Abstract
The frequency of K-RAS mutations ranges between 30% and 48% among the Caucasian, Asian, and European populations and these mutations are predictors of response to EGFR therapies. We sought to determine the expression of K-RAS gene mutations among colorectal cancer patients in PuertoRico. A retrospective study was conducted to determine the expression of mutant K-RAS among colorectal cancer patients in Puerto Rico between April 2009 and January 2011. The mutant expression of K-RAS was found in 39% (n=195) of the Puerto Rican population, and was more common in the age group of 51-69 years (53.8%) and in males (55.4%, p>0.05). Moreover, mutant K-RAS was more commonly found in tumors of the proximal area (43.8%; p=0.03), with distant metastasis (43.3%, p=0.018), with a mucinous histotype (31.7% p>0.05), and in ulcerated tumors (38.8%, p>0.05). K-RAS mutations were observed on codon 12 (87.7%) and codon 13 (12.3%). The most frequent mutation on codon 12 was 12 ASP (39.5%), followed by 12 VAL (25.4%) that is associated with a significant decrease in overall cancer survival. The mutant expression of K-RAS in cases of rectum carcinoma was 39.5%, where the most common mutation was 12 VAL (37.5%). The frequency of K-RAS mutations in the Puerto Rican population here studied was 39% and mutant K-RAS was associated with advanced colorectal cancer stage, mucinous histotype, and ulcerated tumors.
Introduction
Colon carcinoma is the third most common type of malignancy diagnosed in the US. It is also the fourth leading cause of death for males, accounting for 19.7 deaths per 100,000 man, and the second leading cause of death for women accounting for 13.8 deaths per 100,000 females (1). Colorectal cancer is the second most common cancer among Hispanic men and females, accounting for 44.8 cases per 100,000 males and 32.6 cases per 100,000 females (1). In Puerto Rico, its incidence is similar to the incidence of colorectal cancer among Hispanics in US mainland, accounting for 45.4 cases per 100,000 males and 32.1 cases per 100,000 females (2, 3). Advances in cancer therapy have significantly improved cancer survival rates; however disparities are still affecting the Hispanic community (1). A previous meta-analysis summarized the therapeutic considerations for treating colorectal cancer patients with mutant K-RAS; these patients are not benefited from one of the best promising cancer therapies such as the epidermal growth factor (EGFR) therapy (4–6). The K-RAS mutation is a common oncogenic alteration in human cancer that regulates multiple pathways affecting differentiation, cell growth, and apoptosis. Racial/ethnic differences in the status of K-RAS mutation have been reported ranging around 30%-48% among the North American, Asian, and European populations (7–9, 15). Previous studies have reported a significant higher prevalence of mutant K-RAS among US minorities including African Americans (11). Race/ethnicity could be a significant predictor of the presence of mutant K-RAS and should be taken into consideration for treating colorectal cancer patients with mutant K-RAS. The K-RAS mutant status has not been reported for the Puerto Rican population; therefore, there is an urgent need to determine the frequency of the status of K-RAS mutation among the Puerto Rican population. We sought to determine the prevalence of K-RAS mutations in colorectal carcinoma in Puerto Rico and to describe the sociodemographic and clinical variables according to the type of K-RAS mutation.
