Abstract
The trifunctional, bispecific antibody catumaxomab (anti-EpCAM x anti-CD3) is a new therapeutic strategy for epithelial cancer patients with symptomatic malignant ascites. The trifunctionality has the advantage of binding tumor cells and simultaneously two different immune effector cells (e.g. macrophages and T-lymphocytes). The unique binding specifities of catumaxomab induce an effective destruction of tumor cells, including those resistant to apoptosis, through different mechanisms, i.e. perforin-mediated lysis, antibody-mediated phagocytosis, and cytokine release. Moreover, a possible long-lasting humoral and cellular antitumor response was shown in a mouse model with a surrogate bispecific trifunctional antibody binding to mouse CD3. Catumaxomab (Removab®) is the first medication approved by the EMEA (European Medicines Agency) for the treatment of malignant ascites due to EpCAM-positive carcinomas where no standard therapy is available or no longer feasible. The safety profile is acceptable. Adverse events associated with catumaxomab are manageable, generally reversible and mainly related to its immunologic mode of action. Catumaxomab is a promising new therapy for malignant acites even in heavily pretreated patients. Intraperitoneal treatment with catumaxomab plus paracentesis demonstrated clinically relevant benefit in patients with recurrent malignant ascites. Significantly prolonged puncture-free survival (46 vs. 11 days) as well as prolonged time to first need of therapeutic puncture (77 vs. 13 days) was shown in comparison to paracentesis alone (P < 0.0001).
Introduction
Malignant ascites is a common manifestation of different advanced cancers including ovarian, gastric, endometrial, breast, colon and pancreatic cancer and is associated with poor prognosis. 1 Mechanisms leading to malignant ascites are independent on the origin primary tumor.1,2
Malignant ascites is caused by the peritoneal spread of malignant tumor cells leading to increased capillary permeabilization associated with an elevated protein concentration in the peritoneal fluid and impaired lymphatic drainage of fluid owing to obstruction of the lymphatic channels by tumor cells. Vascular endothelial growth factor, secreted by tumor cells, may also contribute to ascites formation by increasing vascular permeability. 3
The clinical management of malignant ascites is a distressing problem. Symptoms like fatigue, depression, anxiety in addition to shortness of breath, abdominal distension, abdominal bloating, anorexia, dyspnea, insomnia, edema, pain, respiratory distress, lower extremity discomfort, reduced mobility and wellbeing are typical in case of symptomatic ascites. Repeated paracentesis is the most common method to improve the quality of life of the patients, resulting in significant but temporary symptom relief. 4 Paracentesis does not prevent ascites re-accumulation, which generally occurs within few days. Risks of paracentesis are infection, peritonitis and bowel perforation. Repeated paracentesis removes large volumes of ascites fluid, resulting in protein loss and hypovolemia with subsequent circulatory problems including hypovolemic shock. Human albumin substitution is therefore often necessary.
Other therapeutic strategies are intraperitoneal and/or systemic chemotherapy (e.g. cisplatin, bleomycin, mitoxantrone), radioisotopes and radioactive labelled monoclonal antibodies5,6 and stimulations of the immune system (like IFN-γ, IFN-α, Interleukin-2, TNF-α).7–11 None of these approaches have been established as standard therapy for refractory malignant ascites because of limited efficacy or severe side effects.8,9
Therefore, new strategies are warranted for patients suffering from malignant ascites.
Most tumors of epithelial origin show an over-expression of EpCAM (epithelial cell adhesion molecule) and in about 90% of ascites samples of ovarian cancer patients, EpCAM-positive tumor cells were detected. 12 Therefore, EpCAM is an attractive target for antibody therapy. 13 Tumor cells in malignant effusions express EpCAM in 70%–100% of those cases that commonly cause malignant ascites, e.g. ovarian, gastric, breast and colorectal cancer. 14 EpCAM is also expressed on cells of normal epithelial tissues. 13 However, within the peritoneal cavity, EpCAM expression is tumor-specific because normal cells in the peritoneal compartment are of mesothelial origin and do not express EpCAM on their surface. 15 Therefore, i.p. administration of antibodies directed against EpCAM offers an advantage of a targeted immunotherapy leading to a direct and specific attack on ascites-causing tumor cells in the peritoneal cavity. Furthermore, all effector cells, like T-cells and accessory immune cells are located in the peritoneal fluid. Thus, especially intraperitoneal treatment of epithelial cancer patients targeting EpCAM seems to be promising.
