Abstract
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides are a group of immune-mediated diseases characterized by inflammation of small vessels, leading to endothelial injury with subsequent tissue damage. Current guidelines recommend induction therapy with rituximab over cyclophosphamide for severe disease activity. In this case series-based review, the authors discuss 3 cases of granulomatosis with polyangiitis (GPA) with proteinase-3 (PR3) disease that deteriorated following induction therapy with rituximab combined with mycophenolate mofetil and high-dose steroids. All 3 patients subsequently required salvage therapy with cyclophosphamide. Our experience suggests there is a temporal window where induction with rituximab is not fully effective, and deterioration or death can ensue. Expert recommendations do not offer a preferential protocol for induction with either rituximab or cyclophosphamide, with some even using a combination of both.
Introduction
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides are a group of immune-mediated diseases characterized by inflammation of small vessels, leading to endothelial injury with subsequent tissue damage. Anti-neutrophil cytoplasmic antibody positivity in the blood is characteristic with 80% to 94% of patients having a positive ANCA at the time of diagnosis. The most common organs injured are the respiratory tract and kidneys. 1 Granulomatosis with polyangiitis (GPA) is associated with proteinase-3 (PR3) ANCA positivity, upper-airway (sinus, eustachian tubes) inflammation, lung nodules, and crescentic glomerulonephritis. Although a wide variety of organ manifestations have been noted. If left untreated, mortality rate is very high, up to 90% at 2 years. 2
The introduction of cyclophosphamide (an alkylating agent) to systemic glucocorticoids dramatically improved outcomes in the early 1980s. Recently, rituximab (an antibody to B cells) was found to be equally efficacious. Presently, the treatment algorithm for GPA involves induction of remission with high-dose steroids and either cyclophosphamide or rituximab for organ or life-threatening diseases. As rituximab is considered less toxic than treatment with alkylating agents, it is preferentially recommended as an initial treatment. While B-cell therapy is preferred as the initial induction agent, expert opinion remains mixed such that cyclophosphamide and even combined cyclophosphamide/rituximab therapy have its proponents.3,4
In this case series, we followed up on 3 patients with PR3 disease who deteriorated following induction therapy with rituximab combined with mycophenolate mofetil and high-dose steroids. All 3 patients subsequently required salvage therapy with cyclophosphamide.
Case 1
A 66-year-old female with a past medical history of chronic sinusitis and multiple sinus surgeries was seen by a private rheumatologist for polyarthritis 2 months prior to admission to our hospital. She was then admitted with worsening joint pain, rash, and persistent sinus symptoms. Examination confirmed polyarthritis with joint swelling, palpable purpura, and 3+ lower-extremity edema. Pertinent laboratory findings included trace proteinuria, hematuria, elevation in serum creatinine at 2.3 mg/dl (0.5-1.2 mg/dl), anemia, rheumatoid factor positivity at 45 International Units (IU) (<14 IU), and PR3 positivity at >8 Antibody Index units (AI) (<1 AI). Ultrasound-guided renal biopsy revealed acute necrotizing pauci-immune glomerulonephritis. Of note, her chest x-ray (CXR) on admission was normal (Figure 1A). She was initiated on 125-mg intravenous (IV) methyl prednisone twice daily (bid) for 5 days, 1 g of mycophenolate mofetil daily, and 1 g infusion of rituximab on day 3 of admission. Her renal function remained stable. Her sedimentation rate and c-reactive protein dropped from 92 to 57 mm/h and from 13 to 2.4 mg/dl (Figure 2), respectively, with improvement in symptoms prior to discharge. She was discharged on prednisone 60 mg and mycophenolate 1.5 g bid. Three days later, the patient was re-admitted to the hospital complaining of dyspnea and cough. She was noted to be hypoxemic. Chest x-ray (day 8 from date of first admission) (Figure 1B) and computed tomography (CT) of the chest revealed multifocal airspace infiltrates. Blood and sputum cultures showed no growth. She was intubated the next day and continued to deteriorate, eventually developing overt macrophage activation syndrome (elevated d-dimer, ferritin, triglycerides, and liver enzymes are consistent with probable macrophage activation syndrome). This was despite therapy with methylprednisolone 250 mg bid, cyclophosphamide 4 mg/kg daily, and mycophenolate. She was put on comfort care and passed away.
