Abstract
This review provides information on the most recent findings concerning presence, origin, and role of kynurenic acid (KYNA), a tryptophan metabolite, in the digestive system. KYNA is an antagonist of both the ionotropic glutamate receptors and the alpha7 nicotinic acetylcholine receptor, as well as an agonist of G-protein coupled GPR35 receptor. Since the GPR35 receptor is mainly present in the gastrointestinal tract, researchers have concentrated on the digestive system in recent years. They have found that KYNA content increases gradually and significantly along the gastrointestinal tract. Interestingly, the concentration of KYNA in the lumen is much higher than in the wall of intestine. It has been documented that KYNA may have a positive influence on the number of pathologies in the gastrointestinal tract, in particular ulcers, colon obstruction, or colitis. Future studies might determine whether it is advisable to supplement KYNA to a human organism.
Introduction
Kynurenic acid (KYNA) is a metabolite of tryptophan formed enzymatically along the kynurenine pathway. The first step in the pathway is catalyzed by tryptophan 2, 3-dioxygenase (TDO) and indoleamine 2, 3-dioxygenase (IDO), enzymes responsible for tryptophan degradation to formylkynurenine. TDO is primarily expressed within the liver and IDO is an extrahepatic cytokine inducible enzyme. The next step in the synthesis of kynurenine is catalyzed by formylkynurenine formamidase. The main end product of kynurenine catabolism is nicotinamide adenosine dinucleotide (NAD+). An another biologically active product, KYNA, is generated from kynurenine by kynurenine aminotransferase.1,2
KYNA presence was first demonstrated in urine by Liebig. 3 Nevertheless, the compound was not thoroughly analyzed until the 1980's and 1990's when researchers indicated that KYNA is an antagonist of ionotropic glutamate receptors.4–8 Subsequently, researchers presumed that KYNA is present in the human brain.9,10 Numerous studies were conducted to investigate the role of KYNA in the physiology and pathology of the central nervous system (CNS). Since both the concentration of KYNA in the human brain and penetration of KYNA through a blood-brain barrier are low, studies of peripheral KYNA gained popularity.
Mechanisms of Action
Receptors affected by kynurenic acid.
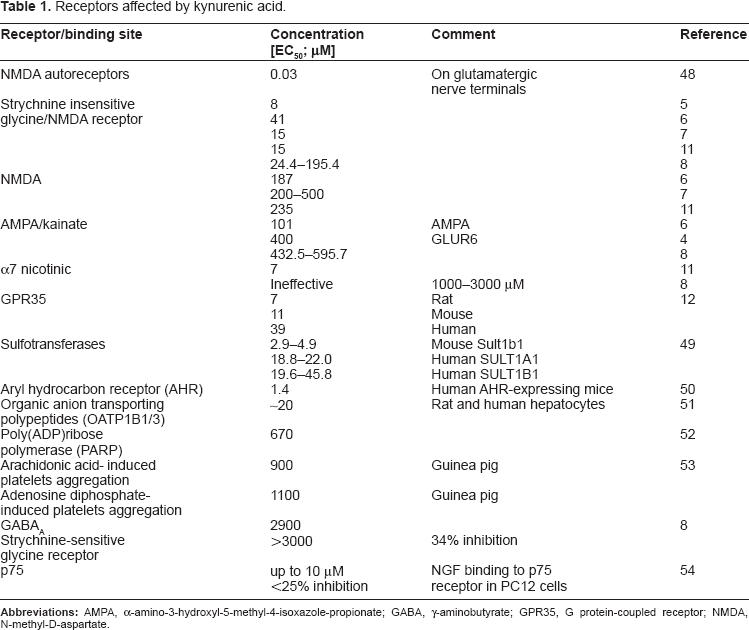
The detailed information on the effect of KYNA exerted by interaction with above mentioned receptors was a matter of several currently published review papers.13–15
Content of KYNA in the CNS and Periphery
KYNA content in tissues and body fluids.

