Abstract
The cellular prion protein (PrPc) is a highly conserved glycoprotein with a still enigmatic physiological function. It is mainly expressed in the central nervous system but accumulating data suggest that PrPc is also found in a broad spectrum of non-neuronal tissue. Here we investigated the cell-type-related PrPc expression in the bovine mammary gland by using immunohistochemistry (IHC), ELISA, Western blot, and real-time RT-PCR. Specific immunostaining of serial sections revealed that PrPc is selectively localized in mammary gland epithelial cells. Particularly strong expression was found at the basolateral surface of those cells showing active secretion. Results obtained by RT-PCR and ELISA complemented IHC findings. No correlation was found between the level of PrPc expression and other parameters such as age of the animals under study or stage of lactation.
T
Materials and Methods
A total of nine adult German Fleckvieh cows ranging from 24 months to 11 years of age were studied. Lactation stage included early and late phases. Immediately after death, mammary gland tissue was removed, prepared, and stored for IHC, Western blot, ELISA, and RT-PCR.
IHC
Tissue samples were immersion fixed in Bouin's solution for 48 hr or with methanol/glacial acetic acid 2/1 v/v for 24 hr. The tissue was then dehydrated in a graded series of ethanol, cleared in xylene, and embedded in paraffin wax using conventional procedures. Five-μm serial sections were cut on a Leitz (Oberkochen, Germany) microtome and mounted on gelatin/chromalaun-coated glass slides. Deparaffinized sections were heated in a microwave oven in 10 mM sodium citrate buffer, pH 6.0 (4 × 5 min at 600 W) to expose antigenic sites. Sections were then rinsed three times in PBS, pH 7.4, and incubated with 1% hydrogen peroxide in doubledistilled water for 10 min at room temperature to inhibit endogenous peroxidase activity. After rinsing in PBS, specimens were treated with 10% normal rabbit serum-blocking solution (Dako; Hamburg, Germany) for 30 min at room temperature. Overnight incubation in a humid chamber at 4C with a PrPc-specific monoclonal antibody (MAb) (L42, diluted 1:50 in PBS) directed against amino acids 145-163 of the ovine PrPc followed.
Specificity and cross-reactivity with bovine species has been shown by previous studies (Harmeyer et al. 1998). On the following day, slides were rinsed and treated with biotinylated rabbit-anti-mouse immunoglobulin (diluted 1:400 in PBS) for 30 min at room temperature (Dako). After a final wash and incubation with streptavidin-biotin-horseradish-peroxidase complex for 30 min at room temperature (Strept-ABC kit; Dako), the reaction product was visualized by using 3,3î diaminobenzidine tetrahydrochloride as a chromogen (Biotrend Chemicals; Cologne, Germany). For simultaneous expression studies, serial sections were probed with either anti-PrPc (1:50) or anti-α-smooth muscle actin (α-SMA) 1:1000 (Dako) antibodies. Finally, sections were counterstained in Mayer's hematoxylin, dehydrated, cleared, and mounted with DePeX (Serva; Heidelberg, Germany). Examination was performed using a brightfield light microscope (DMRBE; Leica, Wetzlar, Germany) and a computer-coupled video documentation system. IHC controls were performed by (a) replacement of the primary antibody with an irrelevant MAb of the same immunoglobulin class, (b) omission of the secondary antibody, and (c) incubation with DAB reagent alone to exclude the possibility of non-suppressed endogenous peroxidase activity. In all control experiments no specific staining was observed. Additionally, sections of bovine spinal cord and dorsal root ganglia were processed in parallel as positive controls for PrPc immunostaining.
