Abstract
Purpose
First-phase ejection fraction has been established as an innovative indicator for detecting early left ventricular systolic changes. This study aimed to use first-phase ejection fraction to assess early changes in the left ventricular systolic function in patients with cirrhosis.
Methods
In this cross-sectional study, 122 patients with cirrhosis were stratified into the following groups based on Child–Turcotte–Pugh scores: group B1 (Child–Turcotte–Pugh class A, n = 35), group B2 (Child–Turcotte–Pugh class B, n = 43), and group B3 (Child–Turcotte–Pugh class C, n = 44). Thirty-nine healthy controls (group A) were enrolled for comparison. Basic information, clinical biochemical parameters, and ultrasound parameters were collected.
Results
Compared with groups A, B1, and B2, group B3 showed significantly decreased left ventricular ejection fraction (all p < 0.05). Groups B2 and B3 showed significantly decreased absolute values of global longitudinal strain compared with groups A and B1 (all p < 0.05). Compared with group B2, group B3 demonstrated more pronounced decrease in absolute values of global longitudinal strain (p < 0.05). First-phase ejection fraction was significantly increased in group B1 compared with that in group A (p < 0.05). In contrast, first-phase ejection fraction was significantly decreased in groups B2 and B3, with group B3 showing a further significant decrease compared with group B2 (p < 0.05). First-phase ejection fraction, ejection fraction, and global longitudinal strain demonstrated significant negative correlations with Child–Turcotte–Pugh class (r = −0.619, −0.429, and −0.608, respectively; p < 0.05). The prevalence of cirrhotic cardiomyopathy was significantly higher in group B3 than in group B1. The area under the receiver operating characteristic curve for first-phase ejection fraction in diagnosing cirrhotic cardiomyopathy was superior to that for ejection fraction and global longitudinal strain.
Conclusion
First-phase ejection fraction sensitively detects early alterations in left ventricular systolic function in cirrhotic patients and may facilitate early identification of cirrhotic cardiomyopathy.
Introduction
Cirrhosis is a chronic diffuse liver injury characterized by severe hepatic dysfunction and multiple late-stage complications. Among these, cirrhotic cardiomyopathy (CCM) represents a significant cardiac complication. 1 The chronic high-output circulatory state in patients with cirrhosis leads to the development of subclinical myocardial injury. A study 2 has shown that CCM is comorbid in approximately 20%–40% of patients with cirrhosis based on the Cirrhotic Cardiomyopathy Consortium criteria in 2020. However, CCM often presents insidiously and is frequently overlooked. Patients with CCM typically exhibit preserved left ventricular ejection fraction (LVEF) at rest, which may mask underlying systolic dysfunction and consequently delay the diagnosis of heart failure. Under conditions of physiological stress, disease progression, or increased cardiac load, these patients can manifest reduced ejection fraction (EF) and even overt heart failure. 3 Consequently, the early identification of alterations in left ventricular systolic function is crucial for monitoring the onset and progression of CCM in cirrhotic patients.
First-phase EF (EF1) is a novel parameter that measures the percentage of change in left ventricular volume from end-diastole to the peak aortic flow velocity. Initially described in 2017, 4 the advantages of EF1 include its noninvasiveness, procedural simplicity, and enhanced sensitivity. This technique has subsequently been applied to evaluate left ventricular systolic function in conditions such as hypertension, aortic stenosis, coronary artery disease, and chronic kidney disease,4–7 demonstrating promising clinical utility. However, the use of EF1 for assessing left ventricular systolic function in cirrhotic patients has not been explored in previous studies. This study aimed to employ EF1 to assess early changes in left ventricular systolic function in patients with cirrhosis.
