Abstract
Background:
Malignant peritoneal mesothelioma (MPeM) is a rare and progressive cancer originating from the mesothelial cells of the peritoneum. In patients with early-stage disease who are suitable for surgery, the treatment of choice is CRS + HIPEC, whereas in advanced-stage patients, systemic treatments are applied. Pemetrexed plus platinum regimens are at the forefront of first-line systemic treatments. Gemcitabine plus platinum regimens are rarely used as first-line treatment for MPeM. The aim of our study is to compare the efficacy of first-line pemetrexed plus platinum with gemcitabine plus platinum regimens in patients with MPeM.
Methods:
In this study, a retrospective analysis was conducted on 48 patients with MPeM who were followed up in our clinic between 2001 and 2025. In our study, 28 patients received pemetrexed plus platinum as a first-line regimen, while 20 patients received gemcitabine plus platinum. The median overall survival (OS), median progression-free survival (PFS), and response rates for both regimens were analyzed. In addition, prognostic factors influencing overall survival were investigated in the entire patient population.
Results:
The median PFS and OS were 11.1 months and 17.0 months for pemetrexed and 8.01 months and 14.4 months for gemcitabine. Although pemetrexed showed numerically higher PFS and OS, the difference was not statistically significant. The objective response rate (ORR) and disease control rate (DCR) were 32.1% and 57.1% for pemetrexed, compared with 25% and 40% for gemcitabine, showing pemetrexed’s superiority in response rates. In the entire patient population, CRS + HIPEC was the main prognostic factor for survival.
Conclusion:
We have demonstrated that the pemetrexed + platinum regimen has better response rates compared with the gemcitabine + platinum regimen in MPeM patients. However, gemcitabine-based regimens can be used as an alternative to pemetrexed in patients with MPeM.
Keywords
Introduction
Malignant peritoneal mesothelioma (MPeM) is a rare and progressive cancer originating from the mesothelial cells of the peritoneum. This condition is characterized by its insidious onset and aggressive progression, often leading to significant challenges in diagnosis and treatment. Due to its rarity, MPeM is frequently misdiagnosed or diagnosed at advanced stages, which complicates treatment options and prognosis. 1
The MPeM exhibits a distinctive histopathological profile alongside notable molecular alterations—most frequently including BAP1, NF2, SETD2, and TP53 mutations, as well as variable expression of biomarkers such as WT1, mesothelin, and PD-L1—which not only aid in diagnosis but also carry significant prognostic and therapeutic implications. 2
The MPeM includes major 3 histological subtypes. The epithelioid subtype accounts for 75% to 90% of cases, has the best prognosis, and closely resembles normal mesothelial cell histology. The sarcomatoid subtype contains densely arranged spindle-shaped cells and may show malignant-appearing osteoid, chondroid, or muscle-like structures. The biphasic subtype contains both epithelioid and sarcomatoid elements, with histological features of both subtypes present in at least 10% of the tumor. In addition, rare histological variants include pleomorphic, desiduoid, small cell, and clear cell subtypes.3,4 Germline genetic alterations that increase the risk of disease due to environmental exposure have been identified as playing a role in the etiology of mesothelioma. The MPeM cases are less commonly associated with asbestos exposure compared with pleural mesothelioma; only 8% of MPeM patients report a history of prior asbestos exposure, while this rate exceeds 80% in pleural mesothelioma cases. 5 Other mineral fibers are also thought to play a role in the disease; in particular, erionite, a silicate fiber, has been shown to be a potent inducer of MPeM. Other potential risk factors include therapeutic radiation, Thorotrast dye used in past angiographic studies, papovavirus, simian virus, and chronic inflammation. 6
