Abstract
Abnormalities of lipid metabolism are common in human immunodeficiency virus (HIV)-infected patients and tend to be accentuated in those receiving antiretroviral therapy, particularly with protease inhibitors (PIs). However, there is a dearth of information on serum lipid profiles and biochemical parameters among treatment-naive HIV-positive patients in our environment. We found that after 24 months of highly active antiretroviral therapy (HAART), there was a significant increase in serum lipids. After 24 months of HAART, renal impairment was associated with a low increase in mean HDL and a high increase in triglycerides (TG). In conclusion, abnormality of serum lipid is common and showed female preponderance among treatment-naive HIV patients in our environment. Patients with HIV infection on HAART should be screened for lipid disorders given their high prevalence as observed in this study, because of its potential for morbidity and mortality in patients on HAART.
Keywords
Introduction
The availability of potent combination antiretroviral regimens has resulted in a dramatic reduction in human immunodeficiency virus (HIV)-1–associated morbidity and mortality in the developed world. 1 However, the optimism generated by such treatment has been tempered by the recognition of an increasing array of adverse metabolic effects, including insulin resistance and glucose intolerance, dyslipidemia, changes in body fat distribution, lactic acidemia, and osteopenia.2–7 Multiple abnormalities in lipid metabolism were reported in HIV-1–infected patients. Changes in blood lipids occur naturally during the course of HIV infection, with decreases early in the infection in both total cholesterol (TC) and high-density lipoprotein (HDL) cholesterol, and increases later in triglycerides (TGs). 8 Infection with HIV-1 is known to increase plasma TG levels by decreasing the number of circulating lipoproteins, a process considered to be the result of reduced lipoprotein lipase (LPL) or by stimulating hepatic lipid synthesis through an increase in hepatic fatty acid synthesis or an increase in re-esterification of fatty acid derived from lipolysis. 9 However, highly active antiretroviral therapy (HAART) also leads to lipid changes with increases in both TGs 10 and TC. 11 While increases in TC during therapy may represent a return to pre-infection levels to some degree, 11 HAART is associated with an increased risk of CVD such as myocardial infarction. 12 Studies of lipid profiles of patients on HAART are either of short duration (less than a year) or based on a small number of patients (less than 100).7–9,11,12 We report on medium-term lipid profiles (up to 2 years) for antiretroviral-naive patients on nucleoside reverse transcriptase inhibitor (NRTI)- and non-nucleoside reverse transcriptase inhibitor (NNRTI)-based HAART and its associated factors in a tertiary institution in North-eastern Nigeria.
Patients and Methods
We undertook a prospective, observational study of HIV-infected patients from a United States President's Emergency Plan for AIDS Relief (PEPFAR)- supported anti-retroviral drug (ARV) clinic who started HAART between August 2008 and July 2009, and who followed the same regimen for 2 years. HAART was composed of two NRTIs plus NNRTIs. The indications for HAART regimen were assessed by the attending physician based on the Center for Disease Control (CDC) 1993 revised classification system for HIV infection and expanded AIDS surveillance case definition for adolescents and adults. 13 Patients enrolled in this study were 18 years and older. Patients took Stavudine (d4T) (one tablet of 40 mg twice daily) and Lamivudine (3TC) (one tablet of 150 mg twice daily). In addition, patients were randomly allocated to nevirapine (NVP) (200 mg daily for 2 weeks and then twice daily thereafter), or efavirenz (EFV) (one tablet of 600 mg once daily at bed time or one tablet of 800 mg once at bed time, depending on whether they had associated opportunistic infection treatment, especially anti-tuberculosis (TB) drugs).
Clinical, anthropometrical and laboratory data were recorded at the start of the study and after 2 years of HAART. Fasting blood glucose, serum TC, TGs, HDLs, low-density lipoproteins (LDLs), urea, and creatinine serum enzyme alanine aminotransferase (ALT) were estimated by an automated clinical chemistry autoanalyzer (Hitachi 902 Roche Diagnostic GmbH, Mannheim Germany). The CD4 + T lymphocyte cell count was estimated by CyFlow Counter (Partec GmbH, Gorlitz Germany). Blood was drawn after a 12-hour fast for the measurement of plasma lipids. Diagnosis of dyslipidemia was based on cholesterol levels ≥ 5.18 mmol/L and TG levels ≥1.69 mmol/L.
IRB approval was obtained from the University of Maiduguri Teaching Hospital, and written informed consent was obtained from participants.
