Abstract
Prion protein (PrPC) was originally known as the causative agent of transmissible spongiform encephalopathy (TSE) but with recent research, its true function in cells is becoming clearer. It is known to act as a scaffolding protein, binding multiple ligands at the cell membrane and to be involved in signal transduction, passing information from the extracellular matrix (ECM) to the cytoplasm. Its role in the coordination of transmitters at the synapse, glyapse, and gap junction and in short- and long-range neurotrophic signaling gives PrPC a major part in neural transmission and nervous system signaling. It acts to regulate cellular function in multiple targets through its role as a controller of redox status and calcium ion flux. Given the importance of PrPC in cell physiology, this review considers its potential role in disease apart from TSE. The putative functions of PrPC point to involvement in neurodegenerative disease, neuropathic pain, chronic headache, and inflammatory disease including neuroinflammatory disease of the nervous system. Potential targets for the treatment of disease influenced by PrPC are discussed.
Keywords
Introduction
Knowledge of the function of prion protein is rapidly expanding. This review examines the role of prion proteins in molecular signal transduction and cell physiology, and considers ongoing questions about the role of prion protein in the nervous system. The involvement of prion protein in non-prion disease as well as potential therapeutic targets is considered.
Prions are the agents that cause a number of forms of transmissible spongiform encephalopathies (TSEs), comprising Creutzfeldt-Jakob disease (CJD), including variant CJD (v-CJD), familial CJD (f-CJD), and spontaneous CJD (s-CJD); Gertstmann-Sträussler-Scheinker syndrome (GSS); fatal familial insomnia (FFI) syndrome and kuru in humans; bovine spongiform encephalopathy (BSE or “mad cow disease”) in cattle; scrapie in sheep; and chronic wasting disease (CWD) in deer and elk. This disease is caused by an abnormal form (PrPSc) of the normally occurring prion protein (PrPC). Since the recognition of prion disease in the early 1980s 1 and the discovery of the molecular basis of the disease, 2 the prion protein itself has proved to be an enigmatic and controversial protein. The association of prions with TSE initially gave prominence to the negative role of prions in cell physiology and disease. The functions of prion protein in normally operating cells are, however, becoming more apparent although the full range of functions has still to be determined. Evidence regarding the involvement of prion protein in cell physiology, signal transduction and long-term potentiation (LTP) in memories, and epigenetic inheritance has been increasing over the past 25 years, and there is now a growing indication that PrPC functions, in both beneficial and harmful ways, as a molecular switch. 3
Prion Protein Structure
Prion disease is the only known disease that can be caused by infection (through the food chain or injection/transplantation) as well as by sporadic and inherited mutations. Prion disease is caused by a self-replicating protein found in normal cells. There is one prion protein in humans, and prion proteins have been found in many mammals and other vertebrates (birds, reptiles, amphibians, fish), 4 as well as fungi, with at least seven discovered so far in yeasts. 5 Interestingly, PrPC has not been found in lower animals (insects, molluscs, protozoa). 6
Prion protein exists in at least two conformational states: first, the cellular α-helix-rich isoform (PrPC) and, second, the prion disease-associated β-sheet isoform (PrPSc) 7 (Fig. 1). In humans, PrPC is a 32-kDa protein, with 253 amino acids encoded by the single-copy PRPN gene, located on chromosome 20. 8 The protein has regions that are highly conserved in all vertebrates. 9 When not in a disease state, the cellular prion protein has two isoforms with 208-209 amino acids: a membrane-bound form and a soluble cytosol (=secreted) form. The membrane-bound PrPC is a glycoprotein, attached by a glycosylphosphatidylinositol (GPI) anchor 10 to lipid rafts on the outer leaflet of the cell membrane, as is the case with most GPI-anchored proteins. The soluble form is not glycosylated. Prion protein has an intrinsically disordered protein (IDP) component, in that the N-terminal end (amino acids 23-121) of the protein does not have a permanent tertiary structure but is flexible (Fig. 1A). The C-terminal end of the protein (amino acids 127-231) has a well-structured globular tertiary structure, folded into three α-helices and two short β-strands. 11
The structure of the disease form of prion protein is not fully known. It has the same primary structure (amino acid sequence) as PrPC, but the secondary structure has more β-sheet regions than the α-helix of PrPC (Fig. 1C) and is able to form amyloid fibrils
12
(Fig. 1D). The isoform of the disease is highly stable, resistant to proteolytic enzymes, and self-replicating.
2
The presence of PrPC is necessary for progression to prion disease and for neurotoxicity to occur, but the actual mechanism that controls the misfolding of PrPC is not known. According to the “protein only hypothesis” of prion disease transmission,
13
the conversion is an auto-catalytic, posttranslational conformation change, with PrPC interacting with existing PrPSc, which acts as a template or “seed” and recruits PrPC to aggregate into the β-form, which acts as a template in its turn, continuing the chain reaction and corrupting more PrPC.
14
The prion disease can be initiated either by introduction of the PrPSc protein (v-CJD, kuru) or by a point mutation in the PRNP gene (s-CJD, f-CJD, FFI, GSS). When PrPC is originally synthesized at the endoplasmic reticulum (ER), the correct folding of PrPC is controlled by chaperone molecules (such as Hsp110, Hsp60, αB-crystallin, Rdj2, Hsp40/Hsp70, Hsp90).
15
Any protein that has been misfolded is handled by the unfolded protein response (UPR) of the cell. This involves the protein being refolded either with chaperones and being degraded by lyso-somes or proteasomes or being confined and isolated in cyto-plasmic inclusion bodies (aggresomes). Interestingly, IDP regions in any protein increase the potential for protein misfolding.16,17 The mechanisms that control the homeostasis of PrPC in the cell are as yet unknown, but the progression to prion disease might be seen as a failure of cellular quality control of the UPR.
Cartoon of the structure of prion protein deduced from NMR, showing the IDP N-terminal end—amino acids 23-121 (yellow dots), the three α-helices (orange), and the short β-sheet section (blue). (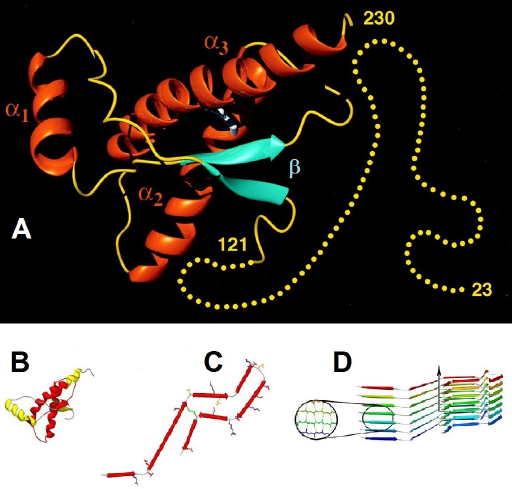
The PRPN gene is expressed in many cells of the body, but is most highly expressed in cells in the nervous system. Likewise, the protein PrPC is ubiquitous in cells of the body, but (as reviewed by Linden and coworkers) 18 is found most abundantly in nervous system cells; in neurons (cell body and synaptic membrane) of the hippocampus, cortex, thalamus, cerebellum, and medulla; and in glial cells, including astrocytes. 19 It is found in high numbers in the neuroimmune network, including small diameter fibers of the skin, sympathetic ganglia and nerves, parasympathetic and enteric nervous system, dispersed neuroendocrine systems, and peripheral nervous system axons. 20 It is also present in high numbers in bone marrow stem cells, lymphocytes, monocytes, macrophages, natural killer cells, dendritic cells, and follicular dendritic cells. 11 In most cells, PrPC is almost entirely membrane bound, with very little found in the cytoplasm. In some cells, however, such as neurons in the hippocampus, thalamus, and neocortex, the cytosol form of PrPC is commonly found. 21 Both membrane-bound and soluble forms of PrPC are found in the cerebral spinal fluid. 22 Membrane-bound PrPC can be secreted from cells into the extracellular matrix (ECM) in exosomes, and Martins and coworkers 23 list a number of mechanisms for the release of soluble PrPC from the GPI anchor at the cell membrane.
PrPC is able to move between cells via two pathways. 24 The first relies on soluble PrPC in extracellular space. The second depends on the presence of the GPI anchor and direct cell-to-cell contact. This shares a common proteolytic shedding mechanism with amyloid precursor protein and other surface proteins, and invokes the mechanism of molecular crowding, 25 thus, occurring when increased density of cells causes increased cell-to-cell communication.
A number of proteins related to human prion proteins have been found in the yeast Saccharomyces cerevisiae as well as in other yeast and filamentous fungi. These proteins spontaneously aggregate in vitro, and display the seeding that is characteristic of prion aggregation. Seven proteins have so far been identified as prions with up to 18 identified as being potentially prions or prion like. 5
The conserved nature of prion DNA sequences in distantly related animals, the ubiquitous distribution of PrPC in many cells of the body, and the concentration of PrPC in nervous tissue, all suggest that PrPC plays an important and widespread role in cellular metabolism, especially in the nervous system.
