Abstract
Capillary leak syndrome (CLS) is a serious disorder characterized by hypotension and refractory systemic oedema. CLS with marked ascites rather than systemic oedema is rare and prone to misdiagnosis and delayed treatment. We report here a case of marked ascites in an elderly male patient with hepatitis B virus reactivation. Following investigations to exclude common diseases that may have accounted for diffuse oedema and hypercoagulable state, anti-cirrhosis therapy failed and severe refractory shock developed 48 hours after admission. The patient developed mild pleural effusions followed by swelling of the face, neck, and extremities. A high cytokine concentration gradient was detected between serum and ascites. Peritoneal biopsy showed lymphoma cells. The final diagnosis was lymphoma recurrence complicated with CLS. Our case suggests that cytokine detection in serum and ascitic fluid may be helpful in the differential diagnosis of CLS. In similar cases, decisive intervention, such as, hemodiafiltration, should be implemented to lessen the likelihood of serious complications.
Background
Capillary Leak Syndrome (CLS) is a rare disorder characterized by acute and severe recurrent attacks associated with a rapid fall in blood pressure (BP) due to reversible plasma extravasation from blood capillaries. 1 The syndrome includes both primary CLS (Clarkson’s disease) and secondary CLS which is often seen with sepsis, severe injury, chimeric antigen receptor T-cell (CAR-T) therapy, engraftment syndrome, ovarian hyperstimulation syndrome, and severe acute pancreatitis. 2 Patients present with sudden-onset anasarca, oliguria, and low BP. 3 Laboratory test results show profound haemoconcentration and hypoalbuminemia. 4 With early recognition and refined fluid management, patients tend to have a favourable prognosis. 4 Nevertheless, in rare circumstance, secondary CLS can be organ-specific (i.e. lung, heart, or intestines) which can confuse the diagnosis and end in a life-threatening situation.5–7
Cancer and chemotherapy are also considered to be important and rare causes of secondary CLS. 8 For example, patients with lymphoma, such as those with diffuse large B-cell lymphoma or T-cell lymphoma, may develop CLS under cytokine therapy (e.g., interleukin (IL)-2, IL-11, interferon), bortezomib, rituximab, CAR-T therapy, and/or autologous stem cell transplantation.9–14 In these patients, ascites and fever were commonly observed. 12 We report here a rare case of a patient with lymphoma who we believe is the first reported case of CLS with refractory ascites.
Case Report
A man in his late 60s was referred to the gastroenterology department at our hospital. He had a 10-day history of diffuse abdominal distention and a three-day history of intermittent fever. His abdominal girth increased quickly and was accompanied by fatigue and loss of appetite without obvious predisposition. He had no swelling in other parts of the body. He had a history of chronic hepatitis B (1.5 years) hypertension (30 years), and carotid artery disease (10 years). Eighteen months previously, he had been diagnosed with diffuse large B-cell lymphoma non-germinal centre B-cell-like (GCB) type, IV stage, B IPI:5 score with B2M, BCL2, MLL2 mutation. At that time, a positron emission tomography combined with computed tomography (PET-CT) scan showed a wide distribution of lymphoma cells in both the abdomen and the left side of the pelvis (Figure 1). Ten months previously, following eight sessions of standard chemotherapies (i.e., six mini-R-CHOP and two rituximab alone) he was classified as being at ‘incomplete remission’ stage. His current medications were as follows: perindopril tert-butylamine (4 mg qd); atorvastatin (20 mg qd); bisoprolol fumarate (5 mg qd); clopidogrel (50 mg qd); tenofovir disoproxil fumarate (3 mg qd). For more than a year, the patient had been reviewed regularly at monthly intervals for levels of MLL2 and B2M detected by liquid biopsy. A week before this current admission, the patient had visited our haematology department and his blood test results, liver and kidney function were within normal limits. He had no history of severe infections, allergies, or toxin exposure.
