Abstract
Meningitis infection is one of the major threats during Hajj season in Mecca. Meningitis vaccines are available, but their uses are limited in some countries due to religious reasons. Furthermore, they only give protection to certain serogroups, not to all types of meningitis-inducing bacteria. Recently, research on epitope-based vaccines has been developed intensively. Such vaccines have potential advantages over conventional vaccines in that they are safer to use and well responded to the antibody. In this study, we developed epitope-based vaccine candidates against various meningitis-inducing bacteria, including Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae type b. The epitopes were selected from their protein of polysaccharide capsule. B-cell epitopes were predicted by using BCPred, while T-cell epitope for major histocompatibility complex (MHC) class I was predicted using PAProC, TAPPred, and Immune Epitope Database. Immune Epitope Database was also used to predict T-cell epitope for MHC class II. Population coverage and molecular docking simulation were predicted against previously generated epitope vaccine candidates. The best candidates for MHC class I- and class II-restricted T-cell epitopes were MQYGDKTTF, MKEQNTLEI, ECTEGEPDY, DLSIVVPIY, YPMAMMWRNASNRAI, TLQMTLLGIVPNLNK, ETSLHHIPGISNYFI, and SLLYILEKNAEMEFD, which showed 80% population coverage. The complexes of class I T-cell epitopes-HLA-C*03:03 and class II T-cell epitopes-HLA-DRB1*11:01 showed better affinity than standards as evaluated from their δGbinding value and the binding interaction between epitopes and HLA molecules. These peptide constructs may further be undergone in vitro and in vivo testings for the development of targeted vaccine against meningitis infection.
Introduction
Meningitis infection is one of the serious threats during Hajj due to its tendency to cause outbreaks and epidemics. 1 According to WHO, this disease affects more than 400 million people who live in the area of “African meningitis belt” (from Senegal to Ethiopia). More than 800,000 people in this area were infected, with a case fatality rate of 10%. 2 In Saudi Arabia, the epidemics of meningitis usually occur during or after Hajj and Umrah seasons, due to massive gathering of people in certain areas. Pattern evolution confirmed that 48% of meningitis cases were reported at the two holy cities, namely, Mecca and Medina. 2 In 2000, Indonesian pilgrims were infected by Neisseria meningitidis serogroup A and W135. Among 253 identified cases from Saudi Arabia, 93 cases were caused by N. meningitidis serogroup W135, while 60 cases were caused by N. meningitidis serogroup A. Statistically, there were nine cases caused by serogroup W135 and six cases caused by serogroup A per 100,000 population. 3
Generally, meningitis can be induced by certain species of virus and bacteria. Three bacterial species, namely, Haemophilus influenzae (45%), Streptococcus pneumoniae (18%), and N. meningitidis (14%), are known to cause the majority of cases, where the case fatality rates vary according to the type of bacteria. The highest fatality rate is caused by S. pneumoniae (19%), while the case fatality rates caused by N. meningitidis and H. influenzae are 13% and 3%, respectively. 4
The Indonesian government requests all Hajj pilgrims to be vaccinated against meningitis bacteria before departing to Mecca. Currently, the government allows the use of Meningitis vaccine only from Novartis, Menveo¯. This vaccine contains polysaccharides from each of serogroups A, C, W, and Y conjugated to a mutant of diphtheria toxin, CRM197, that differs from the wild type by the substitution of one amino acid.5,6 There have been some efforts to develop alternative vaccine candidates that could incorporate broader types of meningitis-inducing bacteria.7–10
Recently, novel approaches have been directed toward the rational design of B- and T-cell epitope-based vaccine, on account of the advancement of recombinant DNA technology, cell culture technique, immunoinformatics, big data projects, and rational design of antigens.11,12 The epitope-based vaccine has several advantages over conventional vaccines, and some are moving forward to the clinical trial pipeline. 12 This next-generation vaccine has high specificity in evoking immune response, high capacity of production, and effective cost of production. Moreover, peptides consisting of epitopes are easily synthesized, purified, stored, and handled. Generally, epitope-based vaccines are also considered safer than traditional vaccines. 12
Immunoinformatics approach has been used to develop subunit vaccine candidates against meningitis-inducing bacteria. Some previous studies generated several vaccine candidates: epitope FMILPIFNV against human leukocyte antigen (HLA) class II from ABC transporter protein of S. pneumoniae and epitope KGLVDDADI against HLA class I from outer membrane protein of N. meningitidis.13,14 The epitopes such as FMILPIFNV and KGLVDDADI were used as standards in this study.
