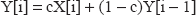Abstract
Objective
To compare the accuracy and reliability of uterine contraction identification from maternal abdominal electrohysterogram and tocodynamometer with an intrauterine pressure transducer.
Methods
Seventy-four term parturients had uterine contractions monitored simultaneously with electrohysterography, tocodynamometry, and intrauterine pressure measurement.
Results
Electrohysterography was more reliable than tocodynamometry when compared to the intrauterine method (97.1 versus 60.9 positive percent agreement; P < 0.001). The root mean square error was lower for electrohysterography than tocodynamometry in the first stage (0.88 versus 1.22 contractions/10 minutes; P < 0.001), and equivalent to tocodynamometry in the second. The positive predictive values for tocodynamometry and electrohysterography (84.1% versus 78.7%) were not significantly different, nor were the false positive rates (21.3% versus 15.9%; P = 0.052). The sensitivity of electrohysterography was superior to that of tocodynamometry (86.0 versus 73.6%; P < 0.001).
Conclusion
The electrohysterographic technique was more reliable and similar in accuracy to tocodynamometry in detecting intrapartum uterine contractions.
Introduction
Electronic assessment of uterine contractions (UCs) in conjunction with fetal heart rate (FHR) monitoring is in widespread use. Information about the timing of contractions is necessary to classify and interpret FHR patterns correctly, and contractility data are relied upon for optimal management of labor.1–3 Commercially available methods of uterine contraction monitoring include external tocodynamometry (TOCO), internal uterine pressure determination (IUPD) and the recently introduced method of electromyography.
The tocodynamometer detects contractions by means of a strain gauge held against the mother's abdomen with a belt. It detects changes in the abdominal contour produced by a uterine contraction, and converts that mechanical signal to an electrical output, which is processed to produce a contraction waveform on the monitor. The IUPD approach employs a pressure transducer inserted transcervically into the uterine cavity. A cable connects the transducer to a bedside monitor, which processes the electrical signal from the transducer and provides a waveform output that directly reflects the intrauterine pressure change created by a contraction.
The information provided by a properly inserted and positioned IUPD is generally more complete and more accurate than data from TOCO. In addition to information about the onset, acme, and completion of a contraction, intensity can also be measured. IUPD is also less likely than TOCO to be affected by patient movement, and works as well in obese as in lean women. However, IUPD has disadvantages. It requires the chorioamniotic membranes to be ruptured, and the cervix to be sufficiently dilated to permit introduction of the intrauterine pressure-sensing device. These limitations restrict its use in early labor, or whenever rupture of the membranes is undesirable. Moreover, the presence of the transducer cable in the uterine cavity poses the risk of infection or, rarely, uterine or placental injury.4–7 Although IUPD gives more accurate information about the intensity of contractions than does the TOCO technique, the clinical value of such intrauterine pressure determinations in the management of labor has been questioned.8,9
The TOCO approach has the advantage of being noninvasive, and can be used in early labor. It is, however, more susceptible to interference from maternal movement or shifting of the abdominal transducer. It can be especially unreliable in obese parturients, when the abdominal fat pad separates the tocodynamometer from the uterus, and in the second stage, when there is often considerable maternal movement. Also, many women find the encircling belt that holds the tocodynamometer or Doppler ultrasound transducer for heart rate monitoring to be uncomfortable. 10
Uterine contractile activity can also be monitored by assessment of the electrical impulses generated by myometrial activity. The electrohysterogram (EHG) can be detected by electrodes placed directly on the uterus,11–13 as well as by surface electrodes on the maternal abdominal wall.13–23 Contractions detected in this manner correspond closely with those determined by traditional clinical methods.18,19,22,23 This noninvasive method of UC monitoring has the potential to provide the ease of use and applicability of TOCO, and to be less susceptible to obesity- or movement-induced perturbation. 23
In prior studies, it has been demonstrated that EHG-derived contraction tracings are more likely to be adequate and are more easily assessed than TOCO contraction patterns. 21 Also, the EHG technique was found to be about 95% sensitive in identifying contractions when compared to an IUPD. 22 We extended research in this area by studying the three available techniques for contraction monitoring simultaneously. This allowed us to estimate the accuracy and reliability of both external methods in relation to the IUPD. We used a fetal heart rate and UC monitoring device (Model AN24, Monica Healthcare Ltd., Nottingham, UK) that employs five standard ECG electrodes applied to the maternal abdominal surface for EHG monitoring. We hypothesized that, in comparison to the IUPD technique, EHG-derived UC displays are at least as accurate and reliable as those obtained from tocodynamometry in identifying the presence and timing of a uterine contraction. The assessment of the fetal heart rate data from the same surface electrodes is described in another publication. 24
Materials and Methods
This prospective three-center study compared the accuracy and reliability of two modes of intrapartum uterine contraction monitoring (EHG and TOCO) with the contraction data obtained from an intrauterine pressure transducer. The latter was considered the “gold standard” device, against which the performance of the other techniques was judged. The study was designed and carried out as part of the requirements for 510k approval of the AN24 monitor by the United States Food and Drug Administration.