Prion Protein and Amyloid
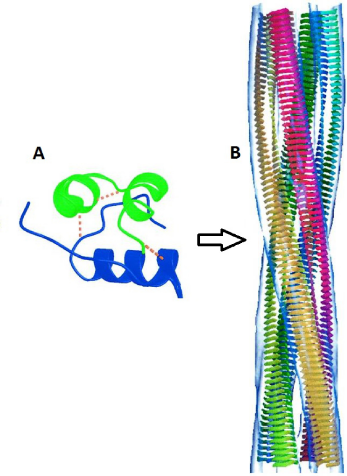
During prion disease, PrPSc is functionally an amyloid form of normal PrPC; indeed, purified PrPC will form amyloids in vitro with detergent, 26 and about 10% of CJD patients and 100% of v-CJD patients show amyloid plaques. 27 Amyloids consist of β-sheets arranged in cross-layers that are repeated many times to form insoluble fibrils 28 (Fig. 2) (described in reviews by Fowler, 29 Greenwald and Riek, 30 and Eisenberg and Jucker). 31 Amyloid disease is characterized by progressive aggregation of the native tertiary protein structure into this very stable β-fibril form. This process appears similar to prion infection, which also recruits PrPC to an amyloid-like PrPSc β-form. This poses the question as to whether all amyloid diseases are in fact prions or at least prion like in their action. Research continues to emphasize the closing gap between amyloid and prion diseases. 32
Amyloid protein has been suggested to be an ancient form of protein structure. 29 Rather than a particular protein sequence that form amyloids, there are many amino acid sequences that have this capability, although, some amino acid sequences are more prone to amyloid formation than others. 5 Amyloid formation may even be a common or intrinsic property of most proteins.
While many proteins have the capacity to form amyloids, they do not normally do so, because of an array of protective mechanisms present in cells, including chaperone proteins that guide and assist protein folding to ensure that amyloid structures do not form and to ensure that proteins achieve their correct functional conformation (see reviews by Bukau et al
33
and Chen and Inouye).
34
Cells also maintain tight intracellular control of temperature and pH to maintain correct protein conformation. When confronted with unfolded or misfolded proteins, the cells invoke the UPR. It is possible that a breakdown in this defense (such as a mutation in chaperone genes)
35
as well as protein denaturation could be responsible for the development of amyloid diseases such as Alzheimer's disease, Huntington's disease, and Parkinson's disease, and amyloid formation in Type 2 diabetes and cataracts. These diseases are characterized by amyloid aggregates that form from particular proteins via a series of intermediate structures.
36
Evolution appears to have selected globular forms of proteins, hiding the amyloid forming sequences in the interior of the protein. While the development of IDP could be seen as an evolutionary step 37 (ie, conserved between organisms, a multitude of functions—especially in cell signaling and molecular binding, more common in complex eukaryotic organisms than simpler prokaryotic organisms), IDPs are also more prone to misfolding than globular proteins.16,17,38 Some of the proteins implicated in amyloid disease, for example, Aβ in Alzheimer's disease and α-synuclein in Parkinson's disease, are IDPs. 38
Functional Amyloids
Not all amyloids are harmful. Non-harmful or useful amyloid structures (functional amyloids) have been identified in diverse organisms, 30 such as bacteria, fungi, insects, and mammals (including humans). Amyloid structures are often used as structural proteins, because of their strength, resilience, and resistance to chemical and enzymatic attack. In bacteria, extracellular amyloids (“curli” fibers in Escherichia coli, and “tafi” fibers in Salmonella) are used as adhesion matrices during colony formation, 39 and in Streptococcus mutans, amyloid is a constituent of the biofilm formation in dental plaque 40 among other functions. 41 Yeasts, such as Candida, use amyloid for cell adhesion, 42 and many filamentous fungi use amyloid fibers (hydrophobin) to strengthen aerial hyphae and spores. 43 Insect eggshells can be protected by amyloids, 44 and some spider silks have an amyloid structure. 45 In humans, functional amyloids so far discovered include the protein PMEL17, which assists in the assembly and transport of the pigment melanin, 46 peptide hormones stored as amyloid structures in pituitary secretory granules, 47 and the suggestion of amyloid involvement in blood clotting. 48 The mitochondrial anti-viral-signaling (MAVS) protein has been found to form amyloid fibrils in cells infected by some viruses, which initiates a signaling cascade to produce interferon to combat the infection. 49 It has also been suggested that amyloids could play a role in memory formation. 50
Functions of PrPC
The full function of PrPC remains elusive, despite decades of research. Many of the proposed roles for PrPC (Fig. 3) are linked to its location, that is, concentrated in nervous tissue cells and localized on plasma membranes, specifically within lipid rafts.
Elucidation of the role of PrPC in the non-disease state has been aided by the study of PRNP knockout mice, as well as overexpression of PrPC in transgenic mice and the expression of PrPC in mammalian cell lines, zebrafish, Drosophila, nematodes, and yeasts. In addition to being non-susceptible to prion disease, null PrPC mice lines have displayed symptoms that ranged from no obvious developmental effects through subtle behavioral and cognitive deficits to severe ataxia, neurodegeneration, and total paraplegia, depending on the type of mutation or ablation. 18 One reason for the variability in the phenotypic effects of PrPC-null mice is the variety of methods used to knockout the PRNP gene (comprehensively reviewed by Linden et al 18 and Onodera et al). 51 Ablation of the PRNP gene using some (but not all) methods resulted in removal of large DNA segments, with the flow on effect of a downstream gene (PRND) overexpressing the Doppel protein, which turned out to be the cause of the observed ataxia. Interestingly, the ataxia could be reversed by PrPC. The variability caused by differences in construction of PrPC-null mice has made pinpointing the function of PrPC challenging.
Prion disease itself is associated with ataxia, loss of circadian activity, p53 regulation, copper and zinc transport dysfunction, and the dysregulation of redox homeostasis through reactive oxygen species (ROS) modulation (as reviewed by Linden et al). 18 These losses in function during prion disease are also suggestive of the role of PrPC in normally functioning cells.
Animal studies have suggested that PrPC plays a role in neuroprotection, neuritogenesis, and neurite polarization,
18
as well as having a role in processing olfactory signals,
52
circadian rhythms and sleep patterns,
53
memory,
54
and behavior.55,56 PrPC-null mice have been shown to have muscle fatigue under stress,
55
decreased mitochondrial number and mitochondrial malformations,
57
and increased superoxide dismutase (SOD) and free radical production.
58
Overexpression of PrPC has been shown to lead to necrotizing myopathy of skeletal muscles
59
and other muscular disorders.
60
PrPC has been found to be neuroprotective and may be involved in muscle strength and endurance via the PPAR pathway.
61
Morel et al
62
also found evidence that PrPC is involved in the differentiation and polarization of epithelial cells. With this wide range of effects attributed to PrPC, the picture that emerges is of a protein with extreme versatility of function, which makes pinning down a unifying molecular basis for the function of PrPC challenging.
Summary of the reported cellular and organism functions of PrPC.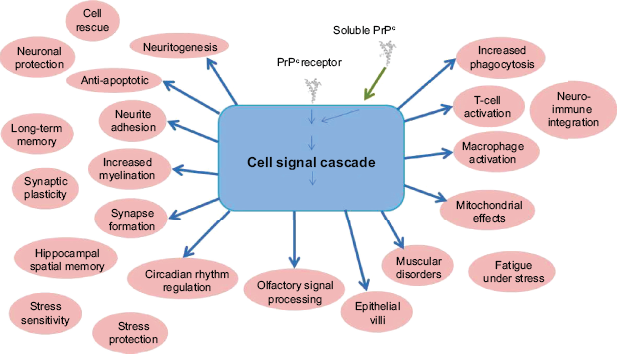
A number of proteins have shown differential expressions (under-expressed or over-expressed) in cells with symptoms of TSE. This finding implies some role for PrPC in the functioning of these proteins. Gawinecka and coworkers 63 found 46 proteins differentially expressed in the proteome of patients with s-CJD, including protein 14-3-3, Hsp90, β-tubulin, SUMO2/3, and stathmin. Most common differentially expressed proteins between s-CJD subtypes were proteins associated with signal transduction and neuronal activity, especially Rab GDP dissociation inhibitor α, which regulates Rab3a-mediated neurotransmitter release.
A well-studied prion is the yeast translation-termination factor Sup35. Like human PrPC, Sup35 carries a prion-determining domain (PrD), which occasionally adopts an amyloid conformation that perpetuates itself by templating the same amyloid conformation on other Sup35 molecules, which then sequesters most Sup35 into insoluble fibers. Prion proteins occur naturally in many wild yeasts and can be a selective advantage when in the amyloid form. 64 The switch from non-prion to prion state occurs at a measurable rate, which is increased during stress. There is a suggestion that prion protein might also be an important switch in mammals, including humans. 32
PrPC appears to be central to neuronal survival and function, because of the many roles that have been suggested for PrPC in the nervous system (reviewed by Westergard et al, 59 Linden et al, 18 Martins et al). 23 These include antiapoptotic effects, 65 neuroprotection,59,66 myelination, neuritogenesis, 67 axon growth, 68 neurite outgrowth,65,69 neurite polarization,67,70 synapse formation, 71 cell-to-cell communication through gap junctions and neurotrophic activity, 72 and LTP.73,74
In the immune system, PrPC affects such processes as T-cell proliferation and phagocytosis among other effects in the nervous and immune systems.6,18,23 PrPC has also been shown to have a role in stem cells (reviewed by Lopes and Santos). 75 Zhang and coworkers 76 have shown PrPC to be highly expressed in hematopoietic stem cells and required for self-renewal, as is the case for neural stem cells. 77 Prion protein is seen as a switching mechanism that controls human embryonic stem cell proliferation, self-renewal, and the fate of cell cycle dynamics. 78
Many of the functions that have been attributed to PrPC could be triggered because of signal transduction.