RT-PCR
Material for RNA extraction was immediately transferred into RNA Later reagent (Qiagen; Hilden, Germany) and subsequently frozen to −20C. Total RNA was extracted by singlestep liquid preparation procedure using peqGOLD TriFast (PeqLab; Erlangen, Germany). After RNA yield determination in a Biostep Photometer (Eppendorf; Hamburg, Germany), a constant amount of 500 ng total RNA was reversely transcribed to cDNA. Random hexamer-based reverse transcription was performed using 200 U of Moloney murine leukemia virus (MMLV) reverse transcriptase (Promega; Madison, WI) according to the manufacturer's recommendations. Light-Cycler (Roche Diagnostics; Basel, Switzerland)-based realtime PCR was carried out in a 10-μl reaction volume using 12.5 ng cDNA. Commercially available LightCycler FastStart MasterPlus SYBR Green I Kit (Roche Diagnostics) was applied according to the manufacturer's instructions. Primer sequences for PrP amplification were as follows: PrP forward 5îAAC CAA GTG TAC TAC AGG CCA and PrP reverse 5îAAG AGA TGA GGA GGA TCA CAG.
Crossing point (CP) of each sample was determined by LightCycler software 4.0 according to the second derivative maximum method. In this method, the second derivative maximum within the exponential phase of the PCR is linearly related to the initial amount of target, where a low CP reflects high expression levels and vice versa. Therefore, CPs computed by the LightCycler software were subtracted from the maximum number of cycles (i.e., 35) performed. Relative mRNA levels can also be computed as x-fold expression calculated by E = 2ΔCP, where E is the x-fold expression, 2 is the efficacy of the PCR, and ΔCP is the difference of CPs between high- and low-expressing animals.
Data in Figure 1 are computed on the basis of relative CPs; therefore, the exponential character of x-fold regulation is not depicted. Data achieved by LightCycler analysis and by ELISA were statistically processed in Sigma Plot and Excel (SPSS Software; Chicago, IL and Microsoft Software; Redmond, WA).
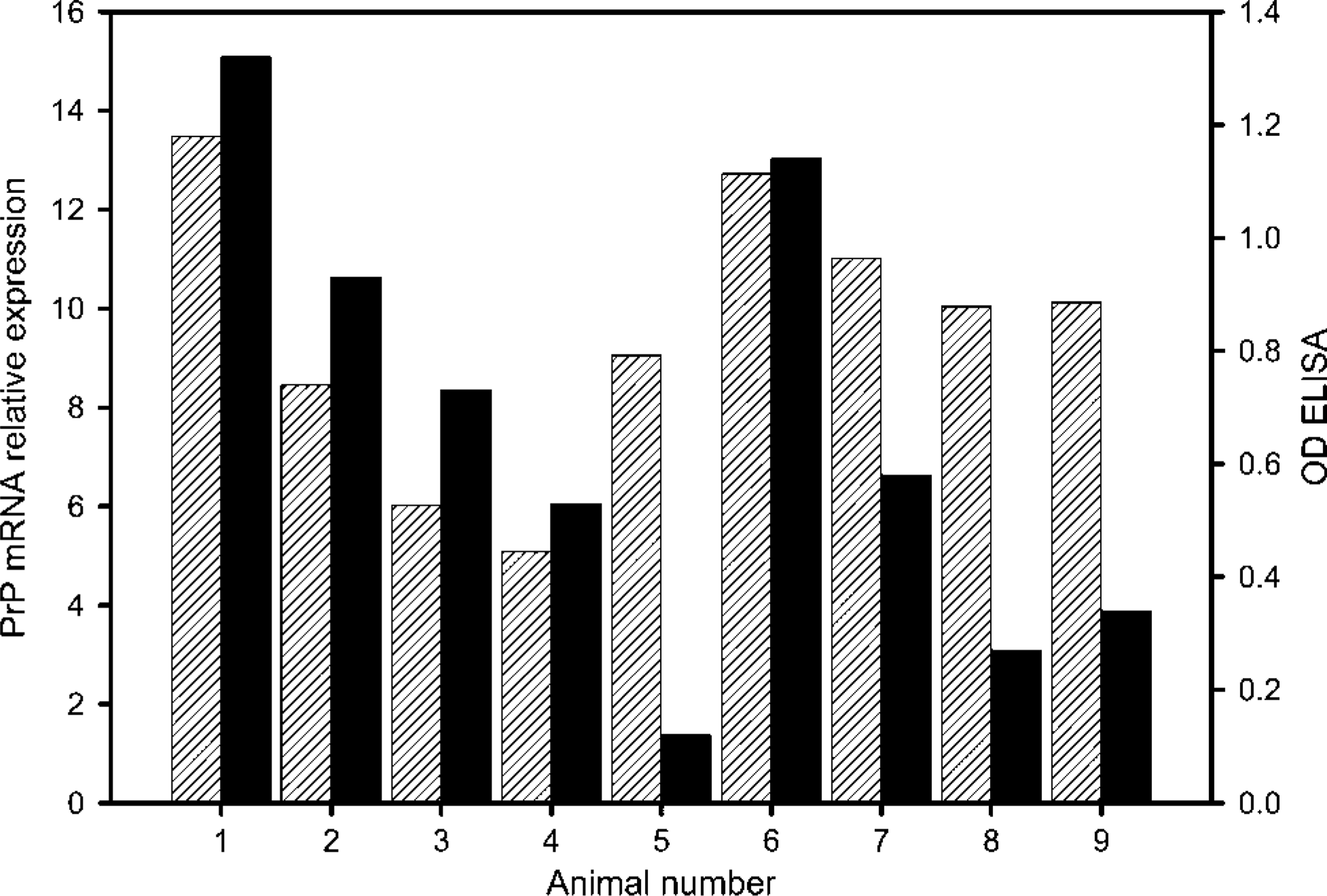