A new therapeutic option is the intraperitoneal treatment with the trifunctional, bispecific antibody catumaxomab, that represents a new generation of monoclonal antibodies. 16 Catumaxomab consists of a mouse immunoglobulin G2a and a rat immunoglobulin G2b part with two different antigen binding sites binding EpCAM on tumor cells and CD3 on T lymphocytes. The intact Fc region provides a third functional binding site, binding and activating selectively Fcγ receptor I, IIa, and III-positive accessory cells. 16 The trifunctionality has the advantage of binding tumor cells and simultaneously two different immune effector cells. The resulting tri-cell complex leads to different mechanisms of tumor cell killing, like antibody dependent cellular cytotoxicity, phagocytosis, apoptosis, cytokine release and perforin mediated lysis.17–19 An activation of the humoral and cellular immune system results by application of this rat/mouse quadromaderived bispecific antibody catumaxomab. 19
Intraperitoneal application of catumaxomab offers the advantage of targeted immunotherapy with a direct and specific attack on the ascites-causing tumor cells in the peritoneal cavity. The intraperitoneal route assures a high concentration of anticancer therapy at the peritoneal surfaces. All effector cells essential for the antitumor activity of catumaxomab, like T cells and accessory cells, are present within the ascites fluid.20,21
Objectives
There is a great need for an effective treatment of malignant ascites. Therefore the evidence of the therapeutic value of catumaxomab as a new therapeutic option is evaluated.
Materials and Methods
Articles and reviews found in PubMed and reference books were evaluated and compared to each other.
Within a pilot study eight patients with symptomatic ascites with histologically confirmed peritoneal carcinomatosis of different solid tumors were included (2 ovarian cancers, 3 breast cancers, 1 adenocarcinoma of unkown primary origin, 1 bronchial cancer and 1 gastric cancer patient). In all patients the EpCAM or HER2/neu antigen was found by immunohistochemical staining. Patients had to have either one prior chemotherapy, were either not considered suitable for chemotherapy or had refused chemotherapy. Exclusion criteria were acute or chronic infections, and relevant concomitant haematological, cardiovascular, pulmonary, hepatic, renal, pancreatic or endocrinal disease. Additional exclusion criteria included leucocyte counts <3 or >13 x 10 9 /L, thrombocyte counts <50 x 10 9 /L, bilirubin levels >2.0 mg/dL and serum creatinine levels >2.0 mg/dL and former antibody therapy. 22
The influence of catumaxomab on CK+ positive cells in bone marrow and blood was analysed within a study with 14 patients with ovarian cancer and symptomatic ascites. Up to 5 i.p. infusions with increasing dosages (10–200 μg) were applied. 23
A multicenter phase I/II dose-escalating study investigated tolerability and efficacy of intraperitoneal catumaxomab application in 23 ovarian cancer patients with malignant ascites containing EpCAM-positive tumor cells. 24 Ovarian cancer patients were treated with four to five intraperitoneal infusions in doses of 5 to 200 μg within 9 to 13 days. Inclusion and exclusion criteria were similar to the pilot study 24
