Abstract
Background:
Worldwide prevalence of dyslipidemia in HIV-infected children on antiretroviral medications (ARVs) is rising due to extensive use of treatment during their entire lives. Dyslipidemia is the potential side effect of ARVs, especially in individuals taking protease inhibitors. The objective of this study was to determine the prevalence of dyslipidemia in HIV-infected children on ARVs receiving care at Kilimanjaro Christian Medical Centre (KCMC) in Tanzania.
Methods:
This was a cross-sectional hospital-based study conducted from September 2015 to May 2016 at KCMC. HIV-infected children and adolescents less than 17 years on ARVs for more than 6 months were enrolled. Blood samples were taken to determine levels of triglycerides (TGs), total cholesterol, lipoproteins (including low-density lipoprotein (LDL) and high-density lipoprotein (HDL)), CD4+ T cells, and viral load (VL). Anthropometric measurements were used to assess nutritional status. SPSS 20.0 was used for analysis. Logistic regression estimated odds ratio (OR) and 95% confidence interval (CI), and
Results:
A total of 260 participants were included in the study; the median age at HIV diagnosis was 3 (interquartile range (IQR) = 1-6) years. The overall prevalence of dyslipidemia was 46.5% with hypercholesterolemia (⩾200 mg/dl) of 11.2%, HDL (<35 mg/dl) of 22.7%, LDL (⩾130 mg/dl) of 7.7%, and hyperglyceridemia (TG ⩾150 mg/dl) of 12.3%. Children aged between 6 and 12 years at HIV diagnosis had 2.7 times higher odds of developing dyslipidemia compared with younger age at diagnosis (OR = 2.7; 95% CI = 1.1-6.6). Patients with advanced (OR = 6.4; 95% CI = 1.5-27.1) or severe (OR = 9.8; 95% CI = 1.2-76.5) HIV-associated immunodeficiency at diagnosis had higher odds of developing dyslipidemia. Protease inhibitor use was associated with higher odds of developing dyslipidemia (OR = 3.1; 95% CI = 1.4-7.1).
Conclusion:
Late diagnosis of HIV at 6 years of age or more, advanced, or severe immunosuppression, and the use of protease inhibitors were independent predictors of dyslipidemia in children on ARVs after 6 months of treatment, and with low HDL levels observed most commonly. Monitoring lipid profiles in children, especially those on protease inhibitors and with advanced/severe immunosuppression at diagnosis, may help in preventing future complications.
Background
The worldwide prevalence of dyslipidemia in HIV-infected children is rising due to extensive use of antiretroviral medications (ARVs). Dyslipidemia reflects abnormal lipid metabolism, commonly leading to hypercholesterolemia and hypertriglyceridemia. Dyslipidemia, hypercholesterolemia, and hypertriglyceridemia are known side effects of ARVs, especially protease inhibitors (PIs).1,2 However, dyslipidemia may also be associated with HIV disease itself 3 and low CD4 counts or percentage prior to initiating ARVs.4,5 The WHO 2016 Guidelines recommend that all HIV-infected children ⩽15 years of age to start on ARVs irrespective of viral load or CD4 count, which might predispose these children to dyslipidemia, especially when they use PIs 6 ; however, the same guideline also recommended initiation of ARVs for adolescent and adult in clinical disease stage III or IV or when CD4 count is less than 350 cell/ml.
Hyperlipidemia is becoming a major concern as children must be on ARVs for their entire lives, putting them in a state of continued lipid derangement which may lead to cardiovascular disease and atherosclerosis.7,8 The prevalence of dyslipidemia in children on ARVs varies worldwide with a previous report as high as 38.3% in India.9,10 In Spain, the prevalence of hypercholesterolemia and hypertriglyceridemia was 27.2% and 39.8%, respectively, 11 whereas in South Africa, dyslipidemia, hypercholesterolemia, and hypertriglyceridemia were observed in 70% of children on ARVs. 1 In a multisite study in Zimbabwe and Uganda, most lipids remained within normal range; however, those on long-term zidovudine (AZT) as 3 NRTI combination had abnormal high-density lipoprotein (HDL) cholesterol which was reported in 88% of participants, 12 while in the same study, low-density lipoprotein (LDL) was significantly higher in those with efavirenz (EFV) than nevirapine (NVP).
