Abstract
New antiretroviral agents that are better tolerated with less side effects and novel resistance patterns are needed at all lines of human immunodeficiency virus (HIV) therapeutic strategies. The CC-chemokine receptor 5 (CCR5) antagonist maraviroc is a member of the novel class of “antiretroviral agents” that prevents the entry of HIV-1 into host cells by blocking the CCR5 coreceptor. In the MERIT (Maraviroc versus Efavirenz in Treatment-Naïve Patients) study in antiretrovial-naïve patients aged ≥16 years with CCR5-tropic HIV-1 infection, maraviroc showed noninferiority to efavirenz for virological endpoints. Evidences from trials suggest that maraviroc is effective at reducing HIV-1 viral load in antiretroviral-experienced and -naïve patients with CCR5-tropic virus, as well as in those with CCR5-tropic virus who have developed HIV-1 resistance to existing antiretroviral regimens. Recent in vitro study demonstrated that maraviroc was also active against CCR5-tropic HIV-2 strains.
Introduction
According to the UNAIDS Report 2010 it is estimated that 33.3 million people worldwide are living with human immunodeficiency virus (HIV), and that ca. 2.6 million people were newly infected with the virus in 2009. This is more than one fifth (21%) fewer than the estimated 3.2 million in 1997, the year in which annual new infections peaked. Interestingly, the number of annual AIDS-related deaths worldwide is steadily decreasing from the peak of 2.1 million in 2004 to an estimated 1.8 million in 2009 (UNAIDS). 1 These epidemiological statistics underscore the need for new and improved therapies to prevent and manage HIV-1 infection. There are currently several classes of drugs available which target different stages of the HIV-1 lifecycle including: nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), 2 non-nucleoside reverse transcriptase inhibitors (NNRTIs),3,4 protease inhibitors (PIs), 5 integrase 6 and fusion inhibitors.7,8 First-line treatment for HIV-1 infection comprises a combination of the above mentioned antiretroviral drugs, known as highly active antiretroviral therapy (HAART).9–11 Nevertheless, the inconvenience of these drug regimens and drug toxicity often leads to poor adherence to therapy and facilitates viral resistance to drug classes. Therefore, treatment options are limited in patients resistant to one or more drug classes, and drug-resistant virus may be transmitted to treatment-naïve patients. New drugs with novel mechanisms of action are therefore being developed, targeting alternative stages of the HIV-1 life cycle, which are effective against both wild type HIV-1 and of resistant HIV-1 strains to commonly used HAART.11,12 HIV-1 entry in to the CD4+ T lymphocytes relies on 2 receptors to infect CD4+ T cells, the primary receptor, and either C-C chemokine receptor 5 (CCR5) or C-X-C chemokine receptor 4 (CXCR4), which serve as coreceptors. Most infections involve transmission of CCR5-tropic virus, but over the course of infection CXCR4-tropic virus emerges in about half of the HIV-1 patients.13–18 A decade after its discovery, maraviroc19–21 is as the first CCR5 antagonist which was given accelerated approval in August 2007 and full approval in November 2008 by the US Food and Drug Administration (FDA) 22 for use in treatment-experienced adult patients with only CCR5-tropic HIV-1 in combination with other antiretroviral therapies and later on by the European Medicines Evaluation Agency (EMEA). 23 Maraviroc inhibits entry of CCR5-tropic virus, but it has little or no activity against CXCR4-using virus. The CCR5 antagonists are a novel class of antiretroviral agent that prevent entry of HIV-1 into host cells by blocking the CCR5 co-receptor. CCR5 is expressed on a number of cells including activated T lymphocytes, macrophages, and dendritic cells, and CCR5-tropic HIV-1 strains are predominantly involved in transmission of the virus. 24
Maraviroc is effective and well tolerated in HAART-experienced and -naïve patients with HIV-1 infections carrying the CCR5 tropism. Data from the maraviroc trials, the MOTIV ATE (Maraviroc versus Optimized Therapy in Viremic Antiretroviral Treatment-Experienced Patients)25–28,31,32 1 and 2 studies and the MERIT (Maraviroc versus Efavirenz in Treatment-Naïve Patients)29,30 study, indicate that maraviroc significantly increases CD4+ cell counts compared with placebo in treatment-experienced patients and to a similar extent as efavirenz in antiretroviral-naïve patients. Even in cases where viral load is not completely suppressed, maraviroc improves immunological response compared with placebo. Maraviroc is a promising antiretroviral agent that does not have any cross-resistance with drugs from other classes.
This review focuses on the main characteristics of maraviroc and the latest data regarding its clinical application in antiretroviral-naïve HIV-1 patients with CCR5-tropism.