In the phase II/III trial, cancer patients (n = 258) with recurrent symptomatic malignant ascites resistant to conventional chemotherapy were randomized to paracentesis plus catumaxomab or paracentesis alone (control) and stratified by cancer type.25,26 Adult patients ≥18 years of age with histologically confirmed epithelial cancer and EpCAM-positive tumor cells in the ascites fluid were eligible for inclusion in this study. Other inclusion criteria were: Karnofsky performance status $60; life expectancy >8 weeks; estimated ascites volume >1 l by CT scan; at least one symptomatic paracentesis within 5 weeks as well as an objectively verified, clinical need for a second paracentesis; refractory or resistant to chemotherapy or standard chemotherapy was no longer feasible. Exclusion criteria were: exposure to cancer chemotherapy or radiotherapy within the previous 28 days; previous treatment with murine monoclonal antibodies; enteral feeding a study entry or ileus within the previous 30 days; ≥70% tumor infiltration of the liver, or portal vein obstruction, or thrombosis.25–27 Patients were randomized 2:1 to paracentesis plus catumaxomab or paracentesis alone (control) and stratified by cancer type (ovarian or non-ovarian cancers). Catumaxomab was administered as an intraperitoneal infusion (over 6 hours) on days 0, 3, 7 and 10 at doses of 10, 20, 50 and 150 μg, respectively. The primary efficacy endpoint was puncture-free survival. Secondary efficacy parameters included time to next paracentesis, ascites signs and symptoms and overall survival.25–27
Discussion
Within a pilot study 8 patients with malignant ascites due to peritoneal carcinomatosis were treated with intraperitoneal application of trifunctional, bispecific antibodies catumaxomab or ertumaxomab, which bound either EpCAM (catumaxomab) or the human epidermal growth receptor 2 (Her2)/neu-antigen (ertumaxomab) on tumor cells. 4 patients received catumaxomab only and 3 patients got a combined application of catumaxomab and ertumaxomab. Treatment consisted of four to six applications within 9 to 23 days with a total amount of 145 to 940 μg. 22 The maximum cumulative ascites volume within an interval of 5 days before enrolment was 500 to 12.800 ml and the number of paracentesis within 90 days before trifunctional therapy was 1 to 11 paracentesis. 7 of 8 patients required no further paracentesis during follow-up or until death with a mean paracentesis-free interval of 38 weeks (median = 21.5, range = 4–136). One patient had an ascites relapse 4 weeks after start of trifunctional therapy requiring paracentesis. Another patient died 17 weeks after treatment due to tumor progression, which was accompanied with an ascites relapse. The clinical effect of trifunctional treatment was significantly correlated with elimination of tumor cells in ascites as defined by no further palliative paracentesis during follow-up or until death in 7 of 8 patients (P = 0.0014). Tumor cell monitoring by FACS analysis or immunocytochemistry showed the elimination of ascites tumor cells during trifunctional treatment. The proportion of EpCAM-or HER2/neu positive tumor cells in ascites samples substantially decreased during antibody treatment. Intraperitoneal treatment with trifunctional therapy within the pilot study was well tolerated. Not a single patient treatment had to be stopped due to severe side effects or adverse events. No hypotensive reaction or organ failure requiring ICU treatment was observed. Most patient had a flu-like syndrome (7 of 8), with fever (maximum = 40.5 °C) lasting for 8–18 hours, chills and fatigue. In 4 patients, allergic skin reactions were observed. Two patients had a local infection at the catheter-insertion site, that could successfully be treated by change of catheter and antibiotics. In 4 patients transient elevation of liver enzyme was detected, which reversed to normal values within 2–5 days without any specific treatment. 22