In Kilimanjaro, Tanzania, the prevalence of lipodystrophy in children on ARV treatment was found to be 27% 13 ; however, the lipid profile was not studied in the previous study by Kinabo et al. 13 Therefore, the aim of this study was to determine the prevalence of dyslipidemia among HIV-infected children on ARVs who receive HIV care and treatment at Kilimanjaro Christian Medical Centre (KCMC) in Moshi, Tanzania.
Methods
Study design and site
This was a cross-sectional, analytical, hospital-based study conducted from June 2015 to May 2016 at Child Centered Family Care Clinic (CCFCC), KCMC in Moshi, Tanzania. KCMC is a referral hospital for the northeastern zone of Tanzania, with a bed capacity of over 600, and provides medical services to a population of approximately 15.7 million people. The patients receive both general and specialist HIV care accordingly.
Study population
The study included all children aged less than 17 years who met inclusion criteria and were confirmed to be HIV infected on ARVs with at least 1 nonnucleoside reverse transcriptase inhibitor (NNRTI) and/or PI in combination with 2 nucleoside reverse transcriptase inhibitors (NRTIs) for a period of at least 6 months prior to enrolment in the study. The exclusion criteria were children on corticosteroids and those known to have metabolic disorders such as diabetes, and nephrotic or nephritic syndrome; however, there was no participant who had any of the exclusion criteria.
Sample size and sampling technique
The minimum sample size (SS) was estimated using a formula by the Survey System (C) 2003 Creative Research and Joint WHO/DGIS (1988) expressed as SS = (Z2 × p(1 – p)) / ε2, where Z = 1.96 for 95% confidence level. A prevalence (p) of 21% was selected based on a study done by Berhane et al, 14 and ε is minimal tolerable error at 95% confidence interval (CI), expressed as a decimal (0.05). The minimum estimated SS was 266 participants. The study used a convenience sampling technique where all children attending their clinic visits during the study period and fulfilling the inclusion criteria were enrolled in the study.
Physical examination
A physical examination was conducted, where the body weight (in kilograms, to the nearest 1 g) and height (in meters, to the nearest cm) was measured for calculating the body mass index (BMI).
Height was measured by using a stadiometer, and the patient was required to stand upright having only the body (buttocks) and head touching the meter rule (m). For those who were not able to stand, length was taken with a Schor board. Weight and height or length was used to calculate BMI
A standardized questionnaire was used to record the information obtained from every patient to ensure uniformity. The questionnaire was pretested during a short pilot study on 10 subjects before recruitment, and these 10 subjects were not included in the study. The challenges identified during pilot were addressed before full implementation of the questionnaire for data collection. The questionnaire for this study included sociodemographic characteristics, HIV stage, information on ARV treatment (initial ARV defined as ARV regime started at first initiation and current ARV defined as ARV regime which the patient was using during the study recruitment, which might be similar to the initial if ARV does not change), duration on ARVs and type of ARVs patient was using (if first-line or second-line regime or combination), record on CD4, and viral load test results. A specific laboratory form was used to order laboratory test which included lipid profiles, CD4 count, and viral load tests. We also recorded duration from the last time ate or given meal to the time of sample collection to calculate the mean time from meal to sample collection.
Diagnostic criteria for dyslipidemia
The plasma concentrations of lipids are known to be influenced by different mechanisms and factors such as age, sex, ethnicity, and use of antiretroviral drugs. For this study, we used a definition for dyslipidemia previously published by other studies,4,7,15 defining dyslipidemia as TG >150 mg/dL (>1.69 mmol/l), TC >200 mg/dL (>5.13 mmol/l), LDL >130 mg/dL (3.33 mmol/l), and HDL ⩽40 mg/dL (<1.03 mmol/l) (see Supplemental Table 1). Calculations were used to change lipids from mmol/l to mg/dl by multiplying cholesterol lipid values by 39 and TG values by 89, as previously used elsewhere. 16 Kilimanjaro Clinical Research Institute (KCRI) biotechnology laboratory use the pediatric and adolescent established chemistry normal range within the study population from health children and adolescent 1 month to 17years old. 17
Laboratory Analysis
Viral loads, CD4 counts/percents, and lipid profiles were performed in the KCRI Biotechnology Laboratory, which supports both clinical trials and basic science research. The Laboratory is equipped with an Abbott M2000rt for HIV viral load testing, BD™ FACSCOUNT for CD4 count and percentage measurement, and COBAS Integra 400 for chemistry analysis. The Laboratory participates in external quality assurance exercises through the AIDS Clinical Trials Group Virology Quality Assurance program, flow cytometry through UKNEQAS, and the College of American Pathology Proficiency Testing for lipid profiles. CD4 counts/percentage cut-off classification according to age was used. We used commercial biochemistry reagent bought from Becton Dickinson (BD) Medical technology which supplies reagent specific for COBAS integra 400 chemistry analyzer used in this study.
