Abstract
Aim
To assess serum Anti-Neutrophil cytoplasmic Antibody (ANCA—PR3 and MPO) and Anti-Saccharomyces Cerevisiæ Antibody (ASCA) levels in autistic children with severe gastrointestinal (GI) disease and to test the hypothesis that there is generalized autoimmunity in a subpopulation of autistic children with severe GI disease and that this autoimmunity is associated with the severity of GI disease.
Subjects and Methods
Serum from 40 autistic children with chronic digestive disease (most with ileo-colonic lymphoid nodular hyperplasia (LNH) and inflammation of the colorectum, small bowel and/or stomach), and 24 controls (13 age matched autistic children with no GI disease and 11 age matched children without autism or GI disease) were tested using ELISAs designed to quantitate ANCA (PR3 and MPO) and ASCA levels. ANCA and ASCA concentration of autistic children with GI disease were compared to GI disease severity (including LNH and erythema).
Results
We previously reported that 6 of the 40 autistic children with GI disease in this study had anti-PR3 ANCA. All 6 of these also had anti-MPO IgG. In this study, we found that 6 of the 40 autistic children also had ASCA and 4 of these children with ASCA also had both anti-PR3 and anti-MPO ANCA. These 4 with ANCA and ASCA had significantly higher GI disease severity, particularly associated with LNH and erythema.
Discussion
These results suggest a general autoimmune response (ANCA and ASCA) in a sub group (high GI disease severity) of autistic children with GI disease. The presence of these autoantibodies may be a useful biomarker for autistic children with severe GI disease.
Introduction
ANCA were originally detected in serum from patients with Wegener's granulomatosis (WG), 1 a disease characterized by necrotizing granulomatous inflammation of the upper and lower airways in conjunction with systemic vasculitis and necrotizing crescentic glomerulonephritis. 2 Following the detection of ANCA in systemic vasculitis, it became clear that ANCA also occurred in other idiopathic inflammatory disorders, 3 including inflammatory bowel diseases or IBD (which include ulcerative colitis (UC) and Crohn's disease (CD)),4,5 in autoimmune-mediated liver diseases, 6 8 in rheumatoid arthritis (RA),9,10 and in systemic lupus eythematosus (SLE).11,12
ASCA of IgG and IgA isotype are present in 60% of cases with diagnosed CD, 13 and directed against phosphopeptidomannans present in the yeast (S. cerevisiae) cell walls.13,16 ASCA may be elicited due to molecular mimicry and priming by a high mannose-containing bacterial or viral antigen or an “auto-antigenic (self) molecule”. 14 It has been observed that levels of ASCA in CD cases are independent of disease activity, duration and/or treatment. 15
Autistic Spectrum Disorder (ASD) is a neurodevelopmental syndrome with onset prior to age 36 months. Diagnostic criteria consists of impairments in sociality and communication plus repetitive and stereotypic behaviors. 17 Traits strongly associated with autism include movement disorders and sensory dysfunctions. 18 Although autism may be apparent soon after birth, many autistic children experience at least several months, up to a year or more in some cases, of normal development—followed by regression, defined as loss of function or failure to progress. 19 21
Children with autistic spectrum disorders (ASD) frequently have accompanying gastrointestinal (GI) symptoms and pathology,22,23 which includes inflammation of the GI tract. 24 27 Many autistic children, particularly those with GI disease, also have a higher propensity for fungal infections.28,29
We previously reported that a significant number of autistic children with GI disease have ANCA (both anti-PR3 and anti-MPO), and that there is a relationship between individuals with ANCA and severity of intestinal disease. 30 We have also reported that a significant number of autistic children with chronic digestive disease have anti-PR3 ANCA, low serum AAT, and high serum PR3, which correlate with high severity of GI disease, suggesting that high PR3 levels may be causing ANCA in autistic children with severe GI problems. 31 Because of this, we hypothesized that the autoimmune response in this sub group of autistic children might be general, so we tested the same population for ASCA.
Materials and Methods
ELISA to Measure Serum ANCA (Anti-Pr3 Antibodies and Anti-MPO Antibodies) and ASCA (IMMCO Diagnostics, Buffalo, N.Y.)
