Abstract
Malignant peritoneal mesothelioma is an exceedingly rare malignant tumor. Herein, we present a case of malignant peritoneal mesothelioma in a 59-year-old Chinese female patient who was stable after treatment for multiple relapses. Imaging revealed massive ascites and an irregular thickening of the peritoneal mesangium. Laparoscopic biopsy revealed heterogeneous cell nests in the parietal peritoneal fibrous tissue, which were confirmed by immunohistochemical staining for Calretinin, WT-1, and D2-40. In terms of genetic screening, BAP1, CSF1R, and other key driver gene variants closely related to malignant peritoneal mesothelioma have been explored in tumor tissues. Notably, CARD11 driver mutation was first found in all malignant peritoneal mesothelioma patients, and ATM A1159T gene mutation found in recurrent focal tissue may be associated with recurrent tumor recurrence.
Introduction
Malignant peritoneal mesothelioma (MPeM) is a rare malignant tumor derived from mesothelial cells that is high invasiveness.1,2 It has been reported that the incidence of MPeM is about 1–2/1 million, and the onset of MPeM is usually between 21 and 40 years old, with a sex bias (male vs female: 1.5–2:1). The surgical excision is the first treatment option for MPeM, based on the National Comprehensive Cancer Network (NCCN) guidelines. For patients who were not eligible for surgery, systemic chemotherapy (Pemetrexed in combination with Cisplatin) and immune checkpoint inhibitors (Nivolumab in combination with Ipilimumab) were commended as first-line treatments. However, relapse is common in patients with MPeM within 1 year of treatment, and the prognosis is not ideal.
Here, we report a case of MPeM in our hospital with multiple tumor recurrences within 5 years of diagnosis. The reason for the long-term survival of patients is considered to be the resection of recurrent tumors and postoperative chemotherapy; however, the use of surgery and drugs also places a great burden on patients’ life treatment and economic status. We now summarize the diagnosis and treatment process of the patient within the 5-year survival time (Figure 1) and conduct Whole-exome sequencing analysis of the tumor primary focus and tissue specimens of two recurrence foci at different times, to explain the cause of recurrent recurrence at the molecular level. The specific details are as follows.
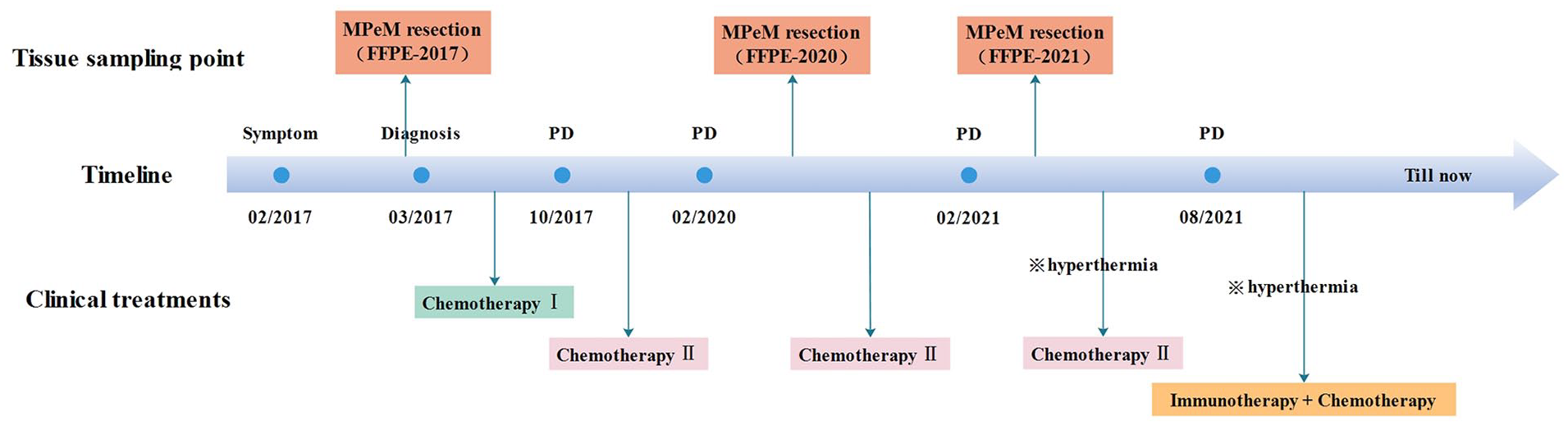
Diagnosis and treatment process. (Chemotherapy I: Pemetrexed + Cisplatin. Chemotherapy II: Gemcitabine. Immunotherapy + Chemotherapy: Camrelizumab + Gemcitabine/Cisplatin (IP)/Pemetrexed).