Chemistry
Maraviroc is a selective, slowly reversible, small molecule which is available as film-coated tablets for oral administration containing either 150 or 300 mg of maraviroc and the following inactive ingredients: dibasic calcium phosphate, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. Maraviroc is chemically described as 4,4-difluoro-N-{(1S)-3-[exo-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]oct-8-yl]-1-phenylpropyl} cyclohexanecarboxamide
33
(Fig. 1).
Chemical structure of maraviroc.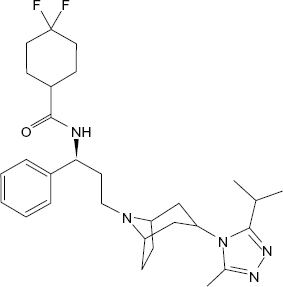
Pharmacokinetics
Recommended maraviroc dosing regimens depending on interactions with other concomitant medications.
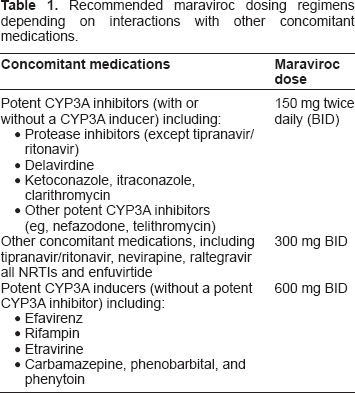
Maraviroc pharmacokinetics was studied in phase I/IIa studies and phase IIb/III studies. The absorption of maraviroc is highly variable with multiple peaks. The mean Tmax was between 2 and 3 hours with individual values ranging from 0.5 to 8 hours (with food). The absolute bioavailability for maraviroc was 23% at 100 mg and has a predicted bioavailability of 31% at 300 mg. The absorption of maraviroc is dose dependent, likely attributed to saturated efflux transporters in the intestine.
Maraviroc binds to both albumin and α1-acid glycoprotein. The plasma protein binding is between 73% and 78% and blood plasma ratio around 0.59 suggesting limited distribution into blood cells. Maraviroc is mainly eliminated by metabolism. In vitro, CYP3A433,37,41 is the main enzyme responsible for maraviroc metabolism (no significant contribution of CYP2D6, CYP3A5 and CYP2B6). 59 After intravenous administration of 30 mg maraviroc (approximately corresponding to exposure after oral administration of 100 mg) total clearance was 44 l/h and renal clearance 10.2 l/h. The terminal half-life was 13 hours after intravenous dose and around 16 hours after multiple oral doses of 300 mg. Maraviroc was excreted upto 19.6% in urine and 76.4% in faeces. Of the excreted quantities, unchanged maraviroc constituted on average 25% in faeces and 8% in urine.34,38–45
Pharmacodynamics
The pharmacodynamics of maraviroc in patients with CCR5-tropic virus was evaluated in two phase II dose ranging studies and two identical phase III studies. In a supportive study, the viral activity was studied in patients with non-CCR5-tropic virus.34,35,46–48
Mechanism of action
It is essential for HIV-1 to bind to the host cell in order to enter it, to complete the replication, and to release new virions to further propagate infection. CCR5 was first characterized as a receptor for MIP-1a, MIP-1b, and RANTES, and was rapidly shown to be the main coreceptor for M-tropic HIV-1 strains and simian immunodeficiency virus (SIV).48,49 Maraviroc as a CCR5 antagosnist acts by selectively binding to the human chemokine receptor CCR5 with a KD of 0.86 nM and inhibiting the interaction of the envelope glycoprotein (gp120) from CCR5-tropic HIV- 1 strains with CCR5. Binding of gp120 to CCR5 is an essential step in the HIV-1 entry process for CCR5-tropic strains. Site directed mutagenesis and computer modeling studies locate the likely binding site of maraviroc to a pocket within the transmembrane region of CCR5. As a consequence of this binding, maraviroc is thought to alter the three dimensional structure of CCR5 such that the viral envelope glycoprotein, gp120, is unable to recognize and bind to the co-receptor.49–51
Antiviral activity in vitro
Maraviroc selectively inhibits CCR5-tropic HIV-1 replication in vitro and was inactive against CXCR4-tropic laboratory-adapted HIV-1 isolates, indicating that the antiviral mechanism of maraviroc is exclusively CCR5-mediated. It also had no significant activity against a range of pharmacologically relevant enzymes, ion channels, and receptors, indicating the potential for a safe clinical profile. 52 Evidences suggests that there is no cross-resistance in vivo and in vitro [phytohemoagglutinin (PHA)-stimulated peripheral blood lymphocytes] between maraviroc and other entry inhibitors.53–56
Modeling studies demonstrate that the CCR5 antagonists bind in a similar pocket of the CCR5 receptor within the transmembrane region. The authors proposed that these compounds may hold the receptor in different conformations, thereby inhibiting entry of maraviroc-resistant variants. Data from two phase II studies in CCR5-tropic, HIV-1 infected patients treated with maraviroc for 10 days demonstrated that at day 11 CXCR4-tropic variants were detected in two patients Clonal analysis suggested that this was probably due to the outgrowth of a pretreatment CXCR4-tropic reservoir, and not due to coreceptor switching under selective pressure from maraviroc.53–56
Drug-drug-interactions
