Abstract
Background:
The feasibility of antiretroviral therapy (ART) monitoring remains problematic in decentralized HIV clinic settings of sub-Saharan Africa. We assessed the rates and correlates of HIV-1 virological failure (VF) and drug resistance (DR) in 2 pre-test-and-treat urban clinic settings of Senegal.
Methods:
Consenting HIV-1-infected adults (⩾18 years) receiving first-line ART for ⩾12 months were cross-sectionally enrolled between January and March 2015, at the referral outpatient treatment center of Dakar (n = 151) and decentralized regional hospital of Saint-Louis (n = 127). In the 12 months preceding plasma specimens’ collection patients at Saint-Louis had no viral load (VL) testing. Significant predictors of VF (VL ⩾ 1000 copies/ml) and DR (clinically relevant mutations) were determined using binomial logistic regression in R software.
Results:
Of the 278 adults on EFV-/NVP-based regimens, 32 (11.5% [95%CI: 8.0-15.9]) experienced VF. Failing and non-failing patients had comparable median time [interquartile] on ART (69.5 [23.0-89.5] vs 64.0 [34.0-99.0] months; P = .46, Mann–Whitney U-test). Of the 27 viraemic isolates successfully genotyped, 20 (74.1%) carried DR mutations; most frequent were M184VI (55.6%), K103N (37.1%), thymidine analog mutations (29.6%), Y181CY (22.2%). The pattern of mutations did not always correspond to the ongoing treatment. The adjusted odds of VF was significantly associated with the decentralized clinic site (P < .001) and CD4 < 350 cells/mm3 (P < .006). Strong correlates of DR also included Saint-Louis (P < .009), CD4 < 350 cells/mm3 (P <. 001), and nevirapine-based therapies (comparator: efavirenz-based therapies; P < .027). In stratification analyses by site, higher rate of VF at Saint-Louis (20.5% [95%CI: 13.8-28.5] vs 4.0% [95%CI: 1.5-8.5] in Dakar) was associated with nevirapine-based therapies (OR = 3.34 [1.07-11.75], P = .038), self-reported missing doses (OR = 3.30 [1.13-10.24], P = .029), and medical appointments (OR = 2.91 [1.05-8.47], P = .039) in the last 1 and 12 months(s), respectively. The higher rate of DR at Saint-Louis (12.9% [95%CI: 7.6-20.1] vs 2.7% [95%CI: 0.7-6.7] in Dakar) was associated with nevirapine-based therapies (OR = 5.13 [1.12-37.35], P = .035).
Conclusion:
At decentralized urban settings, there is need for enhanced virological monitoring and adherence support. HIV programs in Senegal should intensify early HIV diagnosis for effective test-and-treat. These interventions, in addition to the superiority of efavirenz-based therapies provide a favorable framework for transitioning to the recommended potent drug dolutegravir, thereby ensuring its long-term use.
Keywords
Background
Patients with HIV in developing countries have incurred substantial clinical benefits owing to the great efforts in the deployment of antiretroviral therapy (ART). Decentralization in this regard has been markedly instrumental, also in improving the expansion of HIV services to lower-level health delivery units. 1 As many more people harboring HIV infection continue to gain access to care, AIDS-related morbidity and mortality could be dramatically reduced from the current estimates. 2 In addition to providing therapy to patients, the World Health Organization (WHO) 3 has recommended that quantification of viral load be performed routinely amongst persons on ART. However, major gaps in terms of the feasibility and uptake of ART monitoring continue to exist. Notably as many HIV programs in low and middle income countries (LMICs) including Senegal operate under the WHO public health approach. This approach essentially recommends starting HIV-infected individuals on first-line ART regardless of the health system capacities to provide adequate treatment monitoring. 4 In this context, it has been anticipated that levels of therapeutic failures and drug resistance (DR) with associated public health costs would increase. 5 As such, it may be more puzzling to meet the third goal of the Joint United Nations Program on HIV/AIDS (UNAIDS). 6 This goal seeks to reach viral suppression for 90% of patients on ART as anticipated for by 2020 and, ultimately, 95% by 2030.
HIVDR particularly worsens the burden of HIV-1 in sub-Saharan Africa (sSA). HIV-1 resistant strains typically emerge as a result of antiretroviral (ARV) selective pressure at key stages of the virus life cycle. 7 This category of HIVDR is defined as acquired DR (ADR). Greater concerns even stem from the development of pretreatment DR (PDR). This resistance may be acquired from prior exposure to ARV drugs, such as in women under the prevention of mother-to-child transmission (PMTCT) or those re-initiating ART. 8 Large-scale studies have reported worrisome levels of HIV-1 PDR (>10%) in LMICs to non-nucleotides reverse transcriptase inhibitors (NNRTIs),9,10 formerly recommended as part of first-line treatment in combination with 2 NRTIs. These findings explain why countries in sSA are gradually substituting NNRTIs to adopt the WHO-recommended second-generation integrase-strand transfer inhibitor (INSTI) dolutegravir. 11 This potent anti-HIV drug possesses a higher genetic barrier to resistance compared to first-generation INSTI (raltegravir and elvitegravir) and NNRTIs. 12 However, like with any other combinations of ARVs, the long-term use and efficacy of first-line regimens containing dolutegravir would be safeguarded if programmatic factors are efficiently monitored along the continuum of HIV care. 13 The analysis of data on the effectiveness of a decentralized HIV care from sites where this new treatment guideline is being implemented or soon-to-be, would be insightful to HIV programs in sSA, as it would enlighten them on how they can capitalize on the benefits provided by dolutegravir-based ARTs. For example, emphasis should be put on the fact that with less effective ARTs (due to de facto monotherapy, poor drug compliance, or therapy response monitoring) the waiting time to the occurrence of virological failure (VF) or resistance mutation is expected to be shorter and vice versa. 14 In decentralized settings of Senegal, Diouara et al15,16 consistently showed higher rates of VF (23.8%-26%) associated with ADR (15.9%-17.7%) at shorter median durations on ART of 18 and 32 months. Furthermore, data on this intervention type are invaluable to ensure a smooth transitioning to dolutegravir or EFV 400 mg as recommended. 11
Senegal is located on the western coast of Africa and is one of the pioneering countries to institute a national ART program to dispense ARVs. 17 In 2003 patients living with HIV in Senegal started taking ART for free and since 2005, the number of people with HIV on ART has risen from 4407 (9.8%)18,19 to 28 960 (70.6%) in 2019. 2 As of 2008, decentralized HIV clinics other than those in the nation’s capital city Dakar began witnessing upward trends in the proportion of patients starting ART, reaching ~77% in 2019. 20 In spite of these acclaimed efforts, discrepancies in the provision of HIV services may segregate between beneficiaries in Dakar and those at decentralized sites. HIV-1 virological and DR outcomes have been widely studied among patients receiving ART in Dakar,21-25 since the Senegalese initiative for access to ARVs. Related risk factors have also been prospectively examined in a cohort sampled from the same population. 26 In a retrospective cohort design, Ngom et al 27 analyzed factors associated with ART start amongst eligible patients attending the specialized referral center of HIV care in Dakar between 1998 and 2015. However, in the decentralized setup of Senegal, only few studies have addressed the question of HIV-1 ART outcomes15,16 and the associated risk factors.28,29
This study sought to determine the prevalence and correlates of VF and DR to first-line treatment (2NRTIs + 1NNRTI) received for at least 12 months in 2 urban clinic settings of Senegal. That is before the universal eligibility for ART and introduction of dolutegravir in the Senegalese national guidelines and initiation of procurement since July 2019. 30 We stratified our analyses by clinic sites based on the fact that many observational studies in sSA are biased toward urban treatment centers.