This study designed epitope-based meningitis vaccine in silico by using polysaccharide capsule protein of S. pneumoniae, N. meningitidis serogroup A, N. meningitidis serogroup W, and H. influenzae type b and analyzed the complex stability between predicted epitopes and HLA molecules using molecular docking approach. The designed epitopes may serve as promising candidates for the development of epitope-based vaccine against the meningitis-inducing bacteria.
Research Methodology
Tools and materials
This study was conducted in silico. The pipeline used in this study was adjusted and extended from the existing ones.15–19 Polysaccharide proteins of S. pneumoniae, N. meningitidis, and H. influenzae type b were obtained from National Center Biotechnology information (NCBI). The 3D structure of HLA was obtained from Research Collaboratory for Structural Bioinformatics (RCSB). Online and offline software including the latest version of BCPred, 20 VaxiJen v2.0, 21 PAPRoc I, 22 TAPPRed,23,24 Immune Epitope Database (IEDB) 3.0, 25 PEP-FOLD, 26 MOE 2009, 27 and Chimera 1.928 were used in this study.
Procedure
Retrieving protein sequences from database
The sequences of polysaccharide protein of S. pneumoniae, N. meningitidis, and H. influenzae type b were searched in GenBank of NCBI (http://www.ncbi.nlm.nih.gov/). After that, antigenicity of these sequences was predicted using VaxiJen v2.0, which can be obtained at http://www.ddg-pharmfac.net/vaxijen/VaxiJen/VaxiJen.html. 21
B-cell epitope prediction
The B-cell epitope was predicted using BCPred, which can be accessed at http://ailab.cs.iastate.edu/bcpreds/. 20 Antigenicity was also predicted against B-cell epitope afterward using VaxiJen.
HLA class I and class II T-cell epitope prediction from the conserved sequences
Epitopes from S. pneumoniae, N. meningitidis, and H. influenzae type b against HLA class I were predicted using several online softwares including PAProC, 22 TAPPred, 23 and IEDB. 25 Proteasome cleavage site of consensus protein sequence was predicted using PAProC (http://www.paproc2.de/paproc1/paproc1.html). PAProC then generated peptide sequences and score of estimated strength. 29 The peptide binding to the transporter associated with antigen processing (TAP) binding was predicted using TAPPred, http://www.imtech.res.in/raghava/tappred/, which generated protein sequences, their position, and score of predicted binding affinity to each peptide sequence. 23 The antigenicity of peptide sequence generated from TAPPred prediction was analyzed using VaxiJen, 21 and the antigen-bearing sequences were used to predict epitopes that bind to HLA class I and HLA class II using IEDB analysis resource. 25 The binding character of epitopes to HLA was taken into consideration for the selection of the best epitopes. Epitopes with more number of bonds with HLA were considered as better than those with fewer bonds. 16
The 3D structure of epitopes for HLA class I and class II
The 3D structures of the best epitopes for each HLA class I and class II were predicted using PEP-FOLD (http://bioserv.rpbs.univ-paris-diderot.fr/services/PEP-FOLD/) 26 and saved in.pdb format, while the 3D structures of HLA class I and class II were obtained from RCSB (www.rcsb.org). 30
The prediction of coverage population of the selected epitopes
The human population coverage is one aspect that has to be taken into consideration in selecting the best epitopes, besides their ability to bind to HLA molecules. 16 Human coverage population for previously selected epitopes was predicted using IEDB analysis resource for population coverage calculation (http://tools.immuneepitope.org/tools/population/iedb_input). 31
Molecular docking study of HLA–-epitope interaction
The interaction between the best predicted epitopes and HLA alleles was analyzed by means of molecular docking using MOE 2009. The 3D structure of HLA, as a target molecule, and the epitopes must be prepared before docking. Preparation and docking steps were performed according to the established pipeline from previous research.32,33
Results
Protein sequence searching
A total of 4 polysaccharide protein sequences of H. influenzae type b, 3069 sequences of S. pneumoniae, 19 sequences of N. meningitidis serogroup A, and 19 sequences of N. meningitidis serogroup W were retrieved from NCBI. These sequences were subjected to antigenicity prediction in order to estimate the presence of antigen in their sequences. The peptide sequences having the VaxiJen value above the threshold level (≥0.4) were used for further analysis.
B-cell epitope prediction
B-cell epitope prediction.