In addition, the efficacy of intraperitoneal catumaxomab treatment was investigated in 14 ovarian cancer patients with symptomatic ascites on cytokeratin positive (CK+) cells in bone marrow (BM) and peripheral blood (PB) before and after antibody treatment. 23 In this small study population no correlation of marked changes in CK+ cells and outcome was assessed. However, one might suggest a systemic effect by intraperitoneal treatment with catumaxomab, because mean cell number of CK+-cells detected before and after antibody therapy in peripheral blood tended to decrease. 23
CK+ cells were found in (57% of the patients) with a median number of 5 cells/20 ml (range 1 to >100 cells) before and in 42% with a median number of 2 cells/20 ml (range 1–3) after catumaxomab treatment. No marked increase of CK-positive cells in the blood was determined after the antibody therapy. Mean overall survival after antibody therapy was 8 months (range 1–27 months). Only two patients needed a further paracentesis after 2 and after 5 months, respectively. 23
A phase I/II study assessed the maximum tolerated dose (MTD), tolerability and efficacy of catumaxomab in heavily pretreated patients with malignant ascites due to ovarian cancer. In all patients, a sustained reduction in ascites fluid volume after intraperitoneal infusion with catumaxomab was seen. A total of 22 out of 23 patients did not require paracentesis during the observation period (maximum 37 days). Tumor cell reduction in ascites was achieved by up to 5 log (Fig. 1). 24 The MTD was determined for the intraperitoneal infusions at 10–20–50–200–200 μg on days 0, 3, 6, 9 and 13. The pattern of adverse events was determined by the mode of action as the dominating adverse events were those typically associated with the release of cytokines. All patients had treatment emerged adverse events including fever, nausea, vomiting, abdominal pain, lymphopenia and general pain being the most common, and all patients had at least one treatment emerged adverse event considered possibly related to treatment. Most possibly related treatment emerged adverse events were mild or moderate. Transient grade 3 decrease of lymphocytes related to catumaxomab infusion observed in six patients had returned to lower grades within 1 to 8 days. No association with an increased infection rate was observed. 24 There was no apparent dose dependency for the frequency of possibly related treatment emerged adverse events. Cytokine release related symptoms are a well-known side effect of antibody therapies and are regarded as an indicator of efficacy. In the case of catumaxomab, the activation of Fc-receptor-bearing cells besides T cells leads to the additional secretion of cytokines as, e.g. IL-6 or TNFα, which could also be detected in peripheral blood after intraperitoneal application. However, the third valence of trifunctional antibodies is essential because it leads to a substantial enhancement of antitumor efficacy. 24
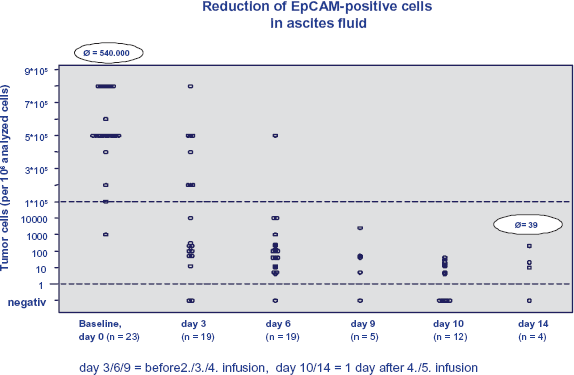
Phase I/II study: reduction of tumor cells in ascites fluid.