Methods
Study design and settings
This cross-sectional study was implemented through the West African Network of Excellence for Tuberculosis, AIDS, and Malaria (WANETAM). The purpose of this multisite regional project was to evaluate the prevalence of transmitted and acquired drug resistance across 6 countries of West Africa. For the recruitment of study participants in Senegal, 2 urban clinic sites were selected: The Ambulatory Treatment Center (ATC) of the University Teaching Hospital of Fann in Dakar (West of Senegal) and the decentralized regional hospital of Saint-Louis (RHS). The ATC is a reference center focusing in the comprehensive management of patients with HIV in Senegal and was created in 1988 under the Senegalese initiative for access to ARVs. 27 The RHS is situated 250 km northwest to the capital city Dakar, in the coastal region also called Saint-Louis. This region represents 1 of the 13 decentralized regions for treating HIV in Senegal.
To improve access to ART, the Senegalese AIDS control program began piloting the universal eligibility for treatment through the program termed TATARSEN (test all, treat all, and retain all in Senegal). 20 This program started in January 2016 in the 5 regions most affected by HIV in Southern Senegal. We therefore discuss the clinical implications of the CD4 T-cell count threshold method for treatment eligibility, with the overall goal of enhancing the uptake of treatment start. Additional details on the characteristics of the sites have been previously described. 31
Study participants and ethical consideration
The largest feasible sample size of 150 was estimated per site with a precision of ±4.8% at 95% confidence interval (CI), assuming an expected prevalence of DR between 5% and 10%. However, only 127 participants were enrolled at the RHS within the time allotted for sample collection. Between January and March 2015, participants were consecutively recruited from both health facilities if they had a positive HIV-1 test, were aged ⩾18 years old and undergoing first-line ART for a minimum length of 12 months. Patients on second-line therapies were excluded, whereas previous exposure to ARV drugs for PMTCT was not an exclusion criterion. A written and signed informed consent was sought from all participants before participation. Ethical and administrative approvals were also obtained from the Senegalese National Ethics Committee for Health Research (n°0278/MSAS/DPRS/CNERS) and the Ministry of Public Health and Social Action (n°00000413/MSAS/DPRS/DR).
For each participant, demographics, laboratory, and clinical information were recorded in a standardized case report form. Self-reported adherence was defined as missed medical appointments at least once within the last 12 months and missed antiretroviral doses at least once within the previous month. All enrolled study participants were offered a viral load (VL) testing. Collected blood and plasma specimens were transported to the reference Laboratory of Bacteriology-Virology (LBV) at the Aristide Le Dantec University Teaching Hospital in Dakar for VL testing and DR genotyping. At the time of this study DR testing was only available in Dakar.
Specimens collection and processing
Ten milliliters (10 ml) of venipuncture blood were collected in EDTA tubes for the enumeration of CD4 T-cells (of the recent 3 months at enrollment) with the FACS count instrument (Becton Dickinson). Remnant blood was centrifuged (2500 rpm) on-site (RHS) and, the plasma layer aliquoted into cryogen vials that were shipped on an ice pack (−20°C) to the LBV. Blood specimens harvested at the ATC were processed at the LBV where they were transported within approximately 3 hours. All plasma specimens were frozen at −80°C until molecular analyses. Quantitation of plasma viral RNA particles was performed using the Generic HIV VL Kit (Biocentric®, Bandol, France) according to instructions from the manufacturer. This PCR-based assay targets the HIV-1 long terminal repeat (LTR) genomic region with a detection threshold of 300 viral RNA copies/ml for 200 µl of plasma volume. 32
HIV-1 RNA retro-transcription-PCR and cycle sequencing
The QIAmp® Viral RNA Mini Kit (250) (Qiagen, Courtaboeuf, France) was used to extract viral genomic RNA from 200 μl of plasma. First-strand cDNA synthesis and first-round PCR amplification were performed in a 1-tube retro-transcription-PCR targeting the partial pol region (reverse transcriptase), which was then subjected to a nested heat-cycling round. PCR reactions were carried out using the in-house protocol from the “Agence Nationale de Recherche sur le Sida et les Hépatites en France (ANRS).” 33 Nested PCR products were confirmed by electrophoresis (~800 bp) and column-purified (PureLink® Quick Gel Extraction Kit, Invitrogen) for BigDye® cycle sequencing (Applied Biosystems®, Courtaboeuf, France). Extension products were precipitated in ethanol/acetate and electrophoresed by capillary. The software SeqMan™ II v5.08 (DNASTAR*) was used to ascertain the base-calling of chromatograms before generating fasta sequences.
Quality assurance and Sequences repository
Specimens from both sites were tested in separate batches and quantification of VL done as collection was proceeding. All molecular analyses were conducted in separate and dedicated rooms and each sample run included positive and negative controls. Redundant sequences, suggesting the presence of contamination, were quality-controlled by computing the pairwise genetic distance per genotyping run on MEGA v6.06. The 27 partial pol sequences generated in this study are available at the European Nucleotide Archive (ENA)/EMBL through the following assigned accession numbers: LT976685-LT976711 (http://www.ebi.ac.uk/ena/data/view/LT976685-LT976711).