Maraviroc is a P-glycoprotein substrate and mainly metabolised by CYP3A4, thus, the interaction potential when co-administered with other antiretroviral agents is large. A number of interaction studies with substrates affecting CYP3A4, P-glycoprotein as well as renal secretion were performed in healthy subjects (Table 1).33,38–45,57–60
Adverse events of maraviroc in clinical trials
Adverse reactions most commonly associated with maraviroc-based therapy in both treatment-experienced and treatment-naïve patients include colds, cough, fever, skin rash, dark urine, weakness, swelling of face, lips, tongue, or throat, gastrointestinal adverse effects including gas and bloating, and dizziness. Because of the potential for hepatotoxicity, caution is advised when treating patients with preexisting liver dysfunction or who are coinfected with hepatitis B or C virus. The analyis of malignancies observed after week 48 did not demonstrate a difference between maraviroc and placebo recipients in both the unadjusted (4.3%–4.5% versus 5.3%, respectively) and exposure-adjusted (4.3–4.5 versus 7.1 per 100 patient-years) incidences at the end of blinded treatment. Whereas exposure adjusted incidences of malignancies were noted to be decreasing after week 48. There was no association of any neoplasm with maraviroc treatment compared to placebo.27,31
Resistance to Maraviroc
The high error rate of HIV-1 reverse transcriptase and rapid turnover of the viral population facilitate the emergence of drug-resistant mutants under conditions of partial drug efficacy. In the case of CCR5 inhibitors, the drug target is a host cell protein that will not undergo mutation in response to CCR5 antagonist therapy. However, viral adaptation to CCR5 inhibitors could involve changes in the viral envelope protein that alters dependence on CCR5. Resistance to maraviroc can occur in either by mutations that allow HIV-1 to use CXCR4 coreceptors (tropism switch) or by mutations that allow HIV-1 to continue using the CCR5 coreceptors, despite blockade by maraviroc. Tropism switch is of particular concern, because the presence of virus that can use the CXCR4 coreceptor has been associated with a more rapid decrease in CD4+ lymphocyte count and faster disease progression. 61 The frequency with which coreceptor switching occurs is unknown. Switching involves the development of multiple mutations throughout gp160, with resulting lowered replication capacity (fitness) and less efficient use of both CCR5 and CXCR4. 62
Recent evidences were presented by Thielen et al 63 and Dimonte et al. 64 Several mutations within gp41 were highly significant for CXCR4 usage; most strikingly an insertion occurring in 7.7% of HOMER-R5 and 46.3% of HOMER-X4 samples (MOTIVATE 5.7% and 25.2%, respectively). 63 Dimonte et al 64 showed that gp120V3 and several specific amino acid changes in gp41 are associated together with CXCR4 and/or CCR5 usage. Determinants of tropism may reside outside the V3-loop, even in the gp41. 64
Maraviroc is a promising new antiretroviral agent that does not have any cross-resistance with drugs from other classes. However, cross-resistance to maraviroc has been described with AD101 com-pound and to viriviroc with other CCR5 inhibitors. However, maraviroc-resistant strains were sensitive to ancriviroc, vicriviroc and aplaviroc.22,31,32 It has been reported that mutations conferring maraviroc resistance accumulate mainly in the V3 loop region (A316T, I323V) of gp120, which binds to the CCR5 co-receptor, but also in other regions of gp120 as well, for instance V1, V2, or V4. 65 Furthermore, resistance to CCR5 antagonists was found to be dependent on several conditions such as the cells used, the availability of CCR5 or the viral strain, generating resistance through different mechanisms (allosteric, competitive, co-receptor switch), and even different complex patterns of mutations. Moreover, the prevalence of maraviroc resistance mutations is low in maraviroc-naïve HIV-1-infected individuals. 66
Tilton et al 67 suggest a model of CCR5 cross-resistance whereby viruses that predominantly utilize the N terminus are broadly cross-resistant to multiple CCR5 antagonists, whereas viruses that require both the N terminus and antagonist-specific extracellular loops changes demonstrate a narrow cross-resistance profile. Among HIV-1 patients who are exhibiting failure of a maraviroc-based regimen, the vast majority appeared to fail with either a CXCR4- or a CCR5-tropic HIV-1 that lacked any in vitro evidence of CCR5 inhibitor resistance. CCR5 resistance can occur, however, resistance to maraviroc requires the emergence of multiple mutations with the V3 and perhaps V4 loops. The relative fitness of this resistant virus remains to be defined but is likely to be given its remarkable ability to replicate in vivo (high viral load) and to replicate in the presence of very low levels of CCR5. It remains to be determined whether patients with viruses capable of utilizing low CCR5 levels are more apt to fail maraviroc regimen due to mutations in Env that enable the use of drug-bound CCR5. If this is the case, cross-resistance to other CCR5 inhibitors may not be inevitable due to differences in how Env can interact with distinct CCR5 domains. 67
