular concern for the disease. Only grade 3 and 4 events were modelled.
There are, however, some difficulties in correctly applying the rate of AEs to the modelled treatment arms. The rates recorded in the trial are subject to decay. The rates are higher during initial treatment and then the effects dissipate over time. However, this rate of decay is difficult to determine and match to the extrapolated model. Therefore, cumulative incidence estimates were converted to constant monthly probabilities, assuming that patients are at risk of adverse events until death.
Costs
Resource use profiles for the management of MDS and AML (blasts >30%) were collected separately using a structured questionnaire (for example, routine follow-up). Additionally, resource use profiles for patients with MDS were collected based on their therapy. Treatment-associated costs (for example, administration of treatment, monitoring) are applied while patients receive the allocated therapy; upon treatment cessation, the treatment-associated costs of BSC are applied until disease progression (transformation) to AML. Subsequently, the costs associated with AML management are applied only for those who progressed.
The unit cost of cytarabine was obtained from public acquisitions (18). The unit cost of azacitidine acid was provided from Celgene. The cost per administration of azacitidine and LDC was obtained from the Instituto Mexicano del Seguro Social (IMSS), the biggest public health institute in Mexico (19). To estimate duration of treatment for azacitidine and LDC, time to discontinuation was estimated using a Weibull function (also provided in the AZA-001 trial). Costs of AEs were based on a published article related to these events in Mexico (20). Additionally, costs for laboratory tests were obtained from the IMSS (19) and the cost for AML management was based on diagnosis-related group also published by IMSS (21). Costs of induction chemotherapy were not considered, because those costs are not likely to be affected by these therapies. Cost estimates were adjusted to 2015 Mexican pesos as necessary (22).
Analyses
Probabilistic sensitivity analyses were conducted by simultaneously sampling from estimated probability distributions of model parameters to obtain 3000 sets of model input estimates (23). The cumulative incidences of AEs were assumed to be distributed as beta random variables. Other estimates were assumed to be distributed as either gamma or normal random variables. If standard errors for model estimates were unavailable, they were assumed to be 20% of their base-case estimates. For each simulation, we calculated the differences between azacitidine and LDC in costs and life-years gained. The 95% credible intervals (CrI) for incremental costs and life years gained (LYG) were calculated based on the 2.5 and 97.5 percentiles of the simulations (24). The uncertainties were expressed graphically through cost-effectiveness acceptability curves that showed the probability that azacitidine is cost-effective (
Deterministic sensitivity analyses were undertaken to explore the effect of azacitidine on the ICER by changing assumptions concerning the values of key model parameters. Key model probabilities and costs were varied across their 95% confidence intervals (if available) or, otherwise, using ±20% of base-case values. Cost-effectiveness was calculated using annual discount rates of 0% (that is, no discounting) and 3% for the deterministic sensitivity analysis.
Results
Table IIa, b, c, presents the base-case results. In the life-time analysis, overall survival was 136.75 and 64.49 weeks for azacitidine and LDC, respectively. OS was projected to be increased on average by 72.26. LYG were 1.38. PFS increases with azacitidine 68.10 weeks (1.30 years). Expected cost including drug therapy, administration and monitoring costs, the expected cost of azacitidine was MX$1,449,040 vs LDC MX$1,430,994. The incremental cost was only MX$68,045. However, drug therapy was lower with azacitidine, while the incremental cost with azacitidine are manly for the increase in the overall survival.
Base-case results - life-time analysis
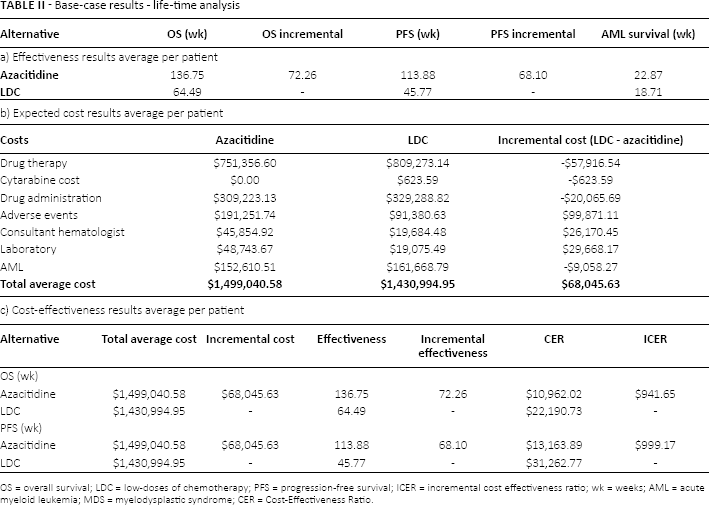
OS = overall survival; LDC = low-doses of chemotherapy; PFS = progression-free survival; ICER = incremental cost effectiveness ratio; wk = weeks; AML = acute myeloid leukemia; MDS = myelodysplastic syndrome; CER = Cost-Effectiveness Ratio.
Average cost effectiveness ratio is lower with azacitidine MXN$10,972 versus MXN$22,190 of LDC. The ICER per life-year gained is only MXN$48,932. This ICER is highly cost effective, according to decision rules in Mexico, the threshold used for acceptable input on comparators is - if it produces ICER ≤1 gross domestic product (GDP) per capita threshold (MXN$180,000/LYG) (26).