Methods
We performed a cross-sectional study to determine the frequency of K-RAS mutations from DNA extracted from paraffin-embedded colorectal tissues between April 2009 and January 2011 by collecting GENOPTIX medical laboratories data. K-RAS mutation analysis includes sectioning of formalin-fixed paraffin-embedded (FFPE) tumor specimens, followed by hematoxylin and eosin staining of the tumor-rich region (confirmed by the pathologist). DNA is extracted from a microdissected tumor section and the presence of 1 of 7 K-RAS mutations is identified by target amplification via PCR and real-time fluorescent signal detection by DxS Scorpions-ARMS PCR. This assay can detect mutant DNA at levels as low as 1%; however, the sensitivity may not be as low as 1%. Possible reasons include excessive DNA fragmentation, PCR inhibitors, improper fixation, embedding, and storage. The presence of any of the 7 K-RAS mutations in codon 12 and 13 were identified by real-time PCR. GENOPTIX data includes data from each of the 6 Puerto Rican health regions. We reviewed a total of 529 reports of K-RAS mutation tests. From these reports, 28 cases were excluded due either to incomplete information regarding the origin of pathology (n=7), or to K-RAS mutant test performed in other tumors (n=21) rather than colorectal carcinoma, including stomach carcinoma (n=1), lung carcinoma (n=15), penile carcinoma (n=1), anal carcinoma (n=1), pseudomixoma peritoneum (n=1), tonsil carcinoma (n=1), and mediastinal mass (n=1). The following information was extracted from the K-RAS reports: age at time of test, gender, Puerto Rican municipality, tumor stage, tumor site, total number of nodules, metastasis, histology, mucinous type of cancer, date of sample collection, date of surgery, date of K-RAS analysis, site of K-RAS, K-RAS result and type of mutation. The patient's confidential information was excluded from GENOPTIX K-RAS reports. For identification purposes and to avoid data duplication, we assigned a unique identification number to each K-RAS report. A total of 501 reports met the inclusion criteria. The patient group included 209 women and 292 men with ages ranging from 25 to 92 years. A large number of the patients were from the Metro-Fajardo region (n=157; 31.3%) followed by the Mayaguez-Aguadilla region (n=147; 29.3%). Overall, 232 colorectal tumors were localized in the colon, 86 in the rectum, and 183 tumors had a non-specified colon location. Mutant K-RAS was found in 195 cases. For the purpose of this analysis, we only included 114 cases with mutations in codon 12 or codon 13. The study was approved by the IRB from the Universidad Central del Caribe-School of Medicine.
Statistical analysis
Univariate statistics (percentage and frequencies) and central tendency measures (mean) were used in order to describe and explore the data. Cross tabulations using a chi-square distribution were performed to compare proportions of sociodemographic characteristics (e.g. age, gender, and Puerto Rican geographic location), tumor stage, tumor site, number of nodules, presences of metastasis, tumor histology, mucinous characteristic, site of K-RAS, and type of K-RAS mutation. The significance level was set at p≤0.05. SPSS 14 software was used to perform all the analyses.
Results
A total of 501 samples were screened, between April 2009 and January 2011, for K-RAS mutation in colorectal cancer tumors (Tab. I). The median age of the patients was of 63.4±12.3 years, and 58.3% of them were males and 41.7% were females. The most common histology type of the colorectal cancer was moderately differentiated (75.4%), 25.9% had a mucinous tumor histotype, and 80.1% of the patients had advanced colorectal cancer (e.g. stages III-IV). The mutant K-RAS was present in 38.9% (n=195) of the patients, more commonly observed among subjects in the age group of 51-69 years (53.8%). Of these mutations 87.7% were located on codon 12, where 12 Asp (39.5%) and 12 Val (25.4%) mutations were most likely reported, and 12.3% were located on codon 13, where 13 Asp mutations were reported. The K-RAS mutation was observed with the following distributions: in 55.4% of males (p>0.05), in 53.8% of patients within the age group of 51-69 years (p>0.05), in 43.8% of proximal tumors (p=0.023), in 43.0% of tumors with distant metastasis (p=0.020), in 31.7% of tumors with mucinous histotype (p=0.083), and in 38.8% of ulcerated tumors (p=0.086). In patients with distant metastasis and mutant K-RAS we found a tendency of having lung metastasis (60%) rather than liver metastasis (40%).