Safety and Tolerability
Assessment of maraviroc pharmacokinetics and tolerability in human subjects is critical for determining safety, appropriate dosing, and feasibility of further efficacy studies. Evidence from a combination of five phase I/IIa double-blind, placebo-controlled, multiple-dose studies with maraviroc alone, and one drug-drug interaction study with oral contraceptives have demonstrated that maraviroc is well tolerated at doses up to 300 mg twice daily. Five of these studies were of 7 to 10 days' duration including dosing at the maximum dose of 1200 mg once daily. Results from safety study conducted for 28 days at the highest proposed clinical dose of 300 mg BID. No serious adverse events were reported and the adverse event profile was similar to placebo.32–43
Hepatic safety and monitoring of liver enzymes is a critical consideration in the development of any new drug. Although several subjects showed increases in hepatic transaminases, there was no clear relationship with dose, and most cases were not significantly above the upper limit of normal. None of the increases in transaminases was associated with increases in bilirubin. 68 Small apparently dose-related increases in total cholesterol and LDL-cholesterol were described by Abel et al. 40 However the clinical relevance of these changes in an inpatient setting where subjects have a change in diet and lifestyle is unclear. Subsequent study examining the safety and toleration of maraviroc 100-mg and 300-mg BID doses in healthy volunteers confirmed that these doses were well tolerated, with no trend changes in liver enzymes, lipid profile, blood pressure, pulse rate or ECG parameters. 40 Abel et al 33 reported differences in maraviroc pharmacokinetics in subjects with hepatic impairment compared with those with normal hepatic function. However, these do not currently support a dose modification since the single 300 mg dose of maraviroc was well tolerated by subjects with normal and impaired hepatic function. Also Ayoub et al 68 suggest that maraviroc does not present significant risks to hepatic safety when taken at the recommended doses in the populations studied (MOTIVATE 1 and 2).
Maraviroc should only be used in pregnant women if the potential benefit justifies fetal risk; lack of pediatric clinical data precludes its use in patients younger than 16 years. 45 Briefly the pharmacokinetic and safety profile of maraviroc ascertained in different reports32–43 support its future clinical development.
Genetic Characteristics in HIV-1 Pregression and AIDs
A number of individuals have genetic mutation that induces resistance or partially “immunity” to HIV-1 infection. A 32-base pair deletion (Δ32) in the CCR5 gene is responsible for this positive effect. Individuals with this mutation either have no CCR5 receptors or have far fewer CCR5 receptors than an average population. This greatly reduces the possibility of HIV-1 infection or progression of the disease to AIDS. Rare cases of HIV-1 infection in individuals homozygous for CCR5Δ32 have been reported. 69 In all these instances, exclusive use of CXCR4 by virus isolates, or presence of V3 variable loop envelope sequences typical of CXCR4-tropic viruses were reported. A so-called HIV mirrorTM technology (National Institute of Health-funded research project) detects the two genes, CCR5Δ32 and CCR2-64I, which are well-known to slow down the HIV-1 disease progression. For instance, Samson et al 70 found CCR5-Δ32/Δ32 in 8 (1.1%) of 704 HIV-seronegative Caucasians compared with 0 of 723 HIV-1 seropositive Caucasians; CCR5-Δ32/Δ32 individuals were not found among persons from Western or Central Africa or Japan. Increased prevalence of CCR5-Δ32/Δ32 also has been found in HIV-1-exposed but uninfected men who have sex with men (MSM), injection drug users (IDUs), hemophiliacs, and uninfected infants of HIV-1-infected mothers. The prevalence of the CCR5-Δ32/Δ32 genotype can be substantial among persons persistently exposed to HIV-1 who have nonetheless remained HIV-1 seronegative.69–78 In cross-sectional and longitudinal study analyses reported by Marmor et al 77 indicate that among high-risk HIV-1 seronegative MSM, CCR5-+/Δ32 (heterozygous) and CCR5-Δ32/Δ32 (homozygous) are associated with protection against HIV-1 infection. These imply that strategies aimed at reducing susceptibility to HIV-1 infection by blocking CCR5 receptor sites need not seek blockage of all receptor sites to achieve a substantial degree of protection. 77