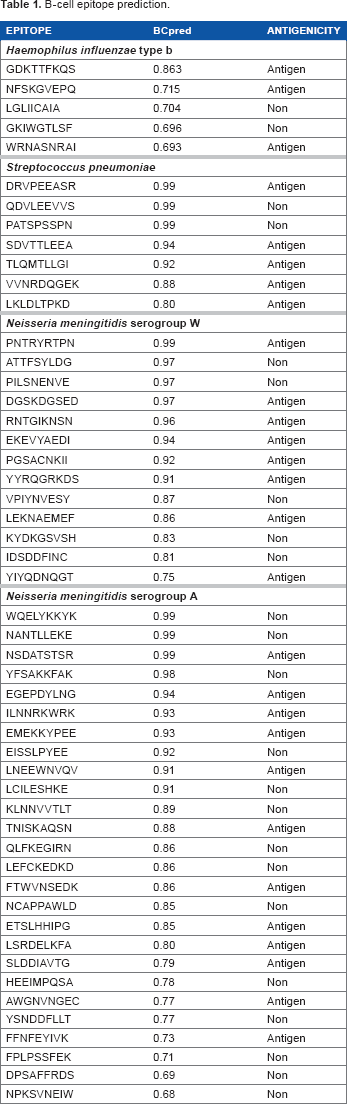
Besides being recognized by B-cells, the peptide sequences must also possess antigen as evaluated by VaxiJen. The peptides with antigenic properties are necessary to raise the immune responses. 18 Not all peptides from each bacteria passed these criteria, as listed in Table 1. H. influenzae protein, S. pneumoniae protein, N. meningitidis serogroup A, and N. meningitidis serogroup W protein generated 3, 5, 12, and 8 probable epitopes, respectively, with antigen attribute. The predicted B-cell epitopes from each bacteria protein, which produced the best BCPred score, were GDKTTFKQS, DRVPEEASR, PNTRYRTPN, and WQELYKKYK.
Identification of T-cell epitopes
T-cell epitopes must be recognized by T-cell receptor (TCR) in order to induce immune response. The epitopes that TCRs recognized are presented by major histocompatibility complex (MHC) molecules on the surface of cells. There are two major types of MHC protein molecules, namely, class I and class II. Cytotoxic T-lymphocyte TCRs recognize endogenous antigen presented on MHC class I, while helper T-cell and inflammatory T-cell TCRs recognize exogenous antigen presented on MHC class II. 35
Identification of MHC class I epitope
The result of TAP prediction.
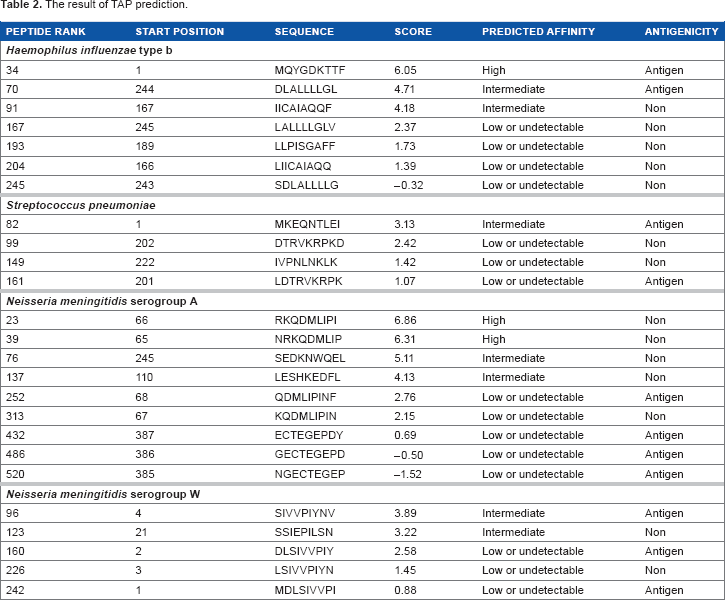
Candidates of T-cell epitope for MHC class I.

