Abstract
Background and purpose
Mitoxantrone (MX) (Novantrone®) is approved in Canada for certain refractory cancers and acute non-lymphocytic leukemias. It has FDA approval as an immunomodulatory agent for use in secondary progressive multiple sclerosis (SPMS). The general aim of this study is to evaluate the efficacy, safety, and tolerability of MX in SPMS.
Experimental approach
A single-centre, open-label, non-randomized study was conducted in patients with a ≥6 month history of SPMS. The primary parameters used to assess efficacy and safety were EDSS scores and the multiple-gated acquisition scan (MUGA) scores, respectively.
Key results
The MX-treatment group experienced a high dropout rate due to significantly reduced ventricular ejection fraction. EDSS scores from baseline to follow-up revealed no statistical difference between active control and MX-treatment groups. Conclusion: MX treatment may not slow disease progression in certain forms of SPMS and may be associated with a high risk of cardiotoxicity.
Keywords
Introduction
Multiple sclerosis (MS) is an autoimmune disease characterized by the progressive destruction of the myelin sheath surrounding axons in the central nervous system (CNS).1,2 MS presents with a diverse array of symptoms, including: vision difficulties, weakness, neuropathic pain, bladder/bowel dysfunction, cognition changes, fatigue, sexual dysfunction, and spasticity.3–6 While the etiology of MS has not been fully clarified, several factors appear to be involved in the pathophysiology of MS: geography, genetics, gender, and environmental triggers (viruses, trauma, and heavy metals).5,7–14 MS is predominantly a disease of young adults with onset most common between the ages of 20 and 45 years.15,16
There are four predominant clinical courses of MS: relapsing remitting MS (RRMS), primary progressive MS (PPMS), secondary progressive MS (SPMS), and progressive relapsing MS (PRMS). 5 Approximately 85% of patients are diagnosed with RRMS, which is characterized by acute disease-symptom flare-ups (relapses) separated by stable symptom-free periods (remissions).5,17,18 Over time, the majority (75%) of RRMS patients advance to SPMS, where the disease symptoms get progressively worse. 5 In addition, relapses and remissions become difficult to separate.5,19 Approximately 15% of patients have an initial diagnosis of PPMS where the patient's disability continually worsens without relapses in symptoms. 5 PRMS is typified as being progressive from the start with the presence of gradually-escalating relapses separated by remissions. 5
While effective medications are available for treating RRMS, very few effective treatments are available for SPMS. Mitoxantrone (MX) hydrochloride (Novantrone®: Wyeth-Ayerst Canada Inc., Saint-Laurent, QC) is a cytotoxic anthracenedione approved in Canada for the treatment of some refractory adult cancers, including prostate cancer and acute non-lymphocytic leukemias.20,21 MX was originally used in cancer therapy due to its potential to produce less cardiotoxicity than doxorubicin, another genotoxic agent. 22 In addition to its use in oncology, the FDA approved MX for use in patients with SPMS, PRMS, and refractory cases of RRMS in October 2000. Its proposed mechanism of action in MS is thought to involve its ability to act as a potent immunomodulator by suppressing macrophage, B-cell, and T-cell activity. 20 Recent clinical trials have shown that MX can reduce both the frequency of MS attacks and the rate of disease progression.23–25
Despite these apparent benefits, the potential for serious cardiotoxicity is an important side effect that must be considered.26–29 Studies have suggested that MX treatment should be used only when expanded disability status scale (EDSS) scores have declined by two points within a year and there is evidence of active inflammatory disease. 30 MX has been shown to cause cardiotoxic effects in cancer patients, however these risks are said to be only ‘slight’ and generally well-tolerated in patients with MS who receive MX as part of their therapy. 31 While the MX associated risk for cardiotoxicity increases with cumulative doses >100 mg/m2,32 the inherent risks of cardiotoxicity emerge at lifetime cumulative doses of 120 mg/m2. 21 Therefore, it has been recommended to employ MX for treatment in only those with very aggressive MS, in which disability has increased and accumulated significantly due to frequent, severe relapses, with many active inflammatory lesions on gadolinium-enhanced magnetic resonance imaging (MRI). 30 With studies finding patients on MX developing ‘temporary and considerable’ cardiotoxicity, or severe delayed (post-treatment) cardiotoxicity, clinicians are becoming increasingly aware of the safety concerns related to the use of MX in patients with MS.33,34 For example, Perini et al (2006) found equal stabilizing effects of MX and cyclophosphamide. 35 Their results indicate the use of cyclophosphamide may be more favorable in terms of cost and safety. 