Methods
Study population
This cross-sectional study was performed at the Second Affiliated Hospital of Dalian Medical University after receiving approval from the local ethics committee (approval no. KY2025239). All patient details have been deidentified. Written informed consent was obtained from all participants. This study was conducted and reported in accordance with the ethical principles outlined in the 1975 Declaration of Helsinki and its 2024 revision as well as the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 8 Patients diagnosed with cirrhosis in our hospital between July 2024 and June 2025 were consecutively included in this study. All enrolled patients met the diagnostic criteria for cirrhosis.9–11 Patients were excluded based on the following criteria: (a) history of cardiovascular disease (including hypertension, heart failure, coronary artery disease, cardiomyopathies, congenital vascular disease, valvular heart disease, and aortic disease); (b) presence of arrhythmias (including atrial fibrillation, frequent premature ventricular complexes, atrioventricular block, sinus arrhythmia, ventricular tachycardia, ventricular fibrillation, and sick sinus syndrome); (c) pulmonary arterial hypertension; (d) comorbid diabetes mellitus, hyperthyroidism, or severe anemia; (e) renal insufficiency; (f) use of medications known to affect cardiac function during the observation period (including beta-blockers, diuretics, and vasoactive agents); (g) history of portosystemic shunt surgery or liver transplantation; (h) hepatocellular carcinoma or other malignancies; and (i) suboptimal echocardiographic image quality or incomplete essential clinical data.
Initially, 163 cirrhotic patients were identified. Of these, 41 were excluded for the following reasons: hypertension (n = 9); arrhythmia (n = 5, including 3 with atrial fibrillation, 1 with atrioventricular block, and 1 with sinus arrhythmia); diabetes mellitus (n = 5); concurrent hypertension and diabetes (n = 4); renal failure (n = 2); use of beta-blockers, diuretics, or vasoactive medications (n = 6); prior portosystemic shunt (n = 5); liver malignancy (n = 3); and inadequate image quality (n = 2). Consequently, 122 eligible cirrhotic patients, including 82 with hepatitis B virus-related cirrhosis, 28 with alcohol-related cirrhosis, 9 with hepatitis C virus-related cirrhosis, and 3 with autoimmune hepatitis-related cirrhosis, and 39 healthy volunteers without underlying diseases were enrolled.
The 122 cirrhotic patients were stratified into 3 groups according to the Child–Turcotte–Pugh (CTP) classification9–11 and assessed for CCM based on the 2020 guidelines: 1 Group B1 (CTP class A) included 35 patients (26 males and 9 females) aged 32–73 years, with a mean age of 57.3 ± 11.2 years. Five patients met the CCM criteria. Group B2 (CTP class B) included 43 patients (30 males and 13 females) aged 42–73 years, with a mean age of 59.3 ± 8.7 years. Fourteen patients met the CCM criteria. Group B3 (CTP class C) included 44 patients (32 males and 12 females) aged 42–78 years, with a mean age 59.5 ± 9.7 years. Twenty-three patients met the CCM criteria. The control group (Group A) included 39 healthy individuals undergoing routine check-ups (29 males and 10 females); their age range was 37–81 years, and the mean age was 58.9 ± 10.3 years.
The 2020 CCM criteria were as follows: (a) Systolic dysfunction was defined based on whether any one of the following criteria were met: LVEF ≤50% and absolute global longitudinal strain (GLS) value <18%. (b) Diastolic dysfunction was defined based on whether at least three of the following criteria were met: early diastolic mitral annular velocity at the septal site (e′) <7 cm/s, ratio of mitral inflow early diastolic velocity (E) to e′ (E/e′) ≥15, left atrial volume index (LAVI) >34 mL/m2, and tricuspid regurgitation peak velocity >2.8 m/s.
Clinical and biochemical examinations
The clinical data and general information of each participant, such as sex, age, weight, and height, were recorded. Body mass index (BMI) and body surface area (BSA) were computed. Heart rate (HR) and blood pressure (BP) were measured following a 15-minute rest period in a calm environment. Biochemical indices, including N-terminal pro-B-type natriuretic peptide (NT-proBNP), were measured. For patients with cirrhosis, the model for end-stage liver disease (MELD) scores were calculated using the following formula: MELD = 3.8 × ln (total bilirubin (mg/dL)) + 11.2 × ln (international normalized ratio) + 9.6 × ln (creatinine (mg/dL)) + 6.4 × etiology (where etiology is 0 for cholestatic or alcoholic liver disease and 1 for all other etiologies), and the presence of hepatic encephalopathy were evaluated according to the West–Haven criteria (grades I–IV).