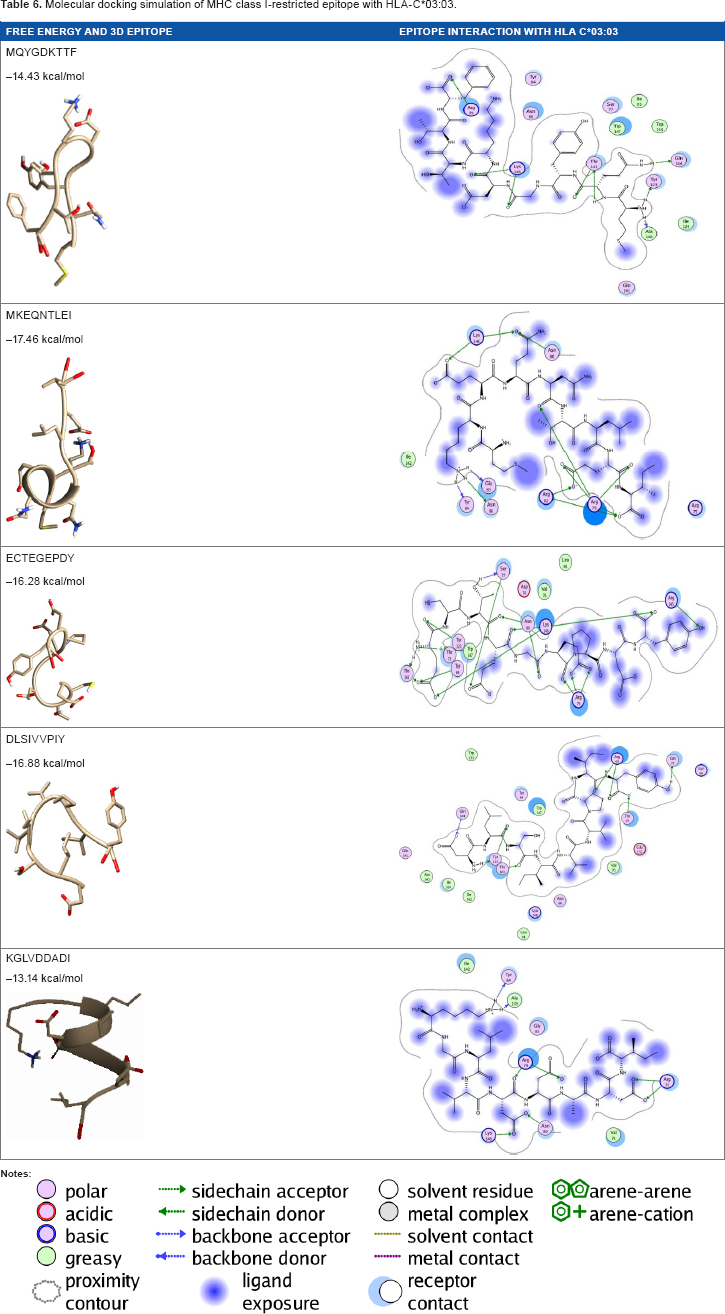
The analysis in stabilized matrix method-based IEDB MHC I prediction tool retrieved several possible MHC I alleles that could interact well with the epitopes from four bacteria (Table 3). Each predicted MHC I allele was given an IC50 value that indicates affinity between epitope and MHC I molecule. A lower IC50 value indicates higher affinity toward MHC molecules. A peptide showing an IC50 value lower than 50, 500, and 5000 nM is associated with high affinity, intermediate affinity, and low affinity toward MHC class I molecule, respectively. Moreover, a peptide is categorized as a binder if it has IC50 lower than 500 nM and is categorized as a nonbinder if IC50 is equal to or more than 500 nM. 37 In this study, the peptides were selected if they possess antigenicity, IC50 value lower than 500 nM, and more than 5 bonds with HLA class I. 16 Our study identified four antigenic peptides (MQYGDKTTF, MKEQNTLEI, ECTEGEPDY, and DLSIVVPIY) as candidates of T-cell epitope for MHC class I that could interact with HLA-B*15:02, HLA-C*03:03, and HLA-C*14:02 (Table 3). Of these three types mentioned, HLA-C*03:03 has the best interaction with the selected epitope candidates. Therefore, HLA-C*03:03 structure was chosen as a model for molecular docking study of HLA peptides.