Relative reactivity of mammary gland tissues of different animals analyzed by RT-PCR or ELISA. PrPc mRNA expression, white streaked bars; optical density (OD) ELISA, black bars. RT-PCR data are computed on the basis of relative crossing points.
ELISA
Samples collected for protein detection in ELISA and Western blot were stored at −20C. To homogenize the samples, 400 mg of mammary gland tissue was mixed in 1.2 ml of 3% glucose-PBS solution (w/v) for 45 sec at 6.5 m/sec in a Ribolyzer (Hybaid; Teddington, UK). For the specific detection of PrPc, microtiter plates (Nunc; Wiesbaden, Germany) were coated overnight with 100 μl (5 μg/ml) MAb P4 (R-Biopharm; Darmstadt, Germany) at room temperature. Plates were then blocked with 3% fetal calf serum in PBS for 30 min at ambient temperature. After a washing step, serial dilutions of homogenized tissue samples (50 μl/well) were added with 50 μl of biotinylated V5B2 antibody (5 μg/ml; R-Biopharm) and incubated for 60 min at 37C in a humidified atmosphere. Subsequently, plates were washed four times and further incubated with 100 μl ExtrAvidin-HRP (Sigma; Mannheim, Germany) at room temperature in a humidified chamber for 1 hr. After a washing step, 100 μl of substrate-chromogen solution (1 mM 3,3î,5,5î-tetramethyl-benzidine and 3 mM H2O2 per liter potassium citrate buffer, pH 3.9) was added to each sample and incubated for 20 min in the dark. Color development was stopped by adding 100 μl of H2SO4. Optical density (OD) was determined on a Sunrise ELISA reader (Tecan; Crailsheim, Germany) at 450 nm.
Western Blot
Before Western blot analysis, tissue homogenates from cattle and sheep mammary gland prepared in the same way as for ELISA were boiled in the appropriate amount of XT sample buffer (BioRad Laboratories; Hercules, CA) and reducing agent (BioRad) for 5 min. An aliquot (20 μl) of each sample was loaded onto the gel. Brain homogenates of healthy cows served as positive controls but were diluted 1:4 prior to loading onto the gel. Brain homogenate from sheep was diluted 1:20. Proteinase K (40 μg/ml; Sigma-Aldrich) sensitivity of PrP was tested by digestion of samples for 10 min at 37C and subsequent inactivation at 99C. SDS-PAGE was performed on 12% polyacrylamide Criterion XT Gels (BioRad) using MOPS running buffer.