Signal Transduction
PrPC Binding Partners
PrPC has a propensity to bind to many molecules, due in part to the IDP nature of the N-terminal end of the protein (see Fig. 1), which lends itself to promiscuous binding.18,79 IDPs depend on molecular crowding to induce compact and stable binding. They function by wrapping around their partner molecules to achieve their final state and can often bind to multiple partners. In general, IDPs have roles in signal transduction, gene expression, and chaperone activity. 79 Up to 45 ligands have been identified as binding to PrPC (see reviews by Westergard et al, 59 Linden et al, 18 Aguzzi and Steele, 80 and Martins et al). 23 In addition, soluble PrPC can act as a ligand for membrane-bound receptors. 23 Although the in vivo relevance of the association has as yet to be determined in all cases, it is possible that many of these ligands could be triggers for cellular signaling.
PrPC has been known for some time to selectively bind to copper ions at the N-terminal region of the protein (reviewed by Vassallo and Hems), 81 with at least four Cu2+ ions binding to each PrPC molecule. The region of the protein that binds Cu2+ is highly conserved between species, arguing for an important role for this property of PrPC. The binding has been linked to a role of PrPC in the regulation of Cu2+ levels and the consequent protection of cells against oxidative stress. 82 This may be linked to an interaction with the calcineurin complex. 83
PrPC has been shown to bind to many molecules, including:
ECM proteins such as laminins
84
and vitronectin,
85
and glycosaminoglycans such as heparin and heparin sulfate;
86
molecules on the outer leaf of the plasma membrane such as 37 kDa laminin receptor precursor (37LRP), 67 kDa laminin receptor (67LR),
87
and ganglioside GM1;
88
molecules on the inner surface of the plasma membrane such as Fyn kinase
89
and neuronal nitric oxide synthase (nNOS);
90
intracellular membrane components such as glutamic acid decarboxylase (GAD),
90
STI1,
91
Bcl-2,
92
and synaptophysin;
90
transmembrane proteins including neural cell adhesion molecule (NCAM),
68
integrins,
85
G-protein-coupled serotonergic receptors (GPCR),
89
and G-protein receptors;
89
transmembrane ion channels such as voltage-gated calcium channels (VGCC),93,94 calcium-activated potassium channels,
95
and two-pore potassium channel protein (TREK-1);
96
cytoskeleton proteins α-tubulin,
90
β-tubulin,21,97 and stathmin;
98
scaffolding proteins GRB2,
99
β-1 integrins,
67
synapsin,
99
Caveolin-1,
89
and protein complex 14-3-3;
100
and chaperones and co-chaperones such as Hsp60,
101
Hop/STI1,
91
αB-crystalline,
102
Rdj2,
103
and clusterin.
104
The binding of PrPC to chaperone molecules leads to speculation as to a possible function of PrPC as a protein co-chaperone, although it is also possible that the binding is linked to the role of chaperones in ensuring the correct folding of a potentially lethal protein.14,15 It is, however, known that PrPC can act as a molecular chaperone for DNA and RNA.105,106 Many of the ligands binding to PrPC are related to cytoskeleton functions, 107 including cell growth (neuritogenesis), differentiation (polarity), and neuron maintenance.
Protein Scaffolding
PrPC is most commonly found in the detergent-insoluble (=lipid raft) domains of the plasma membranes, 108 a region of concentrated cholesterol and sphin-golipids. Some PrPC is also found away from raft regions, in clathrin-coated pits where it is subject to endocytosis. It has been recognized for some time that PrPC traffics between a membrane location and endocytes, especially in neuronal cells. 109 Endocytosis of PrPC and encapsulation appears to be intimately involved in the function of PrPC, and the protein is circulated between the plasma membrane and the cytoplasm rapidly, 18 with a proportion of membrane-bound PrPC being transported in endosome compartments. This may first entail translocation from the lipid raft section of the membrane to a non-raft region 8 before being endocytosed, most probably with the aid of clathrin proteins. Alternatively, Caveolin-1 and caveolae may be involved in endocytosis. 110 The PrPC is then recycled, with much returning to the cell membrane but some being broken down (with lysosomes) and some being exported from the cell (exosomes). Endocytosis from the cell membrane is a common feature of proteins involved in neurotrophic activity and therefore suggests the same role for PrPC.
Lipid raft domains are known to contain a number of receptor molecules, and these domains are closely associated with signal transduction, with the interaction between ligand and receptor activating a signaling cascade. 111 The lipid raft regions of the plasma membrane serve to segregate and concentrate signaling components into discrete locations, which are important sites for relaying information into cells. The location of PrPC in the lipid raft domains and the binding to ligands is additional evidence for a function in signal transduction. PrPC is, however, located on the outer leaf of the plasma membrane and does not have a transmembrane domain, so signal transduction would depend on transmembrane ligands, the partner's transmembrane ligands, or endocytotic pathways (either by clathrin-coated pits or caveolae).
Signal Transduction Cascades
It is now generally accepted that PrPC is active in signaling processes, 112 not merely as a link between extracellular proteins to the cytoplasm but as a signal receptor and inducer of enzymatic activity in the transduction of signals. PrPC acts as a membrane platform for assembly of signaling scaffolds through binding of various ligands and transmembrane signaling pathways, and may modulate the activity of receptor molecules. Despite the known and reported effects of PrPC, the actual molecular mechanisms are still being elucidated. Signal cascades have been reviewed by Linden and coworkers.18,23,112
PrPC binding of laminin molecules results in neuritogenesis, neuronal plasticity, and memory consolidation in rat hippocampus.
113
GPI PrPC colocalizes with 37LRP/67LP, which suggests a complex binding of laminin with 37LRP/67LR + PrPC + other receptors such as integrin, to give a cluster of receptors acting by integrin-mediated signal transduction and/or internalization of the lipid raft (Fig. 4A). This acts to induce cell adhesion, increase filopodia production, and promote directional motility.
23
Interaction of PrPC with ligands at the lipid raft and the subsequent signal transduction and ultimate effect. (A) Binding to laminin receptor (37LRP/67LR), integrins, heparan sulfate proteoglycans (HSPG), and ECM proteins laminin (LN) and vitronectin (VN). (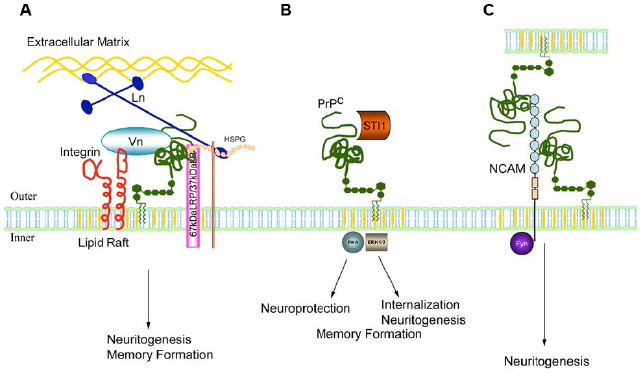
PrPC has been shown to regulate the activity of p59fyn tyrosine (Tyr) kinase through interaction with Caveolin-1 in caveolae of neurites,89,110,114 most likely mediated via interaction of PrPC with NCAM at the lipid raft membrane site. 68 Downstream, this mediates the production of ROS via NADPH oxidase, which in turn acts a “second messenger” to induce a signaling cascade via the extracellular-signal-regulated kinase (Erk1/2) pathway. 115 ROS can also induce cAMP response element-binding protein (CREB), Egr-1, and c-Fos. 116 Erk1/2 promotes calcium flux 117 and neurite outgrowth. 68 It has been shown that endocytosis of the membrane raft complex is essential for this signal transduction. 118
NCAM-induced signaling also activates a range of other signal cascades, including focal adhesion (FA) kinase, intracellular kinase, and mitogen-activated protein kinase (MAPK), as described by Martins and coworkers. 23 This can also occur between cells (Fig. 4C).
The binding of PrPC with secreted stress-inducible protein 1 (STI1) 119 stimulates an increase in cAMP and activation of the cAMP/protein kinase A (PKA) pathway, which facilitates neuroprotection, 120 as well as the Erk1/2 pathway. 119 It is not clear how the bound ligands from the ECM trigger this response, although GPCR and G-proteins may serve as intermediates, controlling cellular redox18,23 (Fig. 4B). This signal leads to a number of cellular outcomes including neuroprotection and cell rescue 120 as well as short-term memory (STM) formation and long-term memory (LTM) consolidation, 121 apoptosis, neuronal death, and neuritogenesis. 18
PrPC has been shown to interact with β1 integrins at the membrane. 67 The consequence of this interaction on cyto-skeleton modulation and neuritogenesis has been reviewed by Alleaume-Butaux and coworkers. 122 In addition, PrPC binds to the ECM glycoprotein vitronectin 85 and may be involved in the neuritogenesis and neuronal differentiation growth of axons in dorsal root ganglia (DRG) during embryogenesis.