The open-method equivalence study took place in three teaching hospitals: Queens Hospital Center and Columbia University Medical Center (New York, NY), and Temple University Hospital (Philadelphia, PA) after approval by their institutional review boards. The study design conformed to the Guidelines of the World Medical Association Declaration of Helsinki. Each hospital used the Model 50XM FHR monitoring system (Philips Healthcare, Andover, MA) for standard internal and external monitoring. The IUPD systems were IPC5000 (Clinical Innovations, Salt Lake City, UT) at Queens Hospital Center and Columbia University Medical Center, and Accutrace (Coviden, Mansfield, MA) at Temple University Hospital.
The study design required the simultaneous use of three methods of uterine contraction detection in each subject. Parturients with a term (≥37 weeks) singleton pregnancy in cephalic presentation who were in the early latent phase of labor or awaiting labor induction were considered potential subjects. Excluded from participation were cases with multiple gestation, known major fetal structural or chromosome abnormality, or malpresentation. In addition, patients with any medical problem that, in the investigator's opinion, would make the patient unacceptable or incapable of taking part in the study were also excluded. A sample size of 70 would have a power of more than 0.9 to identify equivalence in success rate, reliability and accuracy within 10% of the IUPD device, which served as the standard. 25
After recruitment, subjects were monitored with the external Doppler ultrasound technique for fetal heart rate detection and the TOCO system for UC monitoring. The contraction tracing from the TOCO was available to the obstetric team for decision-making. The tocodynamometer position was adjusted as deemed necessary by the obstetric nurse. The maternal EHG electrodes were applied as soon as it was determined that the ultrasound and TOCO external devices were working appropriately. The obstetric team could not access the data obtained by the EHG monitor. These data were transmitted wirelessly to a personal computer at the bedside and stored for later analysis. During labor, a direct scalp electrode and IUPD were substituted for external monitoring in some patients. This change was made at the discretion of the supervising obstetrician when the FHR pattern was abnormal. Patients in whom all three contraction-monitoring systems (EHG, TOCO, IUPD) were used simultaneously became the subjects included in the analysis. Once the IUPD was inserted, each patient had uterine contraction monitoring used continuously throughout the remainder of her labor.
When the internal scalp electrode and IUPD device were attached, their output became the sole data visible to the clinicians. Although we continued to obtain and store the TOCO and EHG contraction data, neither was available for interpretation by the obstetric care team. A member of the research team was, however, tasked with checking the TOCO monitor at least every 20–30 minutes to verify that it was functioning properly.
In total, 138 subjects gave informed consent and were enrolled between December 2009 and June 2010. Of these, 35 were monitored solely by external techniques, four recordings were of insufficient length for meaningful analysis (<30 minutes), and in 25 data from one or more monitors were not stored by the personal computer because of a technical error. These exclusions left 74 laboring women with simultaneously recorded uterine activity data from the three techniques. No subject retracted her informed consent during the study. Data from the three hospitals were analyzed in aggregate.
Differences in the sample characteristics among the three participating institutions were sought using a one-way analysis of variance (ANOVA) for continuous variables and the Fisher exact test for categorical variables. When the ANOVA revealed a significant difference, the Student-Newman-Keuls test was used to identify differences between individual groups.