35 This is a notable interest as MS patients are already at a predisposition for cardiac complications.36–39 While some studies have suggested no adverse cardiac effects, others have shown an increased prevalence of cardiac complications (left and/or right ventricular dysfunction) in MS patients versus controls.39–44 Owing to structural similarity with other anthracyclines, mitoxantrone induced cardiotoxicity is a severe side effect. Studies have shown that anthracyclines induce myocyte death, in a dose dependent and irreversible manner, that leads to the reduction of left ventricular ejection fraction (LVEF) and congestive heart failure. 45 Any patient receiving MX therapy should be notified of not only the cardiotoxic potential of MX, but they should also be informed of other potential complications from their therapy. Other potential complications include therapy-related secondary acute leukemia,46,47 granulocytopenia, 47 menstrual disorders in female patients,47,48 urinary and respiratory tract infections,47,48 and the risk of infertility. 48
The primary efficacy endpoint of the current study was a change in EDSS scores. The principle aim of the study was to evaluate the use of MX in SPMS patients in Manitoba, Canada. Although MX has been approved for treating MS in the United States of America (USA), it has not been approved for SPMS treatment in Canada. Therefore, the use of MX in Canada for SPMS is conducted off-label. The ability to evaluate the use of MX in Canadians with SPMS represents a pivotal step toward the provision of equal treatment opportunity.
Methods
Study design
A single centre, open-label, non-randomized study was conducted in 42 SPMS patients following approval from the Health Research Ethics Board at the Health Sciences Centre in Winnipeg, MB. Written informed consent was obtained for all SPMS patients interested in participating in the study. These patients were subsequently screened for eligibility according to the pre-established criteria outlined in Table 1. All patients were SPMS patients who had been diagnosed with MS ≥ 6 months prior to enrollment according to pre-defined McDonald's criteria, where 3 of 4 MRI criteria must be satisfied and diagnostic criteria are indicated for each of five possible clinical presentations. A total of 42 patients were entered into the study, pre-established as the number of participants required to detect a 50% difference in clinical outcomes, when comparing patients in the study groups. Type 1 and type 2 errors were both set at 0.10 (1 tailed testing) and 0.05 respectively, in order to minimize the risk of falsely concluding non-significant results. 49
Inclusion criteria for patients enrolled in the mitoxantrone study.

Based on the ethical considerations involved in assigning treatment of an off-label drug, the treating neurologist, in detailed consultation with the patient, determined the patient's willingness to receive or refuse MX treatment. The ethical considerations, taken together with the rare nature of the disease and stringent eligibility criteria, accounts for the small sample size from which the study was conducted. All eligible patients interested in participating in the study without receiving treatment were assigned to the active control group (n = 11), while those wanting to receive treatment were assigned to the active MX-treatment group (n = 31). As a result of the ethical constraints imposed on the study, the introduction of a placebo control group with study randomization was not possible. Randomization would have eliminated the choice of eligible patients wanting to receive drug (MX) treatment. Thus, patients assigned to the active control group received no treatment, while those in the active MX-treatment group received 12 mg/m2 infusion of MX every three months up to a maximum cumulative dosage of 120 mg/m2 (or 10 infusions over 27 months). Patients in the MX group underwent baseline assessments of the standardized tests: urinalysis; serum pregnancy test; blood work, including complete blood counts (CBC) and liver function tests (LFT); Multiple-gated acquisition (MUGA) scans to assess left-ventricular function; and EDSS scoring. MRI data was not available due to limitations of availability and cost. During each of the eleven scheduled visits, the MX group underwent standardized tests immediately prior to each successive MX infusion, as well as at ten days post-infusion. Drug-related adverse effects such as nausea, hair loss and injection site reactions were also documented at each successive clinic visit. In order to monitor for potential MX-induced cardiotoxicity, the left ventricular ejection fraction (LVEF) of each patient was assessed through the use of a MUGA scan every six months during the course of the first eight infusions and with every infusion thereafter. MUGA testing was also conducted on all patients on an annual basis for two years post-trial.