Case presentation
Clinical findings
A 59-year-old female with a history of asbestos exposure. On February 11, 2017, she was admitted to the hospital where she lived because of “abdominal distension and weakness without obvious inducement.” Imaging revealed cirrhosis, splenomegaly, portal hypertension, and peritoneal effusion. On February 13, no obvious abnormalities were found in the tumor indicators in the external hospital, abdominal effusion revealed shed cancer cells, and imaging diagnosis indicated a large amount of ascites. Abdominal mesentery slightly thickened, considering that peritoneal lesions could not be excluded, and he was discharged after symptomatic treatment. On March 22, 2017, the patient was transferred to the surgery department of our hospital for laparoscopic peritoneal biopsy. Hematoxylin and eosin staining indicated that heterogeneous cell nests were found in the parietal peritoneal fiber tissue accompanied by degeneration (Supplemental material 1). Immunohistochemical findings of Calretinin (+), WT-1 (±), and D2-40 (+) suggested malignant mesothelioma, CDX-2 (−) excluded tumors from the digestive tract, and ER (−) excluded breast metastases. Combined with immunohistochemistry and clinical findings, the patient was diagnosed with malignant mesothelioma (Figure 2).

The pathological diagnosis of the patient. (The sample obtained this time was named FFPE-2017, which is convenient for interpretation in subsequent detection results).
Since March 2017, the patient has received nine cycles of pemetrexed combined with cisplatin (intravenous injection and abdominal perfusion) in our hospital. Clinical efficacy evaluation in October 2017 suggested disease progression, with increased ascites, local thickening of the peritoneum, and turbidity in the fat space of the mesentery (Figure 3(a)). The replacement treatment plan was gemcitabine combined with local intra-cavity perfusion. After achieving stable disease control, the patient was discharged and scheduled for systematic follow-up assessments. On February 19, 2020, the peritoneal lesion was found to be larger than before, and gemcitabine chemotherapy was administered for four cycles. Due to poor efficacy, laparoscopic peritoneal mesothelioma resection was performed on May 8, 2020, the peritoneal lesions were removed, and pathological examination was performed (Figure 3(b)). After surgery, gemcitabine chemotherapy was continued twice, and the patient was followed up regularly. The re-examination on February 24, 2021 indicated that the tuberculum of the anterior hepatis was significantly larger than that of October 2020 (Figure 3(c) and (d)). Laparoscopic peritoneal mesothelioma resection was performed again on March 8, 2021, and the tuberculum of the anterior hepatis was removed for pathological examination. After surgery, gemcitabine local infusion chemotherapy combined with deep tumor hyperthermia was administered four times. A review in August 2021 showed the progression of the disease and then began immunotherapy combined with hyperthermia and chemotherapy, specifically Camrelizumab combined with Gemcitabine/cisplatin/pemetrexed. Currently, the patient’s disease control is stable. The patient experienced two instances of disease recurrence within 5 years following the initial surgical intervention, and the interval between the two recurrences is less than 1 year (sample: FFPE-2020 and FFPE-2021). Pathological findings of the two recurrences indicated peritoneal mesothelioma and the immunohistochemical results were consistent with those of the primary lesions.