Data processing
The EHG device detects electrophysiological signals from the abdominal skin via five cutaneous ECG electrodes (Blue Sensor VLC-00S; Ambu, Ballerup, Denmark), arranged as illustrated in Figure 1. From these signals the device extracts the uterine activity data continuously. The data were updated every 0.25 seconds (ie, 4 Hz) to match the data rate of the TOCO and IUPD instruments. Data from all three sources were then synchronized to within 0.25 seconds by cross-correlating the simultaneously recorded fetal heart rate data from each source with the fetal heart rate data derived from the fetal scalp electrode, with the highest correlation showing correct synchronization. Using the fetal heart rate, rather than the UC signal, provided the most accurate synchronization.

Arrangement of the five cutaneous electrodes on the mother's abdomen during labor and delivery and the afECG device beside a patient.
To facilitate statistical analysis, the output from the IUPD device, to which the other monitors were compared, was considered to be valid if there was a signal present, and invalid when the signal was absent. The EHG and TOCO outputs were considered valid if contractility data were present at the same time as valid IUPD data were present.
We created an automated software program (written in Visual C++ using Microsoft Visual Studio 2003) that scanned each data set for uterine activity information, and identified periods of interpretable data as well as the location of contraction peaks. Interpretable data were defined as all data points with a stable, well-defined baseline at or above 5% of full scale. A stable, well-defined baseline was defined as follows: the 4 Hz raw data were low-pass filtered using an exponential weighted moving average filter implementing the function:
where X refers to the raw 4 Hz data, Y refers to the output of the filter and c was set to 0.3 to give an effective low pass filter of 0.2 Hz.
The raw data (X[i]) were then classified as stable and well defined if any data point returned to within 10% of full scale of the low pass filtered data (Y[i]) over a two-minute window. Any part of the uterine activity tracing that did not meet these criteria was defined as uninterpretable. During periods of interpretable tracings, a valid individual contraction was defined as a deflection of at least 10% of full scale above the baseline, lasting between 40 and 120 seconds. Any contraction more than 120 seconds in duration was classified as uninterpretable data. Examples of an interpretable contraction tracing from each modality are shown in Figure 2; uninterpretable tracings are demonstrated in Figure 3.

Example of an interpretable UC tracing obtained simultaneously from each modality.

Example of an uninterpretable tracing from each modality.
Statistical Analysis
We calculated two sets of results for each subject: one compared the EHG with the IUPD device; the other compared the TOCO to the IUPD technique. The ratio of these values was determined for each subject. The statistical processing software was Microsoft Excel 2007 running on Windows 7 (Microsoft Corporation, Redmond, WA).
Results from each subject were combined to give an overall mean and two-sided 95% confidence interval (CI) for each statistic, in addition to the EHG/TOCO accuracy and reliability ratios for each subject. Because the ratio data were nonparametric, CIs were determined from transformed data, and then back-transformed for display in the tables.
We assessed the reliability of the uterine activity output of the EHG and TOCO devices by the positive percent agreement (PPA). This is the percentage of time that the test device generated an interpretable UC tracing at the same time as the IUPD device did so. Reliability thus expresses the success rate of each device in creating a valid output in the presence of a simultaneous valid signal from the IUPD system.
Accuracy of the EHG and TOCO techniques was measured in several ways. First, individual contractions were categorized based on which devices had detected each of them, allowing determination of sensitivity, positive predictive value (PPV) and false positive rate of the device output. Second, a timed count of contractions within 10-minute windows was subjected to Bland-Altman analysis,26,27 and, finally, the timing of contraction peaks among the three devices was compared.
The contractions from the external test devices were compared with the IUPD gold standard. For each subject a count was made of contractions detected by both the test and IUPD device, and this was labeled “x.” Correctly detected contractions were defined as those with a peak that occurred within 30 seconds of the peak of the contraction waveform produced by the IUPD. Those contractions detected by the internal device but not the test device (ie, “missed” contractions) were labeled “y”; and contractions detected by only the test device (ie, false positives with respect to the IUPD standard) were labeled “z.” Example traces from each modality for a false positive and a missed contraction are shown in Figures 4 and 5, respectively.

Example of a false positive contraction that begins just after 3 minutes on the EHG tracing and is not seen on IUPD or TOCO obtained simultaneously.

