Abstract
Background
Overexpression of MUC1 predicts poor survival in most cancers. Routine immunohistochemical detection of MUC1 is performed for differential diagnosis in malignant peritoneal mesothelioma (MPM). However, the prognostic significance of MUC1 in MPM has not been determined.
Method
We investigated MUC1 expression and other prognostic factors in relation to survival in 42 patients (20 males and 22 females) for whom archival samples were available, using immunohistochemistry. MUC1 was expressed in 38/42 (90%) patients. Its prognostic significance was statistically analyzed using the Kaplan-Meier method.
Results
High expression of MUC1 (immunohistochemical score of 5-8), was correlated with poor survival in several categories: all subtypes of tumors (p=0.001), male gender (p=0.017), female gender (p=0.001), epitheloid tumors (p=0.001), epitheloid tumors in males (p=0.005), epitheloid tumors in females (p=0.003), and age at diagnosis (AAD) <60 years (p=0.001). Amongst the other clinicopathological variables, univariate analysis also showed that male gender (p=0.007), sarcomatoid histology (p=0.001), peritoneal cancer index (PCI) ≥20 (p=0.013) and AAD ≥60 (p=0.001), correlated with poor survival. Multivariate analysis showed that only AAD ≥60 (p=0.049) was an independent prognostic factor, and that high MUC1 expression significantly correlated with the following categories: all subtypes of tumors (p=0.001), male gender (p=0.002), female gender (p=0.031), epitheloid tumors (p=0.031), and AAD <60y (p=0.012).
Conclusion
AAD and high MUC1 expression in the tumor are indicators of poor prognosis. MUC1 evaluation by immunohistochemistry may serve as a useful prognostic tool in MPM, but may need further confirmation in a larger patients' cohort.
MUC1, also known as episialin, epithelial membrane antigen, or CA15-3 antigen, is a highly O-glycosylated mucin-like transmembrane glycoprotein that is encoded by a gene in position 1q21 (1). MMC1 is a large protein (>200 kDa) with a very large extracellular domain made up of mainly 20 amino acidic tandem repeats, a transmembrane domain and a cytoplasmic tail (2, 3). The extracellular domain that protrudes from the cell surface is responsible for the protective function of MUC1 and is implicated in immunoregulation (4). Moreover, it is believed that the lengthy, rigid, extracellular tandem repeat domain with negative charge is responsible for interfering with both cell-cell and cell-matrix adhesion and hence promoting metastasis and invasion (5). On the other hand, the intracellular domain in MUC1 is implicated in transmembrane signal transduction that takes place through the interaction with the epidermal growth factor receptor by the mitogen activated protein kinase pathway (6). Finally, the intracellular domain also binds to β-catenin with consequent inhibition of E-cadherin-mediated cell-cell adhesion (7, 8).
De novo expression or overexpression of MUC1 has been demonstrated in several malignancies. Tumor cells often loose polarity and change from apical to circumferential staining. During invasion and metastasis, complete coverage of cell membrane with MUC1 is said to take place (9). Compared to non-neoplastic tissues, tumor-associated MUC1 molecules are underglycosylated, leading to demasking of the peptide antigens, thereby facilitating immunohistochemical detection (10). Nevertheless, overexpression of MUC1 in cancerous tumors has been associated with poor prognosis in endometrial adenocarcinomas (11), esophageal squamous cell carcinomas (12), cholangiocarcinomas (13), renal cell carcinomas (14, 15), lung cancer (16), and prostate cancer (17, 18). In malignant pleural mesothelioma there is also an overexpression of MUC1 (19), although the prognostic implication of this glycoprotein is unknown.
In both malignant peritoneal mesothelioma (MPM) and malignant pleural mesothelioma (PM), MUC1 is routinely used in differential diagnostic immunohistochemistry (20–22), along with other markers such as calretinin, HBME-1 and CK 5/6. However, the prognostic value of MUC1 in MPM still remains to be determined. Hence, the current study was designed to investigate the relationship between MUC1 expression and prognosis. We initially examined 2 chemoresistant MPM cell lines (PET and YOU) in order to determine the status of MUC1 in these cells. They both displayed higher expression of MUC1 in comparison to the breast cancer cell line MDAMB-231, which has been reported to have a medium level of MUC1 expression (23). Hence, we proceeded with the analysis of MUC1 expression in tumor samples of patients with MPM.