All reagents and specimens were equilibrated to room temperature before the assay was performed. A 1:51 dilution of the patient samples were prepared by mixing 10 μl of the patient's sera with 0.5 ml of Serum Diluent. One hundred microliters of calibrators (20–200 Eu/ml antibodies), positive and Negative control serums, serum diluent alone, and diluted patient samples were added to the appropriate microwells of a microculture plate (each well contained approximately 150 ng of purified PR3 or MPO). Wells were incubated for 30 minutes (±5 min) at room temperature, then washed 4 x with wash buffer. One hundred microliters of pre-diluter anti-human IgG conjugated with alkaline phosphotase was added to all microwells, incubated for 30 minutes (±5 min) at room temperature, then wash 4 x with wash buffer. One hundred microliters of enzyme substrate was added to each microwell. After approximately 30 minutes at room temperature, the wells were read at 405 nm with an ELISA reader (BioRad Laboratories, Inc., Hercules, CA, USA).
Subjects and Scoring of Severity of GI Disease
Serum from autistic individuals with GI disease was obtained from the Thoughtful House, Austin, Texas. All 40 children in this study with ASD (median age 6 years; range 2–16; 34 male) with gastrointestinal symptoms, were investigated by ileo-colonoscopy. Macroscopic and histological features of the upper and lower GI tract were scored. A point system was developed to assess the severity of GI disease (particularly inflammation). Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points)disease in each area (upper and lower GI) and for scope (macroscopic) and histology of each area. Therefore, the maximum score for GI disease was 12 (3 points each for upper scope, upper histology, lower scope and lower histology). A point system was also developed for severity of lymphoid nodular hyperplasia (LNH). Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points) LNH in each area (upper and lower GI) for a maximum of 6 points. And finally, a point system was also developed for severity of erythema. Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points) erythema in each area (upper and lower GI) for a maximum of 6 points.
Controls
Two control groups (Total n = 24) were studied, including 12 age matched autistic children with no GI disease and 12 age matched children without autism or GI disease (mean 68 months), gender (76% male) and diagnosis (55% regressive onset) matched autistic children with no GI disease; 11 age (mean 72 months) and gender (80% male) matched children without autism or GI disease. Serum and medical history of controls were obtained from the Autism Genetic Resource Exchange—AGRE * .
The Autism Genetic Resource Exchange (AGRE) is the first collaborative gene bank for the study of autism spectrum disorders and one of the world's largest shared resources for the study of autism and related disorders, with a collection of over 900 well-characterized multiplex and simplex families made available to the greater scientific community. Founded by Cure Autism Now (CAN) in 1997, AGRE is currently funded by the National Institute of Mental Health (NIMH) and Autism Speaks (AS), which merged with CAN in 2006.
Serums
Experimental (Thoughtful House) and control (AGRE) serums were frozen at −70 C immediately after collection and cell/serum separation, then stored at −70 C until thawed for use in ELISAs.
Statistics
Inferential statistics were derived from t-test and odds ratios with 95% confidence intervals. ANOVA analysis was used to determine correlation between types of autoantibodies present and severity of GI disease.
Results
Using the ELISAs described above, autistic children with chronic digestive disease, and controls, were tested for serum ANCA (PR3 and MPO) and ASCA levels. Results of a typical ASCA assay are shown in Figure 1. In each assay, positive titers of autoantibodies were determined by comparing experimental and control serum levels with ASCA standards and negative controls (sample diluent alone) (Fig. 2). The Optical Density of the 20 EU standard was the cutoff point necessary to establish a positive titer. For each assay, there were 3 or 4 replicate samples tested in each group (control and experimental), and each assay was repeated at least twice.
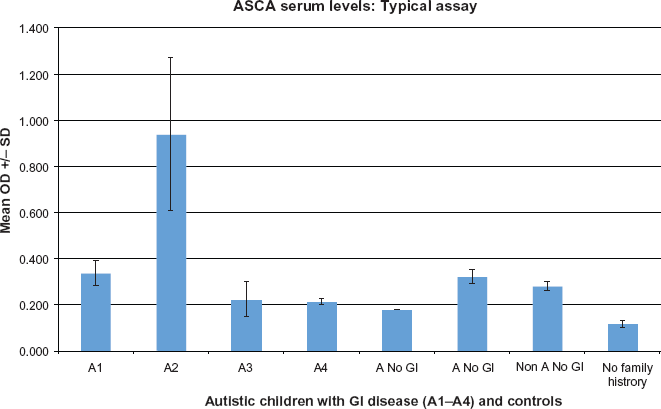
Serum ASCA concentration was measured in a typical ELISA. Four autistic children (A) with GI disease, 2 autistic children with no GI disease controls (A No GI), 1 non-autistic child with no GI disease controls (Non A No GI) and 1 child with no family history of autism with no GI disease (No Family History) were tested. Four replicate samples were tested for each individual.