PrPC is known to interact with ion channels (reviewed by Martins et al),
23
which can then influence the flux of ions across the plasma membrane. There is an interaction between PrPC and TREK-1 channels, which regulate K+;
96
L-type VGCC, which regulate Ca2+;
93
and the NR2D subunit of NMDA (N-methyl-D-aspartate) receptor, which opens a non-selective cation channel (Fig. 5). PrPC also appears to be involved in Ca2+ homeostasis via the purinergic (P2Y) pathway and store-operated calcium channels (SOCCs)
20
(Fig. 6). Calcium ion flux, as well as cytoplasmic ROS, has the potential to influence the mitochondrial retrograde signaling response (reviewed by Butow and Avadhani).
123
In addition, PrPC has been linked to both protein complex 14-3-3
100
and calcineurin B,
83
both of which are known to directly affect the regulation of the K+ leak channel K2P, TRESK, which determines resting membrane excitability
124
and is the only K2P channel upregulated by a Ca2+-dependant pathway. This may give PrPC an indirect role in this ion channel and therefore cellular electrical excitability.
PrPC modulation of the activity of ionic channels. ( PrPC modulation of intracellular Ca2+ homeostasis through G-protein-coupled purinergic (P2Y) receptors, which are activated by ATP and coupled to calcium stimulatory G-protein and to phospholipase Cβ, reducing cleavage of phosphatidylinositol-bisphosphate into inositol-3-phosphate (InsP3). InsP3 promotes a Ca2+ release from the ER; thus, PrPC reduces Ca2+ release, contributing to neuronal protection. Concurrently, there is an increase in the Ca2+ influx through the plasma membrane by activation of SOCCs and increase in the activity of adenylate cyclase (AC).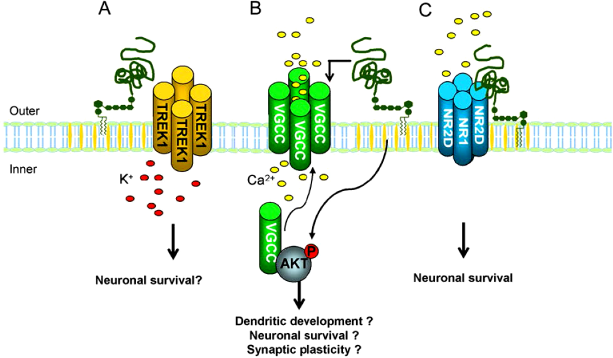
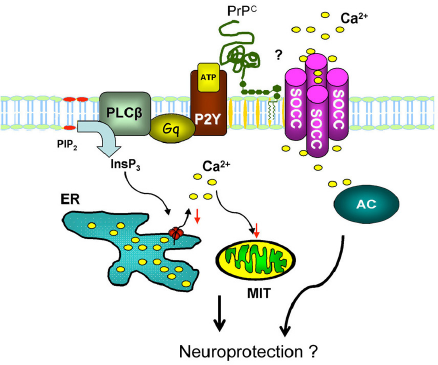
Málaga-Trillo and coworkers 125 have shown that PrPC is essential for cell adhesion and that this occurred through activation of the Src-related Tyr kinase p59fyn and possibly Ca2+ metabolism, leading to the regulation of the trafficking of E-cadherin, a member of surface-expressed CAMs responsible for cell growth and differentiation.
In summary, rather than acting as an explicit single pathway, PrPC has been proposed 112 to act as a sensor within a complex signaling scaffold, activating intracellular signaling cascade networks. PrPC can be seen as acting as a “master controller” in the orchestration of aggregation of proteins at the cellular membrane, with dynamic and complex interactions with multiple ligands in the formation of scaffolding assemblies. PrPC appears to operate as a central protein in a “non-linear” scaffolding system, with each ligand partner coordinating with distinct signaling pathways, which provide coordination with downstream neurotrophic signaling pathways, cytoskeleton modulation, vesicle transport, and communication through ion channels and calcium flux, translating into long-range effects on physiological function. The precise cellular result of the PrPC signal most probably will depend on its cellular setting.
Redox Signaling
One of the earliest and most widely accepted functions of PrPC is the protection that it may afford against oxidative damage. 18 Copper ions are released into the synaptic cleft during neuronal activation, 126 and free Cu2+ can cause an increase in ROS because of redox reactions. PrPC has a high affinity to bind Cu2+ and, hence, limit the formation of ROS. 82 The ability of PrPC to bind copper has thus been suggested as a basis for PrPC function at the synapse 81 where PrPC could transport Cu2+ back to the cell after its release on depolarization. Consistent with this, PrPC-deficient mice showed reduced SOD activity to cope with ROS. 127 As Cu2+, PrPC has been shown to react to H2O2 (an ROS molecule) as a signal, 117 which stimulates a rise in intracellular calcium ion. PrPC has thus been proposed to act as a redox sensor,73,81,116,117 reacting to Cu2+ or ROS, to initiate a signal cascade that, through Fyn kinase, releases cellular calcium ion from ER stores to modulate synaptic transmission, to maintain neuronal activity, and to afford neuroprotection. PrPC also acts to initiate an antioxidant cascade using the glutathione (GSH) oxidant system, which decreases neural sensitivity. 73 If this mechanism is impaired, the result is increased sensitivity of neurons to H2O2, which implicates PrPC as having an important role in neuropathic pain.
In addition to acting as a redox sensor, PrPC is also involved in the regulation of cellular redox equilibrium. 128 The PrPC-Caveolin-1-Fyn pathway induces NADPH oxidase production of ROS,114,115 which triggers the Erk1/2 pathway. 101 It is generally accepted that NADPH oxidase-generated ROS has an important role in signal transduction, particularly in stressed cells. 129
PrPC appears to be central to cellular redox balance and homeostasis. 115 PrPC as a redox modulator may be intimately involved in the regulation of the nervous system at the cell membrane as well as intracellular and ECM. The “harlequin” 3 nature (the biphasic regulation) of PrPC is apparent in its role both as a redox receptor and in the upregulation of the ROS response.
Signaling in the Nervous System
PrPC at the Synapse
PrPC is found ubiquitously in cells, indicating a general cellular function. It is, however, found at higher levels in neurons and is preferentially concentrated at synaptic membrane sites,130,131 mainly presynaptic, and also postsynaptic, where many ion channels are also concentrated. PrPC knockout mice display electrophysiological abnormalities in the cerebellar and hippocampal neurons 132 with decreased neurotransmission function.66,72,74 Progression to prion disease involves loss of synaptic function prior to neural degeneration, 133 and gamma-aminobutyric acid (GABA)A receptors are reduced in some PrPC-null mice (although in others, there may be no change or even an increase). 6 Increased neuronal excitability has been demonstrated with PrPC-null mice, which were more prone than wild-type mice to seizures after administration of a convulsion drug. 134 Re and coworkers 135 showed an increase in acetylcholine release and excitability at the neuromuscular junction. Robinson and coworkers 136 found enhanced synaptic release in Drosophila neuromuscular junction. These factors strongly suggest a role of PrPC at synaptic junctions, in synaptic transmission and neuronal excitability, as does the involvement of PrPC in behavior and memory. 18 This role may be because of involvement in neurotransmitter release (synapsin I and synaptophysin) or because of more general antioxidative or antiapoptotic affects. 82 Some studies of PrPC-null mice have shown lowered LTP at synapses and reduced GABAA receptor, 137 both linked to learning and memory formation. 138 Taken together, these results have been interpreted as demonstrating a modulating effect of PrPC on “neuronal excitability and synaptic activity”. 18 Overall, available evidence suggests that PrPC has a role in modulating neuronal excitability and synaptic activity.
Fournier 131 has summarized the suggested roles for PrPC at the synapse (Fig. 7), including protecting the synapse from oxidative stress (by binding copper ions); scaffolding and endocytosis; signal transduction pathways via activation of (for example) Fyn kinase; neurotransmission by binding to synapsin 1 and synaptophysin; modulation of GABA inhibition and glutamate excitation; and ultimately modulating axonal growth and controlling synaptic plasticity.
PrPC and Memory
The involvement of PrPC mechanisms in the formation of LTM has been proposed by a number of researchers.139,140 The cytoplasmic polyadenylation element-binding (CPEB) protein, found in neurons, has a similar structure to yeast prion protein and acts as a prion when inserted into yeast. In the Californian Sea Hare, CPEB has been shown to be important in synaptic growth and LTM formation.
141
The occurrence of proteins with similar domains to the sea hare CPEB is widespread in eukaryotes and suggests the possibility of prion-based memories being common.
139
Halfmann
32
speculates that the prion switching mechanism has been co-opted by neurons for memory formation. It has been hypothesized
140
that LTM in humans is initiated by electrical stimulation at the synapse, which causes aggregation of PrPC and which holds the synaptic connection together, thus forming the neural circuit associated with the stimulus. The proposal is that the greater the stimulus (traumatic, exciting, etc.), the greater the connection and the long-lasting memory. In support of this, Caiati and coworkers
142
have demonstrated the role of prions in hippocampus plasticity in immature mice. The role of PrPC in memory retention has also been demonstrated in animal models (reviewed by Linden et al).
18
Both LTM and STM have been shown to be influenced by PrPC in hippocampal neurons, via two mechanisms: binding with laminin and binding with hop/STI1.
121
Putative functions of the cellular prion protein (PrPc) at the synapse.