Materials and Methods
Patients and Tissue Samples
This study was conducted with the approval of the ethics committee of the St. Georges Hospital (SESIAH), Kogarah, NSW, in Australia. A total of 42 paraffin embedded tumor specimens of MPM (20 males [48%] and 22 females [52%]), who underwent cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC) between January 2000 and February 2012 were retrieved for further analysis. Chemotherapeutic agents used for HIPEC were cisplatin and mitomycin C, according to the recommended dose. All patients were treated within 3 weeks from the date of diagnosis. The completeness of cytoreduction score (cc-score) was optimal (0-1) for 40 patients (95%), whilst the remaining 2 patients (5%) had a non-optimal score (2-3). Node status was not available for these patients, since the recording system is only currently being set up. Median age of the patients was 57 years (range 23-69). The median follow-up time was 60 months (range 2-72 months). Clinical and pathological features of the patients are summarized in Table I.
Clinical and Histological Features of 42 mpm Patients Screened for MUC1 Expression
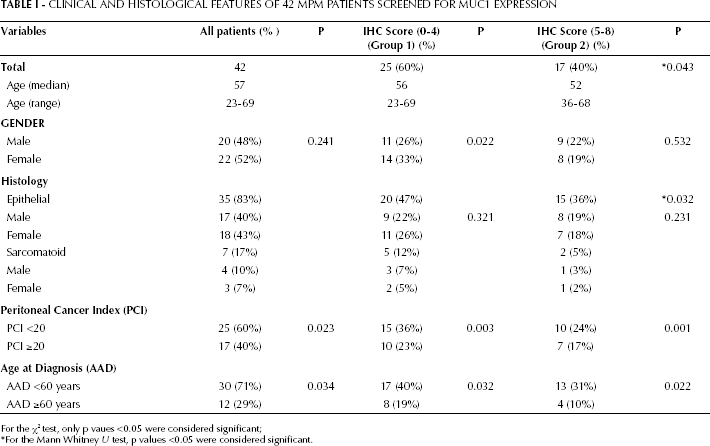
For the χ2 test, only p vaues <0.05 were considered significant;
For the Mann Whitney U test, p values <0.05 were considered significant.
Immunohistochemistry
Staining procedures
Tumor specimens of MPM retrieved from patients were preserved in formalin and embedded in paraffin. Sections of 3 μm were cut and mounted on glass slides. For the detection of MUC1, we used the mouse monoclonal antibody MA695 (NCL-MUC-1, Novacastra laboratories Ltd, Newcastle upon Tyne, UK). Immunohistochemical staining was carried out using automated immunostainers. Briefly, sections were deparaffinized, rehydrated in graded alcohols, and treated for 5 minutes with 1% H2O2. Antigen retrieval was carried out with epitope retrieval solution, DAKO-K5205 for 40 minutes at 98°C and subsequently incubated with the primary antibody (MA695, dilution 1:200) for 45 minutes. The binding of the primary antibody was assessed with DAKO ChemMate detection kit.
Immunohistochemical evaluation and controls
Three blinded and independent evaluations of the stained slides were carried out. Discrepancies were resolved by re-examination of the slides. Immunoreactivity was categorized as previously described (24–26). Brown stains were classified as positive. Positive controls were non-neoplastic peritoneal mesothelium and negative controls were tissues without primary antibody. Briefly, the slides were examined in their entirety under a light microscope and, initially, a proportion score, representing the estimated proportion of positive tumor cells, was assigned (0, none; 1, <15%; 2, between 15% and 30%; 3, between 30% and 45%; 4, between 45% and 60%; 5, >60%). Next, an average intensity score was assigned for each scoring group (0, none; 1, weak; 2, intermediate; 3, strong). The proportion and intensity scores were added to give a total score, which ranged from 0 to 8.
Statistical analysis
Statistical analysis was performed using PRISM (version 5.0). Association between MUC1 expression and clinicopathological characteristics was evaluated using the χ 2 test. Overall survival of patients expressing either high or low MUC1 in the different clinicopathological categories was compared according to the Kaplan-Meier method. Overall survival was calculated from the date of surgery to the date of last follow-up examination or death. Differences between the survival curves were tested using the log-rank test. Univariate and multivariate survival analyses were performed using the cox proportional-hazards regression model. For both the univariate and multivariate models, a p value of 0.05 (with a 95% confidence interval) was used as the cutoff. Only those factors that were significant in univariate analysis were entered into multivariate analysis.