HIV-1 resistance, phylogenetic, and recombination analyses
DR mutations (DRMs) to first-line treatment were identified on the Stanford HIVDR database (http://hivdb.stanford.edu, 2019 version). The level of DR was profiled as low, intermediate or, high. Potential-low-level of resistance was classified as sensitive by WHO interpretation. For subtype identification, all HIV-1 sequences were primarily screened with the online viral subtyping tool Castor v1.0. 34 Multiple sequence alignment was performed with reference sequences using the program MAFFT v7.31 35 and imported to PhyML v3.1 for phylogenetic subtype confirmation. The pattern of recombination was analyzed using the software SimPlot v3.5 and Recco. 36
Statistical analysis
VF was the primary endpoint of the study, expressed as the percentage of cases with detectable viral RNA copies/ml ⩾1000. 37 The secondary endpoint, DR, was defined as the presence of a single or multiple DRM resulting in diminished drug activity. Thus, the prevalence of DR was calculated by dividing the number of isolates with clinically relevant DRMs over the total number tested for VF or with effective resistance genotyping. As of fact, the mutation V75VI often occurs along with the multi-resistance Q151M, but when alone its clinical significance is uncertain so was not considered in the definition of drug resistance (http://hivdb.stanford.edu).
The software Epi-info™ v7.2.1 (Centers for Disease Control and Prevention) was used to summarize data as frequencies. Differences in proportions between the 2 study sites were tested by the χ2 or Fischer’s Exact test when the expected cell count was <5. We employed the R software v3.5.0 (The R foundation for Statistical Computing) for the analysis of continuous variables. The unpaired t-test was used to compare the mean difference between the 2 sites for normally distributed data. For data on a non-parametric distribution, the median with interquartile (IQR) range between the sites was compared using the Mann–Whitney U test.
Binomial logistic regression was implemented with R v3.5.0. The unadjusted association between variables and VF was explored and predictors with a significant likelihood ratio test (LRT) (P < .25) from univariable analysis were entered into a multivariable model. Age, gender, and treatment duration variables were included in the model for their clinical relevance. The ART regimen variable was categorized as NVP-/EFV-based. The overall model fit was examined for the effect of each contributing variable and the parsimonious model selected on the basis of LRT significance. Coefficients’ estimates (ie, adjusted odds ratio, aOR) were presented at their 95%CI. We stratified the full model by site to further examine why outcomes were poorer at the RHS. For DR, all variables were dichotomized before applying the above described analyses. Only clinically relevant DR mutations were considered for regression analysis. Comparator groups were selected based on prior literature knowledge. All tests were 2-sided and the statistical significance level set at a P-value less than .05.
Results
Characteristics of study participants
A detailed description of the participants’ characteristics is given in Table 1. The 278 attendees of the ATC of Fann (54.3%) and the RHS (45.7%) had comparable mean ± SD ages (46.22 ± 10.50 vs 45.29 ± 11.18 years; P = .48, t-test). At both clinic sites, the female subpopulation predominated representing overall 66.9% (186/278), whereof 24.7% (46/186) were on a PMTCT protocol. The prominent mode of HIV-1 transmission was heterosexual (95.7%, 266/278).
Demographics, clinical, and laboratory characteristics of first-line antiretroviral-treated HIV-1 adult patients for at least 12 months in Senegal.
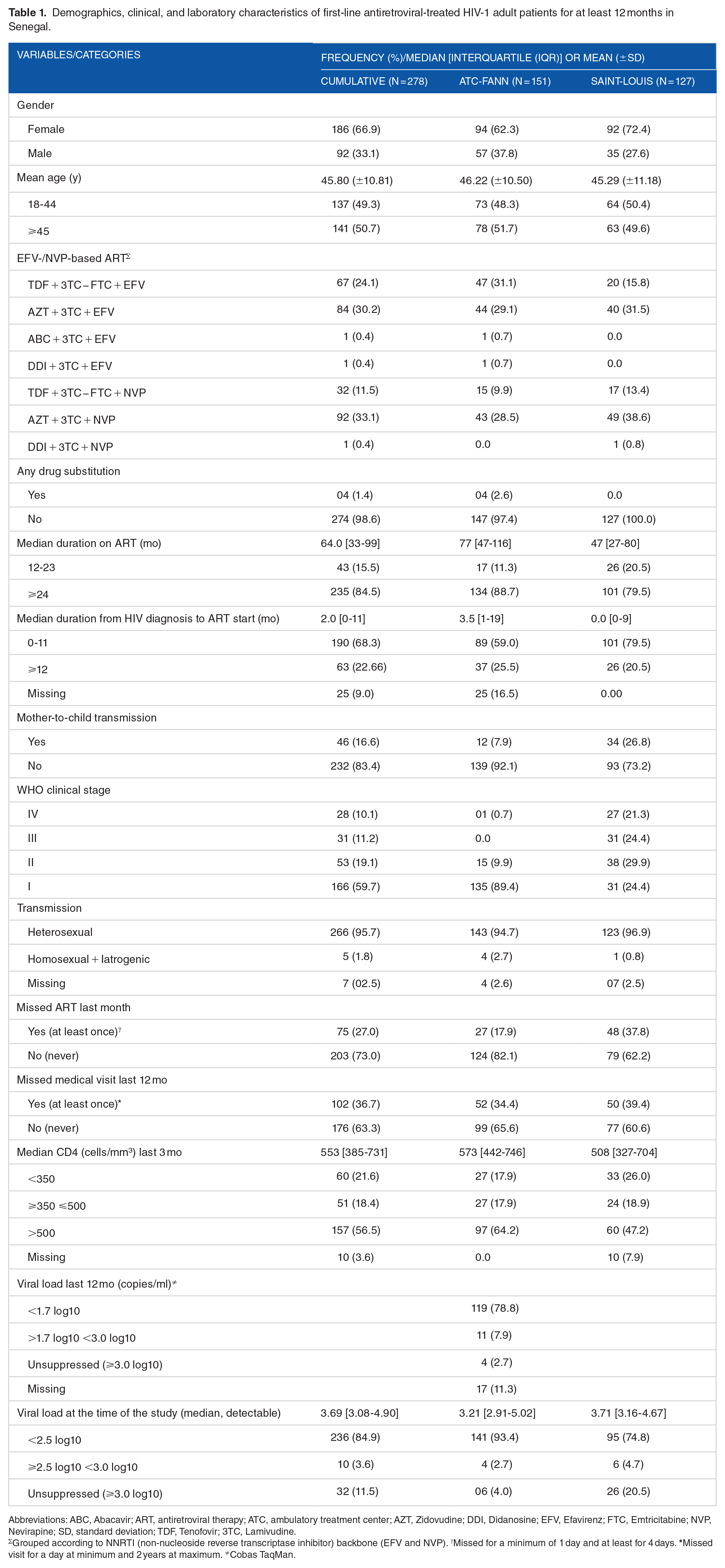
Abbreviations: ABC, Abacavir; ART, antiretroviral therapy; ATC, ambulatory treatment center; AZT, Zidovudine; DDI, Didanosine; EFV, Efavirenz; FTC, Emtricitabine; NVP, Nevirapine; SD, standard deviation; TDF, Tenofovir; 3TC, Lamivudine.
