Abstract
Objective:
To characterize human immunodeficiency virus (HIV)-positive infants among HIV-exposed infants (HEIs) in Nairobi County for public health action.
Methods:
We conducted a retrospective review of records of HEIs’ HIV polymerase chain reaction (PCR) results in Nairobi County in 2015, excluding confirmatory PCR-positive results. HIV-exposed infant registry data were entered into Microsoft Excel and descriptive and associative statistics were calculated with OpenEpi software.
Results:
A total of 5802 HEI records were analyzed, with a median age of 4.5 (interquartile range, 8.5) months; 342 (5.9%) tested positive, 51% were female, and 4420 (76.2%) were seen at maternal and child health clinics. Standard highly active antiretroviral therapy intervention was given to 184 (60.3%) of the mothers of exposed infants, and 178 (53%) of the HIV-positive infants received active antiretroviral therapy. A total of 3464 (59.7%) HEIs had PCR done within the first 2 months, and 119 (3.4%) tested positive. The average turnaround time for PCR samples was 28 ± 22 days, and a total of 191 (3.3%) HIV-positive HEIs were exclusively breastfed for the first 6 months of life.
Conclusion:
The recorded mother-to-child transmission (MTCT) rate of 5.9% in Nairobi County is lower than the 2015 national rate, which suggests the Nairobi prevention of mother-to-child transmission efforts are moderately effective. However, results from this study show that Kenya will struggle to meet the MTCT 2020 target rate of <5%.
Introduction
Globally, there are approximately 2.1 million new human immunodeficiency virus (HIV) infections each year, 150 000 of which are among children. 1 More than 210 000 new HIV infections still occur among infants in sub-Saharan Africa. 2 New HIV infections among children in Kenya declined from 44 000 in 2000 to 12 940 in 2013. AIDS-related deaths claimed 10 390 children in 2013, and 103 000 children remained in need of antiretroviral therapy (ART) by the year 2015. 3 The standard ART regimen is what is stipulated in the ART national guidelines (for children: ABC/3TC/LPV/r or AZT/3TC/EFV or TDF/3TC/DTG); for adults: TDF/3TC/DTG or TDF/3TC/LPV/r or TDF/3TC/ATV/r). There is limited information on new HIV infections among children who live within Nairobi County, especially among HIV-exposed infants (HEIs). This study aimed to characterize the epidemiology of HIV among HEIs in Nairobi County, 2015, using the early infant diagnosis (EID) database.
Methods
Study design
We conducted a retrospective review of records of HEIs’ HIV PCR results in Nairobi County in 2015. According to the Kenya prevention of mother-to-child transmission (PMTCT) guidelines, HIV diagnosis of HEIs is made by a positive virological test using deoxyribonucleic acid polymerase chain reaction (DNA-PCR) test at 6 weeks or as early as possible thereafter. 4
Study population
The study population was all HEIs in Nairobi County, Kenya. The case definition was all HEIs whose HIV PCR test was done and results available in Nairobi County in 2015. Inclusion criteria were all HIV PCR results, excluding confirmatory/repeat PCR-positive results.
Study setting
This study was conducted at Nairobi City-County Health Department, reviewing HEI PCR data of 289 health facilities offering prevention of PMTCT services (Figure 1).
Nairobi City-County map showing constituencies and associated boundaries.
Data collection
We assessed standard maternal and infant data, including demographic information, type of maternal ART received, infant feeding options used, facility-entry point at diagnosis, and infants’ age at testing. Data were abstracted, cleaned, and entered into an Microsoft Excel spreadsheet. Descriptive and associative statistics were calculated using OpenEpi. We conducted a comprehensive audit of PMTCT registry data to check for accuracy and completeness using a standard assessment tool to ensure that the data reviewed were of quality and met the expected threshold of 90% for it to be admissible for use. 5
Ethical review
Both the Nairobi City-County Health Department and the Field Epidemiology and Laboratory Training Program review committees approved this study.
Results
HEI demographic characteristics
A total of 5802 HEI data records were analyzed, with a median age of 4.5 (interquartile range, 8.5) months, and 51.1% were female. The HEIs receiving HIV-negative and HIV-positive results were 5460 (94.1%) and 342 (5.9%), respectively. The proportion of females and males receiving HIV-positive results were 181 (52.7%) and 161 (47.3%), respectively.