Coreceptor Tropism Analysis in HIV-1 Patients
Nowadays, coreceptor tropism analysis for different HIV-1 subtype strains is recommended whenever the use of CCR5 antagonists is contemplated. The HIV-1 tropism frequently changes over the course of the disease. CCR5-tropic virus predominates in early stage of infection and in treatment-naïve patients.79,80 CXCR4 tropism increases both during disease progression and antiretroviral treatment. 79 In later disease stage, CXCR4 tropism emerges in about 20% of treatment-naïve patients. 80 A number of the antiretroviral-experienced patients present CXCR4-tropic HIV-1 variants. 79
Infectious Diseases Society of America guidelines agree that tropism testing should be done before starting any CCR5 antagonist.31,45 Coreceptor tropism testing might also be considered for patients who exhibit virologic failure on a CCR5 inhibitor, eg, maraviroc. Treatment failure is often associated with a switch to CXCR4 tropism.
65
Several pheno- and genotypic methods to establish viral use of coreceptors have been developed, including virus phenotype assays in cell culture and HIV-1-envelope sequence-based predictors.
79
Phenotypic testing was the first HIV-1 tropism assay available, was the exclusive test used in maraviroc clinical trials, and is most widely recommended.31,45,81 However, tropism testing should rule out CXCR4-tropic HIV-1 before treatment with CCR5 antagonists, eg, maraviroc.81,82 Coreceptor phenotype assays, such as the PhenoSense HIV-1 Coreceptor Tropism (Trofile) used in the maraviroc trials, are based on the amplification of HIV-1
Genotypic HIV-1 tropism methods represent the alternatives to phenotypic tropism assays84,85 and Enhanced Sensitivity Trofile Assay (ESTA), which is currently the most commonly used clinical phenotypic tropism assay. 86 For instance, deep sequencing is a highly sensitive technique that can detect and quantify the proportion of non-CCR5 HIV-1 variants, which may emerge and cause virologic failure in patients who receive maraviroc-containing regimens. Reanalysis of the MERIT trial using deep V3 loop sequencing indicates that, had patients originally been screened using this method, the maraviroc arm would have likely been found to be noninferior to the efavirenz arm.87,88
Although phenotypic tests of coreceptor usage are generally considered the gold standard, the expense and long turnaround time (4–6 weeks) of these assays have spurred the development of genotypic methods as an alternative. Genotypic determination of HIV-1 coreceptor usage is based on sequencing the V3-coding region of HIV-1
Non-CCR5 tropism seemed to be overrepresented among clade B-infected individuals, with 74% of the deep sequencing non-CCR5 group consisting of clade B-infected patients, which was higher than the overall clade B composition of 60%. Conversely, HIV-1 subtype C was underrepresented among non-CCR5–tropic HIV-1 patients, at 15%. Deep sequencing and ESTA (Enhanced Sensitivity Trofile Assay) had similar performance in predicting virologic outcome in non-clade B-infected patients.87,88
Genotypic methods are anticipated to be used more frequently in the clinical setting because of their greater accessibility, lower cost, and faster turnaround time than other methods. For the interpretation of V3 loop genotyping, clinically validated systems should be used when possible. Laboratories doing HIV-1 tropism tests should have adequate quality assurance measures. For practical reasons, genotypic population sequencing is the preferred method in Europe. 91
Clinical Maraviroc Trials
There have been various clinical trials of maraviroc to date, among these MOTIVATE (Maraviroc versus Optimized Therapy in Viremic Antiretroviral Treatment-Experienced Patients) 1 and 2 trials and MERIT (Maraviroc versus Efavirenz in Treatment-Naïve Patients) trial being the major basics for maraviroc approval for therapy in CCR5-tropic HIV-1 patients.30,92–94
MOTIVATE studies
MOTIVATE25–28,31,32 1 and 2 are identical Phase III studies comparing maraviroc (once and twice daily) plus OBT (optimized background therapy) with OBT + placebo in treatment experienced, CCR5-tropic HIV-1 infected patients. The studies differed only in the regions in which they were conducted. In MOTIVATE 1 (US and Canada), of the 601 patients enrolled, with the median entry CD4 counts were 150 to 168 cells per mm3 with viral loads of 4.84 to 4.86 log10 copies per mL, 585 were treated. Roughly 90% of patients were male and 80% were white. A total of 40% patients received a regimen containing enfuvirtide, while approximately 70% subjects had two or fewer active drugs in their OBT arm.