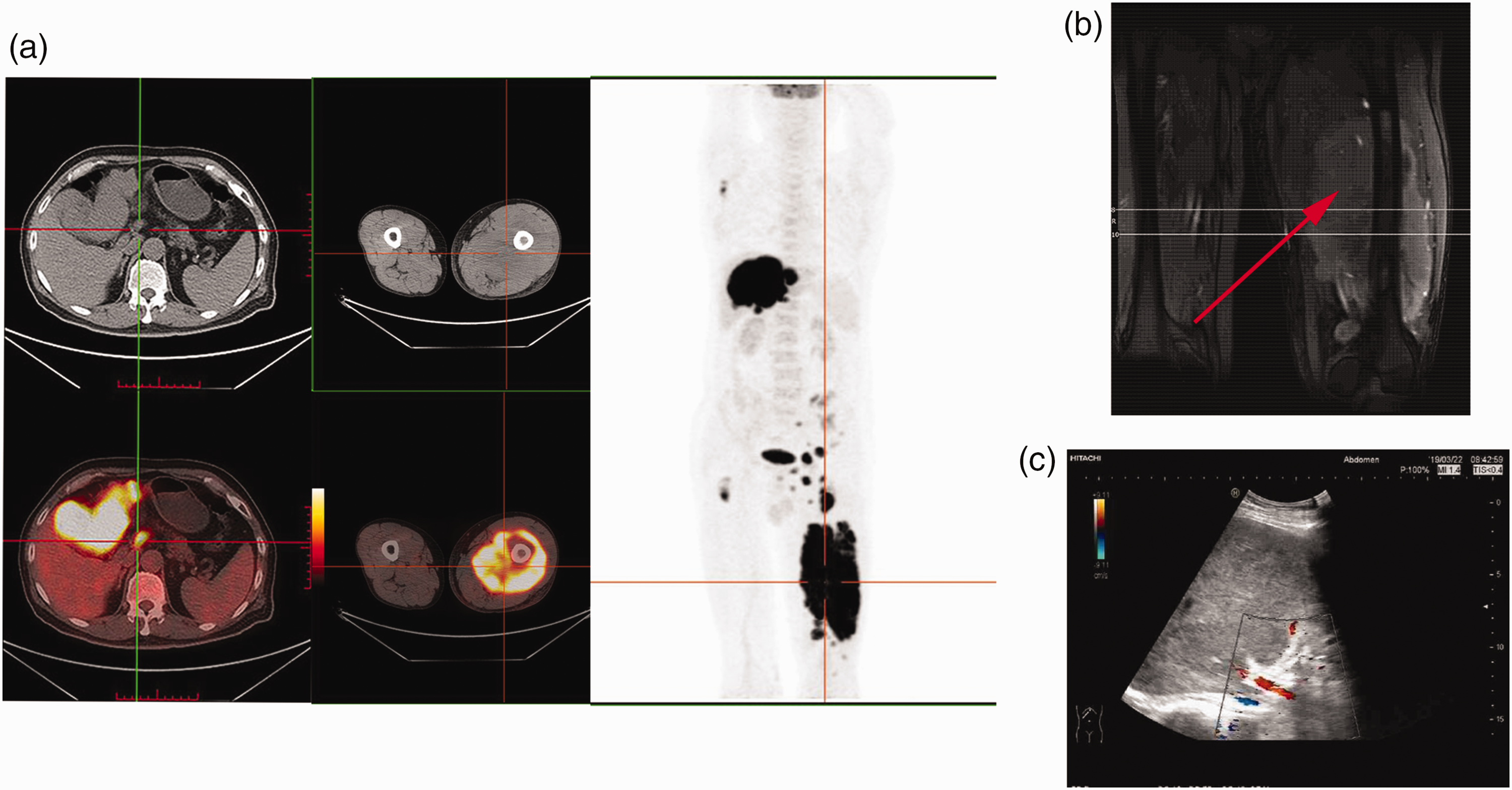
Baseline positron emission tomography-computed tomography (PET-CT), magnetic resonance imaging (MRI) and B-mode ultrasound scans. (a) PET-CT scan showed multiple abnormally enlarged nodes in his left leg, peritoneum, left liver lobe and hilar lymph nodes with a maximum standard uptake volume (SUV) of 19.9. The largest node in the liver was 10.2 × 9.3 cm. (b) MRI image showed a giant soft tissue mass in his left leg and (c) Ultrasound scan on admission showed several hypoechoic nodes and normal portal vein width.
On admission to the hospital, his axillary temperature was 36.7°C and his BP was low (94/52 mmHg). Physical examination showed generalized abdominal distension with shifting dullness (Figure 2a). Lymphadenopathy and hepatosplenomegaly were not detected. The patient’s extremities were cold rather than oedematous (Figure 2b and c). The remainder of the examination was normal. There was no evidence of pallor, cyanosis, jaundice, spider angioma, palmar erythema, clubbed finger, pruritus, tenderness or oedema over joints, rashes, skin mottling, or oral ulcers. His urine was deep yellow and he had a reduced output (approximately 900 ml/24 hours since admission). The patient’s biochemical parameters were notable for hypoalbuminemia (2.9 g/dl), hyperkalaemia (6.4 mmol/l), coagulation dysfunction (prothrombin time 20s), and he had a low estimated glomerular filtration rate (eGFR; 18.0 ml/min, chronic kidney disease epidemiology collaboration [CKD-EPI] equation). Critical indicators of lymphoma increased rapidly, including LDH, ferritin (2305 ug/l), β-2 macroglobulin(14mg/l).