Peritoneal mesothelioma is rarer than pleural mesothelioma and typically presents with nonspecific symptoms in patients. The most common findings are pain, bowel obstruction, and cancer cachexia, which are related to involvement of the peritoneal cavity. 7 Although the disease is largely considered resistant to chemotherapy, the median overall survival (mOS) without treatment ranges from 6 months to 1 year. The standard treatment for MPeM is considered to be cytoreductive surgery (CRS), aimed at aggressively removing the tumors, followed by hyperthermic intraperitoneal chemotherapy (HIPEC) to eliminate microscopic residual cancer cells. 8 When surgical intervention is possible, the degree of cytoreduction achieved and the application of HIPEC are independently associated with better survival. Unfortunately, up to 60% of patients diagnosed with MPeM are unable to undergo surgical treatment. In addition, other factors independently associated with better survival include the presence of the epithelioid histological subtype and the absence of lymph node metastasis. 9
The Peritoneal Cancer Index (PCI) is a reproducible, systematic method used to determine the distribution and burden of the disease. Although PCI can be estimated using computed tomography (CT) imaging before surgery, the most accurate approach is intraoperative assessment. 10 The Tumor, Node, Metastasis (TNM) staging system is less commonly used for MPeM because the disease generally spreads extensively throughout the abdominal cavity, with nodal or extraperitoneal metastasis being rare. However, Yan et al proposed a TNM staging method that correlates the T stage with PCI quartiles. The PCI was classified as 1 to 10, 11 to 20, 21 to 30, and >30, which corresponded to T1 to T4 stages, respectively. Stage I (T1N0M0), stage II (T2N0M0 or T3N0M0), and stage III (T4, N1, and/or M1 disease) were defined accordingly. 11
Treatment options for patients with diffuse MPeM include surgical and/or systemic therapies. Medically operable patients with a good performance status, including those with characteristics such as epithelioid histology and focal disease, are candidates for multimodal therapy. For patients with diffuse MPeM who are not suitable for surgery or who refuse surgery, systemic therapy is recommended. Since MPeM is similar to pleural mesothelioma, systemic treatment recommendations for MPeM have been determined by generalizing data from clinical studies conducted on pleural mesothelioma. 12
In the literature, there are studies on pemetrexed plus platinum as a first-line treatment and gemcitabine as a second-line treatment for patients with MPeM. However, there are very few studies that compare the use of these therapies in the first-line setting. This lack of direct comparative data highlights the need for well-designed clinical trials to establish the most effective treatment protocols for MPeM.13,14 In this study, we aimed to compare the effects of first-line pemetrexed plus platinum and gemcitabine plus platinum on survival and response rates in this rare disease group.
Material and Methods
Patients and study design
In this study, a retrospective analysis was conducted on 48 patients with MPeM who were followed up in Ankara City Hospital, Medical Oncology Department between August 2001 and March 2025. All patients were ⩾18 years old at the time of diagnosis, and MPeM was pathologically confirmed. Clinicopathological characteristics, as well as hematological and biochemical data, were retrieved from the hospital records and reviewed retrospectively. Patients with missing data were excluded.
In our study, patients with non-operable, peritoneal-limited disease were classified as early stage, while patients with inoperable disease or visceral metastasis were classified as advanced stage. For patients who underwent surgery, they were grouped according to the PCI scores in the operative notes, with PCI 1-20 classified as 1 group and PCI 21-39 as another. Tumor response was assessed using computed tomography in accordance with the Response Evaluation Criteria in Solid Tumors (RECIST) criteria. 15
Patients who received cisplatin plus pemetrexed (with or without bevacizumab) or carboplatin plus pemetrexed (with or without bevacizumab) as first-line chemotherapy were classified as the Pemetrexed + Platinum group, while those who received gemcitabine plus cisplatin or gemcitabine plus carboplatin were classified as the Gemcitabine + Platinum group. Carboplatin (area under the curve 5) and pemetrexed (500 mg/m²) or cisplatin (75 mg/m²) and pemetrexed (500 mg/m²) were administered intravenously on day 1 of a 21-day cycle for 6 cycles. Maintenance chemotherapy with pemetrexed after 6 cycles of cisplatin or carboplatin plus pemetrexed was included as first-line treatment. Gemcitabine (1000-1250 mg/m2) was administered on days 1 and 8 in combination with platinum-based agents.
Overall survival (OS) was defined as the time from disease onset to death from any cause or the last follow-up. Progression-free survival (PFS) was defined as the time from treatment initiation to disease progression or death. Objective response rate (ORR) was defined as complete radiological response and partial radiological response. Disease control rate (DCR) was defined as patients who did not progress in post-treatment evaluations. Performance status was assessed using the Eastern Cooperative Oncology Group (ECOG) scale. 16
The laboratory values of the patients were based on those at the time of diagnosis. Leukocytosis was defined as a white blood cell (WBC) count >10 000/mm³, neutropenia as an absolute neutrophil count < 1500/mm³, and hypoalbuminemia as a serum albumin level < 3.5 g/dL. Elevated tumor markers were defined as carcinoembryonic antigen (CEA) > 5 ng/mL, carbohydrate antigen (CA) 19-9 > 37 U/mL, and CA 125 > 35 U/mL.
The reporting of this study adheres to the STROBE (The Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 17 This study was conducted in accordance with the principles of the Declaration of Helsinki (1975), as revised in 2024.
Statistical analyses
The IBM SPSS version 25 was used for all statistical analyses. Histograms and the Shapiro–Wilk test were used to determine normal distribution. Normally distributed continuous variables were expressed as mean ± standard deviation, while non-normally distributed variables were expressed as median (min–max). Two group comparisons of continuous variables were performed using the Mann-Whitney U test. For categorical comparisons, the chi-square or Fisher exact test was used. The Kaplan-Meier survival curves and Cox regression analysis were used for survival and prognostic factors. P-value <.05 was considered significant.
Results
Patient characteristics
The median age of the 48 patients was 57.5 years. Among them, 22 (45.8%) were male, and 26 (54.2%) were female. A history of smoking was present in 43.7% of the patients, while 52.1% had additional comorbidities. The ECOG 0-1 performance status was observed in 47.9% of the patients. Regarding histological subtypes, 34 patients (70.8%) had the epithelioid subtype, whereas 14 (29.2%) had the non-epithelioid subtype. At the time of diagnosis, 35 patients (72.9%) were in an advanced stage. Regional lymph node involvement was detected in 60.4% of the patients. In addition, ascites was radiologically positive in 66.7% of the patients. The demographic and clinical characteristics of the patients are summarized in Table 1.
The demographic and clinical characteristics of the patients.

n: number; ECOG: Eastern Cooperative Oncology Group; min: minimum; max: maximum; CEA: Carcinoembryonic Antigen; CA19-9: Carbohydrate antigen 19-9; CA125: Carbohydrate antigen 125; LDH: lactate dehydrogenase; NOS: subtype not otherwise specified.
Treatment modalities
Among the patients, 11 (22.9%) underwent CRS-HIPEC, while 4 (8.3%) underwent palliative or incomplete surgery. The remaining 33 patients (68.8%) initiated treatment with first-line chemotherapy. The most commonly used HIPEC regimen was cisplatin, administered in 72.7% of cases. Among the 15 patients who underwent surgery, 53.3% had a PCI score of 1 to 20, while 46.7% had a PCI score of 21 to 39. Adjuvant chemotherapy was administered to 8 surgical patients, with cisplatin plus pemetrexed being the most frequently used regimen (50%). Recurrence was observed in all surgically treated patients. As first-line chemotherapy, 28 patients (58.3%) received pemetrexed plus platinum, while 20 (41.7%) received gemcitabine plus platinum. Second-line chemotherapy was administered to 21 patients, with gemcitabine being the most frequently used regimen (28.6%), followed by carboplatin plus pemetrexed (23.8%) and carboplatin plus paclitaxel (23.8%). Four patients received third-line chemotherapy, with vinorelbine being the most commonly used agent. The treatment modalities are summarized in Table 2.
Treatment modalities of patients.

n: number; CRS: Cytoreductive surgery; HIPEC: hyperthermic intraperitoneal chemotherapy; PCI: peritoneal cancer index.