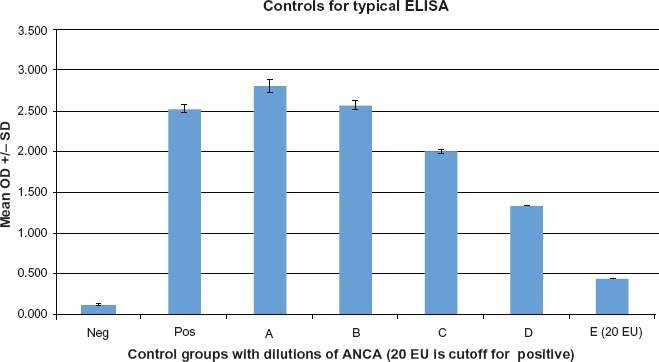
ASCA serum concentration was established for each individual by testing and correlating to known standards of various ASCA titers (A-E) as well as a positive (Serum with known high ASCA) and a negative control (serum diluent alone). Any serums with OD above the OD of the 20 EU (E) control were considered positive.
We found that 6 of the 40 autistic children with GI disease had positive ASCA compared to only one of the controls (autistic and non autistic, age and diagnosis matched children without GI disease) (Table 1) (p < 0.05).
ASCA data.
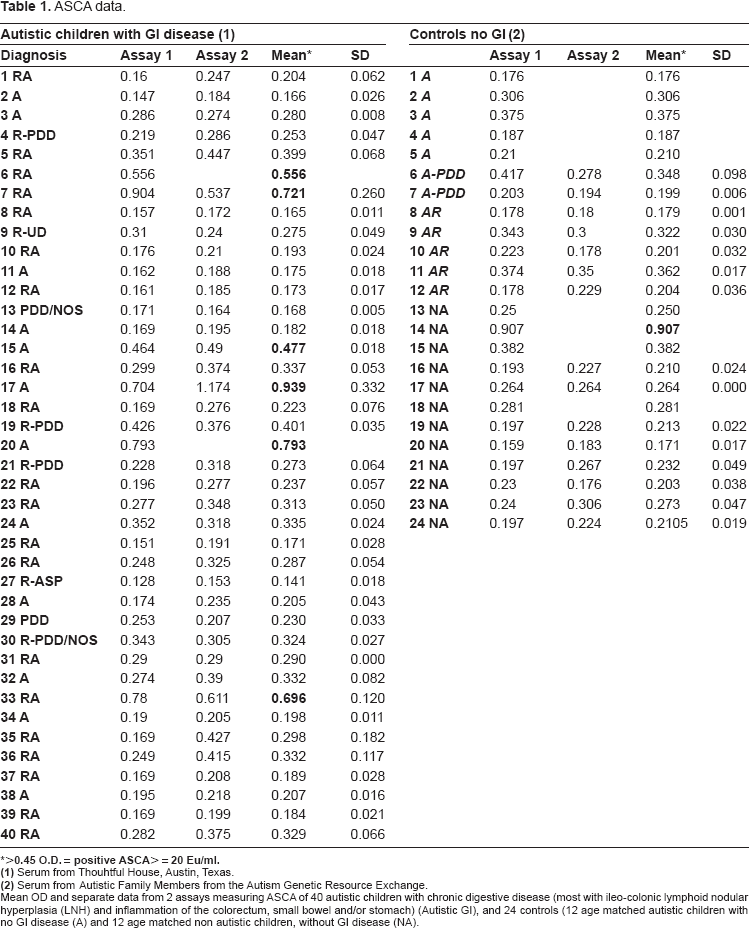
Mean OD and separate data from 2 assays measuring ASCA of 40 autistic children with chronic digestive disease (most with ileo-colonic lymphoid nodular hyperplasia (LNH) and inflammation of the colorectum, small bowel and/or stomach) (Autistic GI), and 24 controls (12 age matched autistic children with no GI disease (A) and 12 age matched non autistic children, without GI disease (NA).
Four of the 6 with ASCA also had both anti-PR3 and anti-MPO ANCA, compared to none of the controls (Table 2) (p < 0.01).
ASCA ANCA comparison.