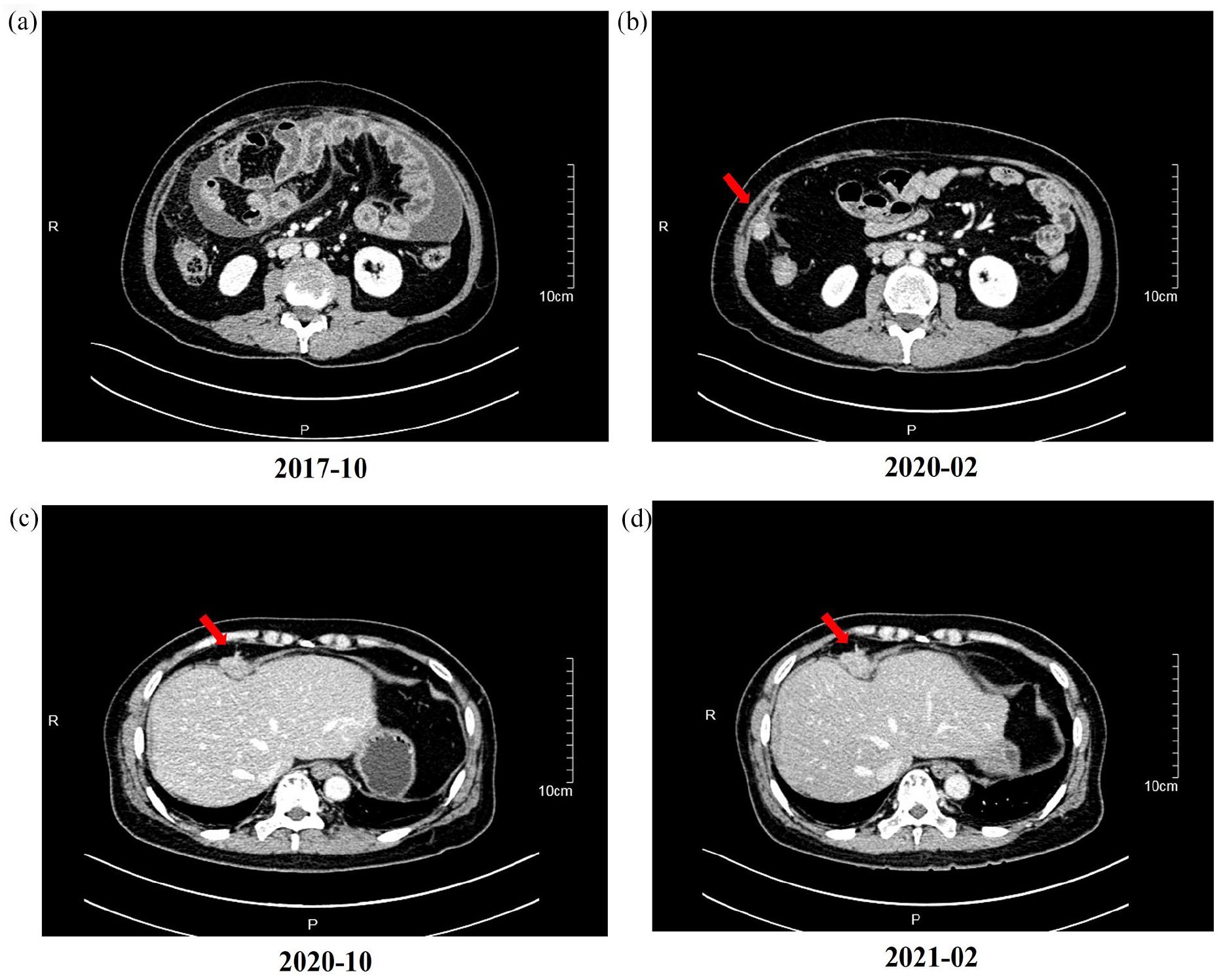
CT image of the patient. (a)–(d) are the imaging findings of patients on October 10, 2017, February 19, 2020, October 9, 2020, and February 24, 2021, respectively; ((a)–(d) all show varying degrees of disease progression; The samples obtained by (b) and (d) were named FFPE-2020 and FFPE-2021).