Specific monitoring parameters were established to safeguard against negative outcomes associated with MX treatment. Any patient receiving MX treatment who presented with a neutrophil count < 1000 cm3, an active urinary tract infection (UTI), an LVEF < 50% or a drop of greater than 10% in LVEF from baseline, were required to prematurely withdraw from the study. Worsening of MS measured by an increase in EDSS score was not an exclusion criteria since all patients enrolled in the study were patients with SPMS and thus, no alternate treatments were available.
All patients received standard blood work (CBC, LFTs), urinalysis, and EDSS measurements at baseline entry to study. The MX-treatment group also received a baseline MUGA scan prior to initiation of the treatment. In addition, the standardized tests (excluding MUGA) were also conducted at each physician-scheduled return to clinic visit.
Results
Comparing baseline patient demographics revealed no statistically significant differences between the groups. The MX-treatment group was comprised of 71% females compared to 55% females in the control group (χ 2 = 0.98, NS). The average patient age was similar in both study groups; 49.2 years in the treatment group compared to 50.4 years in the control group (t = 0.33, NS). Disease status between both study groups was comparable at the study onset with the average baseline EDSS score for the active control group totaling 6.5, compared to an average EDSS score of 6.4 for patients in the treatment group (t = 0.63, NS).
No patients within the control group terminated early, nor were any lost follow up. 20 of the 31 patients (64.5%) enrolled in the MX group prematurely terminated their study involvement due to various reasons, including: moderate to severe cardiac abnormalities, UTI, or quality of life issues, among others reasons, as outlined in Figure 1. Specifically, cardiac complications accounted for 32% percent of the treatment cancellation group (10 of 31 patients), with six of these ten individuals exhibiting cardiac abnormalities that were classified as severe (Fig. 2). Severe cardiac abnormalities were defined as any patient who experienced an ejection fraction decrease of 10% or more from baseline, or who had an ejection fraction that was less than 50%. For the six individuals terminating therapy due to severe cardiac abnormalities, two patients discontinued following their third MX treatment, while four discontinued following their fifth MX treatment. Moderate cardiac abnormalities, such as heart palpitations, pulmonary edema, and tachycardia accounted for study termination in four of the 31 (13%) patients enrolled in the MX group who prematurely terminated their study involvement (Fig. 2).
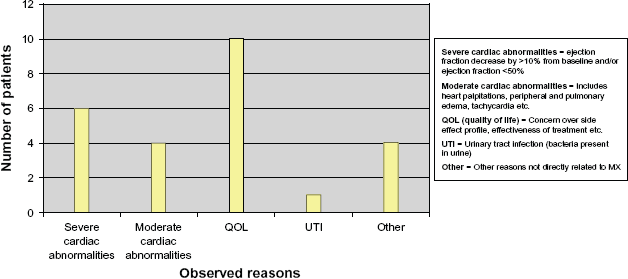
Cancellation group reason for treatment termination.
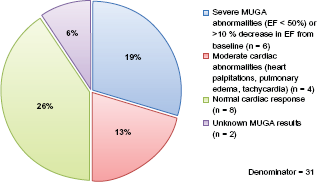
Summary of cardiac complications in cancellation group.