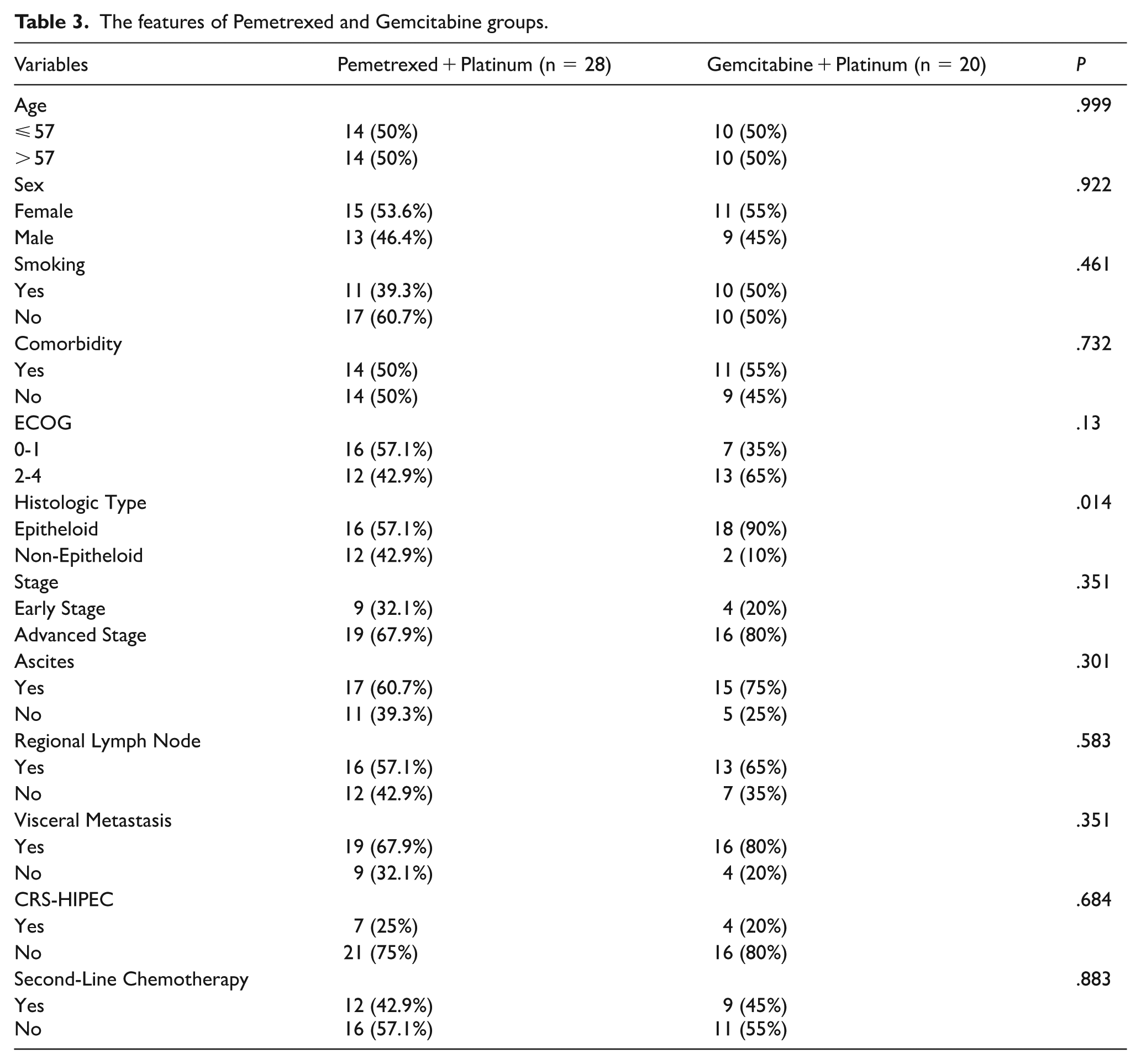
Patients in the Pemetrexed + Platinum group were compared with those in the Gemcitabine + Platinum group based on age, gender, smoking status, comorbidities, ECOG performance status, histological type, presence of ascites, regional lymph node involvement, visceral metastasis, CRS-HIPEC, adjuvant chemotherapy, and second-line chemotherapy. A significant difference between the groups was observed only in terms of histological type. The characteristics of the Pemetrexed and Gemcitabine groups are summarized in Table 3.
The features of Pemetrexed and Gemcitabine groups.