Genetic findings
Whole-exome sequencing (300×) was performed on tumor tissues and paired blood samples of the primary and double recurrence foci in this case, with a view to explaining the cause of recurrent tumor in this case at the molecular level. A total of 230, 226, and 193 somatic gene variations were identified in the primary and anterior and posterior recurrence foci respectively, among which there were 12, 15, and 9 driver genetic mutations respectively (Figure 4). In addition, there were both private and shared mutant genes between the three groups of foci. A total of 118, 97, and 104 shared gene variants were detected between FFPE-2017 and FFPE-2020, FFPE-2017 and FFPE-2021, and the first and second recurrence foci, respectively. A total of 48 common mutated genes were detected in the three foci, including BAP1 and the shared driver mutation of CARD11. In addition, ATM-driving mutant genes were found in the anterior and posterior recurrent foci. We did functional enrichment analysis on a total of 21 driver genes unique to the fore and after the recurrence foci and found that they were mainly related to the biological processes of DNA metabolism and positive regulation of cytokine production (Supplemental material 2), suggesting that the generation of these mutated genes might lead to the increase of tumor metabolic flux. In addition, statistical analysis of the base replacement characteristics of somatic cells at different stages in tumor tissue samples found differences, in which the primary focus was strongly enriched in A>G, G>C, and T>C, while the base replacement of the recurrent focus was doubled to C>A (Figure 5).

Similarity among different lesions based on somatic mutation analysis. (a) Venn diagram illustrating the distributions of genetic mutation in different lesions. (b) Venn diagram illustrating the distributions of driver genetic mutation in different lesions.

Statistics of different types of point mutations. (a) Statistical map of point mutation types. (b) Statistical map of mutation patterns in all samples.
Finally, when analyzing the clonal evolution of tumors to examine the heterogeneity among different foci, it was found that the primary foci and the secondary foci were located on the two branches of the evolutionary tree respectively (the tumor recurrence foci were located on the same branch). According to the evolutionary distance of the samples, the genetic variation of the anterior and posterior foci was less than that of the primary foci and the first foci. It was suggested that there were large commonalities and small differences among subclones within recurrent tumors (Figure 6).
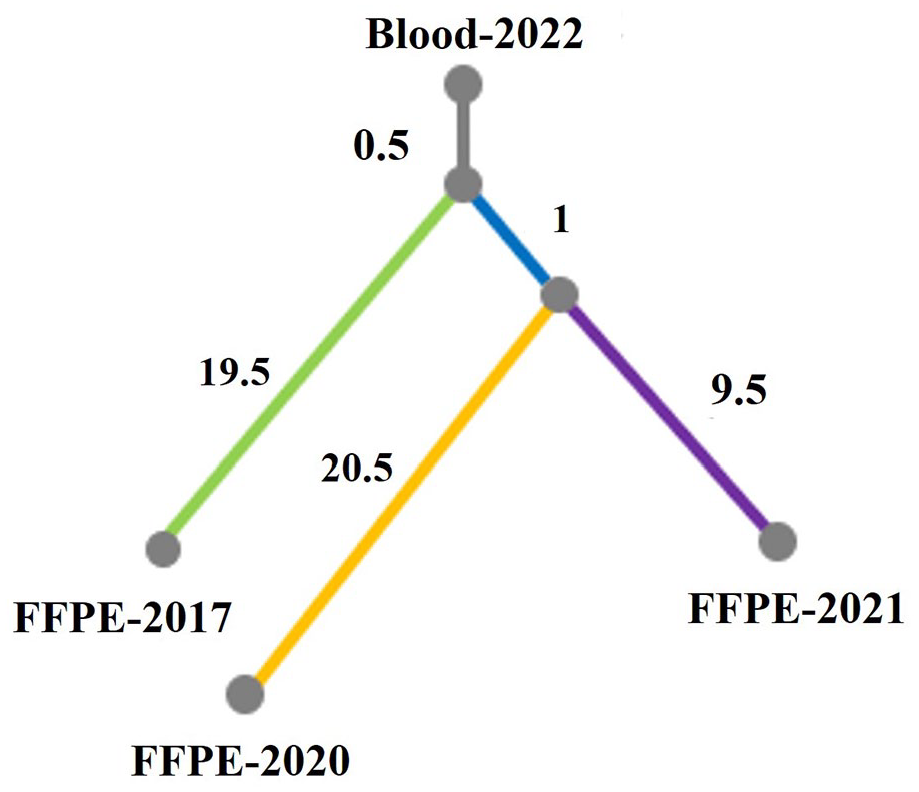
Tumor evolutionary tree.
