Abstract
The purpose of this review was to look at the evidence available for the use of atosiban as a tocolytic in cases of threatened preterm labour. A Royal College of Obstetricians and Gynaecologists Green Top Guideline concluded that there was no clear evidence to show a benefit to tocolysis in reducing perinatal and neonatal morbidity and mortality. Using a systematic literature search, we summarise the evidence available on the use of atosiban for the prevention of preterm birth and compare it with other commonly used tocolytic agents in terms of efficacy, patient preference and drug safety. We conclude that although atosiban appears to be the tocolytic of choice, a clear benefit of using tocolysis in all cases of threatened preterm labour remains to be justified and clinical management should be tailored according to individual needs.
Introduction
Preterm delivery is defined as birth before 37 completed weeks of pregnancy. It is associated with significant morbidity and mortality to the resulting babies, and emotional and financial costs to the parents. Outcome is related to the gestation at which the delivery occurs with babies being born at earlier gestations having the more severe complications. Mortality rates vary from around 20% at 26 weeks 1 to 2.3% at 34–36 + 6 weeks gestation, 2 with increasing rates of respiratory distress syndrome, feeding difficulties and hypothermia and long term developmental problems seen in the more preterm babies.
The rate in the UK is rising. In 2005, 6% of singleton pregnancies and 53% of multiple pregnancies were born prematurely in England and Wales. 3 Worldwide, prematurity affects 9.6% of all births 4 and accounts for approximately 1 million neonatal deaths annually, with low income countries reporting 5 times higher rates of preterm birth than high income countries. 5
In most cases the exact cause of preterm labour or threatened preterm labour is unknown. 5 The reasons are thought to be multifactorial, and vary with gestational age, ethnicity, geography and social factors. 6 There are situations where early delivery is indicated for the safety of the mother or baby such as pre-eclampsia, placental abruption, haemorrhage or intrauterine infection. In other cases, where the cause is less clear, much discussion and research has taken place over past years to determine the benefit of prolonging the pregnancy in spontaneous preterm labour using tocolytic medication.
The Royal College of Obstetricians and Gynaecologists produced a guideline in 20027 looking at the role of tocolytic drugs. They concluded that there was no clear evidence to show that tocolytic drugs improved perinatal or infant mortality or serious neonatal morbidity. The important effect was in reducing the numbers delivering within 7 days. This means tocolysis can be considered if gaining benefit from those days would allow administration of steroids of transfer to a unit with appropriate neonatal care, which are proven to improve outcome.
Different types of tocolytics have been investigated. These include beta-agonists, calcium channel blockers, magnesium sulphate, prostaglandin synthetase inhibitors. 7 Atosiban is an oxytocin receptor antagonist and in this review, we will look at the evidence surrounding atosiban and its role in preventing preterm labour.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Atosiban is a synthetic peptide which acts as a competitive antagonist of oxytocin at human uterine oxytocin receptors. Oxytocin causes uterine contractions through a direct effect on membrane bound receptors in the uterus by increasing the concentration of intracellular calcium in the myometrial cells. It can also lead to cervical ripening by stimulating the release of prostaglandins in the decidual and fetal membranes. Atosiban has been shown to result in inhibition of uterine contractility and a decrease in prostaglandin release. This occurs rapidly, with a reduction in contractions being seen within ten minutes. 8
Atosiban is given as an initial bolus dose of 6.75 mg followed by an infusion of 7.5 mg/ml given at a rate of 24 ml/hour for 3 hours then reduced to 8 ml/hour for up to 45 hours. Total atosiban dose should not exceed 330 mg. Steady state plasma levels are reached within an hour of starting the infusion. 8 Atosiban clearance, volume of distribution and half life are independent of the dose. Once the infusion is stopped, plasma concentrations decline rapidly with an initial (tδ) and terminal (tE) half life of 0.21 ± 0.01 and 1.7 ± 0.3 hours respectively. 8
Two metabolites have been identified, M1 and M3, in the plasma and urine of women. Ml, the main metabolite, is as potent as atosiban in inhibiting contractions in vitro. 8 The ratio of M1 to atosiban is 1.4 and 2.8 at the second hour and at the end of the infusion respectively. 8 Atosiban is detected only in small quantities in the urine with the concentration of M1 detected being 50x higher than atosiban itself. 8 M1 is also excreted in breast milk. 8
Atosiban does cross the placenta. A dose of 300 mcg/min administered at term to a healthy pregnant woman gives a fetal/maternal atosiban concentration ration of 0.12. 8
Clinical Studies
There have been clinical trials to evaluate the efficacy and safety of atosiban. These have looked at atosiban versus placebo, and other tocolytics, most commonly beta-mimetics and calcium channel blockers. Outcomes of interest vary among studies but the primary aim is generally delay to delivery with secondary outcomes relating to neonatal and maternal morbidity.