In MOTIVATE 2 (US, Europe, and Australia), 464 of 475 enrolled subjects received treatment with CD4 counts of 174 to 182 cells per mm3 and mean viral loads between 4.84 and 4.89 log10 copies per mL. Mean change in HIV-1 viral load at 24 weeks was -0.93 log reduction for placebo, and -1.95 and -1.97 for OBT + maraviroc once and twice daily, respectively. Viral load reductions below 50 copies per mL were seen in 45.6% of subjects receiving 300 mg maraviroc QD compared to 40.9% receiving maraviroc BID, and 20.9% receiving placebo. Increase in mean CD4 counts were: 64/mm3 in placebo, 112/mm3 in OBT + maraviroc QD, and 102 cells per mm3 in OBT + maraviroc BID. In contrast to MOTIVATE 1, no increase in liver enzyme elevations was observed in subjects taking maraviroc over those taking placebo. Inclusion of maraviroc and at least one other active antiretroviral, either enfuvirtide or lopinavir/ritonavir depicted here, led to a greater percentage of subjects achieving viral loads <50 copies/mL.26–28,56 In the MOTIVATE trials 43% of patients failing maraviroc exclusively harboured CCR5-tropic viruses.26,27
The results of the trials were pooled for analysis with a total of 1,049 patients (414 maraviroc once daily; 426 maraviroc twice daily; 201 placebo (OBT)). It is notable that 955 other patients who were screened for these two studies were excluded due to dual, mixed or CXCR4 tropism. The primary study endpoint, the mean change from baseline in the log10 HIV-1 RNA levels at 48 weeks, was -1.68 for maraviroc once daily, -1.84 for maraviroc twice daily and -0.79 for the placebo arm. More importantly, the percentage of patients achieving a viral load of <50 copies/mL at 48 weeks was 43% for maraviroc once daily, 46% for maraviroc twice daily and 17% for the placebo arm, which were statistically significant between each maraviroc arm and placebo arm. There were also statistically significant greater mean CD4 cell count increases from baseline in the maraviroc arms (116 cells/mm3 in maraviroc once daily, 124 cells/mm3 maraviroc twice daily, 61 cells/mm3 in placebo). 28 Subanalyses of the pooled MOTIVATE results revealed a treatment benefit of maraviroc in combination with OBT when compared to placebo in combination with OBT.25,26 Importantly, these analyses revealed a significant benefit of using an additional active new antiretroviral agent maraviroc with optimized background therapy (OBT). The results of these studies led to FDA approval of maraviroc for treatment-experienced patients.
MERIT study
The success of maraviroc in the treatment experienced trials and the fact that a CCR5 antagonist would have the greatest potential for effectiveness in populations with predominantly CCR5 tropic virus led to an evaluation of maraviroc in antiretroviral treatment naïve HIV-1-infected patients. The MERIT (Maraviroc versus Efavirenz in Treatment-Naïve Patients) trial was a phase IIb/III, double-blind, placebo-controlled trial that evaluated the safety and efficacy of maraviroc vs. efavirenz in patients with CCR5 tropic virus from Australia, Europe, North America, South America and South Africa. 30 MERIT was a 3 arm comparison study of 300 mg maraviroc (QD), 300 mg maraviroc (BID) versus 600 mg efavirenz QD (each in combination with Combivir [AZT/3TC] 300 mg zidovudine and 150 mg lamuvidine BID).
Similar to the MOTIVATE trials25–28,31,32 patients were initially randomized to receive maraviroc 300 mg once or twice daily or efavirenz 600 mg once daily in combination with coformulated zidovudine and lamivudine. In contrast to the MOTIVATE trials, only 17% of the patients screened for the MERIT study were excluded for having CXCR4 tropic virus. In total, 895 patients were randomized, but an interim analysis found that patients receiving once daily maraviroc fell outside of the prespecified thresholds for non-inferiority in comparison to efavirenz. At week 16, the data and safety monitoring board closed the once-daily maraviroc arm because of inadequate response rates; leaving only the twice-daily maraviroc and efavirenz arms for comparison (total 721 patients). At baseline, the groups had similar median CD4 counts (241 and 254/mm3, respectively) and mean viral loads (approximately 4.9 log copies/mL).