Proteins were then transferred to a polyvinylidene difluoride membrane, Immobilon-P (Millipore; Bedford, MA). Blocking was achieved by overnight incubation of the membrane in 3% casein-PBS containing 0.02% Tween-20 at 4C. Then, biotinylated MAb P4 (5 μg/ml in the case of bovine mammary gland and 2.5 μg/ml in the case of ovine mammary gland) was added for 1 hr at room temperature followed by ExtrAvidin-HRP (Sigma Aldrich) for an additional hour. Chemiluminescence signals were recorded on KODAK imager (Eastman Kodak; Rochester, NY) after application of Super Signal Western Femto (Pierce Biotechnology; Rockford, IL).
Results
Paraffin IHC was applied to localize the normal PrPc in sections of bovine mammary glands by using a well-characterized MAb. Animals included were different ages and in different stages of lactation. The presented results (Figure 2) are typical for multiple experiments and showed specific immunostaining for PrPc in all studied samples. The histological appearance of different parts of the investigated lactating gland varies considerably, and different areas are not all in the same functional state at the same time. This is typical for all investigated glands independent of age and duration of lactation and is illustrated by Figures 2A-2D where the intensity of PrPc expression also varied considerably between individual alveoles. Particularly strong expression was found in such areas (Figures 2A and 2B) where actively secreting lactocytes are cuboidal or columnar in shape and show typical apocrine and eccrine secretory activity. In contrast, in inactive alveoles where the lumen is wide, the walls dilated, and the epithelial layer flattened in shape (low cuboidal lactocytes), expression of PrPc is reduced (Figures 2C and 2D). No expression was found in cells lining intralobular and interlobular ducts (Figure 2C). On the cellular level, prion protein is located in the cytoplasm with preference to the basolateral cell compartment (Figure 2B). In contrast, PrPc is not expressed in cells of the vascular system (endothelial cells, pericytes, and smooth muscle cells), in fibroblasts, and in intramammary leucocytes (Figure 2D and inset). To clarify whether intraalveolar myoepithelial cells contribute to PrPc expression, sequential staining of serial sections with antibodies directed against α-SMA and PrPc was performed. Detailed comparison of serial sections presented (Figures 2E and 2F) clearly show that myoepithelial cells selectively labeled with anti-α-SMA (Figure 2F) do not express PrPc (Figure 2E). Additionally, sections of bovine spinal cord were stained in parallel as positive controls where lamina I and II showed the most intense reaction for PrPc (Figure 2H). Absence of detectable staining of tissue elements in the negative controls (Figure 2G) verified specificity of the applied antibody.
To further validate IHC findings, MAbs with different specificities were used for Western blot and ELISA analyses. Bovine as well as ovine brain homogenates reacted strongly in Western blot and ELISA and had to be diluted 1:4 and 1:20, respectively. By using ELISA, weak but clearly positive results were obtained for bovine mammary gland homogenates of all nine animals. Mean OD value was at 0.7 ± 0.4; thus, protein expression related to OD differed by a factor of 10 between high- and low-expressing animals (Figure 1). However, analysis of selected mammary gland samples by Western blot gave no visible band for PrPc. In contrast, ovine mammary gland samples that were analyzed in parallel by both methods and showed a stronger reactivity in ELISA (data not shown) were positive in the Western blot for proteinase-K (PK)-sensitive expression of PrPc (Figure 3). Obviously, failure of the Western blot technique for detection of PrPc in bovine mammary gland samples was caused by the low amounts of this protein in respective extracts.

Comparative Western blot of selected bovine and ovine mammary gland tissues. Even lanes correspond to proteinase-K-digested samples; uneven lanes correspond to undigested samples. Lanes 1,2 = bovine brain homogenate (1:4); Lanes 3,4 = cow 6; Lanes 5, 6 = cow 7; Lanes 7, 8 = cow 10; Lanes 9,10 = ovine brain homogenate (1:20); Lanes 11,12 = sheep 1; Lanes 13,14 = sheep 2; Lanes 15, 16 = sheep 3.