Description of the Study Population by K-RAS Mutation Status 1
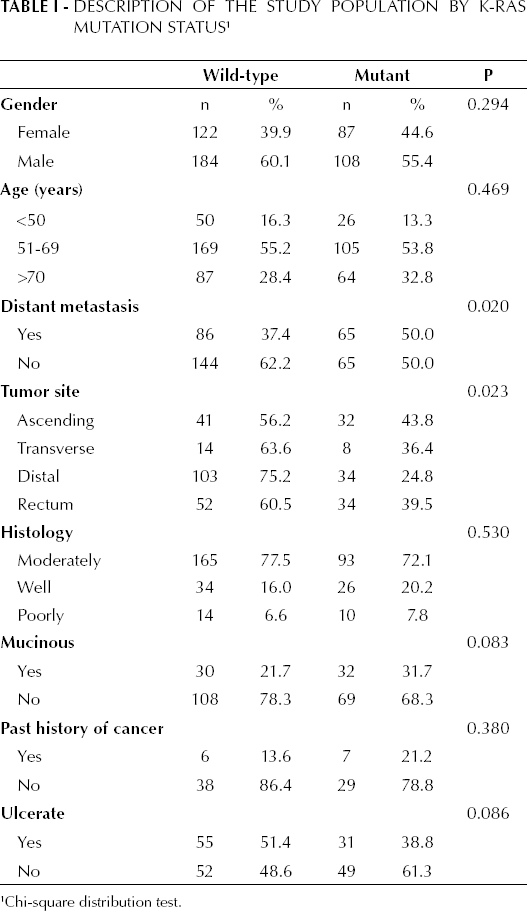
Chi-square distribution test.
Table II shows the K-RAS mutation status in cases of colon carcinoma (31.9%) in which the most common mutation was at 12 Asp (52.2%). In patients with colon carcinoma, mutant K-RAS was observed in 55.4% of the females (p=0.095), in 43.8% of proximal tumors (p=0.017), and in 37.9% of ulcerated tumors (p=0.033). Statistically significant differences were not observed among different tumor histologies and mucinous histotypes and types of K-RAS mutation. The K-RAS mutation status in patients with rectum carcinoma was 39.5%, where the most common mutation was at 12 Val (37.5%). Statistical significance was not found when considering different tumor histology types, ulcerated tumors, and mucinous histotype in patients with rectum carcinoma and mutant K-RAS (p>0.05) (see Tab. III).
Histology, Mucinous Histotype, and Ulcerate Tumors by K-RAS Mutation Status and Colon Carcinoma Only 1
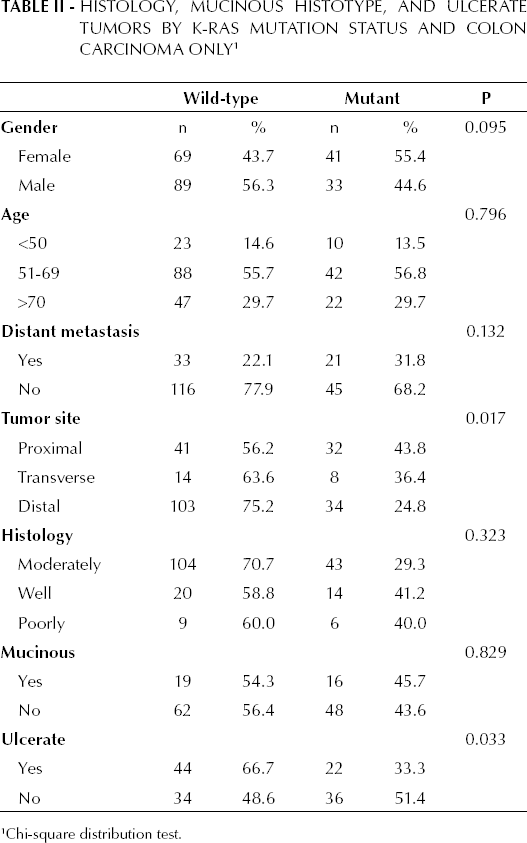
Chi-square distribution test.