Instruments and methods
Ultrasound parameters. A GE Vivid E95 diagnostic ultrasound machine with M5S-D and C1-6 probes was used. We performed an abdominal ultrasound examination to measure the portal vein inner diameter (Dpv) and portal vein velocity (Vpv) as well as the depth of ascitic fluid. The severity of ascites was assessed and classified as follows: mild ascites, depth <3 cm; moderate ascites, depth 3–10 cm; severe ascites, depth >10 cm. After connecting the electrocardiogram (ECG), the following routine parameters were measured: (a) Structural indices. Interventricular septal thickness (IVSTd), left ventricular posterior wall thickness (PWTd), left ventricular end-diastolic internal diameter (LVDd), and left ventricular end-systolic internal diameters (LVDs). The biplane’s Simpson method was applied to measure the left atrial volume (LAV), left ventricular end-diastolic volume (LVEDV), and left ventricular end-systolic volume (LVESV); then, we calculated the LAVI, left ventricular mass (LVM), LVM index (LVMI), stroke volume (SV), cardiac output (CO), cardiac index (CI), and SV index (SVI). (b) Functional metrics. Mitral early diastolic and late diastolic inflow velocities (E and A), the tissue Doppler e′ velocities of the mitral annulus septal wall, calculation of E/A and E/e′, and LVEF (the biplane Simpson’s method).
For measuring GLS, a GE Vivid E95 diagnostic ultrasound machine with an M5S-D probe was selected. Following connection of the ECG, participants were instructed to breathe calmly in the left lateral position and then hold their breath. Three LV-focused views (apical four, apical three, and apical two-chamber) were acquired and stored. It was imperative that the variability of the HR in the three apical views was <5 beats/min, and the frame rate of the images was ≥50 FPS. The dynamically captured images were then imported into the Echo PAC offline analyzer in the DICOM format for analysis. GLS was ultimately derived from software-generated automated tracking, supplemented by manual adjustment of the endocardial border. The GLS values are expressed as absolute values.
For the measurement of EF1, following ECG connection, the apical five-chamber view was acquired. The utilization of spectral Doppler enabled the acquisition of the aortic valve flow velocity profile, thus facilitating the measurement of the time interval from aortic valve opening to peak aortic flow velocity (T1) (Figure 1). Additionally, we measured the ejection time (ET) and then calculated their ratio (T1/ET). Subsequently, the optimized apical four-chamber and two-chamber views were obtained. Left ventricular volumetric analysis was conducted employing the biplane Simpson’s method at two critical time points demarcated by the T1 interval. A schematic representation of EF1 measurement is provided in Figure 2.

Measurement of T1. Double-edged arrow “b” represents full systole. Double-edged arrow “a” represents aortic valve time from opening to peak aortic valve flow rate (T1). T1: time interval from aortic valve opening to peak aortic flow velocity.

Schematic of EF1 measurement. (a) Acquisition of aortic valve flow spectra based on apical five-chamber cardiac views using spectral Doppler and measurement of aortic valve time from opening to peak aortic valve flow rate (T1). (b) Measurement of left ventricular end-diastolic volume and (c) measurement of peak aortic valve flow rate left ventricular volume. Then, EF1 was obtained by measuring the percent change in the volume between end-diastolic volume (EDV) and peak aortic valve flow rate (T1): EF1 = [(LVEDV − LVESVpeak)/LVEDV] × 100%. T1: time interval from aortic valve opening to peak aortic flow velocity; EF1: first-phase ejection fraction; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume.
Formula: EF1 = [(LVEDV − LVESVpeak)/LVEDV] × 100%.