The data and safety monitoring board discontinued the once daily maraviroc arm. This left 721 evaluable patients in the 48 week analysis. In the primary analysis of viral load response <400 copies/mL, twice daily maraviroc was non-inferior to efavirenz. However, in the co-primary endpoint of percentage of patients with viral load <50 copies/mL (65.3% maraviroc, 69.3% efavirenz), the noninferiority criterion was not met. There were also lower virologic response rates noted with maraviroc in high baseline viral load patients, Southern hemisphere patients, black patients and those with non-B HIV-1 subtype. Poorer response rates were observed for maraviroc in black subjects, in southern hemisphere patients, in patients with subtype C virus and in female subjects. This may indicate that maraviroc performs less well in patients with non-B subtypes (prevalent in treatment-naïve patients in Europe).94–96 This could also be related with the Trofile assay, since 23% of those with subtype C had non-reportable viral tropism at screening, as compared to 6% of those with subtype B. 95
At time of treatment failure, 33 (60.0%) subjects in the maraviroc 300 mg BID treatment group and 8 (34.8%) subjects in the efavirenz 600 mg QD treatment group had resistance to zidovudine/lamivudine. One (1.8%) subject in the maraviroc 300 mg BID treatment group and 14 (60.9%) subjects in the efavirenz 600 mg QD treatment group had resistance to efavirenz. And at the time of treatment discontinuation, 40 (31.0%) subjects in the maraviroc 300 mg BID treatment group and 8 (6.5%) subjects in the efavirenz 600 mg QD treatment group showed resistance to zidovudine/lamivudine. One (0.8%) subject in the maraviroc 300 mg BID treatment group and 16 (13.0%) subjects in the efavirenz 600 mg QD treatment group showed resistance to efavirenz. However, the majority of those failing with maraviroc, still had CCR5-tropic virus at time of treatment failure. 30
At 48 weeks, 65.3% of the twice-daily maraviroc group and 69.3% of the efavirenz group achieved viral loads <50 copies/mL. Although the difference in response rates was relatively modest, it failed to meet the preselected criteria for noninferiority because the lower bound of the 97.5% confidence interval was greater than -10% (-10.9%). Interestingly, if <400 copies/mL had been used as the cutoff point for virologic response, noninferiority would have been established (lower bound, -9.5%). At week 96, 12 subjects (28%) with CCR5 tropism at baseline failed with CXCR4-using virus. Considering the mean increases in CD4+ cell count from baseline to time of failure in the maraviroc group including in subjects who had CCR5 tropic virus at baseline and failed with CXCR4-using virus; and the slightly lower incidence of CDC category C infection and malignancy events in subjects receiving maraviroc, it seems that there is no adverse immunological outcome in subjects failing maraviroc with CXCR4-using virus.29,30,32
Upon blinded retesting of screening specimens with the enhanced TrofileTM assay, 15% of patients had CXCR4 tropic virus. Post hoc analysis of the study with these CXCR4 tropic patients excluded revealed that maraviroc twice daily met the non-inferiority criteria with efavirenz (viral load <50 copies/mL: 68.5% maraviroc vs. 68.3% efavirenz). Additionally, response rates for maraviroc in the subgroup analyses improved after reanalysis, particularly in those with high baseline viral load. Somehow, the response rates for maraviroc were higher in the Northern hemisphere, and response rates for efavirenz were higher in the Southern hemisphere. These differences might be attributed to higher adverse event discontinuation rates for efavirenz in the Northern hemisphere and higher default rates for patients receiving maraviroc in the Southern hemisphere. The differences noted by the monitoring board for the once daily maraviroc arm when compared to efavirenz that led to discontinuation of that arm were no longer outside the noninferiority thresholds in the post hoc reanalysis. In both the primary and the post hoc reanalysis, CD4 cell count increases were 26–30/mm3 higher in the maraviroc arm. Subsequent to the reanalysis of the MERIT data, maraviroc was approved for use in treatment-naïve patients by the US FDA. 22
However, according to the study by Vanderkerckhove et al 95 maraviroc has not met the criteria of potency, durability and convenience in a prospective analysis required for first-line regimens, and cannot be advocated for clinical use in treatment-naïve patients. It is already clear that the activity of maraviroc will depend to a large extent on the composition of the virus quasispecies with regard to co-receptor tropism. Due to the new developments in tropism testing and the resulting re-analysis of the samples, the interpretation of the final outcome of the MERIT data becomes challenging. The enhanced TrofileTM assay and the re-analysis of the MERIT data allow us to be more confident about picking up minor variants in the individual patient who, for specific reasons, might benefit from maraviroc (eg, high cardiovascular risk + kidney impairment). The favourable lipid profile and tolerability support the use of maraviroc as a safe alternative in a consolidation or maintenance regimen after achieving full virological suppression, especially in those subjects experiencing side effects on NNRTIs, protease inhibitors or integrase inhibitors. Maraviroc at 300 mg BID dose was safe and well tolerated in treatment-naïve subjects over the 96-week study period. 95 This promising investigation should be explored in future clinical trials.