Atosiban versus placebo
A recent Cochrane review 9 studied the role of atosiban in preventing preterm labour. It identified two trials comparing atosiban with placebo, involving 651 women. No benefit was found in reducing the preterm delivery rate in either study.10,11
Romero et al
10
carried out a multicentre double blind placebo controlled trial involving 531 patients across 37 centres. The primary outcome was time from treatment to delivery or treatment failure, which constituted progression of labour requiring an alternative tocolytic. No significant difference was seen in primary outcome (25.6 vs. 21 days;
Goodwin et al,
11
in contrast, used atosiban or placebo for a maximum time of 2 hours. This is a randomised double-blind placebo-controlled trial involving 120 women between 20 and 36 weeks gestation. Their primary outcome was uterine activity, with a statistically significant decrease in contraction frequency demonstrated by external tocodynamometry with atosiban (55.3% vs. 26.7%;
Other studies not included in the Cochrane review have shown a benefit to atosiban in reducing the delivery rate.
12
Husslein et al included 226 women over 105 centres to immediate or standard treatment with atosiban. Two criteria was used for inclusion, and patients were assigned to the immediate treatment arm if they had one of regular uterine contractions lasting at least 30 seconds at a rate of 4 per 30 minutes or cervical dilatation of 1–3 cm in multiparous women or 0–3 cm in nulliparous women and 50% effacement; those in the standard treatment arm met both criteria. This was a prospective, open labelled randomised trial. There was a significant increase in women undelivered at 48 hours compared to those who received no treatment (77.6% vs. 56.6%;
Atosiban versus beta-mimetics
Three trials15–17 included in the Cochrane review compared atosiban with betamimetics and were regarded as high quality. In total, 1044 women were involved and the authors concluded that there were no significant differences between reduction in delivery at 48 hours, neonatal respiratory distress or admission to neonatal intensive care. However, the rate of maternal adverse drug reaction was higher in the beta-mimetic group resulting in more women ending treatment early. The lower discontinuation rate and comparable efficacy led researchers in Germany to conclude that atosiban is more cost effective than beta-mimetics. 18
The French/Australian Atosiban Investigators Group
16
carried out a multicentre, double-blind, double-placebo, randomised controlled trial comparing atosiban with intravenous salbutamol. This study was conducted across 331 treatment centres in France and 5 in Australia, and utilised computer generated randomisation to reduce risk of bias. Women between 23 and 33 weeks gestation were included, (n = 241), representing an optimal gestation range, but multiple pregnancies were over-represented above 28 weeks in the salbutamol group. No significant difference in numbers undelivered at 48 hours (93.3% vs. 95.0%;
A third multicentre, double-blind, randomised controlled trial compared atosiban to ritodrine.
17
This trial included 252 women at 23 to 33 weeks gestation and the results showed no significant difference in those remaining undelivered at 48 hours (84.9 vs. 86.8%;
Atosiban versus nifedipine
Atosiban has been compared to nifedipine in two randomised controlled trials involving 80 and 63 women respectively.19,20 A meta-analysis of indirect randomised controlled trials has also been done comparing the two drugs. 20
Kashanian et al 19 carried out a randomised trial involving 80 women at 26 to 34 weeks gestation. They did not find a significant difference in the effectiveness of tocolysis between atosiban and nifedipine at 48 hours (82.5% vs. 75%) or at 7 days (75% vs. 65%) but results suggested a trend towards the increased effectiveness of atosiban. As this study was looking at several outcomes, it is likely to be under-powered with just 80 subjects which included multiple pregnancies.