Immunofluorescent labeling (confocal microscopy)
Cell lines (PET, YOU and MDAMB-231) were cultured to near confluency and harvested by trypsinisation. Cells were diluted in culture media (5,000 cells/5 mL) and cultured overnight on sterile cover slips. The cells were washed twice with PBS and fixed in 4% paraformaldehyde supplemented with 0.1% saponin for 30 minutes at room temperature. Cells were then washed twice with PBS and incubated at room temperature for 2 hours with mouse anti-human MUC1 antibody (Serotec, MCA1742) diluted to 1:200 in PBS containing 1% skim milk. Primary antibody was omitted in controls. Cells were then washed twice with PBS and incubated in the dark for 1 hour with goat anti-mouse IgG (Alexa 488, molecular probes, Oslo, Norway) diluted 1:500 in PBS.
Western blot analysis
Cell lines (PET, YOU and MDAMB-231) were grown to 75% confluency. Whole cell lysates were prepared following the manufacturer's protocol (Pierce Biotechnology, Rockford, Illinois, USA). A mouse monoclonal antibody for MUC1 was used (Serotech, MCA1742), diluted to 1:200, and incubated overnight at 4°C. The immunocomplexes were detected with a horseradish peroxidase-conjugated secondary antibody (Santa-Cruz Biotech-sc2031) at a 1:5,000 dilution for 1 hour at room temperature, followed by enhanced chemiluminescence (Perkin Elmer Inc, USA). Uniform loading of protein was confirmed with GAPDH.
Results
Expression of MUC1
Mesothelioma cell lines (PET and YOU)
Detection of MUC1 by immunofluorescence indicated that the PET cell line (MPM cells) had a high expression of MUC1, as indicated by the wide distribution and intensity of the staining, as compared to MDAMB-231 cells (breast cancer cells), which are known for their intermediate staining distribution and intensity. YOU cells (MPM cells), on the other hand, did not show abundant MUC1, but displayed a stronger intensity of fluorescence. When these cells were examined for MUC1 expression using Western blot, both PET and YOU cells showed a stronger band for MUC1, as compared to MDAMB-231. This indicates that PET cells have a higher amount and intensity of MUC1 staining whilst YOU had a lower amount but high intensity of MUC1 expression.
MUC1 expression in MPM patients' samples
Out of a total of 42 tumor samples, 38 (90%) expressed MUC1, although the intensity and proportion of stained cells varied from sample to sample (Fig. 1F-1I). The staining we observed was mainly confined to the cell membrane, although we also found cytoplasmic staining in a few patients' samples.
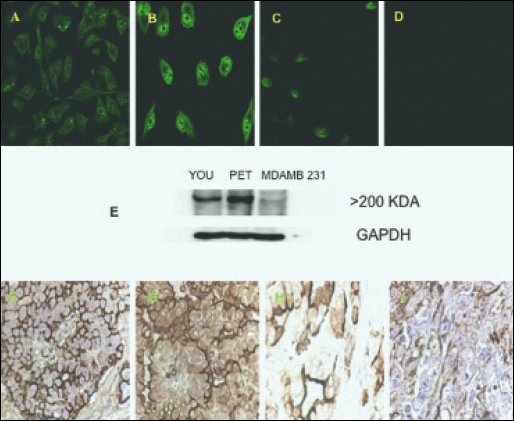
Extent and intensity of MUC1 expression in (A) MDAMB-231 (breast cancer cells), with an intensity that is classified as medium; (B) PET cells (MPM cells) showing a wide distribution and much stronger intensity compared to MDAMB-231 cells; (C) YOU cells (MPM cells) showing a lesser distribution of MUC1 despite the strong intensity; (D) blank containing no primary antibody. Immunofluorescence staining was carried out as outlined in the “Materials and methods” section, using a secondary anti-mouse fluorescent probe Alexafluor488. The images were analyzed using confocal microscopy. (E) Shows Western blot MUC1 bands (>200 kDa) for the 3 cell lines, MDAMB-231, PET and YOU. YOU and PET cell lines show a much stronger band compared to MDAMB-231 (F-I) Show MUC1 staining in 4 different patients; F and G show strong membranous staining, whilst G has weaker staining in the cytoplasm; H and I show less cells stained for MUC1. The slides have been immunostained as described in the “Materials and methods” section and analyzed using the image analyzer Zeiss, Germany (magnification 200 X 30 mm).