HEI clinical characteristics
A total of 3464 (59.7%) HEIs had HIV PCR testing done within the first 2 months of life (per World Health Organization [WHO] guidelines), 6 of which 119 (3.4%) tested positive (Table 1). Most HEIs (4420 [76.2%]) were first identified and services provided to them at maternal and child health clinics/service delivery points, where a total of 246 (5.6%) were HIV-positive. Regarding the feeding options, 207 (4.2%) HEIs who received HIV-positive results were exclusively breastfed (EBF) for the first 6 months of life, and 35 (13.3%) HEIs who continued to breastfeed beyond 6 months of age were HIV-positive.
Demographic and clinical characteristics of HIV-positive HIV-exposed infants, Nairobi County, 2015 (N = 5802).
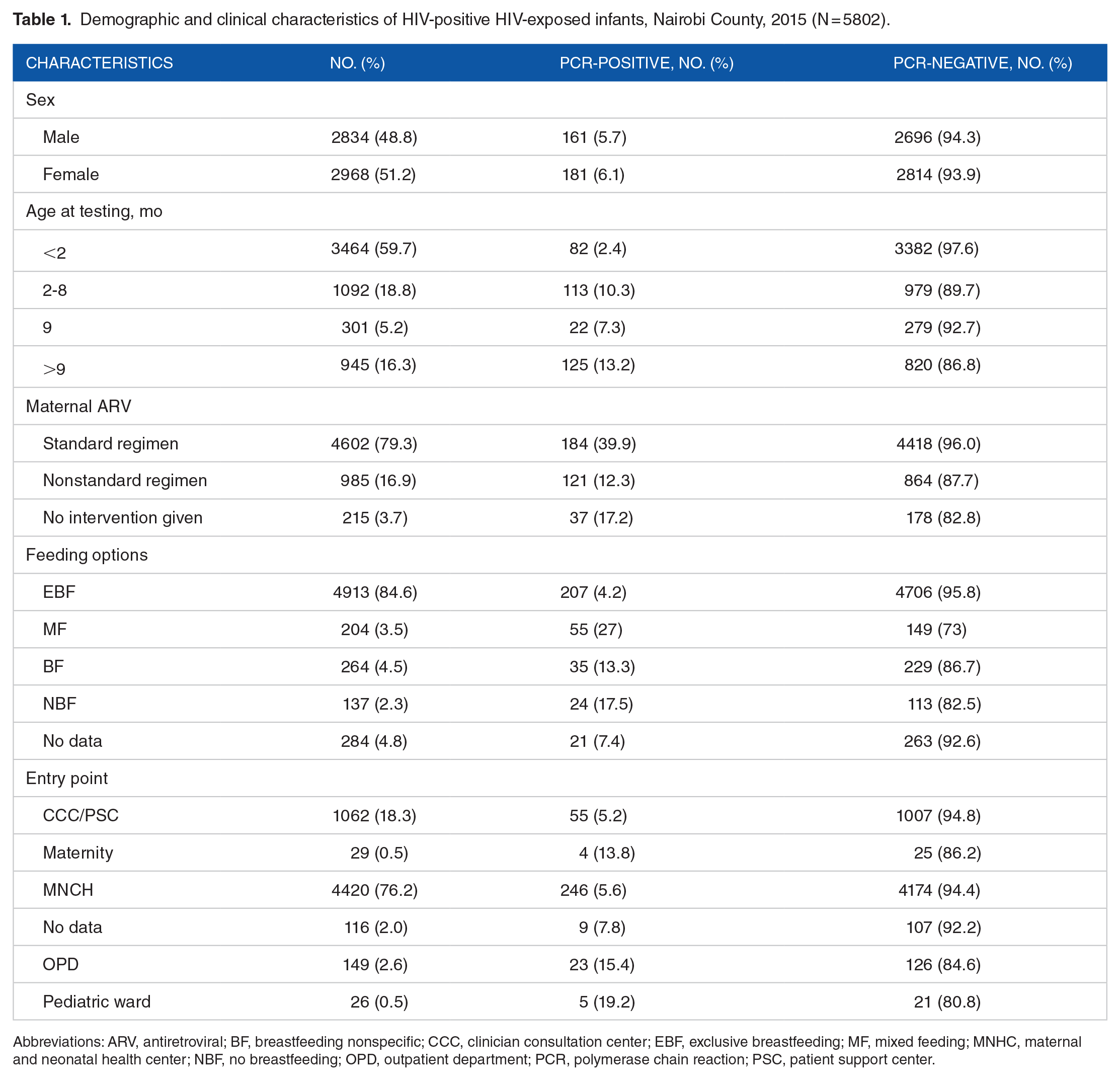
Abbreviations: ARV, antiretroviral; BF, breastfeeding nonspecific; CCC, clinician consultation center; EBF, exclusive breastfeeding; MF, mixed feeding; MNHC, maternal and neonatal health center; NBF, no breastfeeding; OPD, outpatient department; PCR, polymerase chain reaction; PSC, patient support center.