Grouped according to NNRTI (non-nucleoside reverse transcriptase inhibitor) backbone (EFV and NVP). γMissed for a minimum of 1 day and at least for 4 days.
At both treatment sites, the cumulative median duration between HIV diagnosis and therapy start was 2 months (range 0.0-133). Therapy at the RHS was started less than a month (range 0.0-77) before sampling, whereas at the ATC the median time to start was more than 3 times longer (3.5 months, range 0.0-17). More than half of the patients at the RHS took regimens containing NVP (52.8%, 67/127) compared to patients at the ATC (38.4%, 58/151), for a median duration of 47 months (IQR 27-80). In contrast, patients at this latter referral clinic largely received EFV-based regimens (61.6%, n = 93 vs 47.2%, n = 60) for a median span of 30 more months (IQR 47-116) (P < .001, Mann–Whitney U test). Proportions on different treatment arms and between sites differed significantly (χ2; P = .021). Forty-eight of 127 (37.8%) patients at the RHS had skipped ARV pills at least once in the previous month compared to 27/151 (17.9%) at the ATC. Those skipping pills for ⩾2 days at the RHS were 68.8% (33/48), against 29.6% (8/27) at the ATC. Delays in scheduled medical appointments ranged from 1–7 days to 24 and 4 months, respectively.
Compared with Dakar, a higher percentage of participants at Saint-Louis had WHO clinical symptoms of stage III/IV (45.7% vs 0.7%) at study inclusion. Although lower at the RHS, there was no significant difference in the median CD4 count for the 2 study sites (508 cells/mm3, IQR 327-704 vs 573 cells/mm3, IQR 442-746; P = .051, Mann–Whitney U test). The median detectable viral load (⩾2.5 log10 copies/ml) was similar in patients at the ATC and RHS (3.21 log10 copies/ml, IQR 2.91-5.02 vs 3.71 log10 copies/ml, IQR 3.16-4.67; P = .43, Mann–Whitney U test). In the preceding 12 months of the study, patients on care at the ATC had an overall 2.65% VF rate. In contrast, those at the RHS had no prior VL testing.
Predictors of HIV-1 virological failure at ⩾12 months
The overall proportion of patients who demonstrated VF over ⩾12 months on NVP-/EFV-based treatment was 11.5% (95%CI: [8.01-15.86]). The median ART duration did not differ significantly among patients with or without VF (HIV-1 RNA <1000 copies/ml) (69.5 months, IQR 23.0-89.5 vs 64 months, IQR 34-99; P = .46, Mann–Whitney U test). In the unadjusted logistic regression, 7 risk factors were associated with VF (Table 2): drug regimen; ART duration; WHO clinical staging; having missed doses and medical appointments, respectively, in the previous 1 and 12 months(s); CD4 T-cell count and site-of-care.
Binomial logistic regression for predictors of HIV-1 virological failure (⩾1000 copies/ml) in first-line antiretroviral-treated adult patients for at least 12 months at 2 urban treatment settings in Senegal (N = 278).
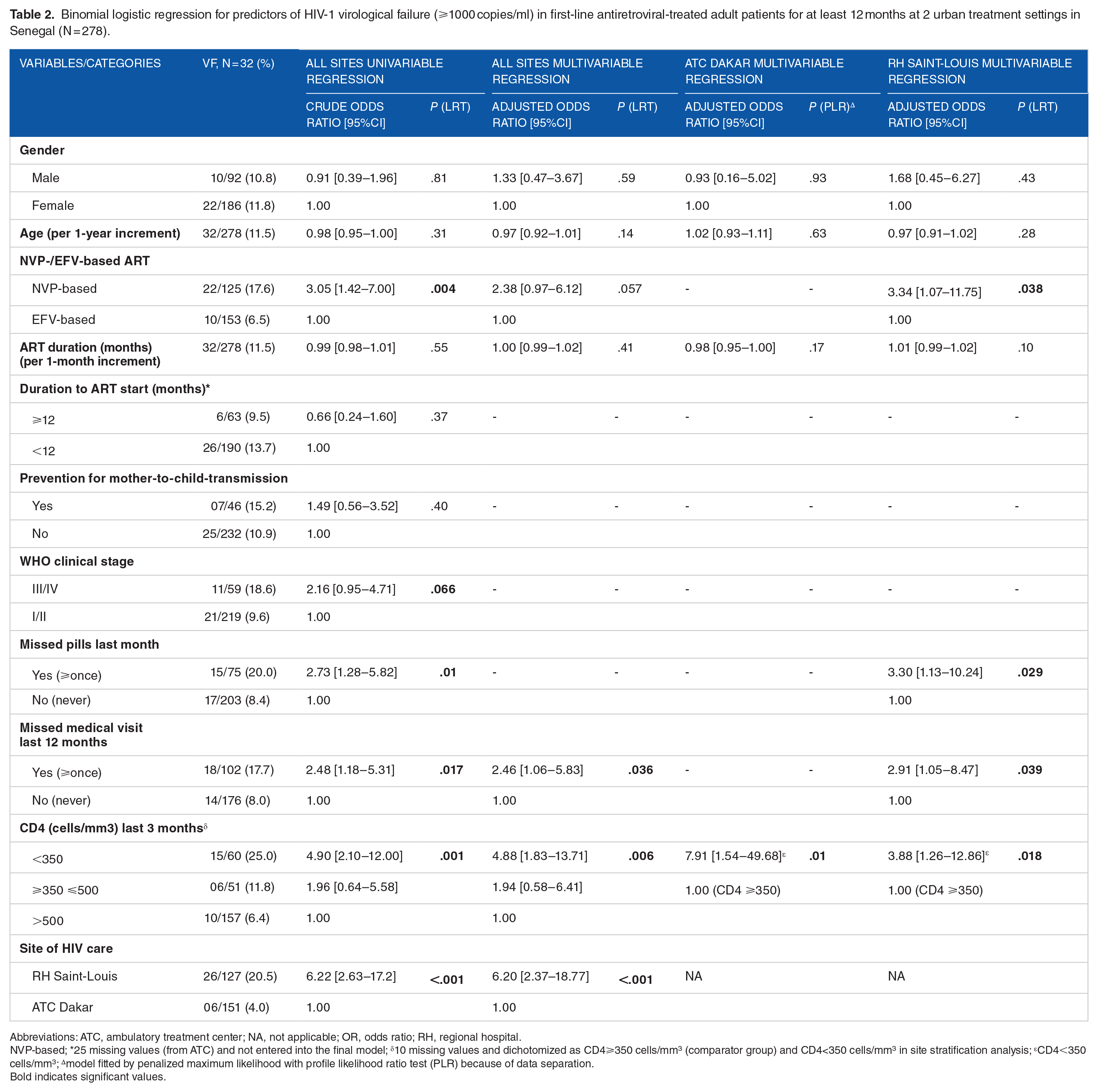
Abbreviations: ATC, ambulatory treatment center; NA, not applicable; OR, odds ratio; RH, regional hospital.