124
(1) Synthesis and anchoring of PrPc in the synaptic membrane. (2) Endocytosis of membrane with PrPc receptors (eg, NCAM, LN, LRP/LR, LRP1, ST1) and ligands, and/or Cu2+. (3) Activation of protein kinases to induce signaling pathways. (4) Interaction with synapsin 1 and synaptophysin in the process of the exoendocytic cycle of synaptic vesicles with effects on glutamate and GABA. (5) Putative soluble PrPC. (6) Postsynaptic PrPC.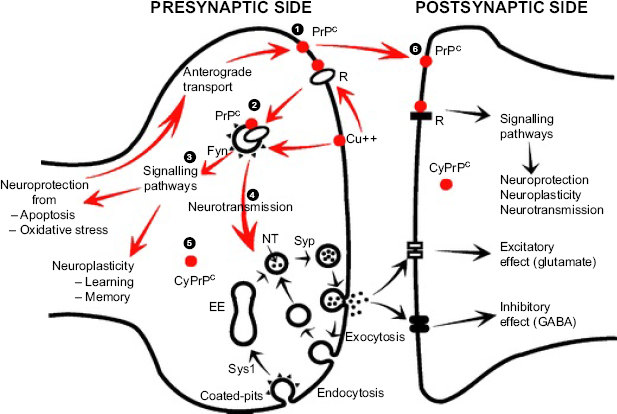
There is also evidence that PrPC has a role in human memory. A change in the amino acid sequence in PRNP was associated with decreased cognitive performance and early cognitive decline in elderly subjects but, conversely, better LTM in healthy young adults. 18 This indicates a strong relationship with this molecular site to cognitive performance.
A β -conformational model of memory formation that involves a functional amyloid has also been proposed, 50 where information (memory) is stored as β-sheet proteins in a prion-like mechanism. The protease resistance of the amyloid ensures the long-term survival of the memory, and the self-propagation of the protein allows transmission to subsequent cell generations.
Long-Range PrPC Signaling
Long-range signaling in the nervous system by PrPC involves neurotrophic activities. Neurotrophic theory is defined by Martins and coworkers 23 as cell-cell communication by cell surface release or presentation of molecules that bind to other molecules present in a target cell. They may be the same cell (autocrine effect), neighboring cell, or distal cell (paracrine effect). These effects depend on the neurotrophic factor and the structure and diffusion properties of the tissue environment.
Retrograde signaling mediated by endosomes has been reviewed by Ibàñez. 143 It is initiated by the binding of nerve growth factor (NGF) to Tyr kinase receptor (TrkA) and subsequent endocytosis of clathrin-coated vesicles containing components of the Ras-mitogen-activated protein kinase (MAPK, phospholipase C-γ, and PI3 kinase pathways).
These endosomes are transported in axons and uncoated in the cytoplasm at their destination. These pathways involve dynein-mediated transport. It is also possible that faster alternative pathways might exist. 143
There is ample evidence of the involvement of PrPC in neurotrophic signaling. 23 Neurotrophic interactions mediated by prions depend on the ability of PrPC to coordinate the assembly of the multi-component scaffold complexes at the cell surface and the endocytosis of these scaffolds. PrPC involvement in neurotrophic signaling has been reviewed by Martins and coworkers, 23 who describe a number of mechanisms for internalization of PrPC and ligand scaffold complexes (Fig. 8). PrPC transport relies on a stable kinesin-dynein assembly to coordinate PrPC clathrin vesicles movement in antegrade and retrograde signaling via microtubules. Prion disease (scrapie) is associated with disruption of assembly of both kinesin and dynein. 61 Erk1/2 signaling induced by ST11 requires the endocytosis of PrPC. 118 This process requires the interaction of ST11 and PrPC, and a functioning kinesin and dynein assembly. 144
There are a number of mechanisms of PrPC signal modulation that operate through neurotrophic activity, including axonal transport, physical transport (involving many signaling molecules and pathways including ATP (purigenic) and chemokines (chemotactic)), 145 and finally, calcium flux and calcium wave transmission through glial transmission. Signal transduction pathways have traditionally been studied in isolation of each other. 143 A more informative approach might be seen as a biosystems approach, integrating prevailing mechanisms. Global SUMOylation, 146 methylation, 147 and the concept of allostasis 148 resulting from biological systems including the hypothalamic-pituitary-adrenal (HPA) axis are examples. Ion channels have been postulated as signal integrators 146 through prion protein modulation of TREK-1 and TRESK (calcineurin level). Prion protein could potentially have a role as a master coordinator of these global responses.
In retrograde transport of neurotrophins, there is a significant role for small p75 receptor (p75NTR). 143 This receptor can bind all the neurotrophins (eg, NGF, brain-derived neurotrophic factor (BDNF)) as well as other ligands. PrPC fragments (106-126) bind with the p75 receptor to act on NADPH oxidase and produce disease. 149 This would imply a role for PrPC in neurotrophic TrkA p38 MAP signaling, involving the induction of TRPC channels in the DRG facilitating heat and cold hyperalgesia. This pathway is important in injury-induced neuropathic pain. 150
The physical separation between axon terminals and their cell bodies can involve relatively long distances to reach downstream affecters in the cell soma (retrograde signaling). The transport of neurotrophic molecules to neuronal bodies has been found to be significant in cell survival responses.
151
These distances also have important implications for pain processing, including chronic pain. The central role of PrPC in long-range neurotrophic signaling may therefore have implications for PrPC in the pain response.
PrPC incorporation into endocytic vesicles. (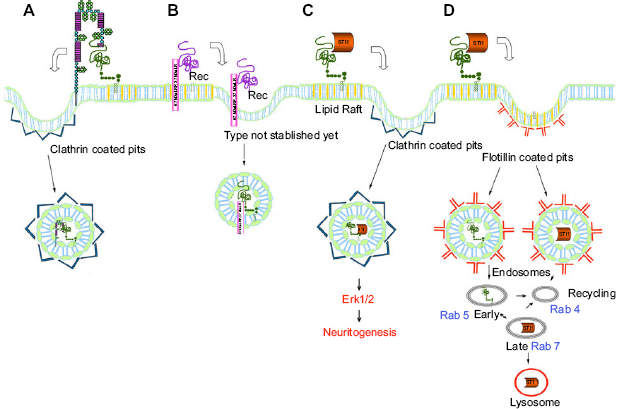
PrPC and Astrocyte Signaling
The PrPC is expressed in glia including astrocytes. 152 Arantes and coworkers 153 have concluded that PrPC has an important role in astrocyte development, morphology, and function. It has a role in astrocyte development via its ligand STI1 and in neuron-astrocyte communication and neuronal survival via its influence on glutamate uptake by astrocytes. It also influences astroglial Na+/K+ ATPase and glutamate. Neurite outgrowth was more prominent in wild-type PrPC astrocytes compared to PrPC-null astrocytes, and null astrocytes showed a more punctate pattern with less fibrillar organization. This is similar to the malformed villi seen in gut epithelial cells 62 and in altered neurite morphology in prion disease. 128
Calcium cytosolic levels are mediated by PrPC via In(1,4,5)P3 and the subsequent release of Ca2+ from ER stores. 154 These Ca2+ variations appear critical for the release of neurotoxic concentrations of the gliotransmitter glutamate, and the regulation of astrocyte signaling glutamate receptors and ATP-activated purigenic receptors (P2Y1). 154 The importance of glutamate toxicity in astrocytes is emphasized by the chronically activated glial cells that surround depositions of PrPSc in prion disease evoked by fragments (106-126).152,154 There is currently no information on the Ca2+ signaling of scrapie-infected astroglia. 154
Dysregulation of the GSH antioxidant cascade initiated by PrPC increases neural sensitivity. 73 If astrocytic glial glutamate transporter 1 (GLT1) is impaired, there will be a build-up of glutamate in synaptic clefts and a resultant neural hyperexcitability and hyperalgesia. 155 PrPC overexpression and disease exhibit the same neural sensitivity.
Given the evidence of PrPC on astrocyte development, together with the role of PrPC in Ca2+ homeostasis and Ca2+ signaling, it would appear that PrPC is involved in glial signaling (glial transmission). Glial communication in astro-cytes is mediated by the gap junction protein connexin 43. Connexin 43 is known to bind to Caveolin-1, 156 which in turn is critical in the signal transduction of PrPC to the Fyn Tyr kinase cascade. 89 Connexin 43 acts to form a functional syncytium between astrocytes through which calcium ion waves can travel. This is described as a “glyapse” by Ren and Dubner, 155 which infers a neuron-glial signaling relationship. They describe this “tetrapartite” synapse between astrocyte, neuron, and microglia as having up to 60 connections. This may be important to glial transmission since recent anatomical studies have shown astrocytes form a polyhedral, three-dimensional tessellated domain with subtended connections of tubulin. 157 This is unique to humans and higher primates, and is associated with long neurite processes in the astrocytes that contain evenly spaced varicosities. Robertson 157 hypothesized that these processes provide an alternative communication pathway across cortical layers that may be involved in consciousness and memory of humans. PrPC may have a significant impact on this process of cell-to-cell communication, since prion disease has a noted and marked impact on memory and cognition. Thus, PrPC may have a role in the putative higher order functions.