Discussion
MPeM is a rare and highly malignant tumor originating from peritoneal mesenchymal cells. 3 in 1908. Its etiology and pathogenesis are unknown, which may be the result of interaction between environmental carcinogenic factors and genetic susceptibility of the organism. Foreign researchers have indicated that asbestos, as a major environmental carcinogen of MPeM (about 33%–50% of patients with asbestos exposure history), can directly damage the genetic material of cells, or indirectly damage the genetic material4–6 by inducing the release of inflammatory factors and activating PI3K/Akt/mTOR signaling pathway.
The reported case initially considered a history of asbestos exposure, but following diagnosis, it was confirmed that the patient had not been exposed to asbestos. The clinical manifestations of MPeM are various, mainly including abdominal distension, abdominal pain, refractory ascites, and some cases may be accompanied by fever, poor appetite, nausea and vomiting, incomplete intestinal obstruction, etc.7,8 This patient presented symptoms such as abdominal distension, discomfort, and fatigue at the first onset, and chose to seek medical treatment after a long period of time. Massive ascites were found during imaging diagnosis. As the disease progressed, the patient began to develop persistent pain in the right side of the abdomen, abdominal CT examination showed typical irregular thickening of the peritoneum, nodular changes, and continuous enlargement of the leading edge of the liver, around the ascending colon and adjacent abdominal wall, clouded adipose space in the mesentery, and abdominal effusion. It can be seen that there were no typical clinical manifestations in the early stage of MPeM, and the patients showed abdominal pain symptoms with the aggravation of the disease in the later stage. The diagnosis of the disease mainly depends on the combination of histopathologic examination and immunohistochemical examination, and neither is necessary. Tissue examination of the patient we reported suggested epithelioid mesothelioma, with positive immunohistochemical Calretinin, WT-1, and D2-40 expressions. Prior to initial diagnosis, the patient exhibited ascites harboring free-floating cancer cells and demonstrated signs of peritoneal metastasis. Despite surgical intervention, complete excision of the lesions was not feasible, and it is likely that microscopic peritoneal foci remained. There was no detected metastasis to the brain, lungs, bones, or liver. Following surgery, the patient was subjected to routine evaluations every 2–3 months, including CT scans and serum tumor marker assessments. The primary method for assessing disease progression was through abdominal CT scans. Serum tumor markers were also employed to monitor disease control. In some instances, serum CA-125 levels were elevated. 9 In this case, however, the tumor marker levels remained stable, with CA-125 levels consistently within the normal range (<23 U/mL), peaking at 12.2 U/mL. In terms of treatment, the patient initially received the standard first-line systemic therapy, consisting of pemetrexed combined with cisplatin. However, due to disease progression or adverse reactions, the treatment regimen was altered to local chemotherapy involving gemcitabine/cisplatin, supplemented by deep tumor hyperthermia. Subsequently, the patient underwent surgical tumor resection. Despite these interventions, the tumor recurred, with notably shorter intervals. So, we use molecular detection techniques to try to explain the recurrence.