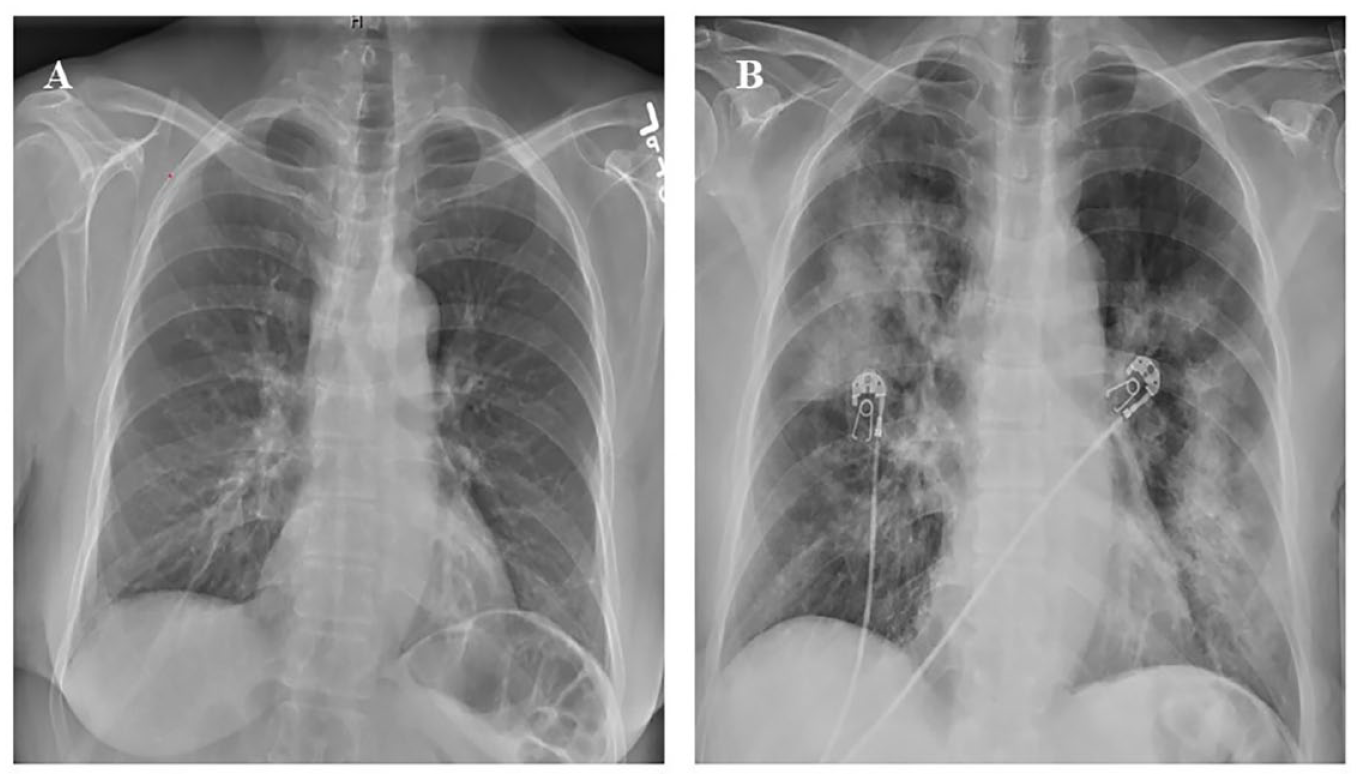
(A) Normal chest x-ray at admission. (B) Chest x-ray revealed multifocal airspace infiltrates.