Maternal characteristics
The maternal highly active antiretroviral therapy (HAART) intervention issued (standard ART regimens prescribed as per Kenya ART guidelines, which adopted additional WHO modifications to PMTCT guidelines to include 12 weeks of infant prophylaxis with nevirapine [NVP]) was 184 (53.8%), and 178 (52%) HEIs received antiretroviral (ARV) prophylaxis (NVP syrup for 12 weeks) against total identified HIV-positive infants.
Sex was not significantly associated with an HEI becoming HIV-positive (odds ratio [OR] = 0.9353, 95% confidence interval [CI] = 0.7383-1.184, P = .5782) (Table 2). Exclusively breastfed HEIs were much less likely to be HIV-positive (OR = 0.119, 95% CI = 0.085-0.1673, P < .00001) compared with mixed-fed HEIs. However, any type of BF was not significantly more protective compared with positivity proportions in non-breastfed HEIs (OR = 0.7196, 95% CI = 0.4085-1.268, P = .2548). Regarding maternal treatment regimen, HEIs of women who received standard regimen ART were significantly protected against HIV infection compared with HEIs of women who received a nonstandard ART regimen (OR = 0.2974, 95% CI = 0.2338-0.3783, P < .00001).
Bivariate analysis of HIV-positive infants among HIV-exposed infants, Nairobi County, Kenya, 2015 (N = 5802).
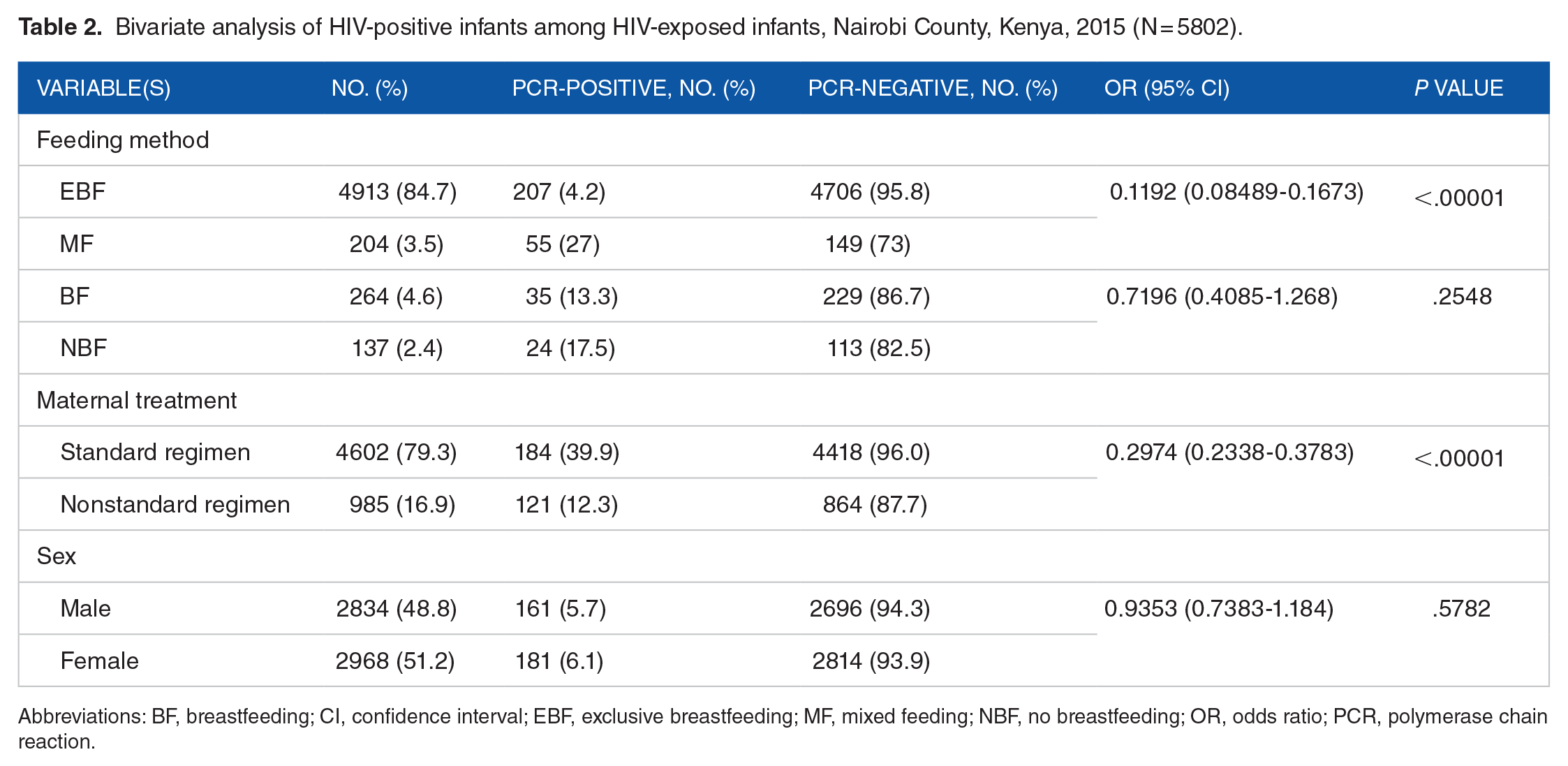
Abbreviations: BF, breastfeeding; CI, confidence interval; EBF, exclusive breastfeeding; MF, mixed feeding; NBF, no breastfeeding; OR, odds ratio; PCR, polymerase chain reaction.
Discussion
Many HIV-positive pregnant women have benefited from PMTCT interventions, with 90% receiving maternal interventions and 83% receiving ARV prophylaxis. 3 However, our results show that despite the interventions in place for PMTCT at Nairobi County health facilities, too many HEIs become HIV-positive. This study found an MTCT rate of 5.9% in Nairobi in 2015, although this is much lower than the 15.1% MTCT rate found in other Kenyan studies. 7 Although multiple studies have noted that more female HEIs are HIV-positive, their findings correspond with ours in that there is no clear relationship between sex and acquiring HIV. 8 As for breastfeeding options, our data show that EBF for the first 6 months of life is significantly associated with HEI remaining negative when compared with other feeding options. These findings are consistent with other studies in Kenya and other parts of east Africa.9-11