The immunohistochemical (IHC) score (describing the area of staining and its intensity) were assigned to 2 groups, one with lower IHC scores of 0-4 (group 1) and the other with higher IHC scores of 5-8 (group 2). Of the 42 patients, 25 (60%) belonged to group 1, and the remaining 17 (40%) were in group 2. The median age of group 1 was 56 years (range 23-69 years) and for group 2 it was 52 years (range 36-68 years). There were 11 males (26%) and 14 females (33%) in group 1, whilst in group 2 gender distribution was almost equal (9 males and 8 females) (Tab. I).
The histological distribution was mainly epithelial for 35 patients (83%), and to a lesser extent sarcomatoid for 7 patients (17%). Out of 35 patients with epitheloid tumors, 20 (47%) were in group 1, of which 9 (22%) were males and 11 (26%) females. The remaining 15 patients (36%) fell into group 2, which had an almost equal distribution of genders, with 8 males (19%) and 7 females (18%). Within the tumors with sarcomatoid histology, out of 7 patients (17%), 5 (12%) fell in group 1, with a male:female ratio of 3:2. However, group 2, which had only 2 patients (5%) bearing tumors with sarcomatoid histology, there was 1 male and 1 female patient.
MUC1 in sarcomatoid tumors was found to be highly expressed in all male patients' samples, whilst in all the females MUC1 expression was low, suggesting a preferential high MUC1 expression in males compared to females.
Within the category of epitheloid tumors, there were no pleomorphic tumors, however there were 5 of tubular and 4 of papillary type. In the category of tubular tumors, there were 3 males and 2 females. All the 3 males expressed high levels of MUC1 whilst both the females had low MUC1 levels. Among the 4 patients with micropapillary tumors, there were 2 males with high MUC1 expression, whereas 1 female had a high expression of MUC1 and the other had a low MUC1 expression. Hence, it appears that epitheloid tumors in males, regardless of their histological variance, seem to express higher levels of MUC1 than in females.
With regard to the peritoneal cancer index (PCI), out of 25 patients (60%) who had a PCI <20, 15 (36%) belonged to group 1, whilst the remaining 10 (24%) fell into group 2. In the PCI ≥20 category, out of 17 patients (40%), 10 (23%) belonged to group 1 and the remaining 7 (17%) to group 2. On a comparative basis, a higher number of patients (60%) had a PCI <20 and, among these, a higher number of patients (36%) had a low expression of MUC1 (group 1).
With regard to the category of age at diagnosis (AAD), out of 42 patients, 30 (71%) belonged to the category of AAD <60 years, of these 17 (40%) belonged to group 1 and the remaining 13 (31%) fell into group 2. Within the category of AAD ≥60 years, 8 (19%) were in group 1 while the remaining 4 (10%) were in group 2. More than twice the number of patients was below 60 years with a slightly higher number of patients in group 1, in this category. In the category of AAD ≥60 years, twice the number of patients were found in group 1. This indicates that, independently of the age group patients belonged to, the majority belonged to group 1 with low expression of MUC1.
Clinical outcome in different clinicopathological categories
Univariate analysis of prognostic factors
Univariate analysis (Tab. II) of different categories of the major clinicopathological variables showed a correlation between poor survival and male gender (p=0.007), tumors with sarcomatoid histology (p=0.001), PCI ≥20 (p=0.013) and AAD ≥60 years (p=0.001). Univariate analysis also showed that high expression of MUC1 is correlated with poor survival in the following categories: all types of tumors (p=0.001); males (p=0.017); females (p=0.001); epithelial tumors (p=0.001); epithelial tumors in males (p=0.005); epithelial tumors in females (p=0.003) and AAD <60 years (p=0.001) (Fig. 2A-2D).
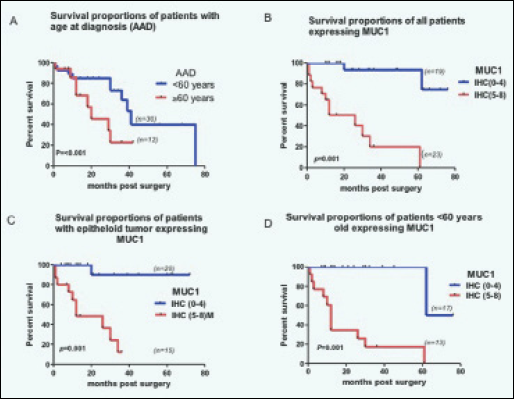
Kaplan-Meier survival curves showing percentage survival in (A) age at diagnosis ≥60 years vs <60 years; (B) all patients with high MUC1 score (describing the area of MUC1 staining and its intensity) (between 5 and 8) and low MUC1 score (between 0 and 4); (C) high MUC1 score vs low MUC1 score in patients with tumors of epitheloid histology; (D) high MUC1 score vs low MUC1 score in the group of patients with AAD <60 years.