Data analysis
Data were checked for completeness and entered into Statistical Package for Social Science version 20 (SPSS Version. 20). Descriptive statistics for categorical variables were summarized using frequency and percentage, and for continuous variables median and interquartile ranges (IQRs). Logistic regression was done to measure the association between dependent and independent variables. All variables with
Results
Sociodemographic and clinical characteristics of study participants
The study population consisted of 260 children. The median age at HIV diagnosis was 3 (IQR = 1-6) years, and 131 (50.4%) were female. Most participants (132, 50.7%) had WHO stage IV disease at first HIV diagnosis; however, the latest CD4 counts did not indicate significant HIV-associated immunodeficiency in most participants (190 (73.1%)). All participants were on ARVs and 107 (41.2%) were taking AZT/3TC (lamivudine)/NVP; only 10 (3.8%) participants were on PIs as their initial regimen (first initiated ARVs regime); however, during the study recruitment, 52 (20%) participants had been on PIs as second-line drugs or changed during the period of treatment (Table 1). This shows that 42 participant had been changed to PI during the course of treatment. Patients on PI-containing regimens had used this regimen for shorter times compared with NVP- and EFV-containing regimens (3.2 years compared with 4.7 and 3.8 years, respectively).
Background information of patients (N = 260).
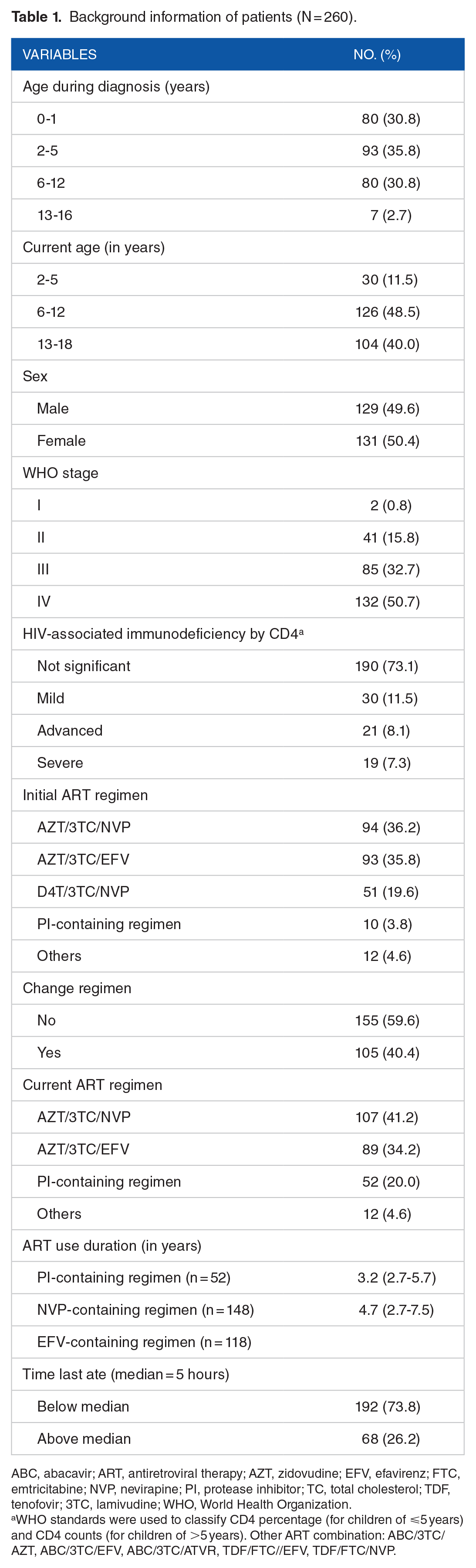
ABC, abacavir; ART, antiretroviral therapy; AZT, zidovudine; EFV, efavirenz; FTC, emtricitabine; NVP, nevirapine; PI, protease inhibitor; TC, total cholesterol; TDF, tenofovir; 3TC, lamivudine; WHO, World Health Organization.
WHO standards were used to classify CD4 percentage (for children of ⩽5 years) and CD4 counts (for children of >5 years). Other ART combination: ABC/3TC/AZT, ABC/3TC/EFV, ABC/3TC/ATVR, TDF/FTC//EFV, TDF/FTC/NVP.
