Abstract
The blood—brain barrier (BBB) facilitates amyloid-β (Aβ) exchange between the blood and the brain. Here, we found that the cellular prion protein (PrPc), a putative receptor implicated in mediating Aβ neurotoxicity in Alzheimer's disease (AD), participates in Aβ transcytosis across the BBB. Using an
Introduction
Alzheimer's disease (AD) is an incurable neurodegenerative disorder that is characterized by the accumulation of neurotoxic amyloid-β (Aβ) peptides in the brain and intraneuronal hyperphosphorylation of the cytoskeletal protein tau (Selkoe, 2011). Clinically, these cerebral manifestations result in memory impairment. Recent evidence suggests that the cellular prion protein (PrPc) can mediate Aβ1–42 oligomer-induced impairment of synaptic function and, furthermore, is required for spatial learning and memory deficits observed in an AD transgenic mouse model (Gimbel et al, 2010; Lauren et al, 2009). However, these results were challenged recently (Balducci et al, 2010).
Cellular prion protein is a glycosyl phosphatidylinositol (GPI)-anchored cell surface protein. Hence, it might require a transmembrane coreceptor to transduce signals into the cell that finally cause Aβ-induced neurodegenerative phenotypes (Lauren et al, 2009). To date this coreceptor has not been identified but a previous report showed that the low density lipoprotein receptor-related protein 1 (LRP1) is capable of internalizing PrPc (Taylor and Hooper, 2007). In addition, LRP1 participates in Aβ1–40 clearance across the blood—brain barrier (BBB) by means of transcytosis (Pflanzner et al, 2011; Shibata et al, 2000; Zlokovic, 2008). Interestingly, LRP1 binds Aβ1–40 with a much higher affinity than Aβ1–42, resulting in greater LRP1-dependent Aβ1–40 internalization rates in endothelial cells (ECs) (Deane et al, 2004). Based on the observation that Aβ binds to PrPc, we set out to determine if LRP1's ability to internalize PrPc has a pathophysiological role in AD pathogenesis at the BBB. By using an
Materials and methods
Primary Mouse Brain Capillary Endothelial Cell Transwell Transport Model
Isolation and cultivation of pMBCECs have been previously described (Pflanzner et al, 2011), and are provided as Supplementary Information.
Amyloid-β transcytosis across pMBCEC monolayers was investigated after inducing high transendothelial electrical resistance. Then, 0.1 nM [125I]-Aβ1–40 and 1 μCi/mL [14C]-inulin (both from Perkin-Elmer, Rodgau, Germany), the latter a marker for paracellular diffusion, were diluted in serum free DMEM/Ham's F12 medium (Gibco, Darmstadt, Germany) + 550 nM hydrocortisone (Sigma, Schnelldorf, Germany) + 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (Lonza, Cologne, Germany) and added to the donor compartment. To inhibit Aβ binding to PrPc, mouse monoclonal anti-PrPc (Ab51.2) hybridoma supernatant was dialyzed against phosphate-buffered saline (PBS) over night at 4°C and used at a final concentration of 2.25 μg/mL. For details on the generation and specificity of monoclonal 51.2 antibody detecting PrPc, please refer to Supplementary Information. Receptor-associated protein (RAP) was expressed in bacteria as a fusion protein with GST (glutathione S-transferase) and was purified as previously described (Martin et al, 2008). RAP-GST, or GST as control, was used at 500 nM to block Aβ binding to LRP1. Amyloid-β transport was studied for 2 hours in the brain to blood and for 6 hours in the blood to brain direction. From each input and at each time point, 10 and 50 μL samples were taken from the compartment the transport was investigated to (acceptor). In all, 10 μL probes were counted on a Wallac Wizard2 2470 automatic γ-counter (Perkin-Elmer) for [125I], or on a Tri-Carb 2800 TR Liquid Scintillation Analyzer (Perkin-Elmer) for [14C]. To investigate the amount of intact [125I]-Aβ1–40 being transported, 50 μL of a 15% trichloroacetic acid solution was added to a 50 μL media sample and incubated for 10 minutes at 4°C before samples were centrifuged at 10,000
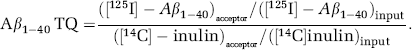
Results represent the mean + s.e.m. of three independent experiments in triplicate.
Coimmunoprecipitation, Sodium Dodecylsulfate-Polyacrylamide Gel, and Western Blot Analysis
For detailed information, please see Supplementary Information.