For further phase II studies, a safe dose below the MTD was determined and set to 10–20–50–150 μg. 24
Because of the very promising results of the phase I/II trial a prospective, randomized phase II/III study in malignant ascites was conducted to show a beneficial effect of catumaxomab treatment. Within this phase II/III study EpCAM-positive cancer patients with symptomatic malignant ascites requiring paracentesis were treated with paracentesis plus catumaxomab versus paracentesis alone (control group) using the standard dosing scheme of four intraperitoneal infusions with 10–20–50–150 μg on day 0, 3, 7 and 10.25,27 Within this prospective phase II/III study efficacy and safety of catumaxomab in the treatment of malignant ascites due to epithelial cancers were assessed. A total of 258 chemotherapy- refractory patients (129 ovarian cancer patients and 129 non-ovarian cancer patients) were randomized: 65.9% patients in the catumaxomab group and 34.1% in the control group for each disease stratum. The primary efficacy endpoint was puncture-free survival defined as time from one day after the last infusion in the catumaxomab group or day zero (for the control group) to the first therapeutic puncture or death, whichever occurred first. For pooled analysis, puncture-free survival was significantly longer (P < 0.0001, HR = 0.254) for the catumaxomab group (46 vs. 11 days in the control group), as well as time to first need of therapeutic puncture (77 vs. 13 days in the control group).25,27 Puncture-free survival for the two strata (ovarian cancer and non-ovarian cancer) is shown in Figure 3. In the ovarian and non-ovarian cancer patients, median puncture-free survival and time to first need of therapeutic puncture were 52 and 37 days for paracentesis plus catumaxomab versus 11 and 14 days for paracentesis alone, respectively (Fig. 2). All differences were statistically significant (P < 0.0001).25,27 The HR for the pooled population was 0.254 (95% CI: 0.185–0.350), corresponding to a risk reduction for puncture-free survival of 75%. These results represent a significant reduction in interventions compared with existing treatment.25–27 A factor of prolongation of 4.2 of puncture-free survival was present for the pooled analysis. This is clinically relevant, considering the potential risk for infection or bowel perforation with each puncture. Although the trial was not powered for overall survival analysis,26,27 the overall survival showed a positive trend for the catumaxomab group (72 vs. 68 days; P-value: 0.0846; HR = 0.723 (95% CI: 0.498–1.048)) and was significantly prolonged in gastric cancer group, which was the largest subgroup in the non-ovarian cancer stratum (71 versus 44 days; P = 0.0313).25–27
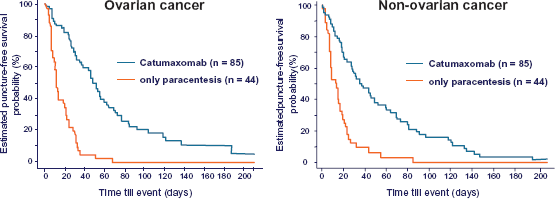
Puncture-free survival in phase II/III trial.
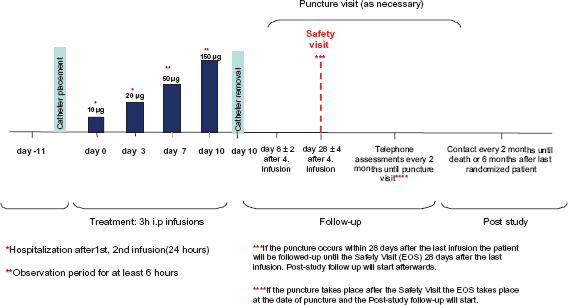
Study design of the CASIMAS study.
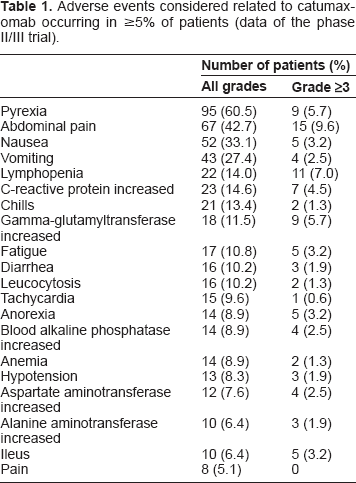
The safety profile was acceptable. Adverse events associated with catumaxomab are manageable, generally reversible and mainly related to its immunologic mode of action. The adverse events of fever, chills last only a few hours and nausea, vomiting and abdominal pain last only a few days. Frequency and severity of adverse events are presented in Table 1. There were fewer ascites signs and symptoms (assessed 8 days after the last catumaxomab infusion or after day 0 in the control group) in catumaxomab treated patients compared with the control group. In six out of 10 symptom categories (anorexia, nausea, early satiety, abdominal pain, abdominal swelling and dyspnea) and in all ascites sign categories (abdominal distension, dull to percussion, shifting dullness, fluid thrill and bulging flanks), these differences were statistically significant (P < 0.05). In addition to benefitting from fewer punctures, patients benefit from a five-fold longer time without burdensome and persistent ascites symptoms. By controlling ascites symptoms catumaxomab helps to improve patients‘ quality of life. 27
Adverse events considered related to catumaxomab occurring in ≥5% of patients (data of the phase II/III trial).