Lipid profiles among study participants
Most participants had normal lipid levels ranging from 77.3% for HDL to 92.3% for LDL. Moderate to severe derangement was observed in all parameters; however, severely low levels of HDL were observed in 59 (22.7%) of participants (Table 2). Lipid profile may be affected by meal or duration of taking food to sample collection, and we found the median (SD) time from last meal was 5.0 (±2.8) hours.
Lipids profiles among study patients.

HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglyceride.
Factors associated with dyslipidemia
Among study participants using NNRTI and PIs, 123 (59.1%) and 43 (82.7%) had dyslipidemia, respectively. In the bivariate analysis, age at HIV diagnosis, HIV-associated immunodeficiency by CD4, and current ARV regimen were significantly associated with dyslipidemia (Table 3). Viral load, age group of the participants, and BMI were not associated with dyslipidemia.
Factors associated with dyslipidemia among study patients (N = 260).
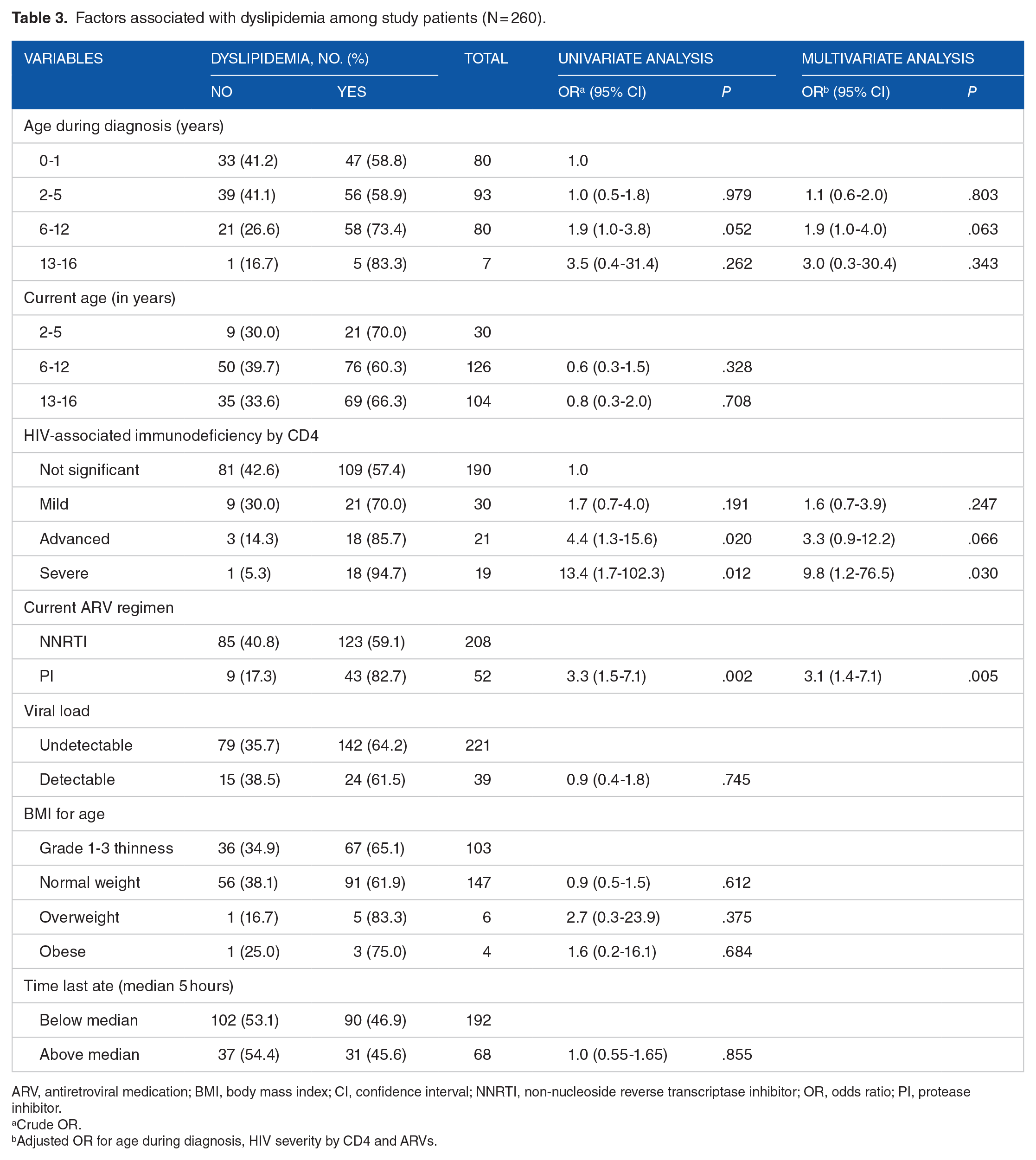
ARV, antiretroviral medication; BMI, body mass index; CI, confidence interval; NNRTI, non-nucleoside reverse transcriptase inhibitor; OR, odds ratio; PI, protease inhibitor.