[125I]-Aβ1–40 Binding Study
Recombinant PrPc (rPrPc) was synthesized as previously described (Leliveld et al, 2008). To study Aβ binding to rPrPc, 96-well plates were coated with the indicated rPrPc concentrations in PBS over night at 4°C on a shaker. Plates were transferred to 37°C for 30 minutes and washed twice with ice-cold PBS. Unspecific binding sites were blocked with 3% bovine serum albumin (Sigma) in PBS (w/v) for 30 minutes at 37°C on a shaker and, subsequently washed thrice with ice-cold PBS. Coated wells were incubated with 0.1 nM [125I]-Aβ1–40 (Perkin-Elmer) in PBS for 30 minutes at 37°C to allow Aβ binding to rPrPc. After five washes with ice-cold PBS, proteins were dissociated by two washes with 0.2 N NaOH for 5 minutes, collected, and recovered [125I]-Aβ1–40 was counted on a Tri-Carb 2800 TR Liquid Scintillation Analyzer (Perkin-Elmer). Counts were normalized to input and expressed in pmol. Results represent the mean ± s.e.m. of three independent experiments in duplicate.
Statistics
All graphs and statistical analyses were prepared using GraphPad Prism 4 software (La Jolla, CA, USA). Data were analyzed by one-way analysis of variance coupled to Newman—Keuls posttest for multiple comparison or
Results
To determine whether PrPc participates in Aβ transcytosis across the BBB, we investigated the transport of [125I]-Aβ1–40 across an
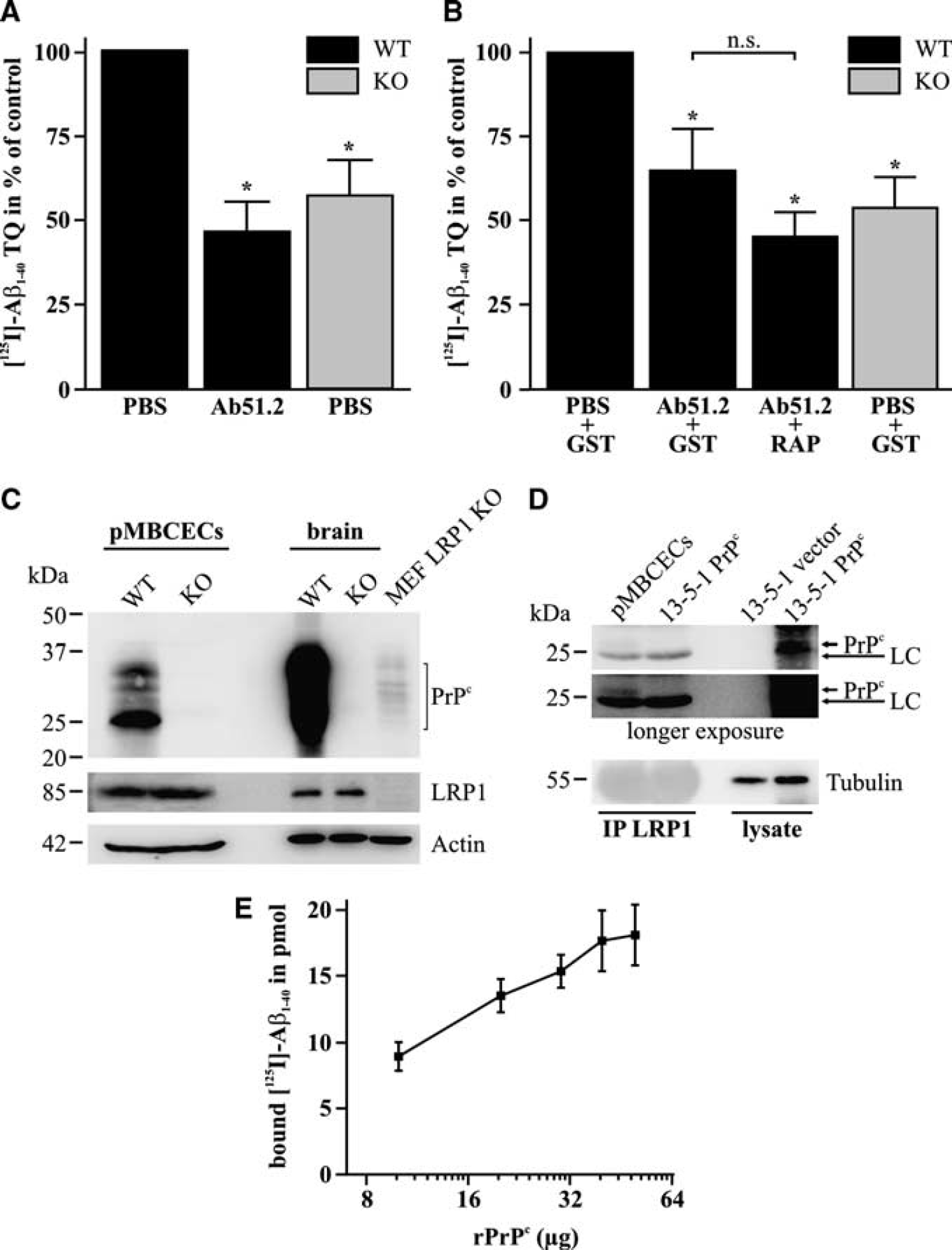
Cellular prion protein (PrPc) enables amyloid-β (Aβ) endocytosis and transcytosis across the blood—brain barrier. (
Discussion
The BBB has an important role in the pathogenesis of AD by governing bidirectional transcytosis of neuro toxic Aβ, thereby, maintaining an Aβ equilibrium that is likely critical to prevent accumulation and aggregation of Aβ species in the brain. Several transmembrane receptors and transporters have been identified to have a role in Aβ transport mechanisms at the BBB (Pflanzner et al, 2010; Zlokovic, 2008), emphasizing the complexity and necessity of these exchanges.
The PrPc belongs to the family of GPI-anchored cell surface proteins. Its role in mediating Aβ oligomer-induced neurotoxicity is not fully resolved due to the lack of intracellular motifs that enable signal transduction into the cell. Hence, it has been suggested that PrPc requires a transmembrane coreceptor to elicit, for example, synaptic dysfunction on Aβ oligomer binding to PrPc (Lauren et al, 2009). To date, the low