There was no statistical significance in EDSS scores between control and MX-treatment groups from baseline to follow-up (χ = 1.6, NS). In addition, there was no significant difference in disease progression between the MX-treatment group and the MX cancellation treatment group (patients who ceased MX treatment) (χ 2 = 0.9, NS). Interestingly, we observed that 6 of the 31 MX treated patients (19.4%) experienced an increase in EDSS score by ~0.7 (indicating disease progression) compared to no increase in EDSS score in the control group. Conversely, two patients (6.5%) in the MX group showed a marked improvement in EDSS score of 0.5, whereas no control patients showed a decrease in EDSS. In addition, there was no significant difference in EDSS scores observed between patients who had received more MX versus patients who had received less.
The time to follow-up for both active control and active group participants was not significantly different. Patients in the active MX-treatment group were assessed every three months following the initial MX infusion and throughout the duration of their MX therapy. Furthermore, cardiac surveillance was continued for all MX treatment patients on an annual basis for up two years post-trial. Control patients were followed up, and EDSS scores were obtained at each physician-scheduled return to clinic visit at Health Sciences Centre consistent with normal standards of care. At least nine months of follow-up data are available for five (45.5%) of the active control group patients and for twenty (71.4%) of the active MX-treatment group patients (χ 2 = 2.3, NS). Complete or partial follow-up data indicates that 15 of 31 (48%) patients in the MX treatment group demonstrated a decline in LVEF while receiving MX, followed by a post-treatment reversion to normal cardiac function (Table 2). Furthermore, it is of interest to note that of these fifteen patients, ten of them were patients in the MX treatment group who prematurely terminated their study involvement due to various reasons including cardiac complications (10 out of 20; 50%).
Reversion to normal cardiac function.

represents an increase in LVEF from previous measurement.
Discussion
In contrast to RRMS, few effective pharmacotherapies have been developed to reduce the continued disease progression associated with SPMS. 50 Some RRMS therapies, such as interferon beta, glatiramer acetate (Copaxone®), intravenous immunoglobulin and plasma exchange therapies have been trialed, producing marginal benefit in patients with RRMS with no noted benefits for SPMS.51–55 In Canada there are currently no therapeutic agents approved for the treatment of SPMS. However, MX has been used off-label in Canada for SPMS based largely on the results of previous clinical trials in the USA.56–59 Several clinical trials examining the efficacy and safety of MX showed promising results for the use of this agent in SPMS.35,60,61 Several studies have shown that patients treated with MX had an improvement in EDSS score, a decrease in annual relapse rate, as well as a significant decrease in lesions on MRI, among patients treated with of MX versus placebo.30,61–65
Previous studies of MX treatment in SPMS reported no cardiotoxicity,34,61 whereas our study demonstrated a strong association to the contrary. Despite our small sample size, severe cardiac abnormalities occurred in six of the 31 patients (20%) receiving MX. Based on the data obtained in our study, patients who discontinued MX treatment due to cardiac events returned to normal baseline cardiac parameters post discontinuation, which concurs with previously published findings. 34 MX induced cellular toxicity has been investigated in previous studies in vitro. A study by Wang et al showed that MX induced a prolongation of action potential duration (APD), in both multicellular preparations and single myocytes. 66 This APD prolongation may account for the proarrhythmic effects contributing to MX induced cardiotoxicity. The cardiotoxic potential of MX was assessed in vivo, and showed numerous morphological changes in the heart following MX treatment.22,67 Shipp et al showed both a dose- and time-dependent depletion of cellular ATP levels following MX treatment of cardiac myocytes. 67 The decline in ATP levels correlates with a decrease in synchronous beating and unequivocal morphological indications of toxicity. 67 These studies provide evidence for the mechanisms underlying MX induced cardiotoxicity.