When evaluating patient responses to Pemetrexed + Platinum and Gemcitabine + Platinum treatments, a complete radiological response was observed in 3.6% of patients in the Pemetrexed arm, whereas no complete response was observed in the Gemcitabine arm. The overall response rate (ORR) was 32.1% in the Pemetrexed arm and 25.0% in the Gemcitabine arm. The DCR was 57.1% in the Pemetrexed arm and 40% in the Gemcitabine arm. Disease progression was observed in 42.9% of patients in the Pemetrexed group and 60% in the Gemcitabine group. The radiological treatment response of the Pemetrexed and Gemcitabine groups is presented in Table 4.
Radiological treatment response of Pemetrexed and Gemcitabin groups.

ORR and DCR were calculated based on the response categories presented in this table.
In our study, the median OS for all patients was 16.9 months (95% CI = 14.7-19.1). The median OS for patients in the Pemetrexed + Platinum group was 17.0 months (95% CI = 13.5-20.6), while the median OS for patients in the Gemcitabine + Platinum group was 14.4 months (95% CI = 3.9-24.9). Although there was a numerical difference between the groups, no statistically significant difference was observed (P = .257). The OS analysis of patients based on treatment regimens is presented in Figure 1.
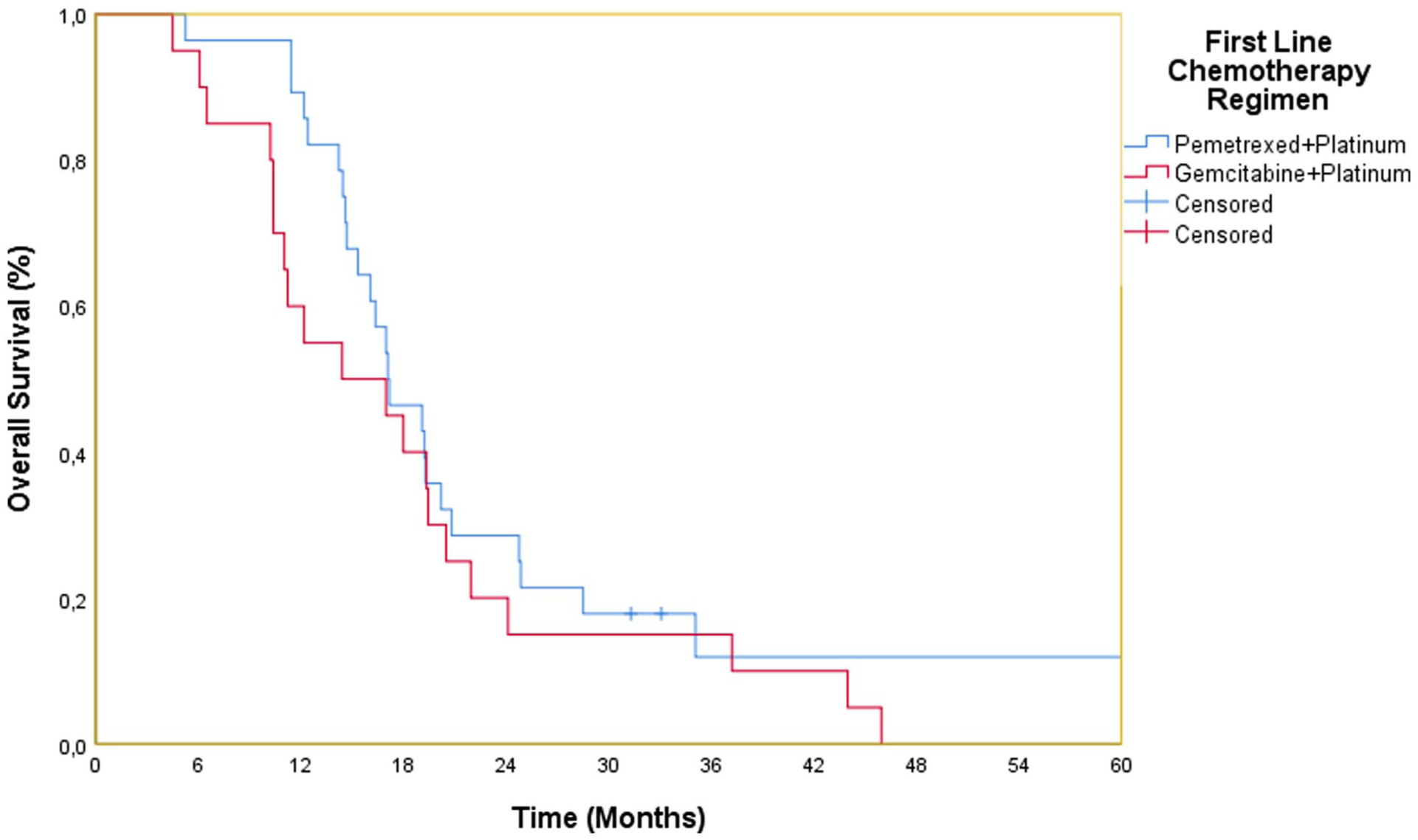
Kaplan-Meier overall survival analysis of patients based on treatment regimens.
The median progression-free survival (mPFS) for all patients in the study was 8.37 months. The median PFS for patients in the Pemetrexed + Platinum group was 11.1 months (8.95-13.2), while the median PFS for patients in the Gemcitabine + Platinum group was 8.01 months (7.6-8.3). Although there was a numerical difference between the groups, no statistically significant difference was observed (p: 0.501). The PFS analyses of the treatment groups are presented in Figure 2.

Progression-free survival rates of the Pemetrexed and Gemcitabine groups.
When patients were analyzed for OS, factors such as age, gender, comorbidities, smoking status, histological tumor type, regional lymph nodes, lymphocytosis, neutropenia, anemia, elevated lactate dehydrogenase (LDH), hypoalbuminemia, elevated CEA, elevated CA19-9, elevated CA125, first-line chemotherapy, and second-line chemotherapy did not have a significant impact on OS. In univariate analysis, ECOG performance status (P = .031), disease stage (P = .043), ascites (P = .017), hypoalbuminemia (P = .023), and CRS-HIPEC (P = .007) were found to significantly affect OS. Multivariable analysis revealed that CRS-HIPEC (P = .009, hazard ratio [HR]: 0.36; 95% CI = 0.174-0.783) was independent prognostic factors influencing OS. The factors influencing survival are summarized in Table 5.
Univariate and multivariate analyses of factors affecting overall survival.

Discussion
In this study, we aimed to compare pemetrexed-platinum and gemcitabine-platinum regimens as first-line treatments for MPeM, a rare and aggressive disease. Pemetrexed-platinum is considered one of the standard first-line regimens, with studies in malignant pleural mesothelioma reporting a median PFS of approximately 5 to 7 months and OS of 12 to 16 months. 18 Retrospective series in peritoneal mesothelioma have similarly demonstrated OS in the range of 13 to 16 months with pemetrexed-based combinations. 9 By contrast, evidence regarding gemcitabine–platinum remains limited. In pleural mesothelioma, gemcitabine-cisplatin has shown median OS ranging from 9 to 13 months,19,20 but to our knowledge, no prospective first-line studies have evaluated this regimen in peritoneal mesothelioma. In our cohort, patients treated with pemetrexed-platinum achieved a median PFS of 11.1 months and OS of 17.0 months, while those treated with gemcitabine-platinum had a median PFS of 8.01 months and OS of 14.4 months. These outcomes are broadly consistent with previous reports of pemetrexed-platinum and suggest that gemcitabine-platinum may also represent a feasible alternative in selected patients. 21
In our study, the primary reasons for receiving gemcitabine instead of pemetrexed in first-line treatment included prior administration of pemetrexed-based therapy in the adjuvant setting, contraindications due to comorbidities such as chronic kidney disease, poor ECOG performance status, unavailability of pemetrexed, and physician preference.
When examining the results of our study, the median PFS and OS were 11.1 months and 17.0 months, respectively, in patients receiving pemetrexed, whereas they were 8.01 months and 14.4 months in those receiving gemcitabine. Although the PFS and OS durations were numerically higher in the pemetrexed group, the difference was not statistically significant. One possible reason for the lack of statistical significance could be the limited sample size.