NVP-based; *25 missing values (from ATC) and not entered into the final model; δ10 missing values and dichotomized as CD4⩾350 cells/mm3 (comparator group) and CD4<350 cells/mm3 in site stratification analysis; εCD4<350 cells/mm3; ∆model fitted by penalized maximum likelihood with profile likelihood ratio test (PLR) because of data separation.
Bold indicates significant values.
Upon multivariable adjustment 3 predictors remained significantly associated with VF (Table 2): self-report of missed medical appointments at least once in the recent 12 months (yes vs no: aOR = 2.46, 95%CI [1.06-5.83]), site-of-care (RHS vs ATC: aOR = 6.20, 95%CI [2.37-18.77]); CD4 count (>500 cells/mm3 as the comparator; <350 cells/mm3: OR = 4.88, 95%CI [1.83-13.71]; 350-500 cells/mm3: OR = 1.94, 95%CI [0.58-6.41]).
Saint-Louis had the highest rate of VF (20.5%, 95%CI [13.8-28.5]), that was associated with ART regimens (NVP-based vs EFV-based; aOR = 3.34, 95%CI [1.07-11.75]); self-report of missed doses in the previous month (yes vs no: aOR = 3.30, 95%CI [1.13-10.24]) and medical appointments in the previous 12 months (yes vs no: aOR = 2.91, 95%CI [1.05-8.47]). The lower CD4 count continued to be strongly predictive of VF at both sites.
Predictors of HIV-1 drug resistance at ⩾12 months
After controlling for age, gender, treatment duration, and other covariates the likelihoods of acquiring DR remained significant in patients: taking NVP-based regimens (vs EFV-based regimens: aOR = 3.83, 95%CI [1.16-15.30]), having CD4 <350 cells/mm3 (vs CD4 ⩾350 cells/mm3: aOR = 15.77, 95%CI [4.89-60.93]), getting care at the RHS (vs ATC: aOR = 4.89, 95%CI [1.44-20.58]). The prevalence of DR at Saint-Louis was 12.9% (95%CI [7.6-20.1]) and significantly associated with taking NVP- instead of EFV-based regimens (aOR = 5.13, 95%CI [1.12-37.35]) and lower CD4 count (CD4 <350 cells/mm3 vs CD4 ⩾350 cells/mm3: aOR = 12.25, 95%CI [3.04-65.93]). Table 3 describes the correlates of DR. High-level DR, compared to low and intermediate levels, was highly predicted for NVP (70.4%) and EFV (59.3%) (Figure 1). FTC and 3TC were also predicted with high-level DR (55.6%), which however is associated with a higher fitness cost due to the point mutation M184V. AZT was predicted with only low (3.7%) to intermediate (22.2%) DR, while high-level resistance was the highest predictor of DR to TDF (14.8%).
Binomial logistic regression for predictors of HIV-1 drug resistance in first-line antiretroviral-treated adult patients for at least 12 months at 2 urban treatment settings in Senegal (N = 273).
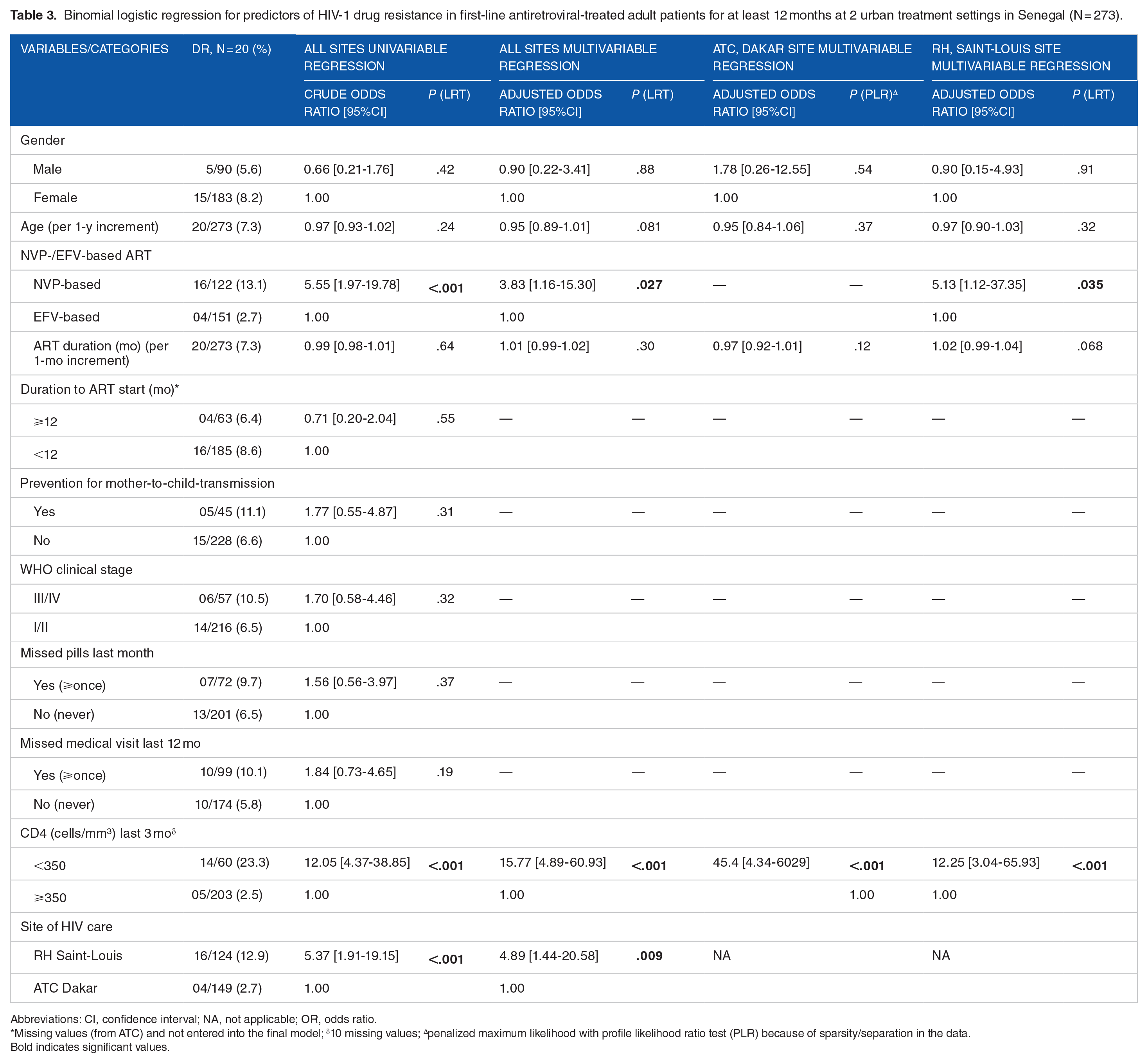
Abbreviations: CI, confidence interval; NA, not applicable; OR, odds ratio.