For the sake of a comparison, the content of KYNA in wet tissue was calculated as follows: ηmol/g = μM.
It should be emphasized that the concentration of KYNA high enough to interact with glutamate, alpha-7 cholinergic, and GPR35 receptors in vitro was detected in lumen of ileum. Also of note, the GPR35 receptor is mainly present in gastrointestinal tissues. 12
KYNA in CNS
Glutamate receptors are mainly located in the CNS. According to electrophysiological studies, KYNA exerts a modulatory effect on neurotransmission in the brain.11,16 Several papers review the action and the potential role of KYNA in the brain.17–19
KYNA in Periphery
Initially, KYNA was found in urine in 1853. 3 More recently, presence of KYNA in urine was described in numerous papers (Table 2).20–26
It was evidenced that KYNA is present in serum where its concentration varied from 0.004–0.06 μM (Table 2). Additionally, KYNA was found in synovial 27 and amniotic fluid. 28
Furthermore, numerous reports concentrate on KYNA content outside of the CNS, specifically focusing on its presence in the digestive system.
KYNA Content in Digestive System
Content of KYNA in the lumen of digestive system.

Interestingly, a much lower concentration of KYNA was found in the wall of the gastrointestinal tract. According to Kuc et al, the concentration of KYNA in the wall of a rat's duodenum, jejunum, and ileum was 0.29 μM, 0.21 μM and 0.28 μM, respectively. 29 Pawlak et al reported a concentration of 0.09 μM of KYNA in rat intestine. 30 The content of KYNA in the wall of a rat's duodenum, jejunum, and ileum was, therefore, 19%, 6%, and 3% of the content of KYNA in the lumen of the appropriate part of a rat's intestine, respectively. As a result, the supposition that KYNA is produced in the wall of the intestine and then secreted to the intestinal fluid in such a high amount seems rather unlikely.
The remarkable content of KYNA amounting to 0.31–0.83 μM and 1.11 μM was found in bile of human and pig, respectively. 29 Unexpectedly, KYNA was detected in the pancreatic juice of pigs with a relatively high concentration (0.76 μM). 29 However, the distribution and activity of kynurenine transaminases in pancreatic tissue, and thus the mechanism of KYNA formation in exocrine acinar cells of the pancreas, have not been elucidated to date.
Based on the data concerning the KYNA absorption in the intestine31,32 and its presence in bile and pancreatic juice, 29 the hepatic-pancreatic-intestinal secretion-absorption functional cycle creating a high concentration of KYNA in intestinal fluid can be suggested. Furthermore, it is highly probable that activity of colon microbiota participates in the maintaining of KYNA in the gastrointestinal content especially in their distal part as well. 29
Properties of KYNA in Pathological States of the Gastrointestinal Tract
KYNA and pathology of gastrointestinal tract.

All in all, scientific reports suggest that KYNA may have a positive influence on the number of pathologies of the gastrointestinal tract, especially regarding ulcers, colon obstruction or colitis. There is some dispute as to whether KYNA exerts a positive or negative action in bowel diseases since an increased level of KYNA in colon neoplasia cannot be interpreted either in favor of or against KYNA, as its mechanism of action in the mentioned diseases is yet unknown. Recently, KYNA's enhancement of the expression of cyclin-dependent kinase inhibitor, p21 Waf1/Cip1, and inhibition of cell proliferation and DNA synthesis in colon adenocarcinoma HT-29 cell line were described. 41
KYNA and the Gut Flora
Most recently, it was indicated that Escherichia coli is able to produce KYNA and liberate it to extracellular milieu. 29 Similarly, KYNA production was shown in cell-free extracts of E. coli. 42 Furthermore, there is allusion to positive correlation between microflora concentration and KYNA content in jejunum and ileum. 29 These findings suggest that gut flora may participate in forming the common pool of intestinal KYNA.
On the other hand, it has also been demonstrated that KYNA affects bacterial growth. Interestingly, low and medium concentrations of KYNA stimulate growth of certain probiotics while KYNA in high concentrations possesses antibacterial properties.43,44
Metabolism of KYNA
KYNA is present in the lumen of rat small intestine in micromolar concentrations, 29 which is sufficient to affect the GPR35 receptor. The sources of KYNA in the gastrointestinal tract are not known, although it seems that, in this instance, KYNA was either produced from tryptophan or delivered with food and other dietary products.
The pathway for KYNA creation in humans was most recently investigated by Hiratsuka et al in young Japanese women who consumed tryptophan in amount of 0.7 g (3328 μmol) per day with subsequent analyzes of various tryptophan metabolites. 20 KYNA production was reported near 10 μmol/day, indicating that transformation of tryptophan to KYNA occurs with an effectiveness of 0.3%. 20 Leklem reported a slightly higher effectiveness of the reaction at 0.42%. 26
The expert report created by the World Health Organization (WHO), Food and Agriculture Organization (FAO), and United Nations University (UNU) recommends a tryptophan dose of 4 mg/kg/day. 45 Therefore, assuming an adult's weight to be 70 kg, a recommended dose of tryptophan per person is 280 mg/day. It can be estimated that either 0.84 mg (effectiveness of 0.3%) or 1.18 mg (effectiveness of 0.42%) of KYNA is created in the human body along the kynurenine pathway from tryptophan.
Therefore, the research suggests that KYNA might be synthesized in the human body along the kynurenine pathway from its precursor, tryptophan. However, there was no analogous research on KYNA synthesis from its immediate precursor, kynurenine, and there is no data regarding the presence of kynurenine in either food or herbs.
Despite some previous research indicating that KYNA might be metabolized to quinaldic acid, 46 we did not observe its formation from KYNA in rat brain slices and liver homogenates (unpublished observation). Nevertheless, there is no data on KYNA accumulation in the body. Numerous scientific papers report that KYNA is present in human urine and hence excreted with it.20–26 Reports of excreted KYNA concentrations vary from 1143.9 μg per day to 5376.6 μg per day (Table 5). Based on the results shown in Table 5, and bearing in mind the fact that estimated production of KYNA from tryptophan in human organism is either 840 μg per day or 1160 μg per day, it could be assumed that KYNA is also absorbed from the digestive system. In fact, there is indication that KYNA can be absorbed from the digestive system into blood circulation. 31
Excretion of KYNA in urine.