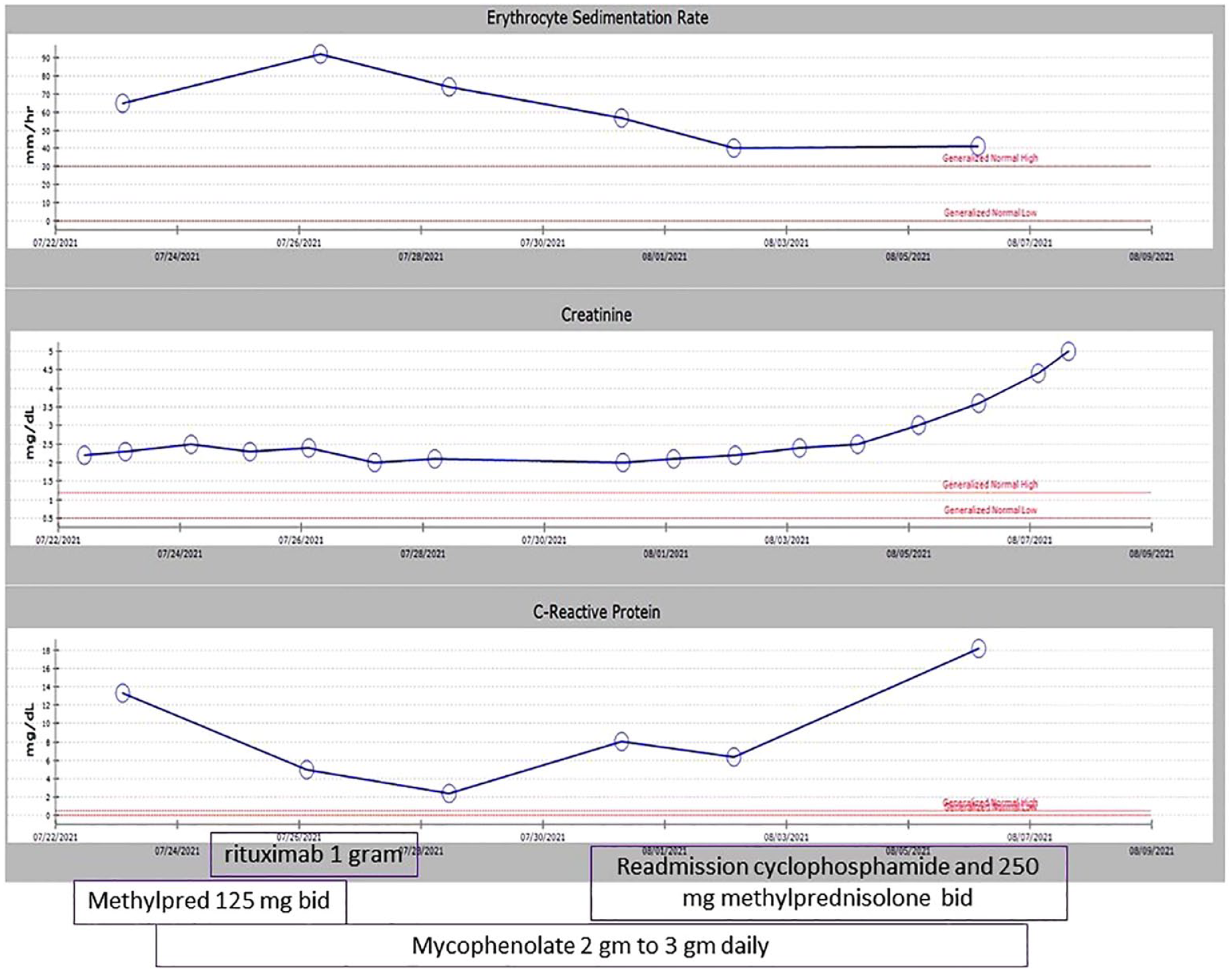
Trend of Erythrocyte sedementation rate (ESR), C- reactive protien (CRP), and creatinine during hospital admission.
Case 2
A 44-year-old male with a history of chronic serous otitis media presented to the emergency department with dyspnea. He was found to have pulmonary nodules and right pleural effusion on a CXR. Thoracocentesis relieved his dyspnea and revealed exudative fluid, which was negative for malignancy. Labs showed ESR 92 mm/h (<15 mm/h), c-reactive protein 34.7 mg/dl (<0.5 mg/dl), positive PR3 level 2.9 AI (<1 AI), and rheumatoid factor 61 IU (<10 IU). He was initially treated with mycophenolate 1.5 g bid, prednisone 40 mg daily, and rituximab infusions 1 g IV 2 weeks apart, and clinicians noticed an improvement of symptoms initially following discharge. However, he came back to the hospital 4 weeks following his second rituximab infusion with worsening shortness of breath as well as increased oxygen requirements at home. He was also found to have a new-onset left-sided pleural effusion, new-onset renal insufficiency creatinine was 2.2 mg/dl from a baseline of 1.0 mg/dl (Figure 3), and urine analysis revealed active urinary sediment (proteinuria, red blood cell [RBC] casts, and white blood cell [WBC] casts). His PR3 antibody was negative, and blood and urine cultures were negative. Thoracocentesis was repeated, and 50 ml of red cloudy fluid was removed. Renal involvement was presumed to be secondary to ANCA-associated vasculitis. He was started on IV cyclophosphamide at a dose of 4 mg/kg for 4 days, then 150 mg orally daily. He was discharged on prednisone 50 mg daily, mycophenolate mofetil 3 g daily orally, and trimethoprim-sulfamethoxazole for pneumocystis prophylaxis. He improved as an outpatient with normalization of renal function and was eventually tapered off cyclophosphamide and steroids. He continued to follow up with rheumatology and pulmonology.
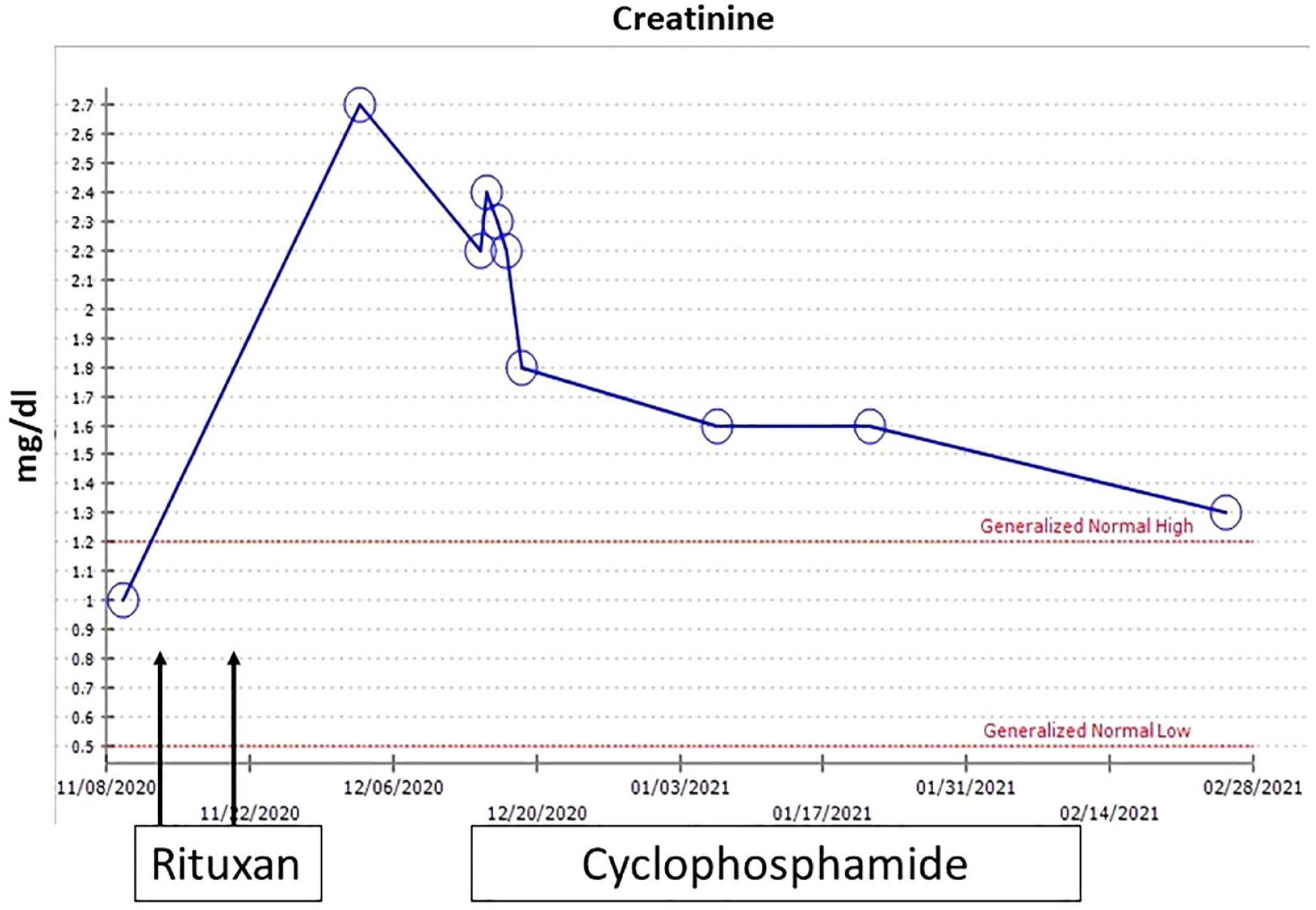
Trend of creatinine during hospital admission.