There also is a role for the transport of neurotrophic pathways involving calcium waves. This pathway potentially has significance in prion signaling in the central nervous system (CNS). This would involve astrocytes and microglial signaling. Calcium wave oscillation and signaling are rhythmic phenomena relying on specific non-linear feedback processes similar to cAMP oscillations, circadian rhythms, and cycle-related kinases, including p53/mdm2 loop. 158 Understanding this calcium wave propagation and messenger system is important for understanding whole cell oscillatory behavior and emergence from inherent ion channel behavior. This pathway was seen in propagating calcium waves from the growth cone to the axonal soma and was sufficient to change the migration pattern of a neuron. 159 In other studies, the near-infrared of pulsed lasers was found to influence axonal growth trajectory, 160 and neuronal transmission was found to be facilitated by the addition of glutamate in brain axons with a resultant biophotonic neural communication. 161 These studies may imply photonic cell-to-cell communication and indicate a future research area in PrPC, given its role in calcium flux and glutamate release.
PrPC and Circadian Rhythms
PrPC has a significant role in circadian rhythms including REM sleep and sleep-wake patterns.162,163 In addition, PrPC-null mice show disrupted sleep patterns. Insomnia can be a symptom of TSE, 164 and the disease FFI is a prion disease. In the rat brain (suprachi-asmatic nucleus (SCN), cingulate cortex, and parietal and piriform cortexes), mRNA of PrPC is regulated in a circadian rhythm, 162 not only in a light/dark cycle but also when kept in constant darkness. Cagampang and coworkers 162 suggest that expression of PrPC corresponds to clock-related transcripts, which may suggest a role for PrPC. Tobler and coworkers 163 show that a lack of PrPC expression is related to sleep fragmentation, and concluded that PrPC is needed for sleep continuity and that PrPC may be a target for the treatment of sleep disorders. Given that a lack of PrPC is associated with loss of circadian rhythms, this is suggestive as to how PrPC expression (or lack of expression) might affect other daily rhythms, such as calcium and vitamin D metabolites, which show different diurnal acrophases and which are involved in cell-cell synchronization. Any role of PrPC may act through an interaction between PrPC and ganglioside GM1.88,165
Genes involved in sleep regulation include melanocortin genes such as proopiomelanocortin (POMC)-tyrosinase, which also affect breathing rhythms, pain, cardiac regulation, and autonomic response. 166 The daily light-dark cycle is known to be the primary signal that entrains circadian rhythms to the earth's rotation. Changing the circadian rhythm can have effects on health and physiology, such as hormonal secretion, body temperature, retinal neural physiology, and gene expression. 167 Variations in melanocortin genes can affect physiologies such as seasonal affective disorder (SAD). 168 Sleep disorders are connected to a variety of broader health risks including cardiovascular disease, 169 obesity, 170 hypertension, 171 and psychiatric and behavioral disorders, 172 which may suggest a role for PrPC in a number of disparate diseases.
PrPC, Immune System, and Phagocytosis
PrPC is also involved in regulation of the immune system, and in the areas of inflammation, autoimmunity, phagocytosis, and neuroimmunity (reviewed by Linden et al). 18 In this function, the expression of PrPC can be either neuroprotective or neurodegenerative, a contrast described as the “harlequin nature of the prion protein.” 3 PrPC has an important function in the modulation of phagocytosis, showing upregulation of NF-KB with a resultant increase in the downstream signaling cascade, which increases phagocytosis. 11 Soluble PrPC was found to stimulate adherence, phagocytosis, and cytokine production (such as tumor necrosis factor (TNF)-α) of monocytes via activation of Erk1/2 and NF-KB. 11 Levels of prion protein expressed in mouse neutrophils have also been shown to be dependent on HPA axis activation during inflammation events. 173 Interestingly, no other bone marrow cell showed this response. This places the expression of the PRNP gene in the neutrophil under the control of the HPA axis. This demonstrates a role for prion protein in integrating the immune endocrine and nervous systems, and there may be a broader role for PrPC in the immune responses linked to the HPA axis.
Available evidence suggests that PrPC also has a role in T-cell activation, 18 in endocytosis and signal transduction,174,175 as well as in the correct functioning of macrophages. 176 PrPC-deficient mice showed decreased pseudopodium extension in macrophages. 177 It has also been demonstrated that prion-like proteins (such as MAVS) behave as switches for signal transduction to initiate protection from infectious disease, such as viral infections.49,178 Prion-like switches could have both positive and negative effects on the immune system to fend off disease or cause neurodegeneration.
Neuritogenesis and Polarity
Neurons develop from neuronal stem cells through a process of neuritogenesis, where multiple neurites elongate and ultimately form axons and dendrites. This necessitates extensions to the plasma membrane, driven by changes to the cytoskeleton, including F-actin and microtubules. 179 PrPC has been known for some time to be involved in neuritogenesis, neurite growth, and neuronal polarity.18,122 The action of PrPC as a receptor to link the extracellular proteins to cytoskeleton, the resulting signal transduction, allows for the modulation of neurite outgrowth, neuronal survival, and synaptic plasticity. 3 Butowt and coworkers 24 found PrPC to be present in the elongating axons of the embryo, increasing during development before declining postnatally. This suggests a role for PrPC in axon growth. In adults, PrPC remains present in areas of ongoing growth or plasticity, such as the olfactory bulb and the hippocampus. 180
Neuritogenesis relies on a number of pathways to remodulate cytoskeleton during neuritogenesis, including the integrin pathway. 179 This pathway is influenced by PrPC, which interacts with β1 integrins at the membrane 67 to modulate the cytoskeleton. Decreased PrPC leads to increased β1 integrin signaling, which in turn has a number of effects. 122 Turnover of FAs is slowed; the RhoA-ROCK-LIMK1-2cof signaling cascade is activated, which in turn triggers conversion of G-actin into F-actin; there is a build-up of ECM fibronectin, possibly via the CREB signaling pathway, which is also regulated by β1 integrin signaling. All of these lead to a decrease in neuritogenesis. In order to initiate neuritogenesis, FAs need to be assembled with high turnover rates, and both G-actin and F-actin need to be present. Normal level of PrPC influences neuritogenesis by its regulatory effect on β1 integrin, illustrating the dynamic interplay between PrPC and β1 integrin in the regulation of neuritogenesis. This would be important in mechanotransduction in the periphery 181 and the CNS. 182
Other mechanisms are also at play with the effect of PrPC on cytoskeleton modulation. According to Zafar and coworkers, 107 up to 40% of the ligands binding to PrPC are related to cytoskeleton functions, including cell growth (neuritogenesis), differentiation (polarity), and neuron maintenance. PrPC has been shown to bind to the ECM protein vitronectin, which influences axon growth. 85 PrPC-null mice, however, are able to grow axons normally, suggesting that there are other mechanisms that can compensate for the loss of PrPC.
Nieznanski and coworkers96,97,181,184 propose that PrPC has a role in α- and β-tubulin oligomerization and competes with microtubule-stabilizing proteins such as stathmin 98 to modulate microtubule stability. The interaction between PrPC and tubulin has also been demonstrated by others, most notably during cell apoptosis,185–188 with Schmitz and coworkers finding that PrPC-null mice showed reduced numbers of neurons with β-tubulin in the hippocampus. 56 PrPC has also been shown to interact with the tau proteins that stabilize microtubules.189,190
PrPC has been shown to mediate calcium-independent homophilic cell adhesion in embryonic cells 125 via signal transduction pathways that reorganize the actin cytoskeleton and mobilize E-cadherin (a CAM) from vesicles to the plasma membrane. CAMs induce signaling events that interact with the cytoskeleton to regulate neurite outgrowth and polarization. 191
The signal transduction of PrPC via Fyn also has the potential to affect microtubule organization, since Fyn appears to be an important player in microtubule organization, at least in T-cells. 192 The interaction between laminin and STI1 suggests that PrPC also has a role in promoting axon growth in the peripheral nervous system. 193 Thus, it appears that PrPC affects multiple mechanisms that in turn control cytoskeleton modulation and neuritogenesis.
Oxidative stress dysregulation and the effect on neuritogenesis by overexpression of PrPC were demonstrated by Pietri and coworkers 128 using a segment of PrPC (amino acids 106-126). This resulted in oxidative injury to bioaminergic neuronal cells. This was characterized by negative effects on neural epithelial cells, neuronal progenitor cells, and neurotransmitter levels, as well as negative effects on serotonergic and noradrenergic cells with resultant negative effects on their neuritogenesis. This resulted in enlarged neurons, terminal varicosities, increased number of budding vesicles, and shorter stumpier neurites. 128 This is similar to the malformed villi seen in gut epithelial cells 62 and altered astrocytes. 153 This varicosity formation is characteristic of the sympathetic fiber distortion in the DRG in neuropathic pain 194 and the delayed neurite basket-like structures resembling varicosities in the periphery and CNS. 195 The mechanism involved would include recruitment of Caveolin-1-Fyn signaling platforms and overstimulation of the activity of NADPH oxidase. There would also likely be an accompaniment of activation on TRPV1, a redox sensor responding to increased ROS 196 and store-operated Ca2+ entry. 197 This mechanism is important in the acute synovial inflammation that results in TRPV1- mediated cell destruction 198 and points to a role of PrPC in modulating inflammatory joint disease.