Outcome Measurement
The mean percentage changes of total TG, TC, HDL and LDL between baseline (month 0) and month X after start of treatment were determined for each individual patient using the formula by Van Leth and colleagues: 14

where X is the study end point at which follow up takes place (24 months) after the start of treatment (baseline or month 0).
A body mass index (BMI) above 25 kg/m2 was considered to be high.
Statistical Analysis
Patients’ data were collected in a computerized database for later statistical analysis with SPSS® version 16.0 for Windows (SPSS Inc., Chicago, IL, USA). Categorical variables were analyzed with the χ
2
test, or the Fisher exact test when necessary. Comparison of quantitative variables prior to starting HAART and at 2 years was performed with the paired student -test or the Mann-Whitney
Results
The clinical parameters of the patients at baseline
Of the 229 patients included in this study, 17 did not complete the programmed follow-up. There were four AIDS-related deaths; the rest defaulted follow-up for reasons unknown to us. The mean age of the study population was 46.3 ± 9.3, males being older than females in general (
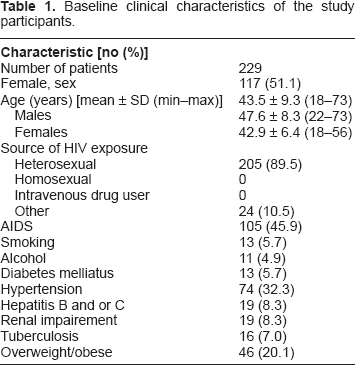
Baseline clinical characteristics of the study participants.
Baseline association between lipid profiles, some metabolic and HIV-associated variables
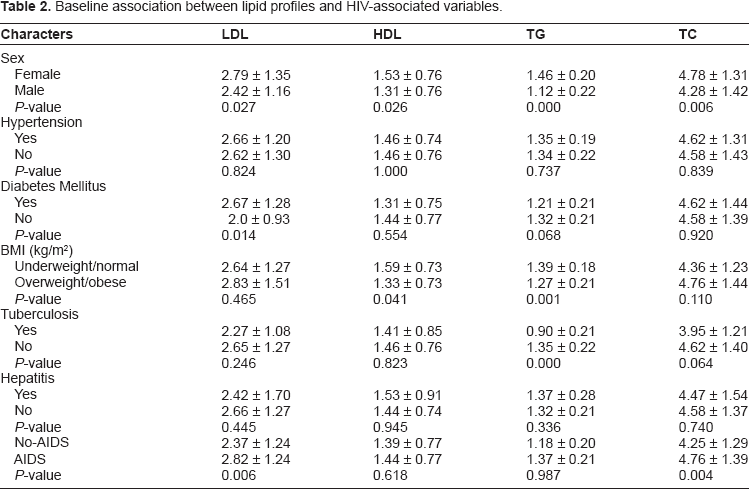
The female gender was associated with statistically significant elevation of all serum lipids at baseline, while no difference was observed between lipid profiles of hypertensive and normotensive cohorts. Diabetes mellitus was associated with a significant elevation in mean LDL. Overweight/obese patients had high TG and low HDL cholesterol. Hepatitis B surface antigen and/or hepatitis C antibody positivity had similar lipid profiles to hepatitis B- or C-negative patients, while the presence of TB was associated with high TG. The presence of AIDS-defining illnesses or immunological AIDS (CD4 count < 200 cells/μL) was associated with a statistically significant elevation in mean LDL and TC, as depicted in Table 2.
Baseline association between lipid profiles and HIV-associated variables.
Changes in the level of serum lipids, CD4 count and some biochemical parameters after 24 months of HAART
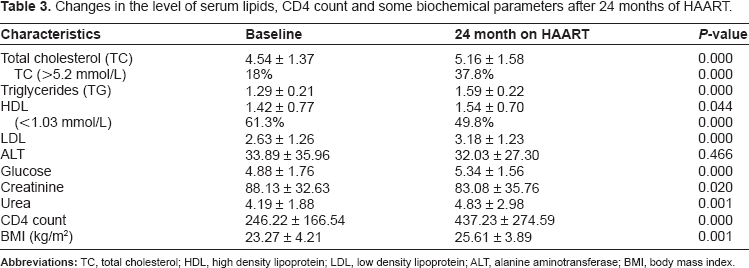
Table 3 shows the changes in lipid profiles over 24 months of HAART. As shown, there were significant increase in TC, TG, HDL and LDL. Fasting blood glucose also increased, as did urea. There was a significant decline in creatinine, but ALT remained unchanged throughout the study period. The CD4 count increased significantly from 246.2 ± 166.5 to 437.2 ± 274.6, and there was an observed significant weight gain as evidence by increase in BMI from 23.3 ± 4.2 to 25.6 ± 3.9. Younger participants (≤46 years) had a significant mean change in their lipid parameters in comparison to older participants (≥47 years) after 24 months of HAART as depicted in Figure 1.