Missing values (from ATC) and not entered into the final model; δ10 missing values; ∆penalized maximum likelihood with profile likelihood ratio test (PLR) because of sparsity/separation in the data.
Bold indicates significant values.
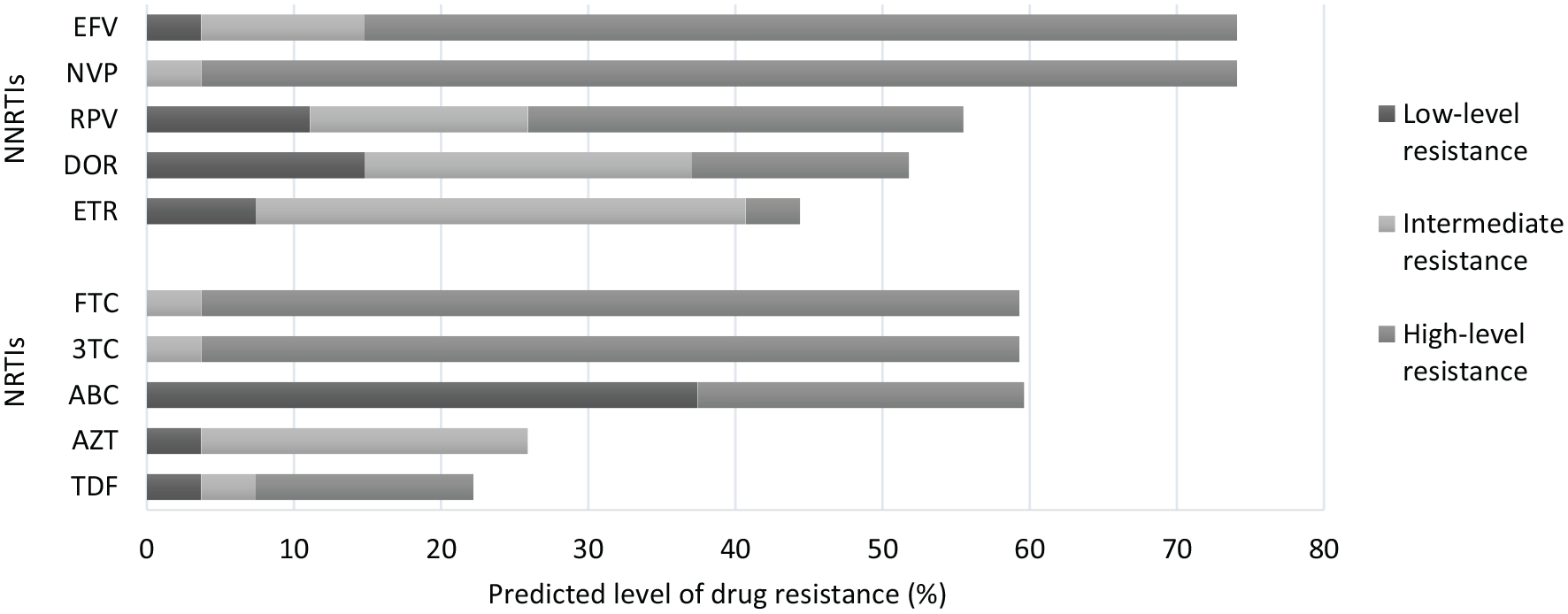
Predicted level of drug resistance to nucleos(t)ide reverse transcriptase inhibitor (NRTI) and non-NRTI (NNRTI) by the Stanford HIVdb.
HIV-1 drug resistance and subtypes
DR genotyping was successful for 84.4% (27/32) samples, whereof 66.7% (18/27) represented circulating recombinant forms (CRF)02_AG, as expected from the phylogenetic analysis. Twenty of the 27 (74.1%) samples successfully genotyped carried at least 1 DRM of clinical relevance (Table 4), giving an overall DR prevalence of 7.2% (95%CI [4.5-10.9]). Almost 3 quarters of patients had DR to NNRTIs (74.1%, 20/27), with more than half experiencing DR to NVP (59.3%). Although higher, resistance was relatively less common with NRTIs (63.0%, 17/27), whereof 51.9% had DR to 3TC, 18.5% to AZT, and 18.5% to TDF. Cross resistance to both classes of inhibitors was seen in 59.3% (16/27) patients. The most frequent resistant genotypes to NNRTIs were K103N (37.04%), Y181CY (22.2%), and A98AG (18.5%). For NRTIs, these were M184VI (55.6%), T215SNY (22.2%), and K65R (18.5%). Thymidine analog mutations (TAMs) were detected in 29.6% (8/27) patients (Table 4). Three of them, including 1 previously on PMTCT (279A, 310A, 1181A) were taking regimens containing TDF, which also selects for TAMs (ie, K70R). Nonetheless, in patient 279A K70R co-occurred with K219E, the TAM variant D67G, and T69D, suggesting undisclosed use of AZT (http://hivdb.stanford.edu). Same for patients 310A, 1181A, and 309A who carried K65R and Y115F strains while on AZT. Resistance across drug classes was frequently seen at 48+ months. The reduced drug activity for the second generation NNRTIs was significantly predicted for rilpivirine (RPV) (15/27, 55.5%), etravirine (ETR) (12/27, 44.4%), and the novel NNRTI doravirine (DOR) (14/27, 51.9%).
Profile of HIV-1 drug resistance mutations in adult patients treated with first-line antiretroviral for at least 12 months with virological failure (⩾1000 copies/ml) in Senegal (N = 27).
