Abstract
Modern medicinal plant drug discovery has provided pharmacologically active compounds targeted against a multitude of conditions and diseases, such as infection, inflammation, and cancer. To date, natural products from medicinal plants remain a solid niche as a source from which cancer therapies can be derived. Among other properties, one favorable characteristic of an anticancer drug is its ability to block the uncontrollable process of cell division, as cancer cells are notorious for their abnormal cell division. There are numerous other documented works on the potential anticancer activity of drugs derived from medicinal plants, and their effects on cell division are an attractive and growing therapeutic target. Despite this, there remains a vast number of unidentified natural products that are potentially promising sources for medical applications. This mini review aims to revise the current knowledge of the effects of natural plant products on cell division.
Introduction
Human beings have long used plants as a medicinal source. Their use has grown more sophisticated with modern chemists using compounds isolated from plants as a basis for generating novel compounds with additional benefits, such as its lower toxicity and potential for combating drug-resistant diseases. Between 1981 and 2010, naturally derived products and their mimics composed an estimated 70% of new chemical compounds reported. 1 Naturally derived compounds with anticancer activity have also been used as the basis for original synthetic analogs, forming their own novel class of chemical compounds. 2
Mammalian microtubules appear to be a common target for naturally occurring toxic molecules produced by a large number of flora, presumably with the original intent of self-defense. Microtubules are a component of the cytoskeleton, found throughout the cell cytoplasm, which is important in the process of mitosis (ie, cell division). Most microtubule-targeting compounds have been discovered in large-scale screens of natural products3,4 (Table 1). Approximately 75% of the available anticancer drugs between 1940 and 2010 were naturally derived products or their mimics. Additionally, of the seven anticancer drugs approved in 2010, almost half of them exert their effects by binding onto microtubules. 1
Selected compounds originally isolated from natural sources that act on microtubules.
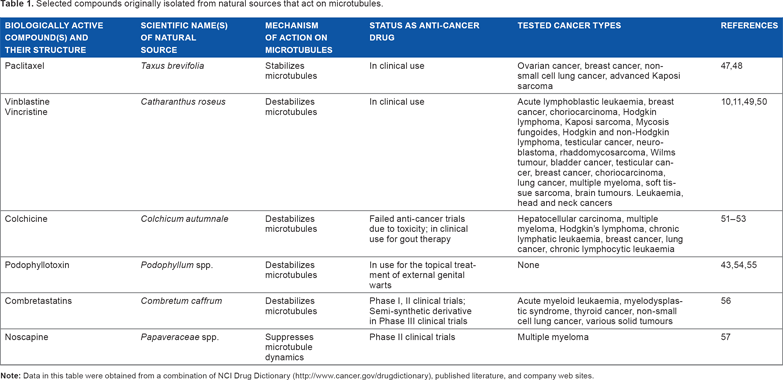
One of the biggest success stories of microtubule-targeted compounds from a naturally derived source is Paclitaxel (commercially known as Taxol), a member of the Taxane family. Paclitaxel is extracted from the bark of the Pacific yew tree (Taxus brevifolia) and acts as an antimitotic drug, by binding to microtubules, thus stabilizing them and arresting cells in mitosis.5–9 Taxol and its derivatives have successfully been used clinically to treat ovarian cancer, breast cancer, and non-small cell lung cancer for almost 40 years, making Taxol the best-selling anticancer drug currently manufactured. Its success has sparked the search for similar microtubule-stabilizing compounds.
Another class of microtubule-targeted compounds from a naturally derived source is the vinca alkaloids, vincristine and vinblastine, which were initially isolated from the Madagascar periwinkle plant (Catharanthus roseus). 10 The vinca alkaloids are microtubule destabilizers and have proven to be particularly effective against hematological malignancies, 11 and their success has generated several semisynthetic derivatives. Semisynthetic and synthetic derivatives may offer advantages over a fully natural source, as the bioactive natural compound may be present only in trace amounts. Natural compounds may instead act as lead compounds, where analogs with higher potencies and lower toxicities may be developed12,13 (Table 2). Natural products are ideal as lead compounds as their chemical structures are complex and diverse (Table 3). The biggest study looking into the isolation of compounds with clinical bioactivity, specifically anticancer activity, from natural sources was done by the National Cancer Institute (NCI) of the National Institute of Health in USA from 1960 to 1980. 14 However, it is estimated that >90% of plant species worldwide remain understudied. Discovery of drug molecules has been limited because of genomic instability and drug resistance characteristics in certain cancer cells. 15 Therefore, modern drug discovery has shifted to personalized treatment of patients, where drugs are selected for specific molecular targets, taking advantage of the vulnerabilities of cancer in a particular patient, leading to increased interest in studying traditional herbs as an alternative source of anticancer drugs because of its multitargeted characteristic. 16
Selected synthetic and semisynthetic compounds originally isolated from natural sources that act on microtubules.