Real-time RT-PCR with PrP-specific primers revealed a specific product with one distinct melting peak. Inspection of PCR products on a 2% agarose gel showed one single band (data not shown). CPs were at a mean value of 10.7 ± 1.71. All samples of the nine animals were positive in real-time RT-PCR. In addition to relative expression of mRNA and protein, x-fold expression was calculated (raw data not shown), and strong individual differences in mRNA expression became obvious (Figure 1). When calculating x-fold expression differences, one should keep in mind that a difference of one CP means that there was twice as much starting material in the sample with the lower CP. Thus, a difference of 8.4 CP between animals 1 and 4 reflects a 337-fold higher expression in animal 1. A correlation between the age of the animals under study and the expressed PrP was not found at the mRNA level (r2 = 0018) or at the protein level (r2 = 0.18). Similar results were obtained for PrPc expression levels and the stage of lactation. Again, no linear correlation was obvious (r2 = 0.04 for mRNA; r2 = 0.02 for protein). Furthermore, PrP-mRNA expression sometimes did not correlate well with the protein level detected in ELISA (e.g., animals 5, 8, and 9).
Discussion
Many studies have addressed the question of PrPc expression in different animal species and distinct organs, but studies on cell-type-specific distribution of PrPc in cattle are rather limited. In particular, data on the PrPc mRNA and protein expression in the bovine mammary gland have not yet been reported. Recently, Tichopad et al. (2003) described tissue-specific expression pattern of the bovine prion gene by real-time RT-PCR using an absolute quantification approach, but they did not include mammary gland. Here we applied IHC, ELISA, Western blot, and RT-PCR in a comprehensive approach to assess PrPc expression in the bovine mammary gland.
MAbs P4 and L42 used for IHC methods were raised against amino acids 89-104 and 145-163 of the ovine PrP sequence and showed significant reactivity to bovine PrP, which differs in two amino acids within each sequence (Harmeyer et al. 1998). The third MAb applied here was V5B2, which was raised against a synthetic peptide comprising amino acids 214-226 (CITQYERESQAYY) of the C-terminal end of human PrPc. Based on the analysis of human brain sections by IHC and testing of human brain homogenates by dotblot and ELISA, Šerbec et al. (2004) claimed that this antibody is specific for human PrPsc. However, the authors presented no data on the reactivity of this antibody with cellular bovine PrP. In our experiments, after initial trials testing all combinations of P4, L42, and V5B2, V5B2 proved to be suitable as detection antibody in a sandwich ELISA optimized for the detection of bovine PrPc. This finding is in accordance with the reactivity of a rabbit antiserum raised against a slightly larger sequence (VEQMCITQYQRESQAYYQR) of bovine PrP that also exhibited no isoform specificity (Takahashi et al. 1999). Furthermore, applicability of this sandwich ELISA has been demonstrated in a previous study on cell-type-specific expression of PrPc in bovine kidney samples (Amselgruber et al. 2006). Compared with the latter study, ELISA signals for mammary glands were relatively weak and indicated that for PrPc in this tissue the extraction efficacy of the applied method was low. Therefore, it was not surprising that ELISA results could not be confirmed by Western blot for which the same extraction procedure was used. Confirmation was, however, clearly possible for ovine mammary gland samples that exhibited distinctly higher ELISA reactivity. Although only mammary gland samples of three sheep were analyzed, this finding indicates that PrPc expression in sheep may be quantitatively quite different from cattle. In ovine mammary glands, Moudjou et al. (2001) found PrPc levels of up to 39 ng/g. Overall, within our study three different MAbs recognizing different epitopes of PrP and three different tests systems, with ELISA being the most sensitive method, were used. Sample treatment with PK constantly eliminated reactivity of the applied antibodies, indicating absence of PK-resistant PrPsc in the samples. RT-PCR was used as an additional method to validate PrPc expression on mRNA level. Samples of all animals were found to be positive. Although we used a relative quantification system for RT-PCR, which in principle allows better comparison and correlation of results achieved by different methods, PrP-mRNA expression did not correlate with the protein level detected in ELISA for all animals (Figure 1, e.g., animals 5, 8, and 9). A similar observation was reported by Ford et al. (2002) for neuronal tissue of mice. The authors explained this result by potential posttranscriptional regulation. However, the different regional distribution of expression found by IHC would also explain this finding because for methodical reasons differences in samples used for each preparation and analysis technique cannot be ruled out.