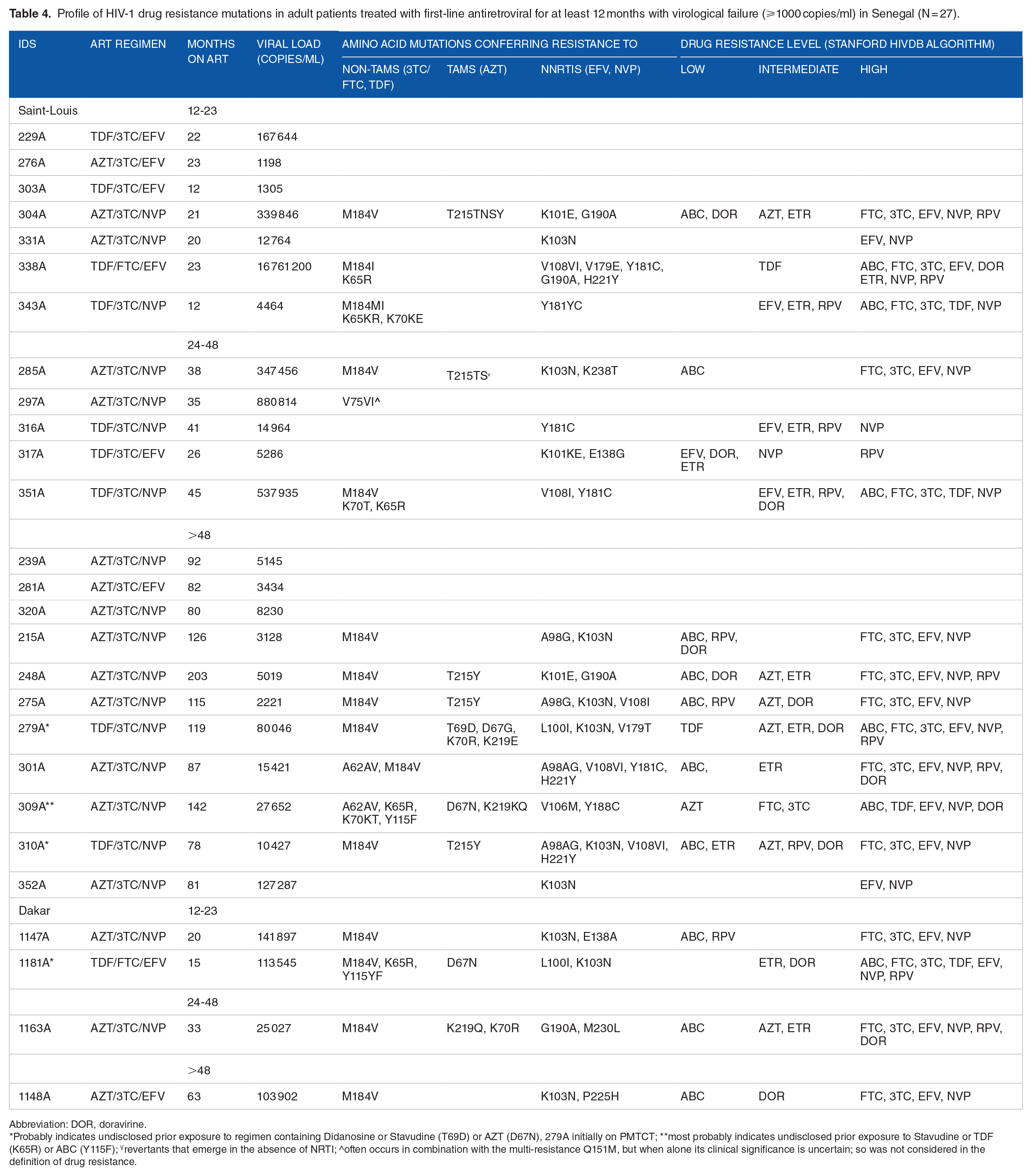
Abbreviation: DOR, doravirine.
Probably indicates undisclosed prior exposure to regimen containing Didanosine or Stavudine (T69D) or AZT (D67N), 279A initially on PMTCT; **most probably indicates undisclosed prior exposure to Stavudine or TDF (K65R) or ABC (Y115F); ۷revertants that emerge in the absence of NRTI; ^often occurs in combination with the multi-resistance Q151M, but when alone its clinical significance is uncertain; so was not considered in the definition of drug resistance.
Discussion
This survey reports a pooled virological suppression rate of 88.5% in patients taking NVP- or EFV-based ART for a median of 64 months (IQR 33-99) at 2 urban clinics of Senegal. In line with the UNAIDS third goal, this rate is reassuring. However, the small difference may reveal existing gaps in the progress toward the 2020 goal. A meta-analysis and systematic review showed rates of 87.7% at 12 months and 83.7% at 24 months on an on-treatment basis in sSA. 38 The multisite study by Aghokeng et al 39 in West-Central Africa (WCA) and Asia found a VF rate of 12.4% at 24 months, which is similar to our estimate of 11.5% but at 64 months. Parallel cross-sectional observations in terms of eligibility criteria revealed 24% VF rate at 36 months in Mozambique 40 and, 41.3% at 33.6 months in Gabon. 41 Our results indicate that a better virological response can still be achieved at a longer median time on treatment. However, variations in terms of the uniqueness of the clinic settings and study design may affect outcomes.
In subgroup analysis of DR, most widespread DRMs among failing patients were 3TC-resistance M184V and NVP-resistance K103N. The multicentric study by Villabona-Arenas et al 13 showed a similar trend, reporting 86.8% of M184V and 49.7% of K103N in 1288 patients failing first-line regimens in 10 countries of WCA. EFV and NVP have a lower genetic barrier, 7 thus facilitating the selection of K103N and Y181C mutations. 13 In our analysis of logistic regression, NVP-compared with EFV-based regimens were significantly associated with VF and DR, mainly at the decentralized clinic site. Tang et al 42 in their meta-analysis found that NVP increases the risk of TAMs and K65R, which potentially confers high-level DR to AZT and TDF, respectively. These 2 NRTIs are still vital for the current ART algorithm, which associates dolutegravir with 3TC/FTC. 11 Hence, they should be keenly monitored as 18.5% study patients with DR to AZT and TDF could be on a de facto monotherapy with dolutegravir. DRMs in patients taking NVP- or EFV-based ART also greatly (44%-55%) affected ETR, RPV, and DOR. The WHO guidelines on PDR released in 2017 8 now recommend the use of dolutegravir-based ARTs in place of NNRTIs (EFV and NVP). 11 These drugs have shown alarming rates of PDR in LMICs, with a yearly odds increase of 17% in WCA.9,10 Despite their small number, the 3 study patients with TAMs while on TDF and, another K65R and Y115F while on AZT hint that PDR to NRTI circulates in our study population.