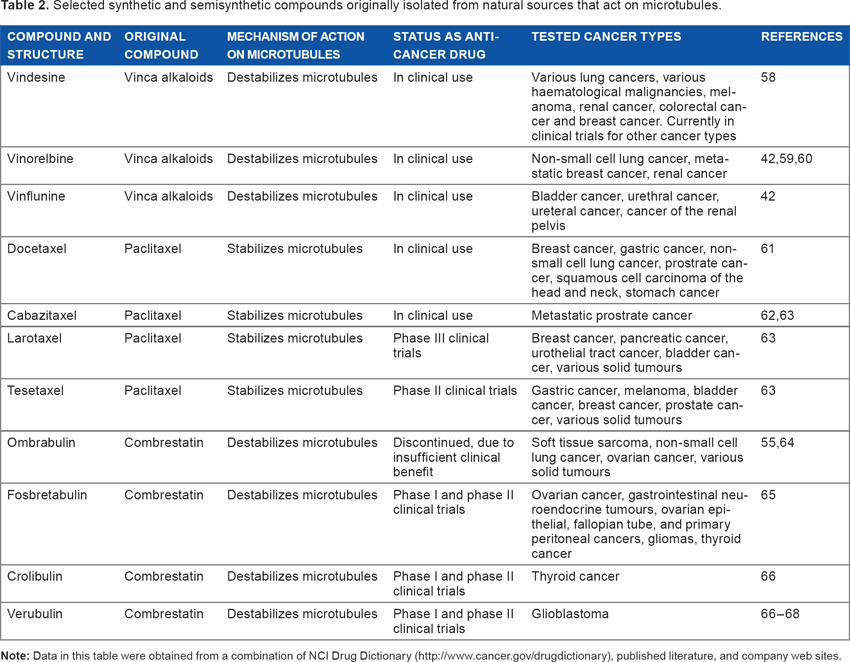
Chemical structures of natural microtubule-targeting compounds and their synthetic and semisynthetic derivatives.
The Role of the Microtubule in Cell Division
Microtubules are a class of the cytoskeletal proteins present in all eukaryotic cells. They form long, filamentous, polymeric structures within the cell, composed of α and β-tubulin heterodimers, of which there are several isotypes. 17 The different isotypes of tubulin in human beings are summarized in Table 4. Microtubules play many roles in eukaryotic cells, including development and maintenance of cell shape, 18 intracellular transport, 19 cell motility,20,21 cell signaling, 22 and cell division. 23
Subtypes and isoforms of microtubules.

Cell division, or mitosis, is a crucial event in the cell cycle that results in the division of a single cell into two identical daughter cells with the equal distribution of genetic materials (Fig. 1). During mitosis, the cytoskeleton forms a superstructure called the mitotic spindle, which facilitates many of the cell division processes. Mitosis involves a series of stages. The initial prophase and prometaphase stages are where there is condensation of chromosomes, which then attaches to the mitotic spindle. The chromosomes then align at the equator of the mitotic spindle (metaphase) before the sister chromosomes segregate into daughter cells (anaphase). The final stage is where the chromosomes decondense and the cells divide fully into two daughter cells (telophase). All the stages of mitosis must be regulated for the proper development and function of a multicellular organism. Central to the function of microtubules is the regulation of microtubule dynamics. Microtubule filaments are able to polymerize and depolymerize stochastically within a cell, in what is termed as microtubule dynamicity. 24 Microtubule dynamics are tightly regulated within cells, through the binding of various regulatory proteins, expression of different tubulin isotypes, and posttranslational modifications of tubulin subunits.25,26 Dynamic microtubules have a very short half-life of a few minutes, or even seconds, whereas stable microtubules have half-lives of minutes to hours. 27
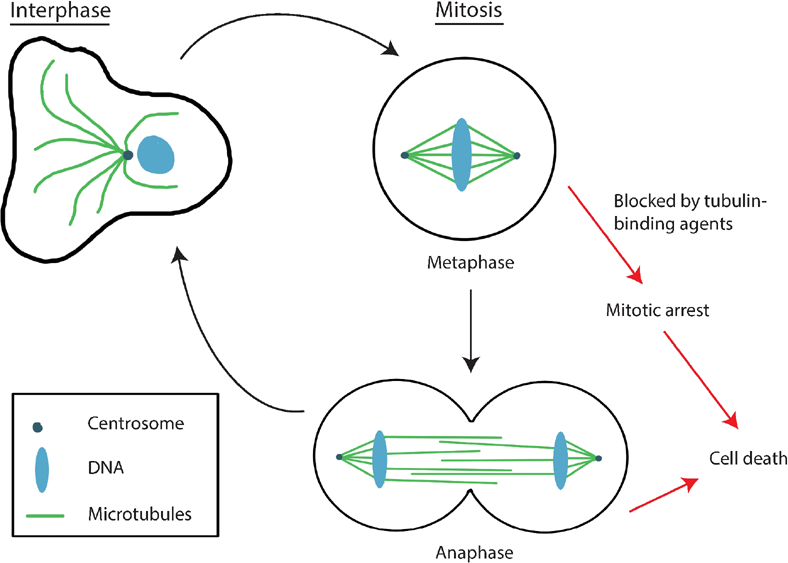
The process of cell division in mammalian cells. This figure illustrates the different microtubule structures present during different stages of the cell cycle. In the interphase stage of the cell cycle, microtubules (green) emanate out from the microtubule-organizing center, the centrosome (dark blue circle), forming an array that extends toward the cell periphery. During the mitotic stage of the cell cycle, the centrosomes are duplicated and separated to form spindle poles, while the microtubule cytoskeleton is reorganized to form a superstructure called the mitotic spindle. The mitotic spindle is responsible for mitotic events such as chromosome congression and chromosome segregation. Two stages of the mitotic stage of the cell cycle are illustrated–-metaphase and anaphase. At metaphase, the mitotic spindle holds sister chromatids (blue) together at the cell equator. At