Regarding response rates, the ORR and DCR were 32.1% and 57.1%, respectively, in the pemetrexed group, whereas they were 25% and 40% in the gemcitabine group. The superiority of pemetrexed in response rates was evident. Moreover, the more aggressive non-epithelioid histological subtype accounted for 42.9% of patients in the pemetrexed group, while only 10% of patients in the gemcitabine group had this histological subtype. Although no statistically significant difference was observed, a higher proportion of patients receiving pemetrexed (67.9%) were diagnosed at an advanced stage compared with 80% in the gemcitabine group.
The primary prognostic factor in MPeM patients is the early-stage detection of the disease and the ability to undergo CRS + HIPEC. 22 In our study, CRS-HIPEC was the main prognostic factor determining survival. Patients who did not undergo CRS-HIPEC had a median survival approximately 9 months shorter than those who underwent the procedure. The initial CRS-HIPEC rates were similar between patients receiving first-line pemetrexed and gemcitabine (25% vs 20%).
Studies on the systemic treatment of pleural mesothelioma, which accounts for more than 70% of all mesotheliomas, have also paved the way for the systemic treatment of MPeM and contributed to the establishment of treatment standards. In the study conducted by Janne et al, pemetrexed monotherapy was compared with pemetrexed plus cisplatin in 25 chemotherapy-naïve MPeM patients. The median OS was 8.7 months in the pemetrexed monotherapy group, whereas it was 13.1 months in the pemetrexed plus cisplatin group. The ORR was 28% when pemetrexed plus cisplatin was used as first-line therapy and 25% when used as second-line therapy. 13
One of the most comprehensive studies on this topic, the Expanded Access Program (EAP), evaluated pemetrexed monotherapy, pemetrexed plus cisplatin, and pemetrexed plus carboplatin in patients with unresectable MPeM. The response rates were 12.5% for pemetrexed monotherapy, 20% for pemetrexed plus cisplatin, and 24.1% for pemetrexed plus carboplatin. The 1-year survival rate was 57.4% in the pemetrexed plus cisplatin group and 41.5% in the pemetrexed monotherapy group, while data for the pemetrexed plus carboplatin group were not available. 23
In a previous phase III study on malignant pleural mesothelioma evaluating cisplatin with or without pemetrexed, the response rate was 41.3% in the pemetrexed plus cisplatin group compared with 26.7% in the cisplatin-only group. The median OS was 12.1 months for the pemetrexed plus cisplatin group versus 9.3 months for the cisplatin monotherapy group. 18
Several small studies conducted in Japan evaluated pemetrexed-based regimens in patients with MPeM. One study investigated first-line therapy with pemetrexed plus cisplatin in 24 patients with MPeM, reporting 2 complete responses and 9 partial responses. The median OS was 15.8 months. 24 Another study evaluated first-line therapy with pemetrexed plus cisplatin in 29 patients with MPeM, reporting a median OS of 15.4 months. 25 The results of these studies were comparable to the median OS observed in our study among patients receiving pemetrexed-based regimens.
In the study conducted by Kitadai et al, both first-line and second-line regimens were analyzed in patients with MPeM. A total of 54 patients received platinum plus pemetrexed as first-line chemotherapy, while 26 patients received it as second-line therapy (including gemcitabine in 12 patients, taxane in 6, nivolumab in 3, and other agents in 5). Among all patients, the median OS and PFS after first-line chemotherapy were 16.6 months and 7.3 months, respectively. The median OS after second-line chemotherapy was 9.9 months. Patients who received ⩾6 cycles of platinum plus pemetrexed as first-line chemotherapy had significantly longer OS after second-line treatment than those who did not (P = .02). 14
There are very few studies evaluating the efficacy of gemcitabine plus platinum as a first-line treatment for MPeM. Most studies have been conducted in patients with pleural mesothelioma, with only a small subset of MPeM patients included in these studies. Nowak et al investigated the first-line gemcitabine plus cisplatin regimen in 53 patients with pleural mesothelioma. The study reported a partial response rate of 33%, stable disease in 60% of patients, and progressive disease in 8%. The mPFS was 6.4 months, while the median OS was 17.3 months. 19 In another study investigating the efficacy of gemcitabine plus cisplatin in pleural mesothelioma, 25 patients were treated, and the ORR was 16%. The mPFS was 6 months, and the mOS was 9.6 months. 26 In a study by Favaretto et al, gemcitabine plus carboplatin was used in patients with pleural mesothelioma, resulting in a 26% partial response rate, with an mPFS of 40 weeks and an mOS of 66 weeks. The toxicity profile was found to be at an acceptable level. 27 The results of these studies were similar to the outcomes observed in the group of patients receiving gemcitabine in our study.