Crude OR.
Adjusted OR for age during diagnosis, HIV severity by CD4 and ARVs.
Children with advanced (OR = 3.3; 95% CI = 0.9-12.2) and severe (OR = 9.8; 95%CI = 1.2-76.5) immunodeficiency at the time of HIV diagnosis had increased odds of having dyslipidemia. Participants who used lopinavir or atazanavir (PIs) had 3.1 times higher odds of developing dyslipidemia (OR = 3; 95% CI = 1.4-7.1) and were significantly associated with dyslipidemia. Other ARVs were not significantly associated with dyslipidemia. However, we observed an interesting pattern between age at which patients were diagnosed with HIV and dyslipidemia, although it was not statistically significant (
Discussion
The increased use of ARVs in children with HIV infection and expansion of PIs motivated us to pursue this study to examine lipid profile derangements. The lipid profile was found to be normal in most children receiving ARVs in our local context. Among children who had abnormal lipid levels, more than one-third had abnormally low HDL levels, followed by elevations in TC, LDL, and TG.
The observed findings in this study were similar to that observed in the multisite study in Uganda and Zimbabwe which indicated the prevalence of cholesterol, TG, and LDL being normal in the majority (75%, 85%, and 100%, respectively). 12 However, our findings differed from the studies done in Latin America and Spain, which observed a higher rate of hypercholesterolemia (20.5% and 27.2%) and hypertriglyceridemia (29.4% and 39.8%), respectively,7,11 and in Spain, which also reported higher rates of lipid profile derangements. 11 These contrasting observations could be due to the differences in age groups, as majority of our study participants were aged between 6 and 12 years and initiated ARVs at age between 2 and 5 years.
The surprising finding from this study was the severely low HDL in 22.7% of participants, making it the dominant abnormality observed, while the prevalence of LDL and TG abnormalities was low, 7.7% and 12.3%, respectively. However, a higher prevalence of low HDL has been reported previously in HIV-naïve children and children changing ARV treatment.4,18 HDL exerts a protective effect against cardiovascular diseases when it is normal or high, causing concern for our study participants. Low HDL cholesterol causes impaired reverse cholesterol transport and may lead to the development of atherosclerosis. 19 LDL and TG levels modulate the effect of low HDL level as a risk of cardiovascular disease. Low HDL with elevated LDL and TG, or low HDL with low LDL and TG, increases the risk of cardiovascular disease as compared with high HDL with elevated LDL and TG, or high HDL with low LDL and TG, which reduces the cardiovascular risk. 20 However, in our study, most children had low HDL with normal LDL and TG, and therefore it is not possible to predict their risk of future cardiovascular disease. Long-term follow-up for these children with low HDL will be important to assess the relationship of low HDL in immunosuppressed children on ARVs and their future risk of cardiovascular disease.
The use of NNRTI-containing regimens as first-line drugs might have also contributed to the lower rates observed in our study. However, with the current recommendation of PI use in first-line regimens in all children less than 3 years old, their use might lead to higher rates of lipid derangement. Lipid derangements usually occur with the long-term use of ARVs, especially those containing PI-based regimens. Very few patients were on lopinavir/ritonavir or atazanavir/ritonavir in our study. Previously, Tanzanian HIV Treatment Guidelines recommended the combination of NNRTI and 2 NRTI to be the first-line treatment of HIV-infected children. However, from 2013, there was a shift to 2 NRTI and PI as the first-line regimen in children below 3 years of age, thus exposing more patients to PIs, with their known link to lipid derangements. It is possible that as more children start to use regimens containing PIs for longer periods of time, more lipid derangements may be observed among our children. Therefore, the current findings may serve as a baseline for future follow-up.