anaphase, the cell elongates the spindle poles move further apart and the sister chromatids move toward the opposite poles. Black arrows indicate the path normally followed by a cell in a cell cycle. When the cell cycle is disrupted at mitosis by tubulin-binding agents, the cell is unable to complete mitosis and follows an alternative pathway (red arrows) where it undergoes mitotic arrest and eventually cell death. All stages of mitosis must be regulated for proper development and function of a multicellular organism. Unregulated mitosis may lead to an overgrowth of cells, as in cancer. The ability to carry out an infinite number of cell divisions is one of the hallmarks of cancer. Blockage of any stage of mitosis may not allow the cells to complete mitosis, resulting in cell cycle arrest and ultimately, cell death.
During mitosis, microtubules are the main components of the mitotic spindle, where microtubule dynamics are increased significantly.27,28 Dynamic microtubules are required for all stages of mitosis: from capturing and congressing chromosomes to the metaphase plate, 29 pulling chromosomes toward opposite poles and initiating anaphase, 30 and finally cytokinesis to complete mitosis.31,32 Microtubule-binding compounds may either stabilize microtubules (promoting growth and not supporting shrinkage of the microtubule filament) or destabilize microtubules (promoting shrinkage and not supporting growth of the microtubule filament). Any alterations in microtubule dynamics will affect the different events in mitosis. For example, if microtubule dynamics are suppressed, chromosomes may not be able to congress to the metaphase plate.33,34 The presence of a single uncongressed chromosome is enough to induce mitotic arrest. 1 Accordingly, altered microtubule dynamics is among the major causes of mitotic arrest. A cell that is arrested in mitosis for a prolonged time may eventually undergo apoptosis, or programed cell death. 35 At present, most of the drugs used to treat cancer target microtubule dynamics in order to arrest cancerous cells in mitosis.
Microtubules–-A Potential Target for Cancer Therapy
Unregulated cell division may lead to an overgrowth of cells, as in cancer. The ability to carry out an infinite number of cell divisions is one of the hallmarks of cancer. 36 Blockage of any stage of mitosis may not allow the cells to complete mitosis, resulting in cell cycle arrest and ultimately, cell death. Microtubules represent the best and most successful target thus far identified in cancer treatment.37–39 Cancer cells are sensitive to microtubule poisons that arrest cells in mitosis because they undergo mitosis more frequently than normal cells. At high concentrations, anticancer drugs that target microtubules may act in one of the two ways. Each approach has different effects, including affecting the polymerized microtubule mass, destabilization of microtubules (decreases microtubule mass), and stabilization of microtubules (increases microtubule mass), dependent on the site of binding on the microtubule lattice. 40 The effects of each compound on microtubules are indicated in both Tables 1 and 2.