A total of 3464 HEIs (59.7%) had HIV PCR testing done within the first 2 months, of which 119 (3.4%) were identified HIV-positive, which is consistent with results from another Kenyan study that found that 42.4% of HEIs had DNA PCR testing done within the first 2 months of life. 12 Most of the 292 (6.3%) HEIs who tested positive were older than those who tested negative, which is consistent with other findings in Kenya. 12 Possible explanations for this disparity are late entry at enrollment into EID program and high dropout rates, which are associated with increased rates of positivity. 13 Reluctance to infant testing may be related to various issues around acceptability. Cultural norms dictating that young infants should not be taken outside the house except for receipt of vaccine (not always co-located with EID) may provide insights into this observation.
Despite the provision of maternal HAART intervention, many HEIs were identified positive, which may be associated with high maternal viral load during pregnancy and delivery, which increases risk of perinatal HIV transmission, even among women treated with ART. 14
Our study showed that the turnaround time for PCR results was 28 days, which is longer than the expected averages of approximately 24 days in other studies, which showed temporal relationships with turnaround time. 15 This has also likely contributed to higher rates of loss to follow-up and poor retention of HEIs to care and delayed initiation into care of HIV-positive HEIs.
Footnotes
Acknowledgements
The authors are grateful to the HIV staff of all the Nairobi County sites who provided data for the study. The authors also thank the leadership at the Nairobi City-County Health Department and the Field Epidemiology and Laboratory Training Program for the support offered in facilitating this study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this paper was provided by Field Epidemiology and Laboratory Training Program, which is part of the Ministry of Health, Nairobi, Kenya.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ABK jointly conceived the study with JR, CN, and EO. ABK and JR analyzed the data and wrote the manuscript with supervision from CN and EO. All authors discussed results and contributed to the manuscript at different stages of development.