This study found that the odds of VF and DR were significantly associated with the decentralized urban clinic site. A finding that concurs with reported poorer outcomes in urban clinics in Cameroon (Yaoundé), 43 Togo (Lomé), and Ivory-Coast (Abidjan), 39 and those in rural or semi-rural settings of sSA.40,41 Hence, the idea that obstacles in effectively delivering HIV services are due to the geographical situation of the clinic does not always apply. Meanwhile, well-structured implementation studies could help evaluate and address these obstacles (in urban and rural sites) as interventions are tailored accordingly. 44
First reports of higher VF (23.8%-26.0%) and ADR (15.9%-17.7%) rates in decentralized HIV settings of Senegal were based on data collected in September 2008 and December 2011.15,16 Another relevant 2-year investigation ended in February 2013, reported 25.3% VF and 19.9% ADR at the decentralized district hospital of M’bour (70 km southwest to Dakar) (Y.M. Adzavon, Personal Communication, December 2013). The 3 studies measured outcomes, respectively, at medians of 18, 32, and 33.5 months, which are shorter than at our site of Saint-Louis (47 months). In 2018, Ba et al 28 sampled patients from another decentralized clinic (Roi Baudouin hospital in the outskirts of Dakar) with a 19% VF rate at a longer mean duration of 60 months. Of note, outcomes at Saint-Louis (even at the latter clinic site) denote a continuous use of failing regimens at decentralized care. If not promptly detected, such failure might be clinically detrimental as DRMs accumulate over time. 45 At Saint-Louis, VL testing became available around 2016 but only went operational in 2018. Additionally, patients at this clinic site had no VL test within the past 12 months of sample collection. These are compelling evidence showing that over the years, viral suppression at decentralized settings in Senegal is alarmingly falling-off the UNAIDS third-90. The health system in Senegal may thus need to be upgraded to the level that would allow effective decentralization of HIV services. Meeting this prerequisite would enable the Senegalese HIV program to effectively transitioning to the potent drug dolutegravir, thus ensuring its optimal use durably. However, future studies would be needed, especially in patients using suboptimal (N)NRTIs first-line ART, to ascertain how decentralized HIV care may affect the extent to which dolutegravir-based ARTs remain efficacious.
In this study, the median time spent on ART did not seem to influence the time-to-occurrence of VF. Failing and non-failing patients had a comparable median time on ART (P = .46). Missed doses of ARV and medical appointments were significantly associated with the odds of VF at Saint-Louis. These findings may be suggestive of behavioral differences among failing and suppressing patients. 14 Bijker et al 46 compared self-adherence to ART in 2 large cohorts prospectively followed up for 3 years in Africa and Asia. After accounting for potential attrition bias, the investigators found that adherence in the African cohort did not wane, but improved consistently over time. They argued that this finding was attributable to non-defaulting or early targeted adherence counseling. Further, inadequate adherence may explain why 6 of the 23 (26.1%) failing patients at Saint-Louis had wild-type viruses as detected by population sequencing. This well-established property does not, however, preclude the existence of preexisting DRMs. In the absence of drug (ie, long interruption ~1 month), reversal to wild-types leads to higher population growth that increases the risk of DR when therapy resumes. 47 Hence, a single VL test may adversely affect adherence support, misclassifying thereby patients failing first-line ART (about ¼) as eligible to more expensive second-line therapies.
Limitations
One strength of our cross-sectional study is the lower VF rate (3.97%) obtained at a longer median of 77 months at the ATC, a finding that contrasts with several studies in sSA. Yet, the interpretation of the present data should be placed in the perspective of several limitations. Baseline CD4 counts were not available. We described VF during the CD4 cut-off policy of 500 cells/mm3 for treatment eligibility. This strategy can delay the start of ART, 48 fostering the decline of CD4 T-cells and lost-to-follow-ups. So a weak immune status at entry may justify the significant association with VF and DR seen at both clinic sites. Even at the ATC, a recent study found majority of patients (69%) with advanced stages of infection (notably with CD4 <200 cells/mm3) at the time of ART start. 27 In a prospective cohort in Dakar, De Beaudrap et al 26 estimated that a 50 cells/mm3 increase in baseline CD4 count reduced the risk of DR. Intensifying the TATARSEN policy in the country could thus mitigate the establishment and effect of resistance. Second, in keeping with the UNAIDS third-90 target the rate of VF at the ATC, although remarkable, did not account for attrition as patients were cross-sectionally sampled. This estimate therefore reflects the effectiveness of first-line ART on an on-treatment basis and not necessarily performance of public health programs. 49 Our study did not also collect qualitative information on clinic attributes and performance. Third, there were no data indicating whether all pregnant women under PMTCT had previous exposure to single-dose NVP or no. Such exposure may have led to overestimating rates of VF and DR to NVP-based first-line ART 50 and makes it less straightforward to differentiate between PDR and ADR. Fifth, selection bias by the treatment site may have affected outcomes as there was no justification to the preferential distribution of NVP-based ARTs at the RHS. Toxicity or stock-outs of EFV could explain these discrepancies, though this information was not documented. Finally, more comprehensive investigations with a higher sample size would be insightful in appraising the degree to which decentralization affects HIV care provision.
Conclusions
Our study stressed that the ART outcomes of patients reflect the inherent challenges of clinic sites where HIV care was sought. At the decentralized regional hospital of Saint-Louis, inadequate outcomes suggest the need to re-inforce the uptake of virological monitoring and adherence support. The superior clinical outcomes at the reference center of Dakar suggest that first-line ART (2NRTIs + 1NNRTI) still retains greater virological efficacy, namely with drug regimens containing efavirenz. Globally, HIV programs in Senegal should focus on prompt diagnosis to improve the TATARSEN policy. Taking account of these key interventions would provide a favorable framework to sustain the transitioning to dolutegravir-based ARTs, which would guarantee the use of this potent drug (dolutegravir) durably in Senegal.
Footnotes
Acknowledgements
The authors acknowledge all patients who consented and donated samples for the study and are also gratefully indebted to all field staffs and local authorities.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received support from the European and Developing Countries Clinical Trials Partnership (EDCTP) who made available funding through the West African Network of Excellence for TB, AIDS, and Malaria (WANETAM). Grant recipient: Prof. Souleymane Mboup. Aristid Ekollo Mbange was supported by the EU-funded AFIMEGQ Intra-ACP doctoral mobility program at the University of Cheikh Anta Diop (UCAD), Dakar, Senegal.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
CTK, SM, HDN, NFNG: Conceptualization. AEM, AD, SL, KNT, MF: Data curation. AEM: Formal analysis. CTK, SM: Funding acquisition. AEM, AAMD, NADD, MF, KNT, SL, AD: Investigation. AEM, AAMD, NADD, HDN: Methodology. HDN, CTK, SM: Project administration. AEM, CTK, SM, HDN: Resources. AEM, AAMD: Software. HDN, NFNG, AD, SL, KNT: Supervision. AEM, AAMD, NADD, CTK, HDN, WFM: Validation. AEM: Manuscript—original draft. AEM, AAMD, CTK, HDN, WFM, NFNG, SM: Manuscript—revision and editing.