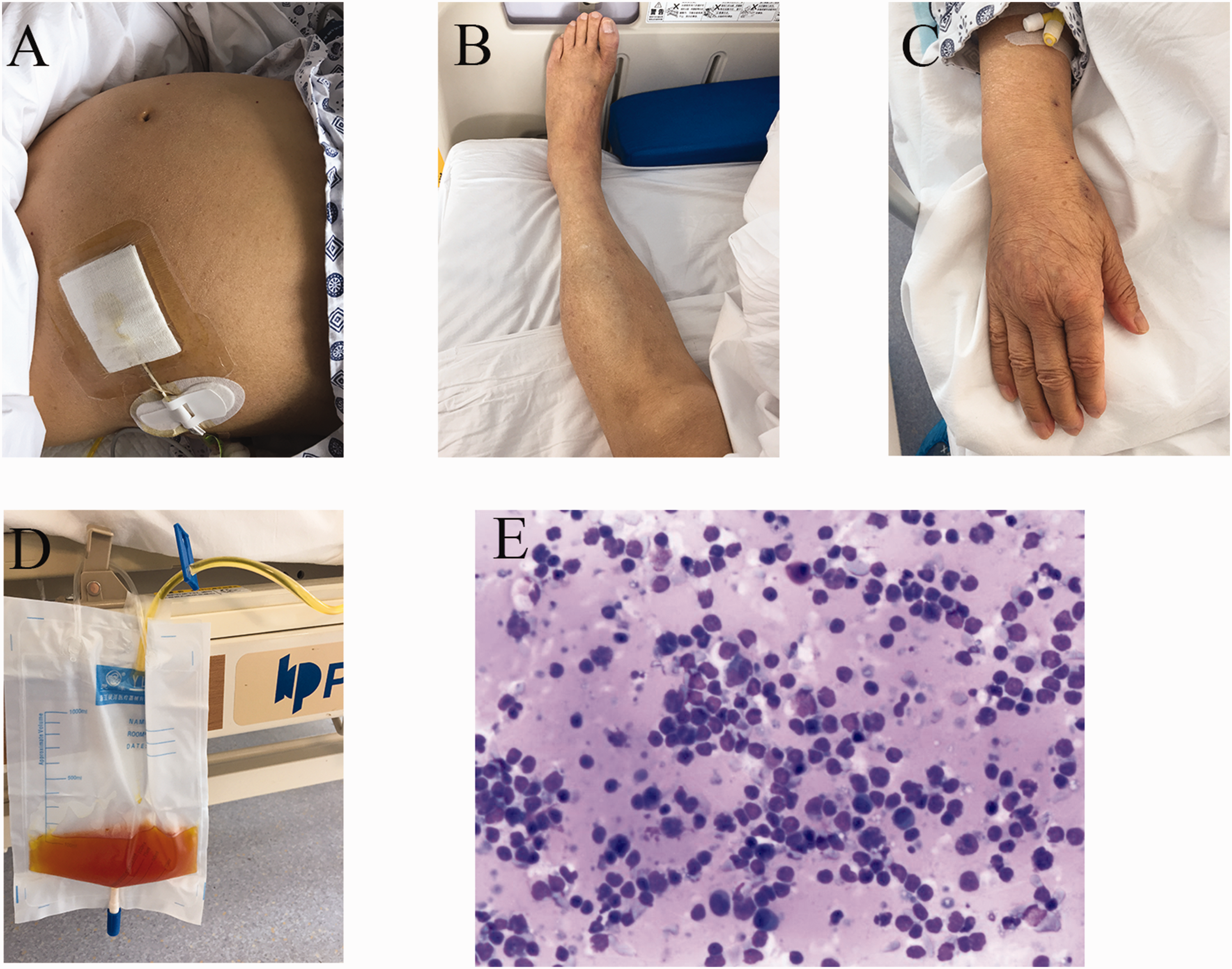
Patient’s physical signs and ascites at Day 7. (a) Massive generalized ascites was seen in the patient’s abdomen. (b) No oedema was seen in the patient’s left leg. (c) Small oedema was seen in the patient’s right arm. (d) Bloody ascites fluid was collected and (e) Cytopathology of the ascites showed non-Hodgkin’s lymphoma cells.
Laboratory test results are summarized in Table 1. Analysis of tumour markers showed increased neuron-specific enolase (NSE; 263 µg/l) and alpha-fetoprotein (AFP; 558 ng/ml). The following were observed: hepatitis B surface antigen (HBsAg) (+); hepatitis B e antibody (HBeAb) (+); Hepatitis B core antibody (HBcAb) (+); hepatitis B virus (HBV)-DNA, 1.3 × 104 copies/ml. The patient’s blood and urine cultures were sterile 72 hours after incubation. Common causes of generalized ascites (i.e., nephrotic syndrome, congestive heart failure, tuberculosis) were ruled out by specific investigations. