Abstract
Objectives
The study was conducted to assess the effectiveness and side effects of vaginal misoprostol (Vagiprost® tablet) in termination of second and third trimester pregnancy complicated with intrauterine fetal death.
Design
A prospective observational cohort study.
Setting
Tanta University Hospital.
Patients
The study was carried out on 324 women with fetal demise in the second and third trimesters. Cases were collected during the period from January 2008 to December 2009.
Intervention
All patients were subjected to history taking, physical examination, Bishop Scoring. Application of 25 μg misoprostol in the posterior fornix of the vagina, this will be repeated every 4 hours over 24 hours. The adverse effects, progress, and outcomes were assessed.
Results
the success rate was 90% and 45% in women with third and second trimesters respectively. The mean induction-termination interval was 8.95 ± 2.63 and 15.3 ± 5.37 hours for women with third and second trimesters respectively. The induction termination interval correlated negatively with the duration of gestation. Approximately, 90% of second trimester and 55% of third trimester women required oxytocin augmentation. The mean value of total required dose of misoprostol was 166.3 ± 7.5 and 120 ± 28.79 μg for women with second and third trimesters respectively.
Conclusion
Vagiprost appears to be a safe, effective, practical, and inexpensive method for termination of third trimester pregnancy complicated with of intrauterine fetal death (IUFD), its effects increase with parity and duration of gestation.
Keywords
Introduction
According to the World Health Organization, the definition of fetal death (which has also been adopted by the United Nations and the National Center for Health Statistics) is a “death before the complete expulsion or extraction from its mother of a product of conception, irrespective of the duration of pregnancy; the death is indicated by the fact that after such separation, the fetus does not breathe or show any other evidence of life, such as beating of the heart, pulsation of the umbilical cord, or definite movement of voluntary muscles”. For the purpose of statistics, however, fetal deaths are classified according to gestational age, and only pregnancy losses that occur at ≥20 weeks' gestation are categorized as fetal deaths 1 Centers for Disease Control and Prevention, National Center for Health Statistics Web site. 1
The management of IUFD poses a dilemma. Although a significant number of these patients will spontaneously go into labour within several weeks, many do not. Moreover, after the diagnosis, the social pressures and emotional aspects of delivery are usually considerable, and the medical consequences of postponing delivery can be significant. Unfortunately, the drug most commonly used for induction of labour, oxytocin is frequently ineffective in stimulating the uterus, especially the preterm one. Within the past two decades, prostaglandins (PGs) have provided an alternative method for induction of labour in women with IUFD. 2
Misoprostol (15-deoxy-16-hydroxy-16–methyl PGE1) has been widely used for cervical ripening and labour induction in various pregnancy conditions, at different gestational ages and using different routes of administration and dosing regimens. 3 Although misoprostol is effective and inexpensive, concern has been raised regarding the widespread use of this agent as a primary or adjuvant agent for labour induction. 4 In spite of these concerns, a large body of evidence exists that shows that the use of misoprostol for labour induction is highly efficacious and safe. 5
Misoprostol is absorbed rapidly when administered orally, vaginally, rectally or intracervically. The vaginal route is advantageous because peak levels are reached slowly and sustained for long and this is associated with fewer side effects.6,7 The vaginal route is more effective than the oral route.8–11 The greater bioavailability of vaginal misoprostol probably explains the clinical results. Zieman et al 12 compared the absorption kinetics of misoprostol with oral versus vaginal administration in pregnant women. It was shown that the systemic bioavailability of vaginally administered misoprostol is three-times higher than that of the oral route when determined by area-under-the-curve. 13 The plasma level was sustained for up to 4 hours after vaginal administration.
The aim of this work was: To evaluate the effect of repeated vaginal administration of small doses (25 μg) of misoprostol in termination of pregnancy in cases of second and third trimester pregnancies complicated with IUFD.
Patients
This study included 324 women recruited from Tanta University Hospital, Obstetrics and Gynaecology department. Cases were collected during the period from January 2008 to December 2009. They were divided into 2 groups:
Group (1): included 160 cases of 2nd trimester pregnancy ≥20 week gestation, as documented by ultrasound examination and complicated with IUFD.
Group (2): included 164 cases of 3rd trimester pregnancy, as documented by ultrasound examination, complicated with IUFD.
Inclusion Criteria
IUFD with gestational age ≤20 weeks, absent spontaneous labour pain and Bishop cervical score <5.
Exclusion Criteria
Contraindications of misoprostol induction are allergy to prostaglandins, and contraindications to vaginal delivery.
Methods
All cases were subjected to: -
History taking, general, clinical examination, ultrasound examination.
Counseling the patient and obtaining written consent.
The induction regimen includes application of Vagiprost 25 μg tablet in the posterior fornix of the vagina every 4 hours (up to 6 doses) after determination of Bishop Score before. Vagiprost (Adwia, El Oubor, Egypt) is licensed in Egypt for Labor induction. The tablet was moistened with moistened with acetic acid before insertion. 13
If the cervix is already ripe (Bishop Score ≥ 6) and the first dose does not lead to effective contractions the subsequent dose could be doubled to 50 or 100 μg after 4 hours.
If no efficient regular uterine contractions occurred after 6 doses, augmentation of uterine contractions was done by oxytocin drip, 4 hours after last misoprostol dose.
Recording the total dose of misoprostol received and the need for surgical interference to remove the retained placenta.
The induction trial was considered successful when induction delivery interval was less than 24 hours.
Failure of delivery within 24 hours is considered “failed trial”, but it's not indication to stop the trial ie, the trial will be completed till termination.
Observation of patients for 24 hours after delivery.
Any complications during induction and 24 hours after delivery were reported.
Results
Out of 164 women third trimester pregnant women having IUFD, induction of delivery succeeded in 148 cases (90.24%). Of these 148 cases, 33(20.12%) required 100 μg of misoprostol with induction delivery interval of ≥ 12 hours, 57 cases (34.76%) needed 125 μg of misoprostol with induction delivery interval of ≥16 hours and 58 cases (35.36%) needed150 μg of misoprostol with induction delivery interval ≥20 hours.
The induction success rate in second trimester pregnant women having IUFD was approximately 45%. All succeeded inductions needed a total dose of 150 ug misoprostol.
Failure rate for second trimester pregnant women having IUFD was 55%, of these cases 15% needed 175 μg of misoprostol with induction delivery interval of ≥24 hours, 10% needed 200 μg of misoprostol with induction delivery interval of ≥28 hours, 5% needed 225 μg of misoprostol with induction delivery interval of ≥32 hours, 15% needed 250 μg. of misoprostol with induction delivery interval ≥36 hours and 10% needed 275 μg. of misoprostol with induction delivery interval ≥40 hours.
Failure rate for third trimester pregnant women having IUFD was 9.75%. All failed induction cases needed 175 μg of misoprostol with induction delivery interval ≥24 hours.
The most serious complications associated with intra-vaginal misoprostol use for IUFD was premature separation of placenta and postpartum hemorrhage. Surgical interference was resorted to for removal of retained placenta. The rate of Surgical interference was 30% (48 cases) among cases of 2nd trimester IUFD (16 cases needed manual separation and 32 cases needed curettage under general anesthesia). On the other hand, the rate of surgical interference was 5% among cases of 3rd trimester IUFD (8 cases) required manual separation.
As regards the incident of side effects, they occurred in 30% 2nd trimester pregnant women having IUFD. The most important side effects were fever in 16 cases (10%), fever and diarrhea in 8 cases (5%), nausea and vomiting in 16 cases (10%) and nausea, vomiting and diarrhea in 8 cases (5%). All side effects happened once induction delivery interval ≥34 hours. There were no complications in 3rd trimester IUFD.
Other results are depicted in the following 4 tables and figures.
Figures (1–4) disclose a significant negative correlation between gestational age and induction contraction interval and between gestational age and induction delivery interval during second and third trimesters respectively.
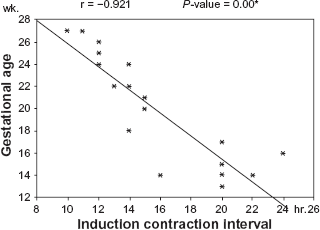
Illustrates the correlation between gestational age and Induction contraction interval in cases of second trimester IUFD.
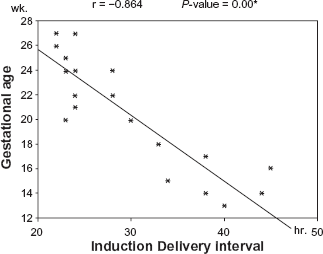
Shows correlation between gestational age and induction delivery interval in cases of second trimester IUFD.
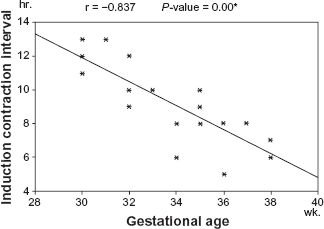
Exhibits the correlation between gestational age and induction contraction interval in cases of 3rd trimester IUFD.