Case 3
A 34-year-old male was evaluated by ENT for ear pain and facial paralysis with serous otitis requiring tube placement. He had lost 25 pounds, and his laboratory studies revealed positive PR3 3.0 AI (<1 AI), rheumatoid factor 23 IU (<14 IU), and c-reactive protein at 8.1 mg/dl (<0.5 mg/dl). He was started on prednisone 60 mg and referred to rheumatology by his ENT physician. His CXR showed bilateral perihilar masses and multiple pulmonary nodules (Figure 4A). Urinalysis and renal function were normal. He was diagnosed with GPA and was started on rituximab 375 mg/m2 infusions for 4 weeks. One month after completing rituximab infusion, he was seen in the clinic with shortness of breath and hemoptysis. Mycophenolate mofetil was added to his treatment. Follow-up CXR (Figure 4B) showed worsening pulmonary infiltrates, and the patient was referred to pulmonology for further assessment. Bronchoscopy was performed and showed erythema, severe inflammation, and areas of ulceration of the mucosa throughout the tracheobronchial tree as well as copious thick secretions. No infectious pathogens were identified on bronchoalveolar lavage. The patient was started on cyclophosphamide infusions 4 mg/kg daily ×4 then oral cyclophosphamide 150 mg orally daily. Proteinase 3 at this time was negative. On follow-up in clinic 1 month later, there was significant improvement of respiratory symptoms and CXR findings. Prednisone was tapered off, and he was continued on cyclophosphamide for 4 months to complete induction therapy.

(A) Chest x-ray showed bilateral perihilar masses and multiple pulmonary nodules. (B) Chest x-ray showed worsening pulmonary infiltrates.
Discussion
Recent guidelines were developed for the treatment of ANCA-associated vasculitis by the American College of Rheumatology and Vasculitis Foundation. In these guidelines, severe disease was defined as a life- or organ-threatening disease, for example, glomerulonephritis or alveolar hemorrhage. Treatment recommendations for an active severe disease included the use of high-dose or pulse steroids in the induction of remission. They also recommend the use of rituximab over cyclophosphamide, as it is considered less toxic, with cyclophosphamide to be only used in cases of a contraindication to or treatment failure with rituximab. 4 These recommendations are based on the RAVE trial (Rituximab in ANCA-Associated Vasculitis), which was a noninferiority study, comparing induction of remission of severe ANCA-associated vasculitis using initial dosing of pulse methyl prednisone 1 g for 3 days coincident with either rituximab versus cytotoxic therapy with daily oral cyclophosphamide at 2 mg/kg/day. The primary endpoint of the study was complete remission and discontinuation of steroids by 6 months. A total of 197 patients were recruited, with 99 patients in the rituximab group and 98 in the cyclophosphamide group. Of those patients, 28 and 27 patients had alveolar hemorrhage from the rituximab and cyclophosphamide groups, respectively. Of those, 16 and 11 patients from each respective group reached complete remission, with no statistically significant difference between both groups. One of the limitations of this study was that patients with severe pulmonary disease requiring ventilator support and those with advanced/severe renal disease with creatinine greater than 4 mg/dl were excluded. 5 Alternatively, recently updated recommendations for induction therapy do not offer a preferential protocol and include rituximab and cyclophosphamide, with some experts even using a combination of rituximab and cyclophosphamide. 3 While rituximab in most cases has less toxicity than cyclophosphamide in both short and long term, our experience suggests there is a temporal window where induction with rituximab is not fully effective, and deterioration or death can ensue. It should be noted that the steroid dose we used was less than that recommended in the 2 published protocols. Cyclophosphamide is an alkylating agent and a potent immunosuppressive, and short-term complications include leukopenia and hemorrhagic cystitis, both can be easily managed and avoided with the use of IV dosing and careful monitoring of peripheral blood cell count, avoiding leukopenia. Major adverse effects such as the development of malignancy or infertility are more related to the long-term treatment much longer than the 3 to 6 months needed to cover for our observed refractoriness to the initiation of rituximab therapy. Cases 2 and 3 had negative cultures on subsequent presentation and responded well to cyclophosphamide, negating the possibility of an infection as the cause of deterioration. Our use of cyclophosphamide with mycophenolate in induction therapy was to maximize immunosuppression following initial treatment failure, based on positive results in previous studies. 6 Also, it is interesting to note that the 2 male patients clinically deteriorated in the context of PR3 negativity following rituximab therapy. This calls into question the concept that ANCA antibodies are directly pathogenic.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patients for their anonymized information to be published in this article, and verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article