There are a number of mitigating factors that could have led to the increase in cardiac abnormalities seen in this study compared to previous studies. One possible factor could be the higher mean baseline EDSS score of the treatment group in this study compared to the lower mean EDSS scores reported in the other studies. The EDSS score spread of patients in the MX treatment group in our study was 6.2–6.6. The majority of other studies that did not report MX induced cardiac toxicity had enrolled patients with a lower mean EDSS score, between 3–6.61,62,65 A higher EDSS score indicates that patients are at a more severe stage in their MS disease progression. Since MS has been shown to be associated with cardiac abnormalities, a more severe stage of the disease may be associated with more severe and established cardiac abnormalities. 39 Therefore, one hypothesis for the differences seen in our study is that patients enrolled in our study may have been at an elevated risk to develop cardiac toxicity due to the MX treatment. However, cardiac abnormalities in the control group were not noted in our study. As a result, our study suggests that the cardiac events reported are directly due to the MX versus any other confounding variables that could affect the outcome.
Another factor that could affect the apparent increase in cardiotoxicity is our use of MUGA scanning rather than the use of an ECHO. The best method for assessment of cardiac function during MX treatment in MS patients has not yet been assessed and there are no longitudinal studies available comparing MUGA and ECHO for efficacy in monitoring for cardiac toxicity. 68 Most of the other studies used ECHO to assess cardiac function in MX treated patients.58,62,65,69 One study conducted by Rashid et al retrospectively analyzed a subset of patients from the SOLVD study, 70 who either had a MUGA or an ECHO scan. 71 Their analysis provided evidence that ECHO analysis is more subjective, as it depends on the inherent skills of the technician, whereas interpretation of the MUGA is computerized. 71 In the current study, MUGA was the most feasible and realistic cardiac monitoring option available. Although ECHO is more commonly used due to the lower costs associated with its use, the MUGA monitoring was more feasible for our study due to shorter waiting times. As an alternative to MUGA and ECHO, other diagnostic techniques may be used to assess cardiac function. The measurement of troponin T, a cardiac biomarker, could further facilitate the measurement of cardiac function.72,73
One other published study has shown cardiac toxicity associated with MX treatment. 58 Montu et al showed cardiac abnormality occurrence was not as high as in our study, with two of the 14 (14.3%) SPMS patients in their study experiencing a decrease in LVEF to below 55%. The two patients were symptomatic and the observed decrease in LVEF was found to be reversible upon MX discontinuation, as we have shown in our study. Interestingly, the other twelve SPMS patients enrolled in the Montu study experienced a decrease in LVEF but MX was not discontinued since this decrease did not yield an LVEF less than 55% in any of these patients. 58 The patients were all monitored by ECHO and were on average younger (mean age = 41.6) than the patients in the current study. The EDSS scores of patients in the Montu study were not indicated. 58
There are a number of factors that may have contributed to the lack of MX efficacy observed in our study. One EDSS-related limitation in this study, as well as other studies, was that all enrolled participants were grouped together regardless of EDSS score (range of 3.5–8). It may be possible to demonstrate the benefit of MX treatment for certain patients depending on the severity of the disease. For example, it may be easier to demonstrate an improved EDSS score in a patient that enters the study at an EDSS of 3.5 versus a patient that is already wheelchair bound at an EDSS of 8.0. As a result, future studies may be well-served by stratifying patients into separate, narrow EDSS categories to delineate the potential clinical usefulness of MX in some patient clusters, while not in others. Also, SPMS patients enrolled in this study may have been in a stable stage of their disease and may no longer have been experiencing relapses since active, relapsing disease was not an inclusion criterion. This may be a deterrent to study design since most of the other studies that found MX to be effective ensured that the patients enrolled had active disease involving relapses and/or remissions versus continued progression. Efficacy may not have been observed in our study if MX efficacy is dependent on patients being in the earlier, less stable stage of SPMS where they are still experiencing relapses. Another variable that may have led to the lack of efficacy observed in the current study was the sole use of EDSS scoring. EDSS scoring can be subjective depending on the individual conducting the measurements and the individual's disability level. Concerns over inter-rater reliability have been eliminated in this study through involvement of only one neurologist rather than multiple neurologists. Furthermore, EDSS may not be able to properly evaluate all aspects of disease progression in response to MX treatment. Additionally, future research may overcome another potential limitation to this study, where it was not observed if EDSS improvements may have been transient during MX treatment, only to potentially revert to baseline scores following MX cessation.
