Abstract
Aim
To assess serum Cu/Zn SOD (Superoxide Dismutase) concentration in individuals with bipolar disorder.
Subjects and methods
Serum from 20 individuals diagnosed with bipolar disorder and 20 age and gender similar controls were tested for Cu/Zn SOD serum concentration using ELISAs.
Results
Serum Cu/Zn SOD levels of individuals with bipolar disorder were significantly higher than age and gender matched controls.
Discussion
These results suggest an association between Cu/Zn SOD serum levels and bipolar disorder.
Introduction
Bipolar disorder is characterized by episodes of mania and depression that are frequently acute, debilitating, and conducive to gross social dysfunction and an elevated suicide rate.1–4 While often lifelong and affecting about 1.5% of the population, the condition responds to, and in some cases is controlled by, pharmacologic treatment–-particularly with mood-stabilizing agents.
Recent findings indicate that the pathologic process of this disorder is involved in complex interactions between multiple susceptibility genes and environmental factors.5,6 Recent advances in genetics, molecular and cellular biology, molecular neuroscience, and imaging technology have provided a variety of new and sophisticated approaches to evaluate the complex features of this disease. These new molecular genetic, molecular pharmacologic, and molecular and cellular biological approaches to the study of the etiology of psychiatry disorders and the mechanism of action of psychotropic drugs have been very instructive in refining our understanding. Given the many widely used advances in investigative approaches and technologies, the term “molecular psychiatry” has been readily adopted to replace the older “biological psychiatry”.
Important among these advances, high density DNA expression microarray technology has recently been widely adapted by scientists in the search for susceptibility genes for bipolar disorder.7–9 Gene candidates coding for proteins related to mitochondrial function and apoptosis, glutamate receptors, markers of GABAergic neurons, molecular chaperones, and oligodendrocyte proteins are differentially expressed in postmortem brain tissue, peripheral blood cells, and olfactory neuroepithelium between control and bipolar disorder subjects.10–12
Oxidative damage results from biochemical interactions between reactive oxygen species (ROS) and target biomolecules. ROS can damage nucleic acids, lipids, and proteins, and figures prominently in the etiology and progression of numerous cancers13–15 as well as coronary and carotid atherosclerosis.16–19
Oxidative stress, occurring as a consequence of imbalance between the formation of free oxygen radicals and inactivation of these species by antioxidant defense system, is capable of causing damage to various cellular and extracellular constituents. The deleterious effects of increased oxidative stress are termed oxidative damage. These effects generally appear after exposure to a relatively high concentration of reactive oxygen species (ROS) and/ or a decrease in antioxidant (AO) defense system against ROS. 20
There are three major families of superoxide dismutase, depending on the metal cofactor: Cu/Zn (which binds both copper and zinc),23,24 Fe and Mn types (which bind either iron or manganese), 25 and finally the Ni type, which binds nickel. 26
Cu/Zn superoxide dismutase (SOD-1) is a key enzyme in the dismutation of superoxide radicals resulting from cellular oxidative metabolism, converting them into hydrogen peroxide, 27 and, as a result, serves a key antioxidant role. In fact, mice lacking SOD die several days after birth, amidst massive oxidative stress. 28 Copper, and zinc participate in SOD enzymatic mechanisms that protect against free radicals and therefore serve an important adjunct role in oxidative balance. 29
Oxidative stress has been implicated as an etiological factor in Bipolar Disorder.30–33 Because Cu/Zn SOD is a major mitochondrial anti-oxidant, this study was designed to determine Cu/Zn SOD serum levels in individuals with bipolar disorder and test the hypothesis that this oxidative stress marker is increased in this group.
Materials and Methods
ELISA to measure serum Cu/Zn SOD (Bender MedSystems, eBioscience, San Diego CA)
All reagents and specimens were equilibrated to room temperature before the assay was performed. A 1:51 dilution of the patient samples was prepared by mixing 10 µl of the patient's sera with 0.5 ml of Serum Diluent. One hundred microliters of calibrators (0.08–2.5 ng/ml Cu/Zn SOD), serum diluent alone, and diluted patient samples were added to the appropriate microwells of a microculture plate (each well contained affinity purified polyclonal IgG to Cu/Zn SOD). Wells were incubated for 60 minutes (±5 min) at room temperature, then washed 4x with wash buffer. One hundred microliters of pre-diluter anti-human Cu/Zn SOD IgG conjugated with HRP was added to all microwells, incubated for 30 minutes (±5 min) at room temperature, then wash 4x with wash buffer. One hundred microliters of enzyme substrate was added to each microwell. After approximately 30 minutes at room temperature, the reaction was stopped by adding 50 µl of 1M sulfuric acid, then the wells were read at 450 nm with an ELISA reader (BioRad Laboratories, Inc., Hercules, CA, USA).
Subjects
Experimental and controls
Serum from individuals with diagnosed bipolar disorder, all fulfilling DSM-IV diagnostic criteria (n = 20; 17 male; mean age 34 ± 12 years), and controls (n = 20; 17 male mean age 47 ± 14 years) was obtained from patients presenting at the Health Research Institute/ Pfeiffer Treatment Center. Most of these individuals were diagnosed using The Mood Disorder Questionnaire before presenting at the Pfeiffer Treatment Center, Warrenville, Il.*
The Pfeiffer Treatment Center is a comprehensive treatment and research center, specializing in the care of with neurological disorders, including bipolar disorder.
Patients (n = 20) for this study were chosen randomly as they presented to The Pfeiffer Treatment Center. At the time of the blood draw, three were taking anti-depressants, two were taking anti-epileptic drugs, four were taking anti-psychotics, four were taking lithium and seven presented not taking any associated drugs. We did not find any significant difference in HGF serum concentration in any of these groups (ANOVA;
Controls were normal healthy volunteers, all employees of the Pfeiffer Treatment Center.