Example of a false negative contraction on the EHG tracing between 9 and 10 minutes.
From x, y, and z for each subject we calculated the sensitivity (correctly detected contractions/contractions identified by the IUPD ie, x/x+y), positive predictive value (correctly detected contractions/all contractions identified by the test device ie, x/x+z), and false positive rate (false positive contractions/count of all contractions identified by the test device ie, z/x+z).
To assess accuracy further, we counted all contractions identified within each 10-minute monitoring window, this being the time frame typically used in clinical practice for specifying the contraction frequency. A Bland-Altman plot of these count values was then constructed. The difference between the external test and IUPD standard device was plotted on the y-axis, against the average of the test and standard on the x-axis. The regression line of this plot was obtained, and the root mean square (RMS) error between the count differences and this regression line was found. Thus, two error values (one for EHG versus IUPD and one for TOCO versus IUPD) were calculated for each recording.
Finally, when determining the timing accuracy of individual contractions, each contraction was marked as either typical (a contraction with a gradual rise and fall, a clear peak, and with no artifact obscuring the contraction shape), or atypical (a contraction that was inverted, double-peaked, misshapen or otherwise poorly defined). Uterine contraction timing accuracy was therefore assessed by determining the time differences between peaks of all typical contractions detected by both the test and IUPD reference technique, giving a mean and standard deviation for the time difference for each external device to compare with the IUPD data.
Results
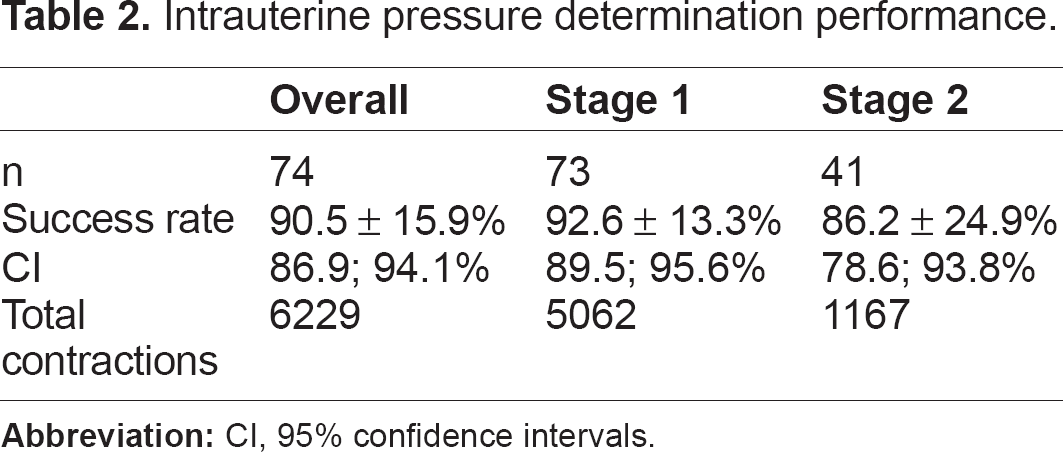
Gestational age and body mass index were not different in subjects from the three study hospitals (Table 1). Those at one hospital were about five years older than the average of subjects from the other two. There was a significant difference in the use of epidural anesthesia among sites, ranging from 61% to 96%. Uterine contraction data were collected from the three simultaneous techniques for a total of 19,282 minutes: 15,694 minutes in the first stage, and 3,588 minutes in the second stage of labor. A total of 6,229 IUPD contractions were analyzed. The success rate for the IUPD standard (ie, the percentage of time it produced a signal) was 90.5% overall (Table 2).
Characteristics of the study sample.

Significantly different from other two groups;
significantly different from Queens Hospital Center.
Intrauterine pressure determination performance.
The reliability (ie, the proportion of time for which the external device contraction form was interpretable when the IUPD contraction was also interpretable) of EHG was significantly greater overall than for TOCO (97.1% versus 60.9%; P < 0.001). This difference was similar in both the first and second stages of labor (Table 3).
Uterine contraction detection reliability (positive percent agreement).

Significantly different from TOCO, P < 0.0001.