Recent immunotherapeutic strategies have begun to reshape the treatment landscape for malignant mesothelioma. The phase II NIBIT-MESO-1 trial evaluated tremelimumab plus durvalumab in unresectable pleural or peritoneal mesothelioma, reporting a median immune-related PFS of 5.7 months and OS of 16.5 months, with durable disease control in a subset of patients. 28 In addition, pembrolizumab has shown promising results, with retrospective cohorts reporting partial responses and durable stable disease, reaching a median OS of around 20 months. 29 Overall, most immunotherapy studies have been conducted in pleural mesothelioma, and peritoneal mesothelioma has generally been included only in small patient subgroups.
Although our study focused on peritoneal mesothelioma, the heterogeneity regarding surgical vs non-surgical patients should be acknowledged; in pleural mesothelioma, the phase III MARS-2 trial showed that adding surgery to chemotherapy did not improve survival, highlighting the ongoing debate on the role of surgery in mesothelioma management. 30
One of the limitations of our study is the small number of patients. However, considering the rarity of this tumor, a review of the literature shows that other studies also involved a limited number of peritoneal mesothelioma patients. A formal sample size calculation was not performed for this study, and therefore, the limited number of patients included may have affected the statistical power of the results. Another limitation is that patients who received immunotherapy as first-line treatment were not included in our study, as the number of such patients was very small and their follow-up periods were short.
Conclusion
Due to the lack of sufficient studies in the literature regarding the use of gemcitabine plus platinum as a first-line treatment for MPeM, we planned to compare the efficacy of gemcitabine plus platinum with pemetrexed plus platinum in our study. We demonstrated that pemetrexed plus platinum showed superiority over gemcitabine plus platinum in response rates. Although pemetrexed plus platinum showed a numerical advantage in PFS and OS, the differences between the 2 regimens were not statistically significant. Therefore, it was shown that gemcitabine may also be effective in patients who cannot receive pemetrexed. The main factor determining OS in our study was whether CRS + HIPEC had been performed.
The MPeM is a rare, progressively advancing disease that is difficult to treat. The main factor positively affecting OS is early-stage diagnosis of the disease and the performance of CRS + HIPEC. Pemetrexed plus platinum regimens remain one of the standard treatments for both epithelioid and non-epithelioid MPeM. Gemcitabine plus platinum, on the contrary, is one of the alternative regimens for first-line treatment.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to all colleagues and staff members of the Department of Medical Oncology at Ankara City Hospital whose support and assistance greatly facilitated this study.
Ethical considerations
The institutional ethics committee approved this study (Ankara City Hospital, Ethics Committee, Date: December 27, 2023/No: E1-23–4508). All procedures followed were by the ethical standards of the responsible committee and the latest Declaration of Helsinki. Ankara City Hospital Clinical Research Ethics Committee approved that informed consent was not required.
Consent to participate
As this was a retrospective study, no patient consent form was obtained.
Consent for publication
Not applicable.
Author contributions
DB served as the corresponding author and jointly designed the study with DU. DU provided mentorship and overarching supervision of the study design and interpretation. SCE, OK, and ST were responsible for data acquisition, while DB and SS conducted the statistical analyses. ŞY contributed to the interpretation and contextual analysis of the results. SEA and GU critically appraised the manuscript and offered substantial intellectual input. OB and EA carefully reviewed the final revision and approved the definitive version for submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data presented in this study are available on request from the corresponding author.