The assumption behind the calculations is that a human being excretes 1.5 liters of urine on a daily basis. 75
KYNA in Food
Content of KYNA in food products.

Average consumption stands for a hypothetical quantity of a certain product that an average human being consumes in one day.
An average KYNA intake from food was calculated based on KYNA content in a gram of wet weight of a certain food product multiplied by an average consumption of a certain food product. The summary of results is shown in Table 6. Based on our estimation, the highest amount of KYNA delivered to the human body with food comes from broccoli. It should be noted, however, that the boiling procedure lowered KYNA content in broccoli by 88%. 31 High amounts of KYNA can be delivered with potatoes. This depends mainly on variety of potato and amount of consumed vegetable as boiling does not significantly affect KYNA content in potatoes. 31 Conversely, consumption of rice and maize delivers remarkably lower amounts of KYNA.
KYNA in Herbs
Content of KYNA in herbs.

KYNA intake stands for a maximum recommended daily dose according to producers’ guidelines multiplied by KYNA content in a g wet weight of a certain herb.
Summary
Reviews of available data suggest that KYNA is present in the lumen of the digestive system in high concentration. Furthermore, KYNA is both produced in the human body from tryptophan as well as absorbed by it from various food and herbs. It can be also produced by gut flora. Numerous researchers suggest that KYNA may play a significant role in the functioning of the digestive system. Interestingly, recent studies indicate that KYNA may possess therapeutic properties when used to treat diseases of the gastrointestinal tract.
Nevertheless, numerous questions still need to be answered. No scientific data exists detailing KYNA excretion levels with feces. Furthermore, the ability of intestinal bacteria to produce KYNA and its subsequent production levels must be investigated. Moreover, existence of KYNA enterohepatic circulation also requires study as KYNA is excreted in bile and can be absorbed from the intestine. Specifically, the effectiveness of absorption in various parts of the digestive system deserves further elucidation.
There is no clear data concerning an optimal daily intake of KYNA by humans and their daily demand for it. Consequently, no data details whether there is the possibility of KYNA shortage in the human organism and, if so, the outcome generated by such shortage. We recommend gastrointestinal tract diseases and states of malnutrition be thoroughly analyzed, as daily exogenous KYNA supply may not be sufficient to cover its losses through, for example, urine.
KYNA's influence on enterocytes also warrants further investigation. And finally, the role of KYNA in tumor development needs be studied.
Future studies may determine whether it is advisable to supplement KYNA to a human organism.
Author Contributions
Analyzed the data: MPT, MT. Wrote the first draft of the manuscript: MPT. Contributed to the writing of the manuscript: MT, PP, JPT, GO. Agree with manuscript results and conclusions: MPT, MT, PP, JPT, GO. Jointly developed the structure and arguments for the paper: MPT, MT, PP, JPT, GO. Made critical revisions and approved final version: MPT, MT, PP, JPT, GO. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
Footnotes
Acknowledgements
M.P. Turski and M. Turska are students, volunteers in the Department of Toxicology, Institute of Rural Health and the Department of Experimental and Clinical Pharmacology, Medical University, respectively.