Statistical analyses
Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) software (version 27.0). Normally distributed quantitative variables were expressed as mean ± SD. Intergroup comparisons were performed using one-way analysis of variance (ANOVA) with LSD test or Bonferroni-adjusted post hoc test under homogeneity of variance. Welch’s ANOVA with Tamhane’s T2 post hoc analysis was applied for heteroscedastic data. Non-normally distributed variables were presented as median (interquartile range) and analyzed using the Kruskal–Wallis H test, with Bonferroni-adjusted pairwise comparisons. Categorical variables were expressed as frequencies (%) and assessed using the chi-square test. p < 0.05 was considered statistically significant. Spearman’s rank correlation was employed to assess relationships between systolic function parameters (EF1, GLS, and EF) and CTP classification. Multiple linear regression analyses were performed with EF1, LVEF, and GLS as the dependent variables and sex, age, BMI, etiology, MELD scores, and ascites status as independent variables to examine the association between CTP class and left ventricular systolic function parameters. Receiver operating characteristic (ROC) curve analysis evaluated the diagnostic utility of EF1, GLS, and EF for CCM in cirrhotic patients. Areas under the ROC curve (AUCs) were calculated and compared. Inter- and intra-observer variability were assessed using intraclass correlation coefficient (ICC) analysis in a randomly selected subset of 30 patients.
Reproducibility assessment
Thirty randomly selected patients were subjected to EF1 and GLS measurements by two independent sonologists for inter-observer analysis. One week later, one sonologist repeated the measurements on the same 30 patients for intra-observer assessment. Intra-observer and inter-observer correlation coefficients were calculated.
Results
Baseline characteristics and clinical biochemical parameters
No significant differences were observed in age, sex, BMI, HR, systolic BP (SBP), or diastolic BP (DBP) between the four groups (all p > 0.05). NT-proBNP levels progressively increased across groups A, B1, B2, and B3 (all p < 0.05). In patients with cirrhosis, compared with group B1, the prevalence of CCM was significantly higher in group B3 (p < 0.05). MELD scores were significantly increased in groups B2 B3 compared with those in group B1 (both p < 0.05). The overall prevalence of ascites was higher in group B3 (95.5%) than in group B2 (88.4%); however, there was no difference between groups B2 and B3 (p > 0.05). The prevalence of hepatic encephalopathy was significantly higher in group B3 (84.1%) than in group B2 (23.3%) (p < 0.05) (Table 1). Among the 122 patients with cirrhosis, a total of 42 patients (34.4%) were diagnosed with CCM using cardiac ultrasonography and exhibited systolic dysfunction and/or diastolic dysfunction. Among them, 10 patients (23.8%) were diagnosed with CCM based on cardiac diastolic dysfunction, fulfilling ≥3 diagnostic criteria for CCM, but had normal systolic function (LVEF >50% and GLS ≥18%). Nine patients (21.4%) were diagnosed with CCM based on systolic dysfunction but had normal diastolic function. Furthermore, 23 patients (54.8%) were diagnosed with CCM along with diastolic and systolic dysfunction, including 9 patients with LVEF ≤50% and GLS <18% and 14 with LVEF >50% but GLS <18%.
Comparison of general clinical data between the four groups.

p < 0.05 versus group A;
p < 0.05 versus group B1; and
p < 0.05 versus group B2.
HR: heart rate; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; NT-proBNP: N-terminal pro-B-type natriuretic peptide; CCM: cirrhotic cardiomyopathy; MELD: model for end-stage liver disease; HE: hepatic encephalopathy.
Conventional echocardiographic parameters
LVESV demonstrated a progressive increase across groups A, B1, B2, and B3 (all p < 0.05). Compared with group A, groups B1, B2, and B3 exhibited significantly increased LVM, LVMI, LVEDV, SV, CO, SVI, CI, and E/e′, alongside decreased e′ (all p < 0.05). Specifically, e′ was significantly decreased in group B3 compared with that in group B1, while LVEDV and E/e′ were significantly increased in groups B2 and B3 compared with those in group B1 (all p < 0.05). Compared with groups A and B1, groups B2 and B3 demonstrated significant increases in IVSTd, PWTd, Dpv, and LAVI as well as significant decreases in Vpv and GLS (all p < 0.05). Notably, compared with group B2, group B3 showed significantly greater increase in Dpv and significantly greater decrease in Vpv and GLS (all p < 0.05). EF was significantly decreased in group B3 compared with that in groups A, B1, and B2 (all p < 0.05). Similarly, LVDd and LVDs were significantly increased in group B3 compared with those in groups A and B1 (all p < 0.05). There were no significant differences in E/A, T1, ET, and T1/ET between the four groups (all p > 0.05) (Table 2).
Comparison of conventional ultrasound parameters and EF1 among the four groups.