This study found that a late diagnosis of HIV infection and low CD4 counts at ARV initiation were associated with dyslipidemia. These findings were different from the study conducted by Chantry and Lapphra, who reported that dyslipidemia was associated with increase in CD4 counts.18,21 This contrast might be explained by the effect of the disease in lipid metabolism and possible liver involvement. It is known that the HIV virus on its own can cause significant lipid derangements. 20 Children infected with HIV usually succumb with high viral loads a few months after infection, and a study done by Calza showed that circulating levels of interferon-α in advanced stages of HIV disease increased production and reduced clearance of TGs, which eventually led to metabolic changes. 3
Although HIV treatment has improved in Tanzania due to the availability of drugs and proper diagnostic tools, HIV infection is still associated with high stigma and delays in presentation to the health facilities for treatment, leading many children to be diagnosed when they develop opportunistic infections as their first presentation. Late initiation of ARVs in advanced disease stages was a contributing factor to higher odds of dyslipidemia observed in this study.
The use of PIs was associated with higher odds (OR = 3.3) of dyslipidemia in our study, similar to previous reports from California. 20 Few of our participants were using PI-based regimens; however, most of them had advanced HIV disease and low CD4 counts at the time of ARVs initiation. PI-based regimens might have potential to cause dyslipidemia in these advanced disease stage patients.
Study strengths and limitations
A major strength of this study is determining the magnitude and risk factors for dyslipidemia in Tanzania, a resource-limited country similar to others in sub-Saharan Africa. A limitation of this study is its cross-sectional design, and the causal relationship between dyslipidemia and ARVs or HIV advanced disease cannot be firmly established. This limits generalization of the study to the global population of HIV-infected children. The use of nonfasting blood samples for measuring dyslipidemia might have affected the observed results, although the lack of fasting would probably have been to increase TG, LDL, and TC. The SS estimated was 266; however, we have been able to recruit only 260 participants during the study period; therefore, less of 6 (2.3%) from the planned SS which we expect could not have a large effect in this study findings.
Conclusion
Most patients in our study had normal levels of TC, TG, and LDL. Late diagnosis of HIV and low CD4 count increased odds of developing dyslipidemia. Patients on PIs were at higher risk of developing dyslipidemia with predominately low HDL levels. These observations provide useful baseline information for follow-up monitoring of lipid profiles in HIV-infected children who anticipate long-term use of ARVs. Future longitudinal or case control study is needed which might be able to follow up and record baseline and changes in lipid profile with time to assess true changes for future recommendations regarding drug effects on lipid changes.
Supplemental Material
Supplement_table_1 – Supplemental material for Dyslipidemia in HIV-Infected Children and Adolescents on Antiretroviral Therapy Receiving Care at Kilimanjaro Christian Medical Centre in Tanzania: A Cross-Sectional Study
Supplemental material, Supplement_table_1 for Dyslipidemia in HIV-Infected Children and Adolescents on Antiretroviral Therapy Receiving Care at Kilimanjaro Christian Medical Centre in Tanzania: A Cross-Sectional Study by Michael E Irira, Rune N Philemon, Joshua Y Mmbaga, Venancia Komba, John Bartlett, Grace D Kinabo and Blandina T Mmbaga in Infectious Diseases: Research and Treatment
Footnotes
Acknowledgements
We would like to extend our sincere gratitude to the study participants, the staff of Child Centered Family Care Clinic and the KCRI Biotechnology Laboratory for their support, especially Remigi Paschal and Linda Minja, and to the MEPI T84HA21123 Team for facilitation to accomplish the study objectives.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Medical Education Partnership Initiative award to the Kilimanjaro Christian Medical University College (HRSA T84HA21123). The study sponsor had no influence in data collection, data analysis, and manuscript writing for publication of this work.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MEI designed the study, participated in data collection, and performed statistical analysis and writing of the first draft manuscript. JYM and VK participated in study design, data collection, and manuscript writing. RNP, JB, and GDK contributed in reviewing the data and writing manuscript. BTM formulated the idea, designed the study, and performed statistical analysis. JB and BTM reviewed, edited, and approved final manuscript for final submission.
Availability of Data and Material
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval and Consent
Ethical approval was obtained from Kilimanjaro Christian Medical University College Research Ethics Committee. The permission to conduct the study was obtained from the hospital administration, the head of Pediatric and Child Health Department, and from the coordinator of CCFCC. Written consent was obtained from the parents/guardians on behalf of their children and assent for older children. Confidentiality was observed by using code numbers instead of names of the patients, and all data were stored unlinked to patient identifiers.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