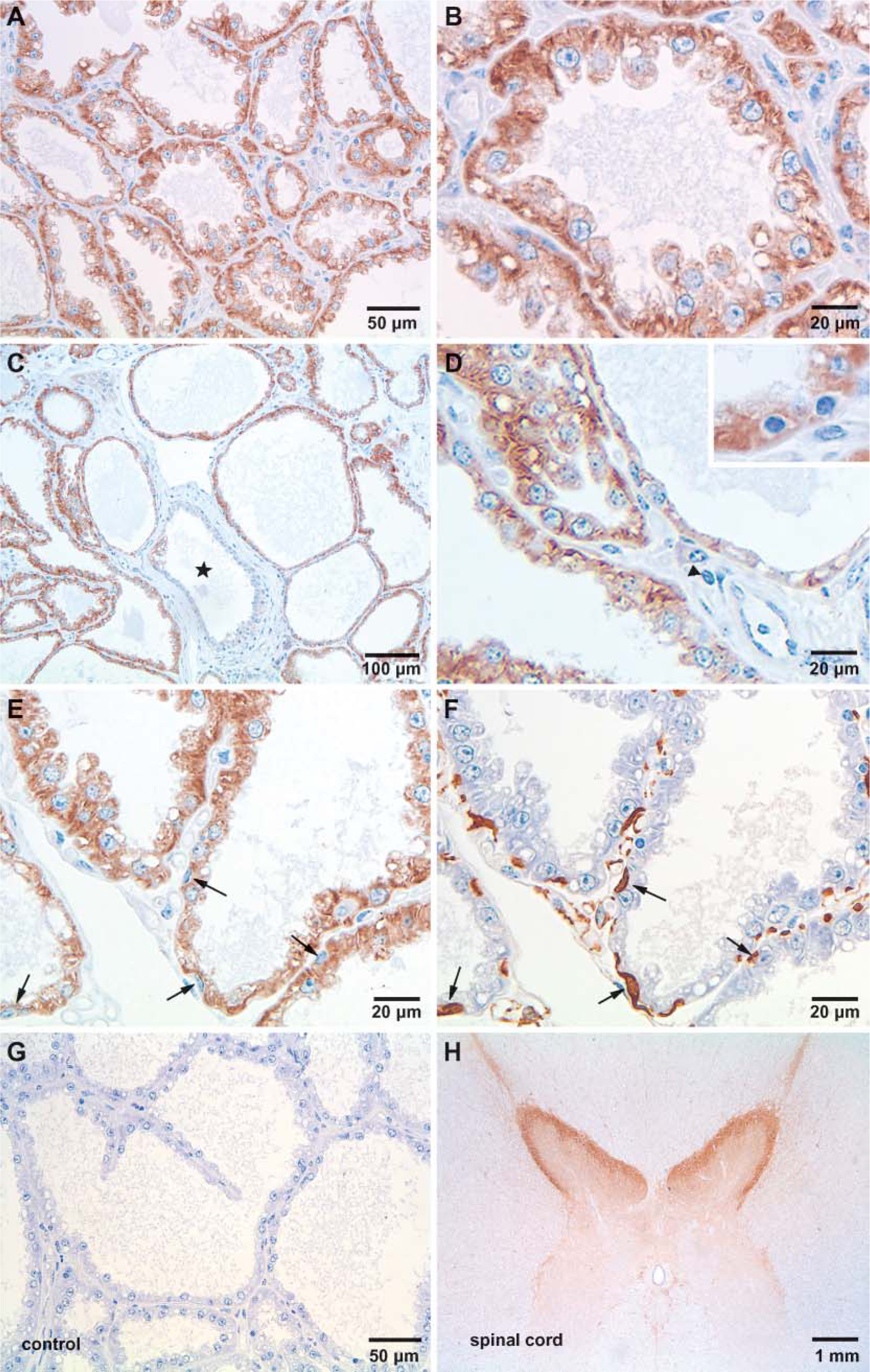
Immunohistochemical localization of PrPc in mammary tissue during lactation. Selective positive staining was exclusively demonstrated in epithelial cells (lactocytes) lining the tubuloalveoli. Stain intensity varies between animals and individual lobules (
IHC clearly showed a strong positive staining for the normal cellular prion protein mainly in alveoles composed of actively secreting columnar cells and a reduced expression in rather inactive alveoles and no reaction in intralobular and interlobar ductules. No expression could be demonstrated in myoepithelial cells. This PrPc expression pattern was independent of age or stage of lactation for all animals. Thus, age and stage of lactation did not correlate with mRNA or with protein levels detected by RT-PCR and ELISA, respectively. This finding indicates that PrPc is expressed at a detectable level only in actively secreting cells, which can be found at different levels throughout the lactation period but scarcely in the prepubertal mammary gland and only at a low level during the dry period.