Changes in the level of serum lipids, CD4 count and some biochemical parameters after 24 months of HAART.
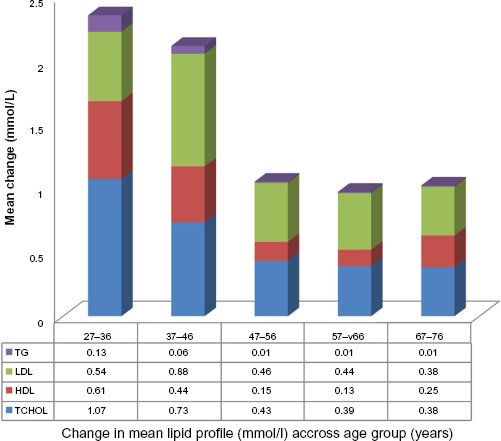
Change in mean lipid profile across different age group (years).
Changes in lipid profiles in some metabolic and HIV-associated variables after 24 months of HAART
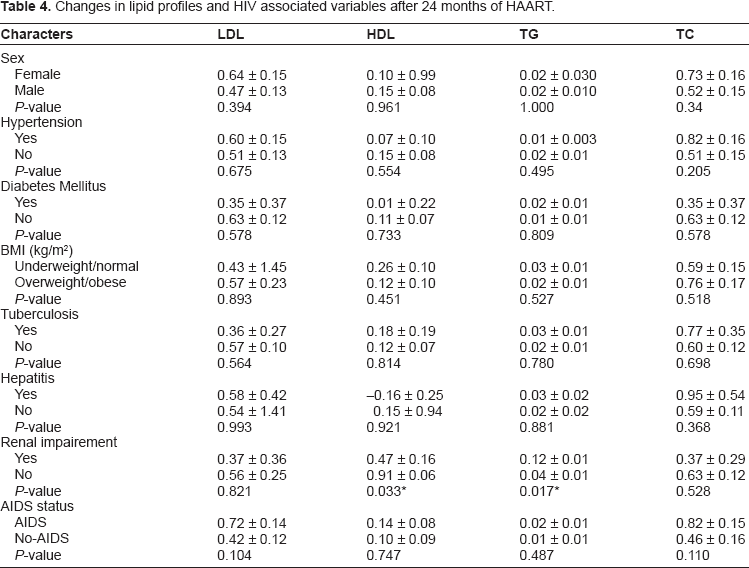
Taking gender and some metabolic and HIV-associated variables into account, renal impairment was associated with significantly low increase in mean HDL and high increase in TG. Patients with metabolic and HIV-associated variables had similar changes in lipid parameters to other cohorts, as shown in Table 4.
Changes in lipid profiles and HIV associated variables after 24 months of HAART.
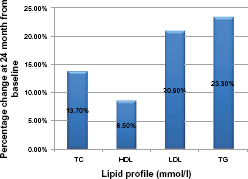
Percentage change in mean lipid parameters after 24 months of HAART
After 24 months of HAART, the percentage mean change of the lipid parameters is shown in Figure 2. The mean TG changed by 23.3% over the study period, while LDL, HDL and TC changed by 20.9%, 8.5% and 13.7% respectively.
Percentage change in lipid profile at 24 months from baseline.