Serums
All serums, experimental and control serums were treated in an identical fashion–-frozen at -70C immediately after collection and cell/serum separation, then stored at -70C until thawed for use in ELISAs.
Statistics
Inferential statistics were derived from t-test and odds ratios with 95% confidence intervals.
Results
Serum from 20 individuals diagnosed with bipolar disorder and 20 age and gender similar controls was tested for Cu/Zn SOD plasma concentration using an ELISA (described above). Each assay was repeated two or more times, with multiple wells for each serum in each assay. Results of a typical assay with assay controls are shown in Figure 1, and Bipolar and control individuals are shown in Figure 2. These data reflect the low variability and high reliability of each assay.
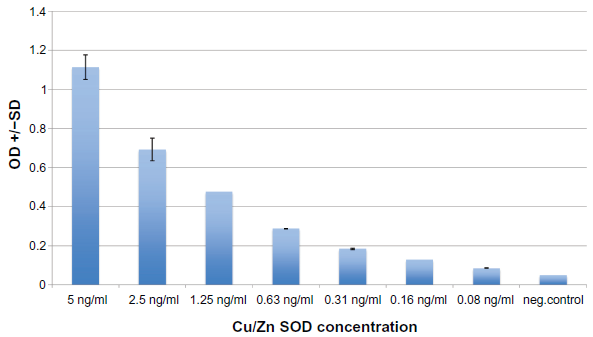
Cu/Zn SOD controls: Typical ELISA.
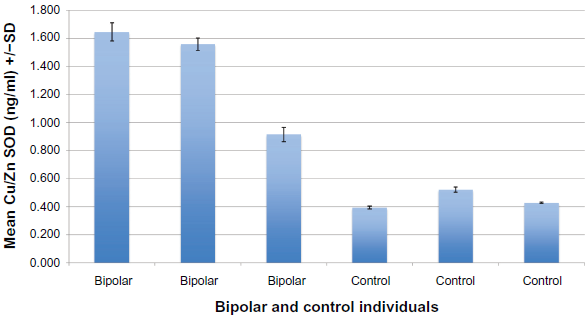
Cu/Zn SOD serum concentration (ng/ml) bipolar and controls: Typical ELISA.
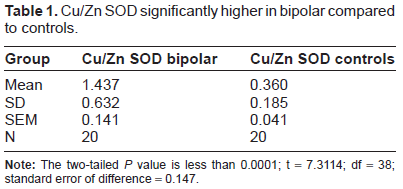
Serum Cu/Zn SOD levels of 20 Bipolar individuals were significantly higher (mean = 1.437 ng/ml) than the 20 non-depressed controls (mean = 0.360 ng/ml) (
Cu/Zn SOD significantly higher in bipolar compared to controls.
We found no relationship between age and Cu/Zn SOD levels (Fisher
Discussion
Oxidative stress has been implicated in the pathogenesis of a diverse group of disease states, and, because the brain has comparatively greater vulnerability to oxidative damage, may be a common pathogenic mechanism underlying many major psychiatric disorders.
This study represents an attempt to assess levels of the oxidative damage expressed by Cu/Zn SOD levels in the plasma of individuals with bipolar disorder, but otherwise healthy adults. As our results demonstrate, compared with similar control subjects, although each individual's Cu/Zn SOD level differed, the mean of those with clinical depression show significantly higher serum levels of Cu/Zn SOD.
We did not find any relationship between pharmacological treatment and Cu/Zn SOD levels. This is most likely because of the preliminary nature of this study. We will update this possible relationship as more patients are studied.
Other studies have shown positive correlations between oxidative stress markers and bipolar disorder34,35 and have suggested a role of mitochondrial dysfunction in the etiology of the disease. 36
Mitochondria-related genes may be abnormally expressed in bipolar disorder. Increasing evidence has shown the presence of mitochondrial dysfunctions such as deletion, mutation and abnormal expression of mitochondria-related genes, increased anaerobic glycolysis, and impaired phospholipid metabolism. Because mitochondrial oxidative phosphorylation is the major resource for the generation of reactive oxygen species, mitochondrial dysfunction indicates that oxidative stress may occur in bipolar disorder and that oxidative stress induced damage may be prevented by mood-stabilizing treatment. 37 This dysfunction may be directly associated with the high amounts Cu/Zn SOD in bipolar individuals observed in this study.
The present data supports the recent finding by Kunz et al 23 that Cu/Zn SOD is raised in euthymic bipolar patients and raises the intriguing possibility that the increase in Cu/Zn SOD levels in bipolar disorder could be mechanistically related to increased oxidative stress associated with mitochondrial dysfunction. Correcting the oxidative imbalance in BD may offer a new means of controlling the detrimental effects of mood episodes on cognition and bodily systems, and, if it also prevented the associated increase in Cu/Zn SOD, it would provide more direct evidence for the hypothesized causal relationship between increased oxidative stress and this potential marker.
The clinical relevance of increased oxidative damage, evidenced in bipolar individuals, is currently unknown, and future studies should appropriately address this deficit. Our data are consistent with the hypothesis that oxidative damage is a potential common pathophysiological mechanism underlying multiple co-morbid conditions in bipolar individuals. Future studies should include measurement of multiple oxidative damage markers to different macromolecules, associated dietary deficiencies associated with these markers, associated severity of bipolar disease, and should address whether bipolar disease remission remediates underlying oxidative damage.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conflicts of interest. The author confirms that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
The author thanks Scott Filer, Executive Director of The Pfeiffer Treatment Center and Dr. Robert deVito, MD for their support and help in this research and manuscript preparation.
The author also thanks Laurie Myers and Kyle Andrews for their technical assistance.