Currently, there are two main classes of microtubule-binding anticancer drugs. These are the microtubule destabilizers, such as the Vinca alkaloids,41–44 and microtubule stabilizers that prevent microtubule disassembly without affecting their polymerization, such as the taxanes. 6 However, studies have shown that various microtubule-targeting drugs, irrespective of their effects on polymerized microtubule mass at high concentrations, all suppress microtubule dynamics at lower concentrations, ie, prevent the growth or shrinkage of microtubules without changing the microtubule polymer mass6,8,33,34,42–45 (Fig. 2). In this way, changes in microtubule dynamics can be used as an indicator of the efficacy of the anticancer activities of a naturally derived compound.
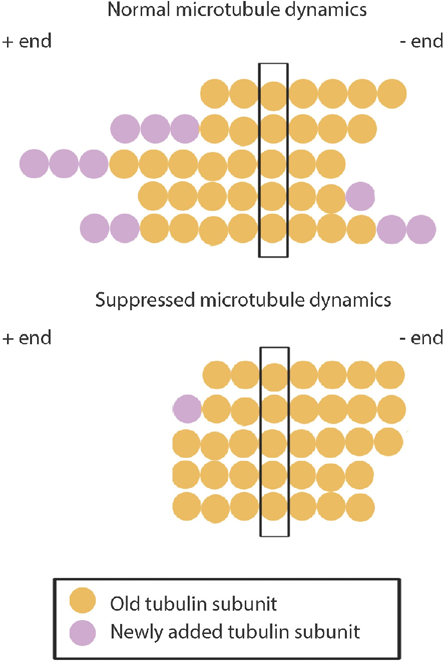
Microtubule dynamic instability. The figure illustrates the growth and shrinkage of a single microtubule, with each row representative of a single time point. Microtubules are composed of stable αβ-tubulin heterodimers that are arranged in a head-to-tail fashion, forming a polar structure. Each heterodimer is illustrated as a single circle. Microtubules therefore consist of two distinct ends: the plus (+) end and the minus (–) end. In vivo, the–-ends are anchored at the microtubule-organizing centers. The + ends are more dynamic than the–-ends, with the microtubule end constantly switching between growth and shrinkage in what is termed dynamic instability. Microtubules are normally very dynamic (top), with tubulin subunits randomly added or lost from both ends. In vivo, microtubule elongation usually occurs in the plus end. When microtubule dynamics are suppressed (for example, through the action of tubulin-binding agents) (bottom), tubulin subunits are rarely added or lost from the microtubule ends.
Conversely, tumors can acquire resistance to microtubule-targeting drugs. Although a discussion on the resistance mechanisms to these drugs is beyond the scope of this review, the possible methods of resistance include multidrug resistance pumps, altered drug binding, altered microtubule assembly, altered tubulin synthesis, and alterations in microtubule-interacting proteins (refer to Fojo and Menefee 46 for a more extensive review).
As with all drugs, the toxic side effects of microtubule-targeting agents must be taken into account. Owing to the physiological functions of microtubules, treatment with microtubule-targeting agents often exhibits myelo-suppression and peripheral neuropathy. The specificity of each compound must therefore be tested. The cancers identified to be susceptible to each drug are illustrated in Tables 1 and 2.
Conclusion
Mitosis is an important stage of the cell cycle, which is deregulated in cancer, leading to uncontrolled cancer growth. An important facilitator of mitosis is the microtubule cytoskeleton. Hence, many anticancer drugs target the microtubule skeleton in order to arrest cancer cells in mitosis, which eventually leads to cell death. Most of these microtubule-targeting drugs act by suppressing microtubule dynamics, which is particularly important for the microtubule function in mitosis. Interestingly, many of the microtubule-binding anticancer drugs are derived from natural sources, including Taxol and the vinca alkaloids, two very successful classes of anticancer drugs. Therefore, there is great potential for the isolation of compounds with similar microtubule-targeting activities from medicinal plants. Future aims for the development of novel microtubule-binding agents are the development of compounds specific to cancer cells, thereby reducing potential toxic side effects, as well as the development of compounds that are able to overcome current drug-resistant cancers.
Footnotes
Author Contributions
Prepared the first draft of the manuscript: INZ. Contributed to the writing of the manuscript: SRD, RR, and AI. Jointly developed the structure and arguments for the paper: INZ, SRD, RR, and AI. Made critical revisions and approved the final version: AI. All the authors reviewed and approved the final manuscript.