Identification of MHC Class II Epitope
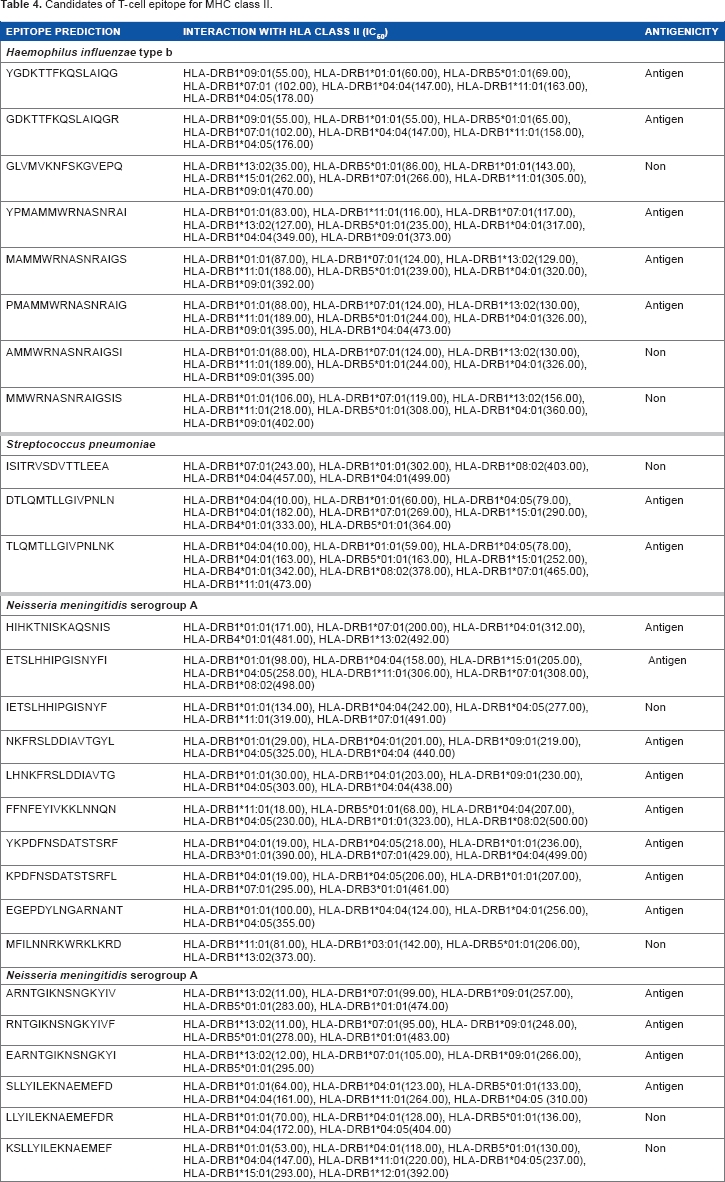
MHC class II molecules present antigenic peptides to stimulate cellular and humoral immunity through the actions of helper T-lymphocytes. Identification of MHC class II-restricted epitope is very important in designing epitope-based vaccine.
Candidates of T-cell epitope for MHC class II.
Coverage population prediction of class I and class II epitopes
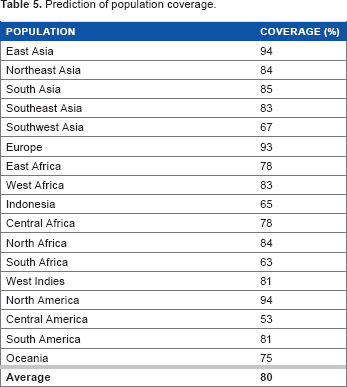
Prediction of population coverage.
Molecular docking
Molecular docking simulation of MHC class I-restricted epitope with HLA-C*03:03.
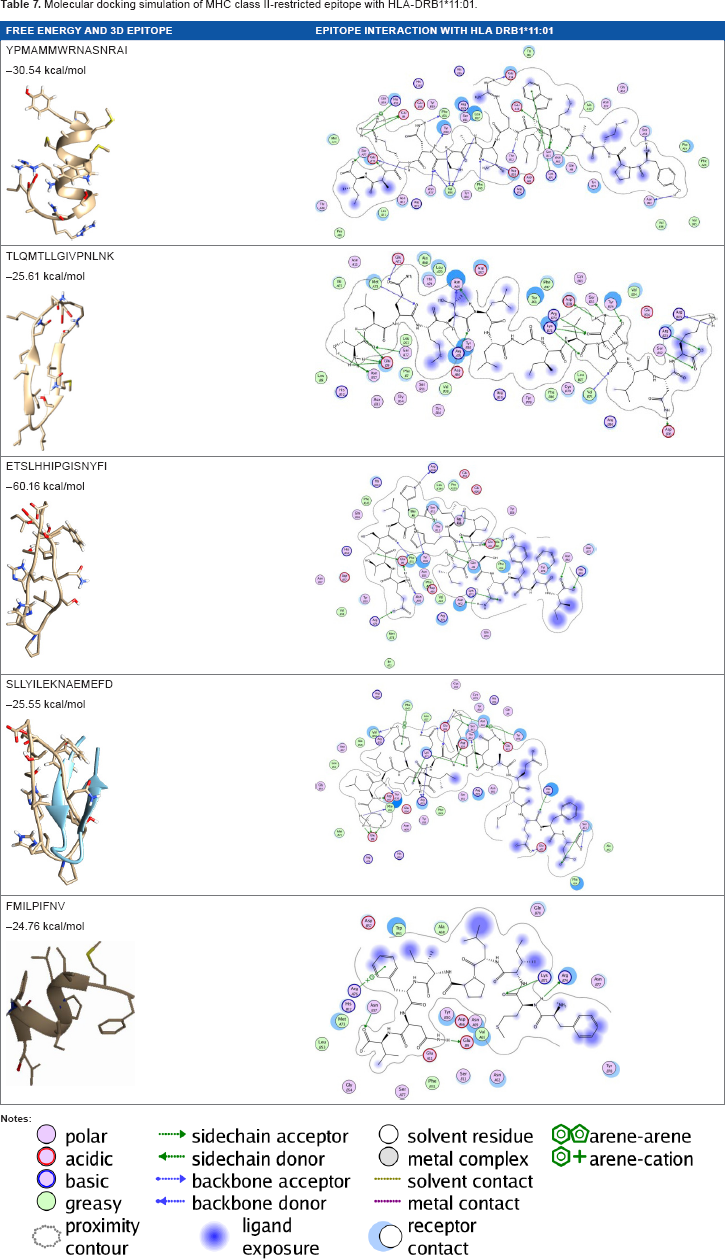
Molecular docking simulation of MHC class II-restricted epitope with HLA-DRB1*11:01.
Discussion
The main objective of epitope prediction is to design a molecule that can replace an antigen in the process of eliciting a relevant immune response. Designed molecules are favorable to use in vaccine production because they are cost-effective and noninfectious in contrast to whole pathogen organisms, which might possess risks to researchers or experimental subjects (animals and humans). This study incorporates immunoinformatics approach to reducing time- and cost-consuming hit and trial sets of wet laboratory experiments. This approach is used for the prediction of antigenic determinants in the capsular protein sequence of H. influenzae type b, S. pneumoniae, and N. meningitidis serogroup A and W.
According to the prediction result of IEDB and molecular docking study, the peptides that passed several criteria of probable epitope such as possessing antigenicity, binder attribute, and good affinity with HLA molecules are MQYGDKTTF, MKEQNTLEI, ECTEGEPDY, and DLSIVVPIY for MHC class I and YPMAMMWRNASNRAI, TLQMTLLGIVPNLNK, ETSLHHIPGISNYFI, and SLLYILEKNAEMEFD for MHC class II. These peptides also passed proteasomal cleavage and TAP binding efficiency prediction, which are of main concerns in designing good epitopes for vaccine candidates. Molecular docking study has been widely used in computer-aided drug design. However, it is now applied to investigate the epitope candidates that could bind MHC class I and class II molecules. Computational immunology is now considered to contribute to vaccine design in the way computational chemistry contributes to drug design. 