The physiological function of PrPc is largely unknown. Studies on cells from wild-type or PrP knockout animals suggest that PrPc protects the cell by controlling copper metabolism governing cell resistance to oxidative stress (Brown et al. 1997; Brown 2001) or by playing an anti-apoptotic role (Brown 2001; Hetz et al. 2003). In light of results presented here, a question arises about a possible function of the cellular prion protein in the mammary gland. The most interesting finding of this study was that lactocytes selectively express PrPc preferentially at the basolateral surface of the cell when they are functionally active. This observation seems to be unusual because lipid raftassociated GPI-anchored proteins are known to be predominately sorted to the apical cell surface (Paladino et al. 2002). Paquet et al. (2004) also found a predominate expression of PrPc on the apical surface of Rov epithelial cells. Morel et al. (2004) demonstrated that PrPc is apical-laterally located at the level of cell-to-cell junctional domains. On the other hand, Sarnataro et al. (2002) reported on basolateral expression of PrPc on polarized Madin-Darby canine kidney (MDCK) and Flp recombination target (FRT) cells. These results were recently reported by Uelhoff et al. (2005). They showed preferential sorting of PrPc to the basolateral membrane of MDCK cells. In contrast, the PrP homolog Doppel was predominately found in the apical membrane. Although functional implications of this observation are unclear, electrophysiological studies and cell Ca2+ measurements support the idea that PrPc absence leads to a compromised Ca2+ homeostasis (Herms et al. 2000). Thus, such a defect finally impinges on Ca2+-dependent neurophysiologic functions such as plasma membrane K+ currents (Herms et al. 2001). Involvement of PrPc in cellular calcium homeostasis is further supported by a recent study (Brini 2003) that convincingly shows important regulatory functions of PrPc on Ca2+ homeostasis in cells transfected with PrPc. Similar interpretations can be drawn from the fact that one of the first events observed in neuronal cells exposed to PrPsc is a substantial endoplasmic reticulum (ER) Ca2+ discharge, followed by upregulation of stress proteins and activation of ER resident caspase-12 (Hetz et al. 2003). Focusing on these data and our results of preferentially basolateral PrPc expression, a possible protective role of PrPc in lactocytes during lactation via regulation of intracellular Ca2+ homeostasis is tentatively suggested. Other functions like protection of the cell by controlling copper metabolism or governing the cell resistance to oxidative stress (Brown 2001; Milhavet and Lehmann 2002) also need to be considered.
In conclusion, selective expression of PrPc in bovine lactocytes demonstrates for the first time that PrPc is abundantly present in bovine lactocytes and that cell type could serve as an interesting model to study the control of PrP gene expression as well as to gain further insight into the physiological function of the cellular prion protein.
Footnotes
Acknowledgements
The project was supported by a grant from the Bavarian State Ministry for Environment, Health and Consumer Protection (ForPrion Project No. 19a) and by the Transmissible Spongiform Encephalopathies (TSE) support programme of Baden-Württemberg (FKZ: 729.59-4/1).
We thank Margit Straka, Karen Müller, and Renate Finkelde for excellent technical support.