p < 0.05 versus group A;
p < 0.05 versus group B1; and
p < 0.05 versus group B2
IVSTd: interventricular septal thickness at end-diastole; PWTd: posterior wall thickness at end-diastole; LVDd: left ventricular end-diastolic dimension; LVDs: left ventricular end-systolic dimension; Dpv: portal vein inner diameter; Vpv: portal vein velocity; LVM: left ventricular mass; LVMI: left ventricular mass index; E/A: mitral inflow early (E) and late (A) diastolic velocities ratios; LVEF: left ventricular ejection fraction; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; SV: stroke volume; SVI: stroke volume index; CO: cardiac output; CI: cardiac index; LAVI: left atrial volume index; e′: early diastolic mitral annular velocity at the septal site; E/e′: mitral inflow early (E) and early diastolic mitral annular velocity at the septal site ratios; GLS: absolute value of the global longitudinal strain; EF1: first-phase ejection fraction; T1: the time interval from aortic valve opening to peak aortic flow velocity; ET: ejection time; T1/ET: ratio of the time interval from aortic valve opening to peak aortic flow velocity (T1) to ejection time (ET).
EF1 parameters
EF1 was significantly increased in group B1 compared with that in group A (p < 0.05). Conversely, EF1 was significantly decreased in groups B2 and B3 compared with that in group A (both p < 0.05), with group B3 showing a significant decrease compared with group B2 (p < 0.05) (Table 2).
Correlation analysis
EF1, EF, and GLS demonstrated significant negative correlations with CTP class (r = −0.619, −0.429, and −0.608, respectively; p < 0.05). Multiple linear regression analysis demonstrated that the CTP class was significantly correlated with indices of left ventricular systolic function, including EF1, GLS, and EF (all p < 0.05) (Table 3).
Associations of risk factors with left ventricular systolic dysfunction parameters: a linear regression analysis.

Bold values indicate p < 0.05.
LVEF: left ventricular ejection fraction; GLS: global longitudinal strain; EF1: first-phase ejection fraction; BMI: body mass index; MELD: model for end-stage liver disease.
ROC analysis
The AUCs for diagnosing CCM in patients with cirrhosis were 0.917 (95% confidence interval (CI): 0.850–0.983) for EF1, 0.895 (95% CI: 0.840–0.949) for EF, and 0.902 (95% CI: 0.837–0.966) for GLS. EF1 demonstrated superior diagnostic performance compared with GLS and EF (both p < 0.05) (Figure 3 and Table 4).

ROC curves obtained from different ultrasound parameters to diagnose CCM in patients with cirrhosis. ROC: receiver operating characteristic; CCM: cirrhotic cardiomyopathy.
Diagnostic accuracy of EF1, GLS, and LVEF.

EF1: first-phase ejection fraction; GLS: global longitudinal strain; LVEF: left ventricular ejection fraction; AUC: area under the curve; CI: confidence interval.
Repeatability test
EF1 and GLS had high degree of intra- and inter-observer reproducibility (Table 5).
Reliability analysis of GLS and EF1 parameters.