PrPC and Body Symmetry
When an embryo consists of three or four cells, it begins the process of producing body symmetry. This is controlled by intracellular Ca2+ concentrations that vary throughout the embryo.181,199 High expression of PrPC in the nervous system of the embryo compared to the adult 180 and the role that PrPC plays in the control of Ca2+ flux implicate PrPC as having a role in the development of symmetry. This is critical for diseases of symmetry, such as Parkinson's disease, cervicogenic headache, 200 transformational headache, 201 and some familial migraines. Parkinson's disease has part of its genesis in polymorphisms of several axonal guidance genes in the embryo, and this genomic difference is predictive of age of onset and absence of adult Parkinson's disease. 202
PrPC Signaling Integration
It appears likely that PrPC may have an influence on a variety of coordinated responses to stimuli. One such response is mechanotransduction 203 where a localized mechanical response can elicit a global mechanical and chemical signaling response. PrPC is known to interact with integrin pathways and thus influence these signaling cascades and cytoskeleton. Integrin pathways are also known to coordinate the response to mechanical stimuli in mechanotransduction, 204 which influences ion channel activation. PrPC also binds to and influences TREK-1 and Ca2+-activated ion channels. 96 Thus, PrPC would seem to be directly involved in mechano-transduction, which may be important in mechanotherapy 203 and mechanobiology of the brain, 182 where ion channels can have a significant effect. Mechanical force is also important in membrane dynamics, cytoskeleton and microtubule modulation, and potentially in microtubule-coded communication. 44 This mechanism would involve posttranslational modifications of tubulin and, hence, the cytoskeleton. Antegrade and retrograde mechanical signaling has been proposed to play a role in information transfer and neural plasticity. 44 As a consequence, PrPC may also be crucial to the signal transduction pathways involved in mechanotransduction, because of its role in microtubule assembly and the function of the cytoskeleton in neurons.
PrPC may also have a role in the global modification of proteins by small ubiquitin-like modifier (SUMO) proteins (or SUMOylation). The SUMOylation of K+ leak channels and nuclear and perinuclear targets produce a global response via NF-kB 205 and PPAR-γ. 206 SUMO 2/3 is also heavily involved in regulating the p53 response to DNA damage by SUMOylation and deSUMOylation, an important process to protect genomic integrity. 207 SUMO/sentrin-specific proteases (SENP) involved in the deSUMOylation of p53 reduce apoptosis and increase the antioxidant SIRT1/SUMOylation. 207 PrPC is a candidate target for SUMOylation because of its involvement with p53, its regulation of ion channels, and redox modulation. Patients with prion disease (s-CJD) showed a consistent decrease in expression of SUMO2/3, 63 which also points to a role of PrPC in SUMOylation, as does the regulation of the myocyte enhancer factor 2 (MEF2) by calcineurin and Ca2+ flux. 208 This is an example of a complex physiological process being tightly regulated by posttranslational modification by a switch that activates or inhibits synapse formation. 209 The role of PrPC in the protein pathways involved in DNA repair is an area of future research.
Three yeast prion proteins (PSI+, URE3, RNQ+) have been implicated in non-DNA, epigenetic inheritance (reviewed by Fowler). 29 That is, the aggregated form of the protein is passed to and persists in daughter cell phenotypes, often demonstrating positive selective characteristics in the rapidly changing environments in which yeasts can find themselves. In humans, PrPC is thought to be involved in the chromatin modification through interaction histone H3. 210 Histone changes have been linked to epigenetic inheritance in humans (reviewed by Greer and Shi) 211 as well as chronic pain. 147
PrPC, Non-Prion Disease, and Potential Treatments
PrPC has a significant role in nervous system signaling, responding to extracellular stimuli by binding to ligands, then coordinating a response to these stimuli by ligation with other proteins in the signal scaffold, and initiating signal cascades. This function makes PrPC a potential therapeutic target, with implications for health and disease beyond the scope of prion disease and its rarer familial and sporadic variants (GSS, FFI, etc.). PrPC involvement in disease may extend to subtle dysfunctions of the protein and its ligands that could be of genetic, epigenetic, or environmental origin. Modulation of PrPC function would, therefore, appear to be a potential treatment intervention. Accordingly, diseases such as insomnia, chronic pain (including headache), chronic inflammation (including autoimmune disease), and neurodegenerative disorders are discussed with reference to PrPC regulation and dysregulation.
PrPC, Neuroendocrine, and Disease
It is increasingly apparent that PrPC has a role in the regulation of the neuroendocrine secretion of the pituitary molecule POMC in an animal model. 212 POMC is also regulated by p53, which is a target of PrPC. Oversecretion of PrPC over long periods resulted in destruction of POMC secretory granules by crinography (lysosome mediated). 212 POMC is a precursor molecule in the formation of melanin and hormones ACTH, αMSH (an inhibitor of NF-kB), β-opioid, and thyroid; and is therefore involved in energy homeostasis, autonomic regulation, pain regulation, and the pain and anesthetic response of red-headed women. 213 Importantly, αMSH is an inhibitor of NF-kB, which may also be upregulated by PrPC via ROS signaling. Taken together, these evidences point to a role for PrPC in melanocortin-inspired disease. As p53 can be activated by various stressors, including sun exposure, inflammation, and aging, this may implicate PrPC as having a role in such diverse disease responses as inflammation (including asthma), 214 energy and weight homeostasis, 18 recovery from brain injury, 59 and myopathy. 215 Further, PrPC may also be involved in other melanocortin diseases such as collagen-related disease (of the eye), 216 pigmented collagen disease of the synovium (involving p53 regulation of monocytes), 217 and thyroid disease. 218 The link between PrPC, αMSH, and NF-kB suggests that the neuroprotective role of PrPC could play a part in the anesthetic response of elderly patients who suffer postanesthetic dementia (Alzheimer's disease),219,220 possibly involving the role of PrPC in cytoskeleton organization,21,122 an important focus for further investigation.
Another possibility in relation to the interaction between PrPC and the melanocortin system (as hypothesized by Hernandez) 116 is the close proximity on chromosome 20 (in humans) of the PrPC gene to critical pigmentation genes, including genes for agouti signaling protein (ASIP), attractin (ATRN), and melanocortin 3 (MC3) neural anti-inflammatory receptor. 221 This proximity may link pigmentation to regulation of PrPC and hence to prion disease in animals such sheep and rodents. If extended to humans, this would point to an interrelationship between PrPC gene expression and PrPC-regulated disease, especially given the effect of the temporary disruption of the cytoskeleton during general anesthesia 222 and the role of PrPC interacting with MAPs and microtubule assembly and disassembly.
In addition to the two mechanisms for anesthesia vulnerability discussed above, a third area of vulnerability to anesthetically induced neurodegeneration related to PrPC function could be proposed. It has been demonstrated that there is a link between protein 14-3-3, calcineurin, and PAR-1/MARK pathway,223,224 which results in the coupling of microtubule dynamics and neuronal excitability through TRESK channels. 225 TRESK is the ion channel most sensitive to anesthetics such as halothane and isoflurane, mediating the suppression of wakefulness, awareness, and memory. 226 PrPC has also been linked to protein 14-3-3 100 and calcineurin B 83 giving a further link (via TRESK channels) to cytoskeleton dynamics. Thus, it might be expected that PrPC would have a role in postanesthetic disease vulnerability. Implications for treatment include the screening of patients undergoing anesthesia for TRESK polymorphisms and the targeting of PrPC for neuroprotection in relation to anesthetic-induced disease.
PrPC and Neurodegenerative Disease
There are clear common features between prion diseases and amyloid neurodegenerative diseases (Alzheimer's disease, Parkinson's disease, Huntington's disease). They share neuropathic symptoms such as synaptic dysfunction, neuron loss, and neuropeptide signaling disintegration. The diseases are all progressive conformational diseases, which involve the misfolding of proteins from the native form to an amyloid-like β-form that accumulate (aggregates) in the CNS as amyloid fibrils. All may have a genetic component, and all show similarities in gene expression pathway changes during the disease process, most commonly, the MAPK/Erk1/2 signaling pathway. 227 It is likely that progression to neurodegenerative disease may (in part) be because of a failure in the chaperone-mediated UPR. 228 It has also been established that (at least in the case of Alzheimer's disease in mice) 229 the amyloid disease can be experimentally transmitted via amyloid protein.
Initiation of the conformational disease depends on a switch from the native state of proteins to the misfolded state. Triggers for this switching are largely unknown, but Iram and Naeem
230
have reviewed the five most important aggregation mechanisms, viz,
reversible aggregation of protein monomers, transient conformational change leading to aggregation, chemical modification (eg, oxidation, proteolysis) leading to aggregation, nucleation-induced aggregation, where the “seed” monomer eventually reaches a critical size and can then rapidly accumulate further monomers (as in progressive prion and amyloid disease), and aggregation initiated by a surface-changing protein conformation.