Univariate Analysis and Prognosis of Different Clinicopathological Categories for 42 mpm Patients
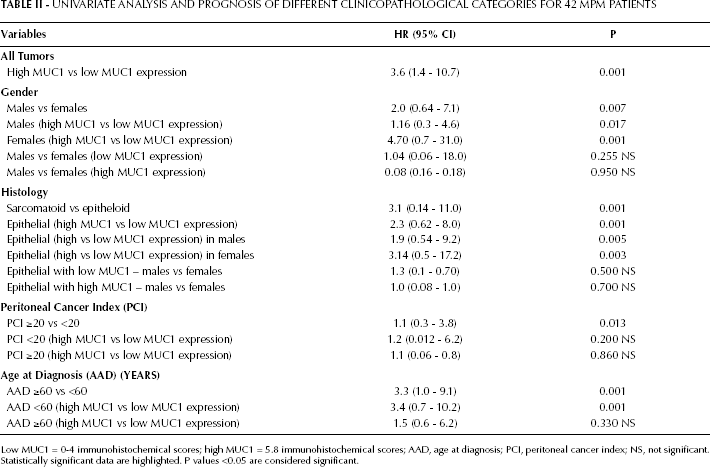
Low MUC1 = 0-4 immunohistochemical scores; high MUC1 = 5.8 immunohistochemical scores; AAD, age at diagnosis; PCI, peritoneal cancer index; NS, not significant.
Statistically significant data are highlighted. P values <0.05 are considered significant.
Multivariate analysis of prognostic factors
We then performed a multivariate analysis (Tab. III) of overall survival using a Cox proportional-hazards model to determine survival within the major clinicopathological categories. The results indicated that only AAD ≥60 years was an independent prognostic factor (p=0.049; HR=3.3; 95% confidence interval [CI], 1.0-9.1). With regard to the expression of MUC1 in the major clinicopathological categories, the following categories showed a statistically significant difference: all subtypes of tumors, p=0.001, HR=4.0 (95% CI, 1.9-11.4); male gender, p=0.002, HR=1.3 (95% CI, 0.6-5.2); female gender, p=0.031, HR=5.2 (95% CI, 0.4-8.2); epithelial tumors, p=0.031, HR=2.6 (95% CI, 0.9-8.9); and AAD <60 years, p=0.012, HR=3.4 (95% CI, 0.9-11.4).
Multivariate Analysis of Different Clinicopathological Variables for 42 mpm Patients
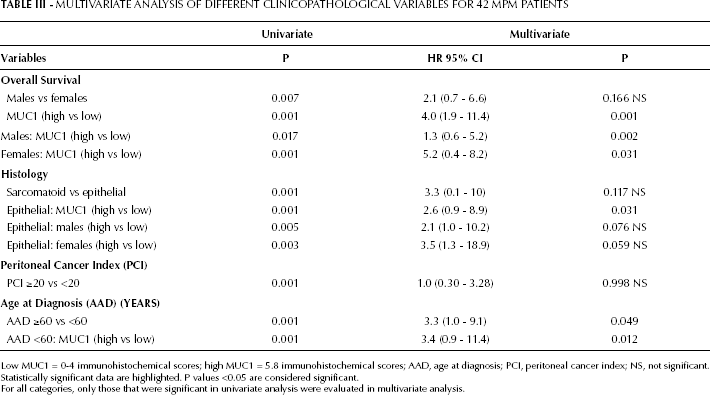
Low MUC1 = 0-4 immunohistochemical scores; high MUC1 = 5.8 immunohistochemical scores; AAD, age at diagnosis; PCI, peritoneal cancer index; NS, not significant.
Statistically significant data are highlighted. P values <0.05 are considered significant.
For all categories, only those that were significant in univariate analysis were evaluated in multivariate analysis.