Al Omari et al
20
included 63 women between 24 and 35 weeks gestation in a randomised controlled trial using intravenous atosiban and oral nifedipine. No significant difference in rates of delivery at 48 hours (77.4% vs. 81.3%;
Similar results were obtained in the meta-analysis.
21
No significant difference between delivery rates at 48 hours were found between nifedipine and atosiban, however, there was a trend to reduction in delivery rate at 48 hrs in the nifedipine arm. This contradicts the findings by Kashanian et al. A significant decrease in neonatal respiratory distress was seen in the nifedipine arm of the meta-analysis but this was not confirmed by Al-Omari et al who did not find any difference in neonatal outcomes. Neonatal outcomes were not assessed by Kashanian et al. Maternal side effects were similar in both comparative studies and not commented on by Coomarasamy et al. Kashanian et al showed an significantly increased rate of maternal side effects in the nifedipine group (40% vs. 17%;
Concerns exist as to the reliability of an indirect comparison for making conclusions that will aid clinical practice. The direct trials mentioned did not use a blinding format and so taken together, the quality of the available evidence could be improved to draw comparisons between atosiban and nifedipine.
Other
Research has also looked into the timing of administration of atosiban. A Cochrane review suggested no benefit from long-term atosiban maintenance therapy following an episode of threatened preterm labour.
22
This review included only one trial
14
involving 513 women. This was a multicentre, double-blind, placebo-controlled trial. Patients were included after initially responding to atosiban therapy. Thereafter, they were assigned to atosiban maintenance or placebo, both administered subcutaneously, until the end of week 36. The primary outcome was time to first recurrence of labour and there was a significantly increased period of gestation in the atosiban group (36.2 days vs. 28.2 days;
Difficulties have been identified throughout the studies which should be considered when interpreting the results. Since atosiban works by acting on oxytocin receptors in the myometrium, its effectiveness may be gestation dependent, with more receptors available with increasing gestations perhaps explaining why atosiban may be more effective at later gestations. This has been shown in published studies 10 but the exact reason for the findings has not been determined.
There is also some difficulty in clinically diagnosing threatened preterm labour, so selecting appropriate patients for treatment is challenging. This was addressed by a randomised trial comparing immediate atosiban use with standard administration when either regular uterine contractions or cervical dilatation were confirmed. 12 Only 50% of women in the latter group received tocolytic medication, demonstrating the high probability that many women presenting will not go on to show signs of labour. Those given atosiban immediately had a lower delivery rate within 48 hrs compared to those who received it after demonstrating signs of labour. There was no significant difference in delivery rate after 48 hrs or gestational age at delivery.
The lack of good quality clinical studies into tocolysis and preterm labour is highlighted in the RCOG Consensus Statement on Preterm Birth. 23 They recommend future research should be carried out with randomised placebo controlled trials, focussing on tocolytic benefits in general and a direct comparison of atosiban and nifedipine. However, with the current advice available stating that tocolysis should be considered to gain time for proven therapies that will improve outcome (steroids and transfer for neonatal facilities) further placebo controlled trials may be controversial to carry out. We must also be careful to evaluate results comparing efficacy of different agents in view of the common practice of rescue tocolysis. This practice has the potential to skew results.
Safety
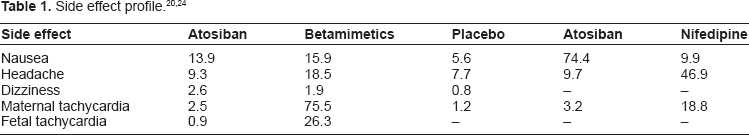
There is no report of serious adverse effects of atosiban use. It is associated with minor side effects in particular nausea, hyperglycaemia, headache, dizziness and palpitations. Since data do not strongly recommend one tocolytic over another on the basis of delivery delay or outcome, maternal tolerance is often considered as a significant consideration. Table 1 summarises the rate of adverse effects. Atosiban has a lower rate of adverse effects than the alternative tocolytic medication and is therefore better tolerated by patients. There is no evidence of a relationship between atosiban and fetal distress and it may be useful in the management of acute fetal hypoxia suggested by a cardiotocograph. 23
Patient Preference
Administration of atosiban is associated with minor side effects but these are less than other tocolytics. In a comparison of atosiban with beta-agonists, there was less cessation of treatment due to side effects in the atosiban group.9,18 The overall rate of atosiban cessation due to maternal side effects is thought to be 0%–1%. 26
For those in preterm or threatened preterm labour, the reason for tocolysis is for benefit to be gained from administration of steroids or transfer to a more suitable neonatal unit.7,27 When counselling women, the evidence is strong for the benefit of steroids. The transfer for experienced neonatal care is more upsetting to parents to deal with but the benefits to neonatal outcome are proven. In these situations, most parents would be expected to accept minor side effects and inconvenience for the overall improved neonatal morbidity.