The accuracy of EHG was not inferior to TOCO (Tables 4–6). The sensitivity (the proportion of contractions recognized by the external device when the reference IUPD method identified a contraction) for EHG was 86%, versus 73.6% for TOCO, a highly significant difference (P < 0.001). There was no significant difference in the overall positive predictive value (the proportion of all contractions detected by the external test device when the internal reference technique recorded a contraction) for EHG or TOCO (78.7% versus 84.1%, respectively; P = 0.052) or the false positive rate (21.3 versus 15.9%; P = 0.052).
Uterine contraction detection, sensitivity.
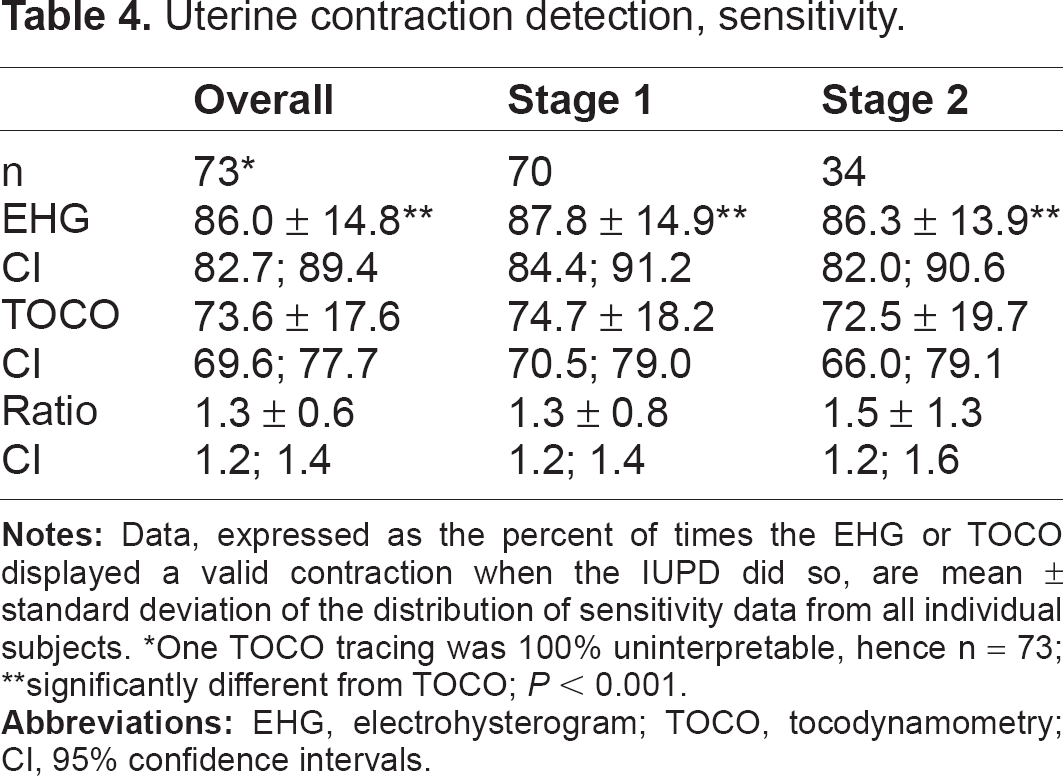
One TOCO tracing was 100% uninterpretable, hence n = 73;
significantly different from TOCO; P < 0.001.
Uterine contraction detection, positive predictive value.

One TOCO tracing was 100% uninterpretable, hence n = 73;
not significantly different from TOCO; P = 0.052.
Uterine contraction detection false positive rate.