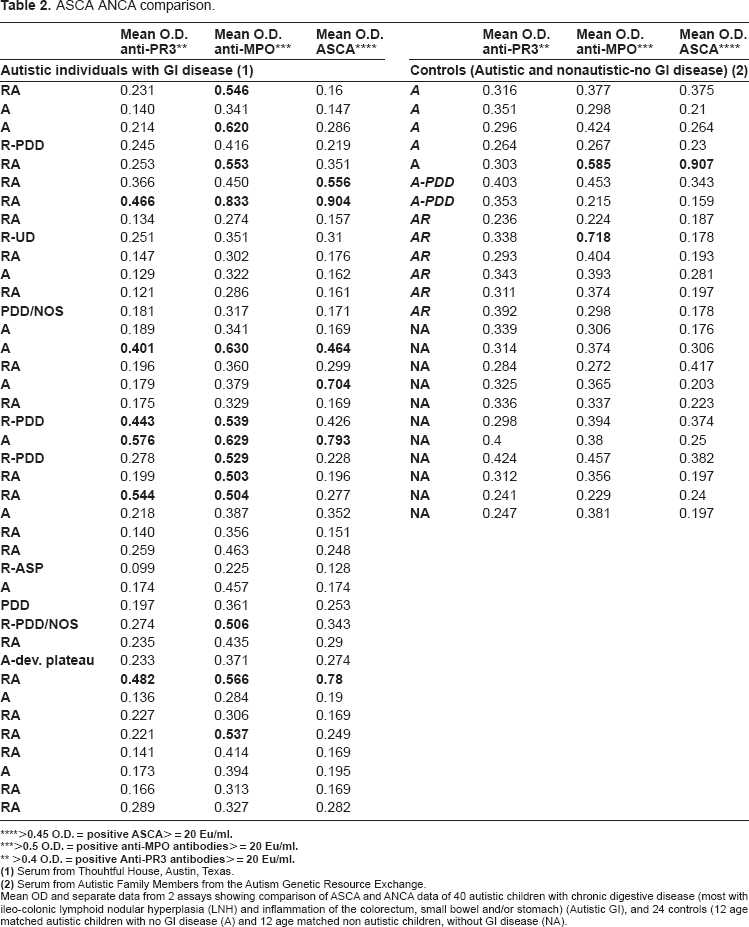
Mean OD and separate data from 2 assays showing comparison of ASCA and ANCA data of 40 autistic children with chronic digestive disease (most with ileo-colonic lymphoid nodular hyperplasia (LNH) and inflammation of the colorectum, small bowel and/or stomach) (Autistic GI), and 24 controls (12 age matched autistic children with no GI disease (A) and 12 age matched non autistic children, without GI disease (NA).
The 4 children with both ANCA and ASCA had significantly higher total GI disease severity (p < = 0.005), including severity of erythema (p < = 0.001) compared to those without the 3 types of autoantibodies. Although not significant, there was also a difference in severity of LNH between the two groups (p < = 0.1) (Tables 3 and 4).
Autoimmunity compared to severity of disease.
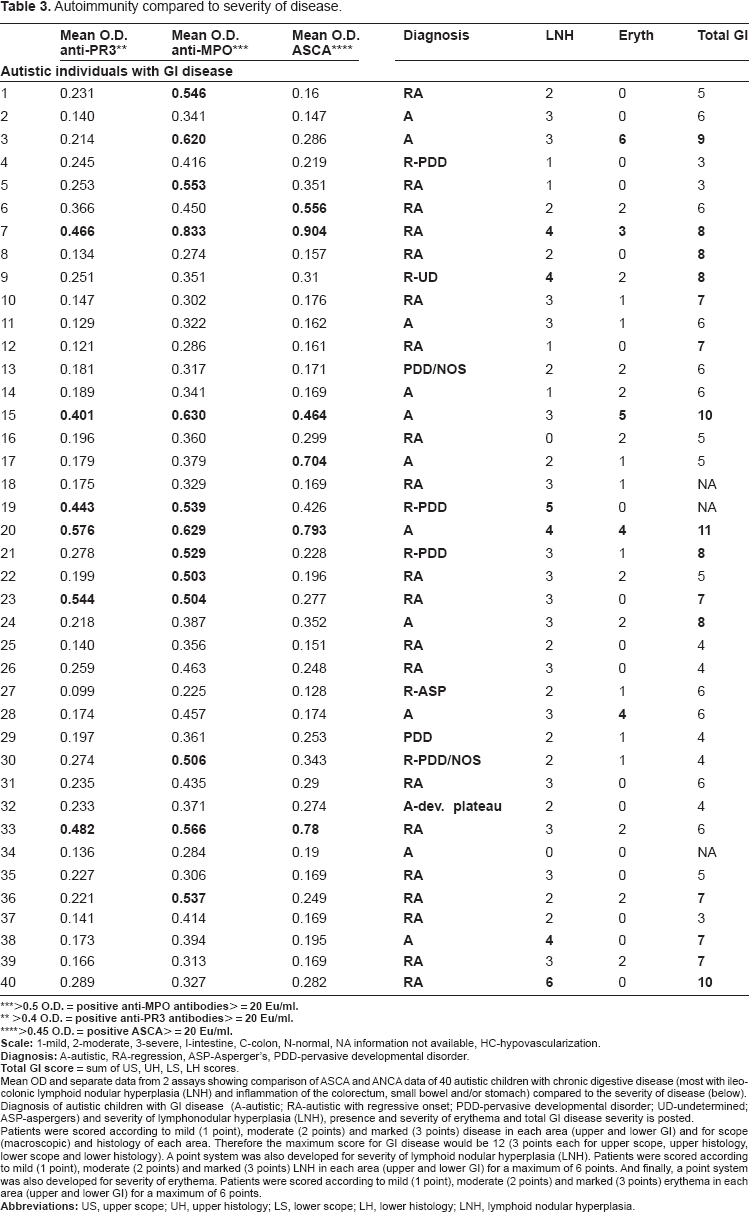
Mean OD and separate data from 2 assays showing comparison of ASCA and ANCA data of 40 autistic children with chronic digestive disease (most with ileo-colonic lymphoid nodular hyperplasia (LNH) and inflammation of the colorectum, small bowel and/or stomach) compared to the severity of disease (below). Diagnosis of autistic children with GI disease (A-autistic; RA-autistic with regressive onset; PDD-pervasive developmental disorder; UD-undetermined; ASP-aspergers) and severity of lymphonodular hyperplasia (LNH), presence and severity of erythema and total GI disease severity is posted.
Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points) disease in each area (upper and lower GI) and for scope (macroscopic) and histology of each area. Therefore the maximum score for GI disease would be 12 (3 points each for upper scope, upper histology lower scope and lower histology). A point system was also developed for severity of lymphoid nodular hyperplasia (LNH). Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points) LNH in each area (upper and lower GI) for a maximum of 6 points. And finally, a point system was also developed for severity of erythema. Patients were scored according to mild (1 point), moderate (2 points) and marked (3 points) erythema in each area (upper and lower GI) for a maximum of 6 points.
Autoimmunity vs. severity of disease.
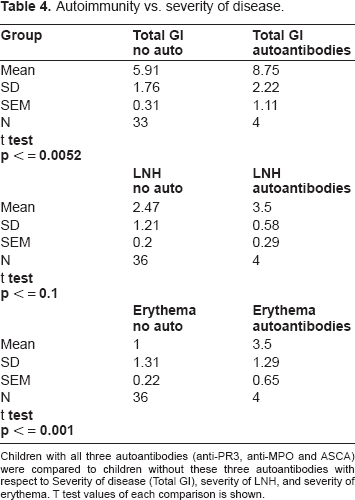
Children with all three autoantibodies (anti-PR3, anti-MPO and ASCA) were compared to children without these three autoantibodies with respect to Severity of disease (Total GI), severity of LNH, and severity of erythema. T test values of each comparison is shown.
We also found that when ANCA (anti-MPO and anti-PR3) as well as ASCA are present (compared to anti-MPO alone, anti-MPO and antiPR3 alone) there is a higher correlation to high severity GI disease in autistic children when considering Total GI disease (p < 0.08) and the presence and severity of LNH (p < 0.02) (Table 5).
Relationship between autoantibody combination and severity of disease.