Regardless of possible contributing factors, the statistics from our study present important safety issues which must be considered before initiating therapy. In terms of the primary efficacy endpoint (EDSS), MX did not significantly improve disease outcome when compared to the control group. Specifically, no statistically significant differences in EDSS scores from baseline to follow-up were noted between active control and active MX-treatment groups, questioning the overall benefit of treatment. Interestingly, more individuals in the MX-treatment group (6 out of 31 patients) demonstrated non-significant increases in EDSS scores than in the active control group (0 out of 11 patients). Therefore, the results presented in our study suggest that the potential for MX-induced cardiotoxicity and worsening of disability must be factored into the decision as to whether to initiate treatment or not. Although MX induced cardiac toxicity is not new, it has previously not been reported as being reversible. However, when used at much lower doses in MS than for oncology, our study demonstrates that the observed cardiac toxicity is reversible. Previous studies have shown a potential for MX induced cardiotoxicity reversal.60,74 Taken together, these results may suggest that the cardiac toxicity reported for MX use in MS patients may be over-reported.
The results presented from this Canadian study seem to contradict those presented in larger clinical trials conducted in the USA. Despite similarities in study design, the larger trials demonstrated that MX had a positive effect on disease progression and relapse rates, while this study showed no significant variation in disease progression between treatment and control groups. In addition, the results of this report identified a significant frequency of cardiotoxicity associated with the use of MX that was not evident in the US trials. These conflicting findings raise an interesting question as to whether there is an inherent variability in MS presentation between Canadian and US patients, and if so, could future treatment decisions be affected by the geographic region of patients?
Conclusions
The current study demonstrates the need to individualize treatment to the specific needs and aspects of each patient. This study identifies important factors which clinicians must consider when determining whether to initiate therapy with MX. Although results of this study demonstrated such exponential rates of cardiac abnormalities associated with MX therapy, recommendations cannot be made to change current MX prescribing practices due to the inherent variability of MUGA, small sample size, and reversion to normal cardiac function of several patients who experienced cardiac abnormalities. These results should however raise awareness of the potential occurrence of cardiac abnormalities, whether permanent or reversible, especially in patient populations similar to the patients enrolled in this study. Further research is required to identify whether patients with more severe progressive SPMS are at higher risk of cardiac complications with MX, and whether MX even provides any sort of benefit in this patient population.
Dealing with a chronic disease has its challenges but dealing with the long term consequences of a drug-induced adverse event which could have been prevented can severely impact the quality of life in individuals already engaged in a struggle with this disease induced dilemma. Therefore, a balance between disease management and quality of life must be considered for all potential candidates of MX therapy. If a decision to initiate MX therapy is made, judicious cardiac monitoring is strongly recommended throughout the treatment duration, especially in MS patients who are already predisposed to cardiac abnormalities. Based on the results of our study, the suggested cardiac toxicity may be a reversible dose-dependent effect.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgements
The authors would like to acknowledge the contribution of Wyeth-Ayerst Canada Inc., Saint-Laurent, QC for supplying drug to conduct this study.
Abbreviations
MX, Mitoxantrone; MS, multiple sclerosis; SPMS, secondary progressive multiple sclerosis; EDSS, expanded disability status scale; MUGA, multiple-gated acquisition scan; CNS, central nervous system; MHC, major histocompatibility complex; RRMS, relapsing remitting multiple sclerosis; PPMS, primary progressive multiple sclerosis; PRMS, progressive-relapsing multiple sclerosis; MRI, magnetic resonance imaging; ATP, adenosine triphosphate; CBC, complete blood count; LFT, liver function tests; LVEF, left ventricular ejection fraction; UTI, urinary tract infection; IFN-β, interferon beta; APD, action potential duration; ECHO, echocardiogram.