In the study comparing immediate administration of atosiban with treatment after specific criteria of contraction duration/frequency or cervical changes were met, patient satisfaction was also studied. 12 This identified a significant increase in satisfaction in those that received atosiban early despite no difference in admission time, gestational age at delivery, mode of delivery or numbers receiving steroids. This is probably due to the high levels of anxiety that exist in these situations and patients feel more reassured if they perceive preventative action to be taken even if the outcome is the same.
Place in Therapy
Atosiban is licenced for use as a tocolytic. Its efficacy is similar to other tocolytic drugs but with a better side effect profile. Administration can provide time to allow transfer for suitable neonatal facilities or prescription of steroids. The clinical difficulty is when to give atosiban and to whom.
Current prescription advice 8 is to give atosiban in those with regular uterine contractions of at least 30 seconds duration at a rate of ≥4 per 30 minutes or cervical dilatation or 0–3 cm and effacement of ≥50%. The gestation should be between 24 and 33 completed weeks and with normal fetal heart. The patient should be ≥18 years.
Controversies exist as to the benefit of administering atosiban outwith these criteria. It is shown that late preterm births result in increased neonatal morbidity, although the rate of neonatal mortality is extremely low.2,28 The rates of respiratory distress, sepsis work ups and phototherapy are all increased leading to longer hospital stays and increased numbers requiring intensive care admission. Would tocolysis provide some benefit in these circumstances? The argument against however is that no significant benefit has been shown to steroid administration after 34 weeks and since that is the main indication for tocolysis, its use in late preterm labour is not indicated.
For threatened preterm labour before 24 weeks the evidence for atosiban use is limited. A small study of 40 women in threatened preterm labour between 18 and 24 weeks were given atosiban or placebo. 29 This showed a slight prolongation of pregnancy in the treatment group (17.1 weeks versus 15.6 weeks) and a cessation of contractions within 12 hours. The numbers in this study are small and routine use cannot be implied from this data. The numbers of women suitable for tocolysis in the previable group is very small as strict criteria need to be adhered to in order to prevent complications such as infection.
Little evidence exists for the use of atosiban in cases of preterm prelabour rupture of membranes (PPROM). Studies have looked at the use of other tocolytics in PPROM and found no benefit in prolongation of pregnancy,30–32 with one study showing a 2.5 fold increase in chorioamnionitis after long term tocolysis in women with PPROM. 33 No studies were found directly looking at atosiban use in patients with PPROM. In selected cases where benefit can be gained from prolonging the pregnancy without detriment to the fetus or mother, short term use of atosiban could be considered. Further clinical studies are required to look at this issue.
Two case reports exist showing use of atosiban for prophylactic prevention of preterm labour;34,35 both following transabdominal cervico-isthmus in women with poor obstetric histories. The first used atosiban from 24–32 weeks and the second 16–32 weeks. Both had successful outcomes. These are unusual cases and consideration of such treatment must be on an individual basis.
Conclusions
Atosiban appears to be the preferred tocolytic for use in view of its improved tolerability and equal efficacy to the alternative agents. Still the discussion continues, however, as to the overall use of tocolysis in cases of threatened preterm labour.
Clinical judgement of each individual case should be used in deciding whether to commence atosiban therapy. If harm is unlikely to occur with prolongation of pregnancy, the benefit from gaining time to allow steroid administration or transfer for neonatal facilities should be the outcome used to determine use.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.