Possible pathogenic mutation genes including BAP1, CDKN2A, BRCA2, NF2, TP53, WT1, etc. have been detected through genetic testing of MPeM patients by next-generation sequencing. The BAP1 mutation. BAP1 is a carboxy-terminal hydrolase of ubiquitin protein that has been established as a tumor suppressor, using its deubiquitination activity to regulate many biological processes, which is most closely related to the occurrence of MPeM. 10 For example, BAP1 binds to IP3R3 to deubiquitinate and promote apoptosis. If BAP1 expression is inactivated, cells are unable to complete the apoptosis process, allowing DNA mutations to persist, leading to the development of tumors. 11 Baumann et al. 12 found that MPeM patients with embryogenetic BAP1 mutation had a much higher outcome than MPeM patients without embryogenetic BAP1 mutation, with an overall survival (OS) of 10 years versus 9 months and a 5-year survival rate of 53% versus 5.2%. BAP1 as a therapeutic target has become the focus of current research. In a multicenter, single-arm, open-label Phase II clinical study of patients with relapsed or refractory MPeM, the EZH2 inhibitor tazemetostat was found to have a 32.8% disease control rate in patients with BAP1 mutation (2 years follow-up). The results of progression free survival (PFS) at 18 weeks (95% CI 1.6–114.3 weeks) and OS at 41 weeks (95% CI 1.6–120.9 weeks), which have not been publicly published, have the potential to be an effective drug in the future for MPeM patients with BAP1 mutation. 13 In this study, genetic mutations of CSF1R and ATM were also detected, which have been found in previous studies to be related to the occurrence of tumors, including prostate cancer, breast cancer, glioblastoma, gastrointestinal stromal tumors, and other tumor tissues. 14 The combination of CSF1/CSF1R can promote tumor formation by recruiting tumor-associated macrophages. However, the discovery in MPeM patients was again after the first report by Lu et al. 15 The ATM gene is a tumor suppressor gene that initiates the apoptotic pathway of damaged cells to maintain genomic stability when cells are damaged. Once the ATM gene is disabled, its cell cycle checkpoint function is weakened, and the cell’s self-repair ability is impaired, which leads to the occurrence of diseases such as malignant tumors. 16 At present, somatic mutations of ATM gene have been found in various tumors such as lung adenocarcinoma and gastric cancer, among which ATM R3008C/H/L was found to be a recurrent mutation in TCGA data. To this end, we will find in this case, the ATM (p. A1159T) and ATM (p. H3008R) made of protein 3D structure prediction (http://missense3d.bc.ic.ac.uk/missense3d/) (Figure 7), Protein cavities and channels are crucial to function. Ligand recognition, binding, transport, and enzyme catalysis all require cavity weight arrangement. It is speculated that ATM A1159T may be another recurrent mutation, which leads to recurrent tumor occurrence in patients. In addition, molecular mutation of CARD11 as a driver gene was detected in all three tumor samples, which has not been found in MPeM patients in previous studies, and whether it is related to the occurrence and development of MPeM needs more research.

ATM p.A1559T (a) and ATM p.R3008H (b) three-dimensional protein structure prediction.
Conclusion
Here, we report a long-term survival of a female MPeM patient who responded well to pemetrexed plus cisplatin but developed disease progression 3 years after initial treatment and had a high recurrence rate after complete resection, which severely affected the patient’s quality of life. The mutation of ATM A1159T gene in the patient’s recurrent foci may be related to the recurrence of the patient’s tumor. To the best of our knowledge, this is the first time MPeM cases reported.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X241271818 – Supplemental material for Case report: ATM A1159T mutation in malignant peritoneal mesothelioma may be associated with tumor recurrence
Supplemental material, sj-docx-1-sco-10.1177_2050313X241271818 for Case report: ATM A1159T mutation in malignant peritoneal mesothelioma may be associated with tumor recurrence by Ying Li, Jintao Yang, Bei Zheng, Na Yan, Wenjuan Yang, Xinling Xie, Guoshu Xu and Meiling Zhang in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
The authors would like to thank all the medical staff of Litongde Hospital of Zhejiang Province for their assistance in patient diagnosis and treatment. We would also like to, thanks for the case discussion from members of the Medical Department of the Dian Diagnostics Group Co., Ltd.
Author contributions
G.S.X. and, M.L.Z. conceived of and designed the study. They and colleagues Y.L., B.Z., W.J.Y., and X.L.X. treated patients and recorded their clinical information. J.T.Y. and N.Y. performed the literature search, analyzed, and interpreted the data, and wrote the manuscript. All the authors have read and approved the final manuscript.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Basic Public Welfare Research Program of Zhejiang Province (LGF20G030004).
Competing interests
Author JT Yang and N Yan were employed by Dian Diagnostics Group Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki. This study was approved by the Zhejiang Tongde Hospital Biomedical Research Ethics Review (acceptance number No. 2022-144 (K). Written informed consent was obtained from all subjects prior to the commencement of the study.
Consent for publication
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