Discussion
Previous reports have demonstrated that HIV-infected patients exhibit multiple abnormalities in lipid metabolism both in pre-HAART9,15–18 and post-HAART eras, especially with the use of protease inhibitors (PIs).19–21 Although the mechanisms that induce lipid abnormalities associated with potent antiretroviral therapy remain elusive,22–24 several pathophysiologic models have been proposed to explain the development of dyslipidemia in HIV-infected patients, involving several proposed interactions between the virus, antiretroviral therapies, and host factors.18,25,26 Metabolic derangement associated with the use of HAART calls for clinical concern about its use. Hyperlipidemia, defined as an increase in TG and TC levels, was observed in this study, consistent with a previous report. 27 The baseline hypercholesterolemia was similar to findings by Bernasconi 27 of 19% on a non-PI based regimen. However after 24 months on non-PI based HAART, the prevalence of hypercholesterolemia increased to 37.8%, which is comparable to 54% on a PI based regimen. 27 The results of this study contrast reports suggesting that dyslipidemia is mainly due to the use of a PI based HAART regimen. Patients who demonstrate elevated TC and/or TG levels should be treated appropriately, in order to prevent the development and progression of atherosclerotic heart disease, stroke, and pancreatitis. The mean HDL determined before commencement of HAART in this study was 1.42 ± 0.77 mmol/L, higher than the cohort from a study on Nutrition for Healthy Living (NFHL) conducted in the United States, 28 Spain, 29 and Nigeria. 30
Decreased levels of HDL occur at an early stage of HIV infection. Low HDL is a well-recognized independent risk factor for adverse cardiovascular outcomes and this has even been shown to be true in HIV-infected individuals, irrespective of other risk factors. 31 Several studies have shown that an HDL increase is associated with a significant decrease in mortality from coronary heart disease (CHD), independent of changes in LDL. 32 Other studies have associated the risk of cardiovascular disease (CVD) with low concentrations of HDL.33,34 HDL dyslipidemia (serum mean HDL < 1.03 mmol/L) as defined by the National Cholesterol Education Program (NCEP-ATP III) declined by 11.5% after 24 months on HAART in this report. NNRTI regimens have been noted to have additional protective factors against low HDL. 35 This is especially strong with NVP. 36 This anti-atherogenic effect of a NVP-based regimen may be related to its protection against oxidative stress. 37 In a resource-limited setting, where access to PIs may be limited due to cost, this study has shown that a NVP-based regimen may offer the additional benefit of stabilizing lipid profiles, particularly in terms of enhancing HDL cholesterol.
Changes in blood lipids occur naturally during the course of HIV infection, with decreases early in the infection in both TC and HDL cholesterol and increases later in TGs. 38 However, HAART used in our cohort also leads to lipid changes, with increases in both TC 11 and TGs, 10 as previously documented. While increases in TC during therapy may represent a return to pre-infection levels to some degree, 11 the increased incidence of hypercholesterolemia after 24 months in our study may suggest that HAART is associated with an increased risk of myocardial infarction. 12
The strengths of our study include the longitudinal design and 24 month follow-up, combined with our exclusion of pre-treated patients, and data once patients switched from one type of therapy to another. The main limitation of our study is the potential for confounds, which are inherent in every observational study.
The absence of any association between any of the lipid parameters and hypertension may be related to the duration of the hypertension, the degree of blood pressure control or the degree of immune suppression. Even in previously diagnosed hypertensives, who have been on medication or who have a positive family history of cardiovascular events in a first degree relative, there were no significant correlations between hypertension and abnormal lipid profiles, especially low HDL. 31 This suggests that HIV infection constitutes an additional and independent cardiovascular risk in hypertensive patients.
Studies have reported elevated TG was shown to positively correlate with interferon alpha, in patients with advanced disease/opportunistic infection. Markedly reduced immunity had to delayed clearance of plasma lipids due to reduced lipoprotein lipase activity.39,40 We found no significant association between hepatitis serological status and abnormalities of the lipid profiles. Other studies have reported low LDL, low TC and low HDL in HIV-positive patients. In particular, those co-infected with hepatitis C and with advanced stages of liver disease tended to have low TC.41–43 Though the findings were not significant, absolute values of these parameters were lower in those with hepatitis infection. TB is a common opportunistic infection in HIV/AIDS patients. 36 With an HIV/TB co-infection, there is a synergism between the two infections that leads to progression of the two diseases and high mortality if not treated. As revealed in this study, HIV patients co-infected with TB had a significantly higher mean TG levels, and high but insignificant TC levels, compared with HIV-positive patients who were not co-infected. This may indicate an exaggerated state caused by HIV/TB co-infection.
Author Contributions
Analyzed the data: BD, IK, AH. Wrote the first draft of the manuscript: BD, IK, AH, AD, AS. Contributed to the writing of the manuscript: BD, MA, AA, IK, BA, BF. Agree with manuscript results and conclusions: BD, IK, AH, AD, AS. Made critical revisions and approved final version: BD, IK, AH. All authors reviewed and approved of the final manuscript.
Funding
Authors disclose no funding sources.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
Footnotes
Authors disclose no potential conflicts of interest.