35 The algorithms for epitopes identification served by IEDB and molecular docking study have increased the overall efficiency in epitope discovery for vaccine research.
These epitopes also showed good population coverage (80% in average) and reached above average values in East Asia, North America, and Europe population. The high value of population coverage is needed to minimize the complexity of putting different epitopes in the development of vaccine. 31 The predicted epitopes can be synthesized for further in vitro and in vivo assays.
Conclusion
We have predicted numerous antigenic peptides from the capsular protein sequence of H. influenzae type b, S. pneumoniae, and N. meningitidis serogroup A and W, which would be beneficial for effective vaccine development against meningococcal diseases. Results indicated that MQYGDKTTF, MKEQNTLEI, ECTEGEPDY, and DLSIVVPIY are potential vaccine candidates that have considerable binding with MHC class I alleles, while YPMAMMWRNASNRAI, TLQMTLLGIVPNLNK, ETSLHHIPGISNYFI, and SLLYILEKNAEMEFD are the potential candidates for MHC class II-restricted T-cell epitopes. These epitopes also had low energy minimization values that favored the stability of the epitope-MHC allele complex. However, experiments using model animals should be performed to verify their suitability to be included in a vaccine formulation against meningococcal diseases.
Author Contributions
Supervised this research: USFT and HZ. Worked on the technical details: AM. Gave technical assistance to the whole process: AAP. All the authors are responsible for writing this manuscript.
Footnotes
Abbreviations
Acknowledgment
The authors thank Directorate of Research and Community Engagement, University of Indonesia, for providing facilities for this research.