Discussion
MUC1 is one of the several immunohistochemical markers that are routinely detected in differential diagnosis of MPM (20–22). However, the prognostic value of this marker has not been reported for MPM. Many types of cancers with overexpression of MUC1 are linked to poor prognosis (11–13, 15). Our initial investigation of MUC1 expression in chemoresistant MPM cells (PET and YOU) provided a preliminary indication of which cells had a higher secretion of MUC1. The impact of MUC1 on chemoresistivity, and therefore its effect on prognosis, has been demonstrated in a number of cancers, such as breast and ovary cancer (27–29).
In MPM a number of factors, such as gender, histology, PCI, AAD and estrogen receptors have been reported to affect prognosis (30). In the current study we examined the impact of MUC1 expression on prognosis in correlation with various clinicopathological categories along with a few other known prognostic factors, such as gender, histology, PCI and AAD. Univariate analysis revealed that in all tumor samples examined high expression of MUC1 predisposed patients to poor survival. A similar survival disadvantage was also found for patients with high MUC1 expression within each gender, as well as for patients with epithelial histology and AAD <60 years, thus indicating that MUC1 expression determines a worse prognosis in these categories of patients. Multivariate analysis revealed that MUC1 was an independent prognostic factor in all types of tumors, both in males and females, in epitheloid tumors and for AAD <60 years. Similarly, amongst the known prognostic factors, only AAD ≥60 stood up as an independent prognostic factor, whereas gender, histology, and PCI were only significantly correlated to survival in univariate analysis.
In the current study, both males and females expressing high levels of MUC1 were predisposed to poor survival, however the HR for females with high expression of MUC1 was much greater than for males (HR for females=4.7, HR for males=1.16). Likewise, in epitheloid tumors, similar HRs were found for females and males (HR for females=3.14, HR for males=1.9). Hormonal regulation of MUC1 has been demonstrated in a number of organ systems, especially the endometrium, where it plays a vital role in embryo implantation (31). Recently, progesterone-dependent control of MUC1 has also been shown in human endometrium (32). In human cancer cell lines, addition of estrogen and progesterone is associated with increased MUC1 expression (33). MUC1 upregulation by androgens has also been shown in breast cancer cell lines (34) and in prostate cancer cells (35). Hence, the observed greater risk of females in the high MUC1 expression group may be related to the steroid hormone levels found in this group and, therefore, needs further investigation. It is also known that MUC1 is linked to estrogen receptors (ERs) through its cytoplasmic tail (36), and hence the activation of ERs by estradiol may be responsible for cellular proliferation. MUC1 is also linked with a number of other growth factors, such as EGFR, ERB2, PDGFR, and FGFR (37), and these receptors may provide tumor cells with further stimulus to proliferate. Hence, female patients with high expression of MUC1 may be at a higher risk of disease progression and death than males.
Although gender disparity is observed in both MPM and PM, currently no plausible explanation seems to exist for this difference. In the present study, although female patients with high MUC1 expression are at greater risk of death than males (as shown by their HR), a higher number of females are in the low-MUC1 expression group (33% of females vs 26% of males). Furthermore, in the category of tumors with epithelial histology, which predispose patients to better prognosis as compared to sarcomatoid histology, a similar scenario exists, with a higher percentage of females (26% of females vs 22% of males). Although the difference between females and males in the low-MUC1 group is not large, it may contribute to gender disparity. We did not carry out a Kaplan-Meier analysis for the sarcomatoid category due to the small sample size.
Although it has been suggested that a high PCI is linked to poor survival in MPM (38), our current study did not show such a correlation. PCI is an indication of the extent of cancer spread in the 13 quadrants of the abdomen, however, it may not indicate the aggressive nature of the tumors. MPM is a cancer that is often diagnosed late in the course of the disease and, hence, a high PCI may indicate how late in the disease process the cancer has been diagnosed rather than the aggressive nature of the cancer. Therefore, it may not have any bearing on prognosis and this seems to be the case with our current analysis. This is also in agreement with other studies that examined the impact of PCI on survival in MPM (39, 40). The majority of the patients (90%) had a cc-score of 0-1 after cytoreductive surgery and hence any residual disease spread over time will be dependent on the expression of oncoproteins, such as MUC1, that may dictate the rate of spread. Hence, survival after treatment may not be dependent on PCI at all.