Histology, Mucinous Histotype, and Ulcerate Tumors by K-RAS Mutation Status and Rectum Carcinoma Only 1
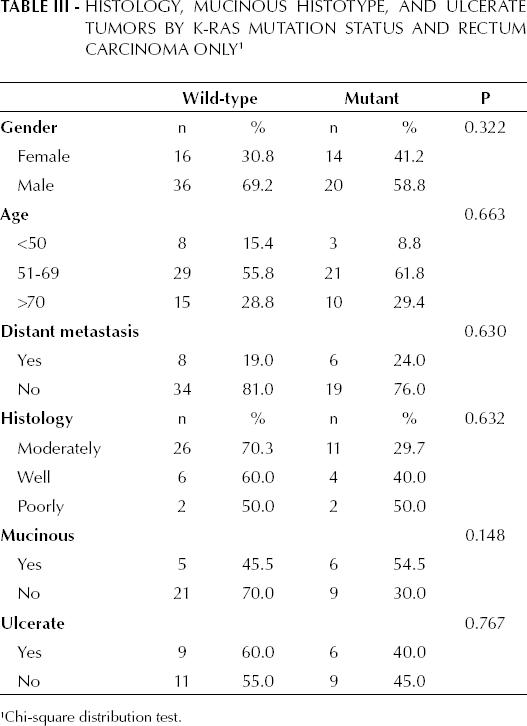
Chi-square distribution test.
Discussion
This is the first population-based study performed in Puerto Rico for the analysis of K-RAS mutations in patients with colorectal cancer. Overall, the mutation of K-RAS was detected in 39% of the population. This result is similar to other studies where the K-RAS mutation was reported in about 30%-40% of the population considered (12). The distribution of the K-RAS mutation in the Puerto Rican population was 88% in codon 12 and 12% in codon 13. The most common mutation at codon 12 was at 12 Asp (39.9%), which is similar to the frequency found in Austrian (31.2%), Slovenian (38.5%), and Brazilian (39.0%) populations (13–15). The second most common mutation found at codon 12 was at 12 Val (25.4%). Previous research has suggested that the 12 Val mutation increases the risk of recurrence or death in patients with colorectal cancer, as well as decreases the overall survival (14). No significant differences were observed between genders, but a tendency suggested a slightly higher K-RAS mutation frequency among females (55.4%; p=0.095). Mutant K-RAS was found in 39.5% of rectum carcinomas, in which a 12 Val mutation was observed (37.5%). Similarly to previous findings, a K-RAS mutation was observed in proximal tumors (p=0.023) and in tumors with distant metastasis (p=0.020) (15, 16). Conversely, one study did not find any statistically significant correlation between proximal tumor location, higher tumor grade, and absence of peritumoral lymphocytic inflammation and mutant K-RAS (17). As reported by previous research we found that the mucinous histotype is more commonly detected in codon 12 (91.7%), however it did not reach statistical significance in our study (18, 19). In accordance with what reported by Tie et al in 2011 (10), we found that metastasis to the lung was commonly reported in patients with mutant K-RAS; therefore, we believe that chest imaging should be performed to detect lung metastasis among patients with colorectal cancer (10, 20).
In conclusion, the prevalence of K-RAS mutation in the Puerto Rican population (39% in colorectal carcinoma patients) is similar to that reported by other studies. A higher prevalence of the 12 Val mutation in rectal carcinoma and a trend towards a higher mutation rate in females are the main differences between this cohort and the results reported in previously published studies. Also, the finding that different patterns of metastasis progression in colorectal carcinoma were determined by K-RAS mutation may help in generating better surveillance strategies and better patients' outcome.
This study only considers the presence of mutant or wild-type K-RAS in DNA extracted from paraffin-embedded colorectal tissues provided by GENOPTIX medical laboratories data from April 2009 to January 2011. This study analyzed a large representative sample of the Puerto Rican population; however it may not be possible to generalize its results to other populations. The data provided cannot be linked with colorectal cancer overall survival. Further investigation should be performed to evaluate the impact of K-RAS mutation in overall survival and response to treatment.
Footnotes
Acknowledgements
This study received statistical support from Data Management and Research Support Unit and Puerto Rico Clinical and Translation Research Consortium.