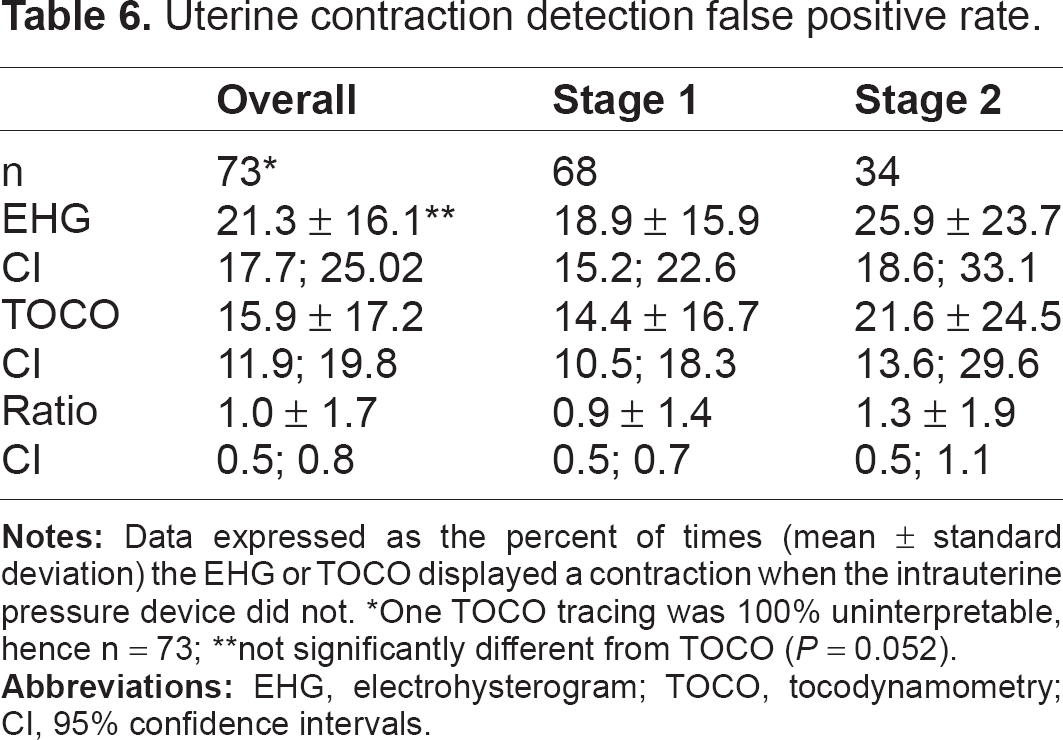
One TOCO tracing was 100% uninterpretable, hence n = 73;
not significantly different from TOCO (P = 0.052).
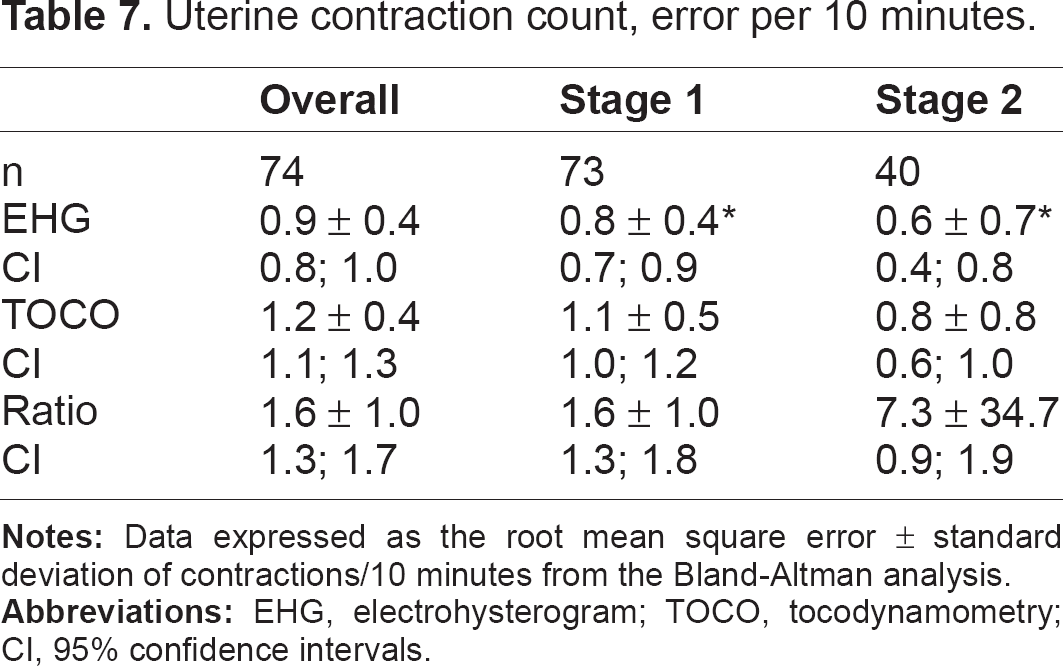
Table 7 shows the RMS error between the EHG and TOCO devices and the internal transducer standard for a count of contractions occurring in each ten-minute window of a recording. The overall EHG error was significantly lower, at 0.88 contractions/10 minutes, compared to 1.22/10 minutes for TOCO (P < 0.001). The difference was accounted for by less error in the first stage; during the second stage there was not a significant difference. The mean difference in the timing of contraction peaks between the external devices and the IUPD standard (Table 8) was 4.5 seconds for EHG and 1.5 seconds for TOCO (P = 0.001).