In conclusion, maraviroc improves immunological response and has shown favourable pharmacokinetic and safety profiles in patients with high cardiovascular risk or in those co-infected with tuberculosis or hepatitis. Long-term studies are needed to confirm whether therapeutic expectations resulting from clinical trials with maraviroc translate into a real benefit for HIV-1 infected patients for whom traditional antiretroviral therapies have failed or are not suitable and in antiretroviral-naïve HIV-1 patients with CCR5 tropism.
Recent Investigations
Symons et al 97 reported the the impact of maraviroc on viruses with different co-receptor preferences in a patient with a dual/mixed (D/M) infection. Dual/mixed-tropic HIV-1 strains are predominant in a significative proportion of patients, though few information is available regarding the genetic characteristics, quasispecies composition, and susceptibility against CCR5-antagonists of the primary-isolates. This case report demonstrates that dual-tropic viruses, capable of using both co-receptors in phenotypic assays, can be inhibited by maraviroc if they have a CCR5 co-receptor preference in vivo. 97
Svicher et al 98 conducted an in vitro study and their results indicated that in both pure-X4 and R5/X4(+)-isolates, extensive prevalence of X4-using species was observed. In vitro selection-experiments with CCR5-inhibitor maraviroc showed no-emergence of X4-tropic variants for all R5- and R5(+)/X4-isolates tested. Their study shows that dual/mixed-tropic viruses are constituted by different species, whereby those with characteristics R5(+)/X4 are genotypically and phenotypically similar to the pure-R5 isolates; thus the use of CCR5-antagonists in patients with R5(+)/X4-tropic viruses may be a therapeutic-option.
HIV-1 and HIV-2 genomes differ by about 50% to 60% at the nucleotide level. Such differences may be correlated with differential response to some antiretrovirals, as observed with the natural resistance of HIV-2 to non nucleoside reverse transcriptase inhibitors, the fusion inhibitor enfuvirtide, and the decreased susceptibility to some of protease inhibitors. 99 Visseaux et al 99 investigated the HIV-2 susceptibility to maraviroc in a PBMC model, including 13 HIV-2 CCR5-tropic, 2 HIV-2 dual-tropic and 2 HIV-2 CXCR4-tropic clinical isolates. HIV-2 CXCR4-tropic virus was highly resistant to maraviroc, but the agent was active in in vitro against HIV-2 CCR5-tropic viruses. 99
Summary
Maraviroc is a CCR5 coreceptor antagonist eligible for the treatment of patients infected with CCR5-tropic HIV-1 and is also approved for this population who are antiretroviral-naïve or have been pre-treated with conventional antiretroviral therapies as well. Administered orally twice daily, maraviroc, in combination with optimized background therapy regimens, showed good virological and immunological efficacy.
Maraviroc was noninferior to efavirenz (each in combination with zidovudine/lamivudine) for the primary virological endpoints at 48 weeks in the MERIT study of antiretroviral therapy-naïve patients aged $16 years with CCR5-tropic HIV-1 infection. Maraviroc demonstrated noninferiority to efavirenz on both primary virological endpoints and demonstrated sustained virological efficacy in this patient population and improved immunological markers for up to 96 weeks. Data from the phase III programme of maraviroc, which includes the MOTIVATE 1 and 2 studies and the MERIT study, indicate that maraviroc significantly increases CD4+ cell counts compared with placebo in pre-treated patients and to a similar extent as efavirenz in antiretroviral-naïve patients. Even in cases where viral load is not completely suppressed, maraviroc improves immunological response compared with placebo.
Maraviroc usage is not recommended in individuals with dual/mixed- or CXCR4-tropic HIV-1 as efficacy was not demonstrated in a Phase II study of this patient group. Since less tropism data are available for non-B subtypes of HIV-1, tropism determination for these subtypes may have a greater degree of uncertainty. Maraviroc was generally well tolerated by both treatment-experienced and treatment-naïve patients with CCR5-tropic HIV-1 infection. Recent in vitro study reported that maraviroc was active against CCR5-tropic HIV-2 strains as well. Resistance to maraviroc has been reported, however, the prevalence of maraviroc resistance mutations is low in maraviroc-naïve HIV-1 infected individuals.
Footnotes
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