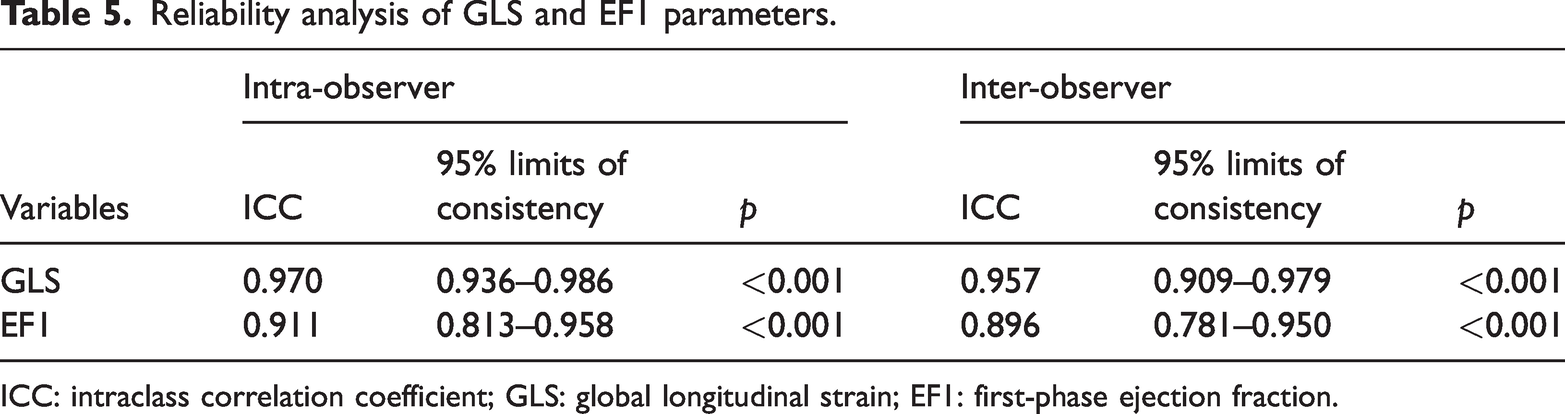
ICC: intraclass correlation coefficient; GLS: global longitudinal strain; EF1: first-phase ejection fraction.
Discussion
In cirrhotic patients, hepatic decompensation triggers a multifactorial cascade involving bioactive metabolite accumulation, systemic inflammation, malnutrition, and portal hypertension-induced vasodilator release.2,12 These mechanisms collectively induce visceral arteriolar vasodilation, systemic vascular resistance reduction, and afterload diminution,2,12 thereby activating sympathetic neurohormonal pathways and elevating cardiac output to establish a hyperdynamic circulatory state.2,12,13 Consequently, compared with the control group, the SVI and CI in groups B1, B2, and B3 were significantly elevated (all p < 0.05). However, chronically sustained hyperdynamic circulation may ultimately precipitate high-output heart failure, induce compensatory cardiac remodeling, and lead to CCM. This study demonstrates the potential for CCM manifestation even in group B1. Critically, the prevalence of CCM was significantly higher in group B3 than in group B1. Early assessment of left ventricular functional alterations in cirrhotic patients is essential for monitoring CCM onset and progression as well as for optimizing clinical outcomes.
In the 2020 CCM guideline, 1 both LVEF and GLS are validated parameters, where resting LVEF ≤50% or absolute GLS values <18% provide direct diagnostic evidence of CCM. The present study utilized LVEF and GLS to assess left ventricular function across CTP classifications and compare it with that in healthy controls. Although compared with group B1, group B2, and control group, the LVEF in group B3 was significantly decreased (all p < 0.05), almost all cirrhotic patients maintained LVEF ≥50%. This apparent paradox stems from compensatory augmentation of myocardial contractility in response to hyperdynamic circulation-induced ventricular wall stress, which maintains the LVEF due to progressive depletion of cardiac diastolic functional reserves.4,14,15 When exposed to physiological stress or cirrhotic progression, these patients may manifest reduced EF and overt heart failure. 3 GLS demonstrates superior sensitivity over LVEF for detecting early systolic dysfunction 16 and provides a more accurate assessment of left ventricular systolic function in cirrhosis. 17 In our study, compared with groups A and B1, the absolute GLS values significantly decreased in groups B2 and B3 (all p < 0.05), aligning with Isaak et al.’s findings, 18 which showed that increasing CTP scores in cirrhotic patients are correlated with progressive systolic dysfunction. Notably, divergent findings persist regarding GLS alterations in advanced cirrhosis,17,19 with some studies reporting preserved or even increased GLS in late-stage patients with poor prognosis.17,19 Given these limitations in conventional metrics, we introduced EF1 as a novel index for assessing left ventricular systolic function.