Uterine contraction count, error per 10 minutes.
Uterine contraction detection, timing accuracy.
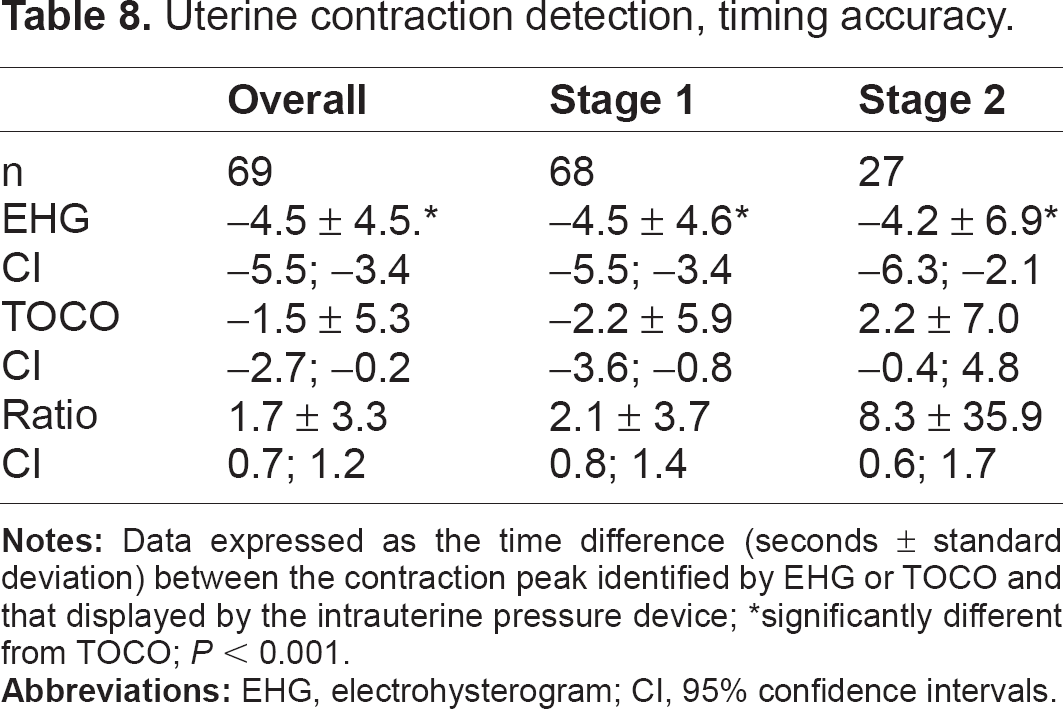
significantly different from TOCO; P < 0.001.
Discussion
In this study we assessed the reliability and accuracy of two modes of external uterine contraction detection in comparison to intrauterine contraction monitoring with a pressure transducer. The IUPD proved a useful comparison technique. It produced an interpretable signal in about 90% of the 0.25 second sampling intervals, and generated more than 6000 contractions over about 320 hours of monitoring.
The EHG device was very reliable in that it displayed interpretable contraction data about 97% of the time when the IUPD did, and the performance of the EHG did not deteriorate during the second stage. By contrast, the significantly lower TOCO reliability was only about 61% overall, and the mean of the individual EHG/TOCO reliability ratios in the second stage (2.9) suggests that EHG monitoring is especially reliable relative to TOCO during that time. It appears that EHG signals are less affected by the maternal movement and expulsive activity of the second stage than TOCO. The overall false negative rates were similar: 10.3% for EHG and 12.3% for TOCO.