Liu and Zhao 231 have used an algorithm to predict regions of various proteins that might correspond to “switching regions”. They confirmed the five PrPC regions reported by Kuwata and coworkers 232 as potential sites for the conformational change of PrPC to PrPSc. The switching mechanism and the switching regions of proteins are obvious targets for intervention and therapy, particularly before the protein attains its permanent irreversible conformation, prior to frank disease onset. The implications for drug therapy of conformational disease to circumvent or reverse the misfolding have been discussed recently, 233 as has the potential role of low-level laser therapy (LLLT). 234
Once established, treatment to reverse the protein misfolding in neurodegenerative diseases has proved very difficult, although a number of targeted drugs may eventually be found to be effective235,236 as may immunotherapy.228,237 Recently, laser has been shown to detect insulin fibrils and the fibrils of Alzheimer's disease and Parkinson's disease. The authors attribute the photon absorption to the juxtaposition of aromatic amino acids in the fibril and also suggest that laser may remove the amyloid. 238 Laser has been shown to be effective in treatment of Alzheimer's disease in the mouse model239,240 and in tissue culture, 241 and is thought to have potential for treatment of human-misfolded protein diseases. 242
The relationship between PrPC and both Parkinson's disease and Alzheimer's disease is increasingly becoming a target for research, as reviewed by Prusiner 243 and Chrobak and Adamek. 244 PrPC has been shown to act as a receptor to the amyloid beta (Aβ) protein of Alzheimer's disease, which suggests a synergy between the two proteins. PrPC appears to be neuroprotective against the build-up of α-synuclein, β-amyloid, and tau plaques, 245 possibly as a result of its influence on phagocytosis and neuroregulation. 11 PrPC has also recently been suggested as a therapeutic agent in the treatment of Alzheimer's disease.246,247 The relationship between PrPC and Alzheimer's disease has been the subject of a number of recent reviews.132,248,249
PrPC and Diseases of Asymmetry
Diseases involving asymmetry are good candidates for research into the role of PrPC in disease. These would include Parkinson's disease, 202 asymmetrical headache 200 (including migraine with aura and cervicogenic headache), complex regional pain syndrome (CRPS), 250 and posttraumatic stress disorder (PTSD), which involve asymmetrical amygdala volume (right larger than left). 251
Genetic polymorphisms of a set of axonal guidance and nuclear import genes are predictive of age of onset, and presence and absence of Parkinson's disease. The role of PrPC in axonal guidance and proliferation is well established. 67 Application of a redox-modulating intracranial laser device has been shown to be effective in neuroprotection against Parkinson's disease in an animal model. 252 A similar mechanism may be in place in humans, and testing for this gene may point to preventative strategies against Parkinson's disease.
During phagocytosis, there is a release of near-infrared wavelengths 400-820 nm. 253 These wavelengths have been found to be neuroprotective when applied to the brain in the prevention of Parkinson's disease in an animal model. 252 In addition, neurogenesis has been demonstrated when exogenous pulsing photons (800 nm) are applied to an axon, which may mimic the near-light emitted by cells as communication from nearby cells. 160 The neurogenesis is also influenced by membrane-anchored proteins (axonal guidance by PrPC). Upregulation of phagocytosis by PrPC would result in an increase in endogenous photons. Exogenous photons applied to other body areas, apart from the brain, in the same animal model 252 result in neuroprotection through an abscopal effect. 234 Interestingly, asymmetric disease is associated with abnormal left-right symmetry of global photon emission from the body. 254
PrPC and Pain
Prion disease is accompanied by neural sensitivity and decreased resistance to environmental stress, which may indicate a role for PrPC in regulation of chronic pain syndromes. PrPC is expressed in many cells of the neuroimmune network, 18 including lymphocytes and macrophages, peripheral axons, sympathetic C fibers and ganglia, 20 gut epithelial tissue; 62 and in the CNS, including the hippocampus, thalamus, and cortex (particularly, SCN and cingulate cortex, which are involved in sleep diurnal rhythms and cognition). Since chronic pain is considered to be a dysregulated immune response,255,256 there exists a potential role for PrPC in the modulation of chronic pain that could include chronic sympathetically mediated pain syndromes such as neuropathic pain, cervicogenic headache, migraine with aura, and CPRS. 257
Chronic pain is a widespread problem affecting quality of life and productivity. For example, cervicogenic headache, a subset of sympathetically mediated chronic pain, is a major burden in treatment clinics and daily life activities, 200 because of the lack of responsiveness (25% of cases) to classical treatment including medication, surgery, and physiotherapy.258,259 TRESK polymorphisms have been recently found to be involved in headaches 260 and response to anesthetics, 226 and TREK-1 polymorphisms have been found to be involved in polymodal pain syndromes. 261 The interaction of PrPC on TREK-1, VGCC, and TRESK, through the interactions of calcineurin 83 and Ca2+ flux, 23 means that PrPC may have a role in K+ channel regulation and is thus a potential treatment target. The ability of the organism to compensate and adapt to severe ion channel dysfunction gives a role for PrPC to be involved in compensatory pathways.
The implications for treatment of chronic and unresponsive pain that would evoke a PrPC response and that may modulate prion function would include treatments that change the redox status including drugs and light energy. For example, LLLT affects the oxidant/antioxidant balance through intracellular signaling cascade (reviewed by Wu and Xing). 262 Photons are absorbed by the cytochrome-c-oxidase and increase ROS in the mitochondria and the cytoplasm, leading to signal transduction via NF-kB and the Erk1/2 and the PI3K/Akt pathways. 262
PrPC has a role in modulation of the cytoskeleton, through interactions with integrins, stathmins, and tubulins (see above). Morel has also shown that overexpression of PrPC results in disruption of microtubule architecture and the consequent shortening of intestinal villi and the homeostasis of epithelial renewal. Pietri and coworkers 128 found that overexpression of PrPC (106-126) in serotonergic and norad-renergic neurons resulted in altered neurite extensions with increased budding vesicles and changes to the cell body shape with contorted swellings that resembled varicosities. In neuropathic injury (in an animal model), there is a disruption of cytoskeleton structure in the dorsal root ganglion, with the formation of sympathetic varicosities, which is important as the mechanism behind neuropathic pain behaviors. 194 This is a result of abnormal communication between sensory neurons and sympathetic fibers in the DRG. Therapeutic interventions aimed at restoring homeostasis in cytoskeleton architecture, such as LLLT, are becoming increasingly important in the treatment of neuropathic pain where microtubule disruption causes reversible varicosity formation and provides relief from chronic pain.263,264
PrPC and Insomnia
One of the characteristics of prion disease is insomnia, and FFI is one of the TSE variant diseases. This suggests a role for PrPC in regulating sleep and disruption of diurnal rhythms such as SAD. Melanocortin and p53 pathway regulation are involved in insomnia, 166 which is characterized by a maladaptive stress response (see above). As PrPC is known to interact with p53 and melanocortin and (more broadly) is thought to modulate the cellular stress response, this points to a mechanism for PrPC in the regulation of sleep. A decrease in GABAergic inhibition has been considered by Palagini and coworkers 170 as contributing to insomnia. Fournier 131 has found a role for PrPC in the synapse to regulate GABA release. Thus, there is likely to be a role for PrPC in sleep homeostatic mechanisms, particularly in vulnerable populations, including vulnerability to SAD. 168 In terms of treatment, light therapy has proved effective in both SAD 168 and depressive disorders. 242
PrPC and other Diseases
Owing to its signal transduction with multiple pathways, PrPC may have involvement in a number of other disparate diseases, including:
Diseases involving collagen, which would include hypermobility diseases (encompassing joints and nerve impairment),
265
muscle and tendon repair, and chronic muscle and tendon diseases because of PrPC involvement in β1 integrin pathway signal transduction
67
—mechanotransduction pathways. Treatment implications involve mech-anotherapy
203
and, possibly, regulation of mesenchymal stem cell activity.78,266,267 Autoimmune diseases, including diseases of Tyr kinase Lyn
268
(eg, systemic lupus erythematosus, rheumatoid arthritis) through the regulation of purigenic signaling
23
by PrPC. Other neurodegenerative diseases including schizophrenia and bipolar disorder105,269 through PrPC action on Ca2+ homeostasis.
Conclusion
PrPC is an enigmatic protein. Although originally described in terms of infective disease, the extent of its myriad functions is becoming increasing evident, especially in the nervous system, where it is essential for correct system function. PrPC interacts with multiple partners to act on a number of targets simultaneously. PrPC is seen as a master controller of cellular signaling, acting as a receptor and scaffold for multiple ligands and initiating a number of signal transduction pathways. It has been proposed to have synaptic, gap junction and short-range signaling functions including redox modulation and calcium ion flux homeostasis, as well as a role in neurotrophic, purigenic, and chemotactic signaling. As this review indicates, knowledge of the role of PrPC in physiology and disease will become increasingly important in the design of novel treatments. The relationship of prion protein to other amyloid forming proteins and its potential involvement in diseases disparate from prion disease itself, possibly involving the conformational switching and mis-folding of proteins, opens up a potential area of research for targeted therapies such as drug therapies and LLLT.231,233,234 As an extension of our current work, we have further identified some conditions as having a potential PrPC involvement, including chronic pain, neurodegeneration, inflammation, and autoimmune disease. The wide range of interactions in which PrPC participates suggests multiple targets for therapeutic interventions, including treatments aimed at the PrPC interaction with redox potential mechanisms, Ca2+ flux, and cytoskeleton modulation.
Author Contributions
Conceived and organized the review: AL. Wrote the first draft of the manuscript: AL, BB. Contributed to writing the manuscript: AL, BB, RA. Agree with manuscript results and conclusions: AL, BB, RA. Made critical revisions and approved final version: AL, BB, RA. All authors reviewed and approved of the final manuscript.