Age is a prognostic factor in a number of cancers such as prostate (41, 42) and colon cancer (43). Our earlier studies with estrogen receptors (in press) suggest that in MPM the age of diagnosis is an important determinant of patient survival. Within the 2 AAD groups that we examined, MUC1 expression had a significant effect on prognosis for patients <60 years old. For patients with an AAD >60 years, the levels of MUC1 expression did not show any significant difference in multivariate analysis. Upon closer examination, we observed that this group is exclusively made up of males, whereas all the females ≥60 years old had lower MUC1 expression. Generally, males that are <60 years old have higher levels of androgens and lower levels of estrogens (44) and hence the differential influence of these hormones on MUC1 expression may be responsible for this high HR. All females ≥60 years old displayed a lower expression of MUC1, and this may suggest that the low steroid levels (45) found in this group may be modulating MUC1 expression. However, this needs further investigation. Without considering MUC1, in multivariate analysis an AAD >60 years is an independent prognostic factor for poor survival.
Although and AAD ≥60 years is an independent prognostic factor for poor survival, our analysis with MUC1 seems to indicate that an AAD <60 years is significant for MUC1-expressing MPM patients. Generally, patients who are ≥60 years old have co-morbidities associated with MPM and, hence, they may be more at risk of an early death than those <60 years old at diagnosis. The analysis of MUC1 expression indicates that a high MUC1 expression in those <60 years old is associated with poor prognosis. As outlined earlier, this may be due to hormonal factors that can affect MUC1 expression.
In malignant mesothelioma, the histology of the tumor plays a crucial role in patient survival after surgical resection and chemotherapy. Generally, tumors with epitheloid histology confer better survival to MPM patients (46, 47). Our results indicated that survival for patients with epitheloid tumors is dependent on MUC1 expression, and this finding extended to both genders. This may indicate that rather than the morphology of the mesothelial cells, it is the level of oncoproteins that dictates survival in MPM cells. Although there are no studies showing that the epitheloid morphology is related to lower MUC1 expression, the current study shows a higher percentage of patients (47%) with lower expression of MUC1 as compared to a lower percentage (36%) with high expression of MUC1.
Sarcomatoid tumors generally have poor prognosis (48, 49), and hence a higher MUC1 expression may be expected. However, in the current investigation MUC1 expression seems to be high only in sarcomatoid tumors in male patients, whilst it is low in females with the same type of tumors, indicating that MUC1 expression may be hormone-dependent, as reviewed earlier. Hence, males with tumors with sarcomatoid histology may be at greater risk of death than females. Similarly, when examining the variants in epitheloid tumors, both tubular and papillary types seem to have higher expression of MUC1 in males than females, which is a further indication that androgens and estrogens may be at play in these tumors. Hence, regardless of tumor histology or sub-variant types in epitheloid tumors, MUC1 seems to be highly expressed in males, which may partly explain gender survival disparity. Since there were only small numbers of sarcomatoid tumors as well as variants in epitheloid tumors, we did not carry out separate survival analysis.
The molecular organization of MUC1 is complex, with a number of functions carried out also in normal cells. MUC1 acts as a lubricant and a barrier, protecting cells from dehydration, proteolysis and infection. In cancerous cells, though, MUC1 acquires additional functions which provide the cells with chemoresistivity (increasing barrier function) (50), and loss of cell-cell and cell-matrix adhesion (interfering with integrin mediated adhesion) that favor invasion of tumor cells into the underlying stroma, lymph and blood vessels (51–53). Furthermore, overexpression of MUC1 also enables the cell to evade apoptotic processes through various mechanisms (54–55). Hence, the overexpression of MUC1 translates into the poor prognosis observed in the current study. Thus, data on patient's MUC1 expression might have an impact on decisions regarding therapeutic methods, resection safety margin, and follow-up periods. Multivariate analysis has confirmed that a high MUC1 expression is an independent prognostic factor in these malignant tumors. In addition, other investigators showed that in a number of cancers, such as colon, prostate, and renal cell carcinoma, high MUC1 expression leads to poor prognosis. Taken together, these results indicate that MUC1 overexpression can cause cancer cells to be more aggressive and, thereby, lead to poor prognosis.
Hence, in conclusion, we report for the first time that expression of MUC1 is a novel independent prognostic factor in MPM. MUC1 may be a useful marker for choosing the therapy and predicting patient outcome in MPM. Those who show high expression of MUC1 should be monitored more frequently and special therapeutic regimes should be formulated to meet the needs of this class of high-risk patients. In addition, the finding that MUC1 is an independent prognostic factor may also lead to the development of a new therapy that targets MUC1. Currently, several therapies that target MUC1 are at various stages of development for a number of cancers (56–58) and these may eventually be used for treating MPM.