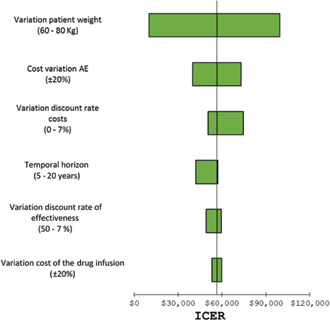
Tornado diagrams were created based on one-way sensitivity analyses performed by applying the upper and lower boundary given previously around each of the parameters in the model. Figures 2, 3 and 4 present results of the sensitivity analysis with tornado diagram, probabilistic sensitivity analysis and acceptability curve, respectively. In general, the results are relatively insensitive to the parameter changes reflected in the various scenarios. Assuming life-time benefit (time horizon = 20 years) and a time horizon of five years, the estimated cost-effectiveness of azacitidine acid compared with LDC ranged from MXN$42,020 per LYG to MXN$56,969 per LYG.
OS = overall survival; PFS = progression-free survival.
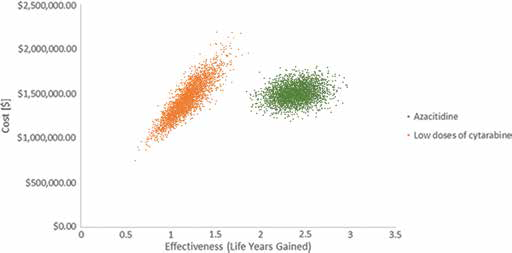
Probabilistic Sensitivity Analysis.
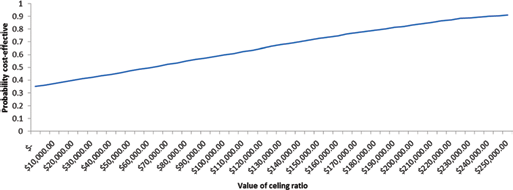
Acceptability Curve.
The most sensitive parameter of this analysis was the patient's weight, expected cost-effectiveness ranged from MXN$10,254 per LYG to MXN$99,489 per LYG. Varying the administration cost and the AML cost had a minimal effect on the ICER because patients in the model spent a small amount of time in the AML health state before death.
The probabilistic sensitivity analysis showed that azacitidine was a cost-effective option in 96.49% of the simulated cases MXN$180,000/LYG willingness-to-pay. The probabilistic sensitivity analysis confirmed the robustness of the results of the model.
Discussion
Our study is the first economic evaluation published for higher-risk MDS in Mexico. We assess to determine the economic value of azacitidine in the treatment of higher-risk MDS. When compared separately with LDC, the mean ICER for azacitidine was MXN$56,471 per life-year gained. The results of our analysis demonstrate that the economic value for treatment of higher-risk MDS patients with azacitidine is within the range of currently reimbursed medicines in Mexico.
Several studies have assessed the clinical benefits of azacitidine compared with CCR and have shown clinical superiority, but economic evaluations are lacking. A Canadian study comparing azacitidine with CCR options (BSC, LDC, standard chemotherapy [SDC]) confirmed the superiority of azacitidine in terms of cost-effectiveness, with a global ICER of CAD$84,395/QALY gained, which was below the 3xGDP WHO threshold (38,710×3) (27) for Canada. The study also showed the superiority of azacitidine compared with each of the other treatment options, with the following ICER values: CAD$84,395/QALY gained for BSC, CAD$88,786/QALY gained for LDC and CAD$28,501/QALY gained for SDC (14). This alternative was not included in our analysis due to not being used in the clinical practice.
A recent study compared azacitidine with decitabine and found that azacitidine was a cost-effective treatment for MDS according to US National Healthcare Input data (28), with a comparative gain of 0.171 more QALYs and savings of €15,890 over a 2-year period. However, the relevance of the study is limited, mainly because survival data were retrieved from two different phase III trials and no direct comparison was made.
A key strength of the present analysis is the use of the AZA-001 trial data. Because that trial had no cross-over, the statistically significant overall survival advantage (9.4 months) for azacitidine compared with the alternatives is a clinically significant increase. The comparator used in the clinical trial closely reflects the standard-of-treatment options available to higher-risk MDS patients in Mexico, thus providing robust clinical evidence of efficacy for “real-world” treatment options.
Limitations of this study should be noted. First and foremost, the evaluation was based on assumptions regarding the treatment effects of azacitidine on OS beyond the reported end-of-follow-up in the AZA-001 trial. It is recommended to consider the fact that this approach is based on a mathematical model which depends mainly on the accuracy of available data in the moment of the analysis and should be treated as such. Also, in the absence of end-of-life survival data from the AZA-001 study, survival curves were extrapolated using the Weibull distribution. Considering that major clinical benefits are visible in the long term and that our results are difficult to collate due to the fact that MDS is an orphan disease, conclusions have to be made with caution.
Second, data on the costs of adverse events were not available from the trial and were obtained from secondary sources. We conservatively estimated the costs of AEs based on a study based on patients with colorectal cancer (20). The values we used in the model are therefore likely to overestimate the costs of AEs and, on balance, are therefore conservative. In any case, model results were tested in the sensitivity analysis, so that any lack of precision in the cost estimates is not likely to have materially biased our findings one way or the other.
Compared with LDC, azacitidine represents a cost-effective treatment alternative in patients with MDS from a Mexican perspective.
Footnotes
Financial support: We appreciate the unrestricted support provided to carry out this project to the Laboratory Celgene, S. de R.L. of C.V.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