In this study, EF1 was significantly increased in group B1 compared with that in group A (p < 0.05). Conversely, EF1 was significantly decreased in groups B2 and B3 compared with that in group A (both p < 0.05), with group B3 showing significantly greater decrease compared with group B2 (p < 0.05). Myocardial wall stress in healthy individuals in group A occurs during early systole, near the T1 of the left ventricular pressure waveform, coinciding with the peak aortic flow, when myofibrillar contraction velocity is maximal. 4 However, in group B1, when myocardial contractile function is fair, EF1 shows a rising trend due to the activation of the sympathetic nervous system and the activation of myocardial β-AR that induces enhanced contraction of cardiomyocytes.20,21 During the progression to group B2 and group B3 cirrhosis, hepatic decompensation drives the accumulation of bioactive metabolites and systemic chronic inflammatory responses, which synergistically disrupts cardiomyocyte signaling pathways through oxidative stress and cytokine cascades, ultimately inducing myocardial dysfunction. 2 At these two stages, the compensatory activation of both sympathetic nervous system and renin–angiotensin–aldosterone system (RAAS) reaches its threshold, where their capacity to enhance CO and induce vasoconstriction becomes insufficient to counteract visceral vasodilation and the loss of effective circulating volume. This persistent overload has been shown to trigger β-adrenergic receptor (β-AR) downregulation and functional uncoupling, which ultimately results in receptor desensitization and impaired signal transduction.22,23 Furthermore, chronic left ventricular pressure–volume overload exacerbates interstitial fibrosis, inducing increased myocardial electrical conduction heterogeneity and electromechanical dyssynchrony, thereby aggravating the loss of coordinated contractility. 24 Thus, EF1 tends to decrease during the decompensation period. The significant negative correlations between systolic function parameters (EF1, EF, and GLS) and CTP class reflect progressive impairment of cardiac contractility resulting from declining hepatic function and sustained hyperdynamic circulation. ROC curve analysis confirmed the superior diagnostic accuracy of EF1 for CCM compared with GLS and EF. This finding substantiates that EF1 may enhance sensitivity for detecting early systolic changes.
In this study, the E/A ratio revealed no significant intergroup differences (p > 0.05). Compared with the control group, all cirrhotic subgroups exhibited markedly elevated E/e′ and diminished e′ (both p < 0.05). Increased E/e′ and decreased e′ reflect progressive depletion of diastolic reserve capacity in the hyperdynamic state. This phenomenon has also been observed in other etiologies of heart failure with preserved EF.25,26 Both parameters provide greater sensitivity than E/A in detecting diastolic dysfunction. Compared with group A, groups B1, B2, and B3 demonstrated significantly increased LVM and LVMI (all p < 0.05). Compared with groups A and B1, groups B2 and B3 exhibited significantly increased IVSTd, PWTd, and LAVI (all p < 0.05). Compared with groups A and B1, group B3 showed significantly increased LVDd and LVDs (both p < 0.05). The aforementioned data not only illustrate the early changes in systolic and/or diastolic function in patients with cirrhosis but also the gradual changes in cardiac structure under the influence of prolonged hyperdynamic circulation.
Limitations
This was a single-center study with a relatively small sample size; therefore, future research involving larger cohorts is necessary to validate our findings and establish more precise cutoff values. We also plan to conduct studies using larger cohorts that would allow more detailed subgroup analyses. EF1 measurement requires high image quality, and some patients were not enrolled because of poor image quality. The exclusion of patients with hypertension and diabetes and those receiving beta-blockers, diuretics, or vasoactive agents during the study observation period restricted the generalizability of our findings to a real-world cirrhotic population.
Conclusion
EF1 may effectively assess early left ventricular systolic function changes in patients with cirrhosis.
Footnotes
Acknowledgments
We appreciate Prof Yang’s guidance regarding statistical methods and the help provided by Prof Li for correcting possible grammatical and spelling errors in the manuscript.
Author contributions
G.S.L. and B.B.Y. provided the study concept and design. X.Y. and X.W. performed the examination. X.Y and X.W. collected the clinical data. G.N.J., J.Y.Y., J.Y.N., and X.W. analyzed the data. X.Y. wrote the manuscript. G.S.L. and B.B.Y. revised the manuscript and data. All authors read and approved the final manuscript.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024. The experimental procedures complied with relevant privacy protection laws and guidelines. This research was approved by the hospital’s ethics committee (Ethics number: [2025] the Second Affiliated Hospital of Dalian Medical University, Ethics Approval Number [239]). All experiments were conducted in accordance with the relevant guidelines and regulations, and informed consent was obtained from each participant.
Funding
Not applicable.