density LRP1 is the only transmembrane protein that has been shown to mediate PrPc internalization through clathrin-coated pits through interactions between the extracellular domains of both receptors (Shyng et al, 1994; Taylor and Hooper, 2007). LRP1 endocytosis is facilitated through its cytosolic NPxYxxL motif (Li et al, 2000; Pflanzner et al, 2011), a highly conserved domain that also functions in signal transduction (Martin et al, 2008).
Genetic evidence supports that LRP1 mediates Aβ1–40 transcytosis across the BBB (Pflanzner et al, 2011). Due to the fact that LRP1 is required for PrPc endocytosis (Taylor and Hooper, 2007), we investigated whether PrPc is involved in Aβ1–40 transport across the BBB. Strikingly, on genetic deletion of PrPc, we found that Aβ transcytosis in the brain to blood direction was reduced by ~50% in our primary
Since PrPc does not contain intracellular motifs that facilitate receptor internalization, we investigated whether PrPc facilitated Aβ1–40 transport across the BBB might be coupled to LRP1-mediated Aβ1–40 transcytosis. We have previously shown that inhibition of ligand binding to LRP1 with RAP or knockin mutation of the NPxYxxL endocytosis motif reduces LRP1-mediated Aβ1–40 transcytosis across the BBB by ~50% (Pflanzner et al, 2011). Assuming that Aβ transcytosis by LRP1 and PrPc occur independent of each other, RAP treatment should have a clear additional effect on Aβ transcytosis compared with PrPc antibody alone. However, RAP did not further decrease Aβ transport compared with anti-PrPc alone (Figure 1B). In line with this observation and LRP1's ability to internalize PrPc (Taylor and Hooper, 2007), we therefore hypothesize that Aβ1–40 transcytosis across the BBB could occur through a mechanism in which both receptors cooperate to (1) increase the amount of available Aβ-binding sites and (2) compensate for the lack of a functional endocytosis motif within PrPc, to enhance transcytosis efficiency. RAP treatment has been shown to interfere with both LRP1-mediated Aβ transcytosis and PrPc endocytosis through clathrin-coated pits in separate studies (Pflanzner et al, 2011; Taylor and Hooper, 2007). Taken together, these findings suggest that the role of PrPc in Aβ transcytosis across the BBB is potentially linked to LRP1. This is further emphasized by the fact that LRP1 coimmunoprecipitates with 27 kDa PrPc
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