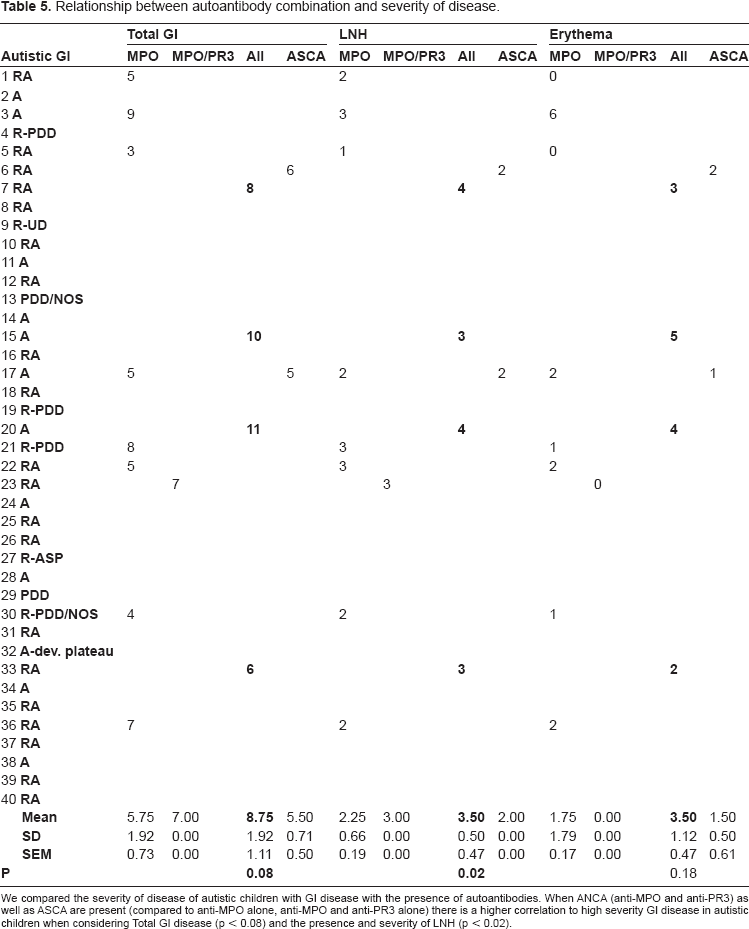
We compared the severity of disease of autistic children with GI disease with the presence of autoantibodies. When ANCA (anti-MPO and anti-PR3) as well as ASCA are present (compared to anti-MPO alone, anti-MPO and anti-PR3 alone) there is a higher correlation to high severity GI disease in autistic children when considering Total GI disease (p < 0.08) and the presence and severity of LNH (p < 0.02).
Discussion
Immune dysfunction has been reported in autistic children including autoimmunity to central nervous system (CNS) proteins.32,34 This has led to speculation that exposure of the developing neuronal system during critical periods of aberrant immune activation may result in the brain pathology of ASD. Neuroactive compounds, that share immunomodulatory properties, have been implicated in the disease process. For example, elevated platelet serotonin levels are observed in approximately one third of children with autism. 43 45 Analysis of data from small but representative groups of ASD patients has shown that approximately 30%–70% of autistic patients have circulating anti-brain autoantibodies, including autoantibodies to a serotonin receptor, 36 myelin basic protein 35 and unknown antigens from adult brain tissue extract. 46
Separate epidemiologic studies suggest that a family history of autoimmune disorders is more common among children with autism than healthy control children.38,39 There is also increased incidence of asthma, allergy, autoimmune psoriasis and Type I diabetes in mothers of children with ASD, 40 and first degree relatives of children with autism and Aspergers are more likely to have an autoimmune disease compared to controls.41,42
While significantly higher levels of autoantibodies are detected in autistic patients when compared with controls, the pathophysiological significance of these antibodies is uncertain. Moreover, their presence has not correlated with an anticipated pathological effect. For example, autoantibodies to myelin basic protein are frequently detected in autistic children, but no signs of associated demyelination have been described, 47 and anti-brain autoantibodies have also been found in patients with neurological disorders other than autism, as well as in normal individuals. This has raised the question of pathogenic significance and disease-specificity of the antibodies found in the serum of patients with autism, hypothesizing that the production of these antibodies may be secondary to innate CNS pathology and may simply be a marker of an event in the CNS that allows for the presentation of self-antigens. 48
An association with autoimmune enteropathies with specific antibodies targeted to gut epithelial cells has been shown in ASD, 49 and two separate epidemiologic studies suggest that a family history of autoimmune disorders is more common among children with autism than healthy control children.50,51
Up to this point, however, it has been unclear whether individual autistic children are positive for more than one antibody. We have previously shown that a subset of autistic children with GI disease have ANCA.30,31
The data presented here demonstrates that a small sub group of autistic children with severe GI disease have ASCA and most of these individuals also have ANCA to both PR3 and MPO. These results also suggest that this group has a generalized autoimmunity, which includes antibodies to PR3, MPO and S. cerevisiae, and, in combination, these antibodies are related to the severity of their GI disease, suggesting that autistic children with these three autoantibodies may be susceptible to the most severe GI disease that the presence of these antibodies may be a marker for this sub group of autistic children.
Disclosures
The authors report no conflicts of interest.