The sensitivity of EHG to detect interpretable contractions traced by the IUPD was 86%, significantly better than the TOCO sensitivity of about 74%. Bland-Altman analysis comparing both external methods with the IUPD showed a significantly lower RMS error for EHG. The positive predictive values for the two test techniques were not different. These results show that EHG was not inferior to TOCO in accuracy; in fact, EHG performed better than TOCO in some respects. The sensitivity and PPV of EHG when compared to IUPD in our study (86.0% and 78.7%, respectively) were similar to those observed by Jacod et al 20 (94.5% and 88.3%).
Both external devices displayed false positive contractions, ie, contractions that appeared on the tracing when there was no corresponding contraction on the IUPD record. There was a nonsignificant trend toward more frequent false positive contractions with EHG than with TOCO (21.3% versus 15.9%); both techniques had a higher false positive rate in the second stage. Whether these contractions produced by the external devices are artifactual, are normal-strength contractions not detected by the IUPD, or are contractions of an amplitude too low to be detected by the IUPD is not known. We did not attempt to correlate the bedside clinical assessment of the contractions with their appearance on the tracing. The high frequency of these false positive contractions emphasizes the need to integrate uterine palpation and the parturient's subjective assessment of a contraction with electronic representation of contractions by any external technique in clinical decision-making.
There was a small time-offset in the identification of the contraction peak by both external devices compared to the IUPD. This averaged about 1.5 seconds for TOCO, and 4.5 seconds for EHG. This three-second difference, although statistically significant, is unlikely to have any clinical impact. It is equivalent to 1.5 mm on a standard 3.0 cm/minute tracing, and would not affect the interpretation of contraction frequency or of an accompanying fetal heart rate deceleration.
Both the EHG and TOCO are capable of identifying the onset, peak and end of uterine contractions. Neither, as configured in commercially available machines, provides a measure of the intensity of the contraction. For that reason, some obstetricians prefer the IUPD technique, especially when labor is being induced or augmented. Other investigators have demonstrated that EHG has a high sensitivity for detection of contractions, but its potential to provide information about contractile force is less certain.18,19,22 While there is legitimate disagreement about the importance of quantifying the amplitude of contractions in the management of labor,8,9 IUPD carries risks that external techniques do not, and its use in early labor or when chorioamnionitis is suspected is limited. Based on our results, and those of others,12,19–23 EHG monitoring is a reliable and reasonable alternative to TOCO when an external technique is desirable during labor. EHG monitoring has also been shown to have potential use in other aspects of care, including identification of term and preterm labor,12,15,17,19,20 and as the information contained in its recordings is better understood it will likely occupy an important niche in the continuum of obstetric care.
Footnotes
Author Contributions
WRC participated in the design of the study, oversaw study activity at Queens Hospital Center, prepared the first draft of the manuscript, and contributed to and approved the final version; SO recruited and oversaw the monitoring of study subjects at Queens Hospital Center, managed the study documentation there, and approved the final submitted version of the manuscript; SH recruited study subjects at Temple University, managed the study there, and approved the final manuscript; FM recruited study subjects at Columbia University Medical Center, managed the study there, and contributed to several versions of the manuscript; MS participated in organizing the study at Queens Hospital Center, recruited subjects there, contributed to manuscript drafts, and approved the final submitted version; RB oversaw the management of the study at Temple University and approved the final manuscript; BSS participated in the conception and design of the study, helped to edit several drafts, and approved the final manuscript; JMH was the senior statistical analyst and edited and approved the final manuscript; BRH-G participated in the conception and design of the project, assisted with the statistical analysis, edited drafts of the manuscript, and approved the final version.
Funding
This research was supported by a grant from Monica Healthcare, Ltd., Nottingham, UK.
Competing Interests
Wayne R. Cohen and Barry S. Schifrin have been paid consultants to Monica Healthcare, Ltd.; Barry S. Schifrin is a paid consultant to Philips Healthcare division of Koninklijke Philips Electronics N.V.; John M. Himsworth was previously employed as a biomedical engineer by Monica Healthcare, Ltd.; Barrie Hayes-Gill, a biomedical engineer, is an Executive Director and shareholder at Monica Healthcare, Ltd.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.