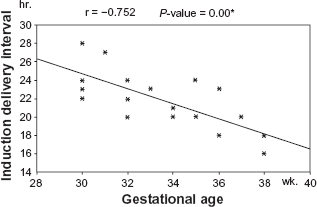
Reveals the correlation between gestational age and induction delivery interval in cases of 3rd trimester IUFD.
Discussion
Studies demonstrated that the optimal intravaginal dose of misoprostol is 25 μg taken every 4 to 6 hours. Higher doses or shorter dosing intervals are associated with a higher incidence of side effects, especially hyperstimulation syndrome.14,15
The current investigation was conducted to assess the effectiveness of vaginal misoprostol (Vagiprost® tablet) in termination of second and third trimester pregnancy complicated with intrauterine fetal death. The study confirmed the presence of inverse correlation between parity and induction contraction interval. In addition, inverse relatioship was instituted between parity and induction delivery interval. Furthermore, another significant negative correlation was established between parity and total required dose of misoprostol. These results agree with those of Chittacharoen et al; Caliskan et al and Yilmaz et al.3,13,16 Meanwhile, that result contradicts that of Auxiliadora de Aquino and Cecatti. 17
The mean values of parity, induction contraction interval, induction delivery interval and total needed doses of misoprostol (in micrograms) between cases in the 2nd and cases in the 3rd trimesters IUFD.

The present study exposed the presence of significant negative correlation between gestational age and induction contraction interval; between gestational age and induction delivery interval and between gestational age and total needed dose of misoprostol. These findings mode with those of Nakintu. 18
In this study, the induction contraction interval in primipara and second para was significantly lower than that in nullipara. Similarly, the mean of induction contraction interval multiparas was significantly lower than that in nullipara. However, there was no significant difference between the induction contraction interval in primipara, second para and that in third and fourth para. We found also, that the mean value of induction contraction interval in cases of 2nd trimester IUFD was significantly higher than that in cases of 3rd trimester IUFD. This agrees with the reports of Nakintu; 18 and Bugalho and his colleagues. 19
The induction contraction interval in relation to the parity in women with 2nd trimester IUFD.

The induction contraction interval in relation to the parity in cases of 3rd trimester IUFD.

The contemporary work shows that the induction delivery interval period in cases of 2nd and 3rd trimesters IUFD in nullipara was significantly higher than the corresponding values in women with previous history of childbirth. Approximately, 80% of patients required total misoprostol doses less than 200 μg to be delivered, and the maximum dose of 275 μg was needed by only in 10% of cases. These results differ from those of other authors.20,21 The total required dose of misoprostol in cases of 2nd and 3rd trimesters IUFD in nullipara were significantly higher than that required in parous women. Increased parity has no significant effect on the mean value of the required dose.
The rate of induction success in 2nd versus 3rd trimesters pregnancy with IUFD.

The success rate was 45% in cases of 2nd trimester IUFD compared with 90% in cases of the third trimester IUFD. This result agrees with that of many other authors.22,23 Bugalho et al found that the success rates range from 67 to 100%. 24
The need for oxytocin augmentation was significantly higher in the 2nd trimester (90% of cases) than that in 3rd trimester cases with IUFD (55% of cases). Nyende 20 found that 20% of his cases required oxytocin augmentation to complete the induction of labour.
The necessity for surgical interference because of retained placenta was 30% in cases 2nd trimester IUFD compared with 5% in 3rd trimester IUFD. Likewise, the occurrence of side effects during induction was significantly higher in cases 2nd trimester IUFD.
As regards complications, we found them in 30% of cases of 2nd trimester pregnant women having IUFD but none in cases of third trimester. The most important side effects were fever in 10%, fever and diarrhea in 5%, nausea and vomiting in 10% and nausea, vomiting and diarrhea in 5%. All side effects happened once induction delivery interval ≥34 hours. Hofmeyr and Gülmezoglu speculated that the most serious complications associated with intra-vaginal misoprostol use for IUFD are premature separation of placenta, postpartum hemorrhage, and rare events such as uterine rupture and amniotic fluid embolism. Gastrointestinal side effects: up to 35% of women will experience nausea, vomiting, or diarrhea. Pyrexia and shivering: this may occur in up to 7% of women. 25
Last but not least, we believe that vaginal administration of Vagiprost is a very effective drug for termination of pregnancy in cases of IUFD, its effects increase in direct proportion with parity and duration of pregnancy. Its use is an alternative to other treatments to terminate pregnancy in the second trimester for women with fetal death.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