Over 80% of these patients (86% ovarian and 81% non-ovarian cancer) received all four planned infusions.
Puncture free survival was longer (48 days) in patients with (81%) cytokine-release-related symptoms during catumaxomab treatment than in patients without (19%) cytokine-release-related symptoms (27 days), although this difference was not statistically significant (P = 0.1546). 28 The median overall survival showed a positive trend for the catumaxomab group (median 72 versus 68 days, respectively; P = 0.0846; HR = 0.723 (95% CI: 0.498–1.048)).
Pharmacokinetic analyses showed a high variability between subjects. The mean elimination half-life was 2.13 days. The concentrations of catumaxomab in ascites fluid and in plasma increased with the number of infusions and the doses applied in most patients. Plasma levels tended to decline after achieving a maximum after each dose. 29 However, analysis of CK+-cells indicate a systemic effect of intraperitoneal treatment with catumaxomab. 23
The observed safety profile includes frequent adverse reactions such as fever, chills, nausea and vomiting that are assumed to be caused by a release of proinflammatory cytokines, hepatic and hepatobiliary disorders, and a temporary drop of peripheral lymphocytes. These reactions are expected due to the mode of action of catumaxomab. Another frequent adverse event is abdominal pain. Most adverse events were transient and usually resolved without sequelae within several days or a few weeks. The development of human anti-mouse antibody (HAMA) and human anti-rat antibody (HARA) is a well-known consequence of the administration of xenogenous antibodies. Most patients developed a HAMA/HARA response one month after catumaxomab therapy. The clinical significance of HAMA/HARA formation is not yet assessable. However, the available data demonstrated that the antibody can be administered safely at doses not exceeding the MTD. The addition of pain- and fever-reducing medication may in general improve the tolerability of catumaxomab.25–27
This study was the first prospective, randomized clinical trial in malignant ascites to demonstrate a beneficial effect of treatment.
Due to the convincing results of the phase II/III trial, catumaxomab (Removab®) is the first medication approved by the EMEA (European Medicines Agency) for the treatment of a malignant ascites due to EpCAM-positive carcinomas where no standard therapy is available or no longer feasible.
Data suggest that catumaxomab could be even more effective if used at an earlier stage in the treatment of epithelial cancers.
An ongoing two-arm, randomized, multi-center, open-label phase IIIb study (CASIMAS study) investigates the safety of a 3-hour intraperitoneal catumoxomab treatment with or without premedication of prednisolone in patients with malignant ascites due to epithelial cancer (Fig. 3). Rationale of this trial is the evaluation of attenuation of side effects with prednisolone premedication without decline of efficacy. Safety and tolerability of prednisolone premedication using a composite safety score calculated from the frequency and intensity (CTC-grade) are assessed. Inclusion and exclusion criteria are similar to the criteria of the phase II/III study. Primary endpoint of the study is the puncture-free survival for efficacy. Secondary endpoints are time to next ascites puncture, overall survival, number of ascites punctures, further safety end-points, ascites-related symptom scores, quality of life analyses and immunomonitoring. 230 patients are planned within the CASIMAS trial.
In conclusion, intraperitoneal treatment with catumaxomab plus paracentesis demonstrated clinically relevant benefits in patients with recurrent malignant ascites due to epithelial cancers. Catumaxomab showed typical pattern of manageable and reversible adverse events that are mainly related to its immunologic mode of action. However, catumaxomab is an efficient new therapy for malignant acites even in heavily pretreated patients. The induction of long-lasting anti-tumor immunity may provide an additional benefit of the intraperitoneal therapy with catumaxomab and should be further investigated in larger clinical trials.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. Fresenius Biotech GmbH is developing and testing catumaxomab. Pauline Wimberger and Rainer Kimmig have received honoraria, research funding, and other remuneration from Fresenius Biotech GmbH. The peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
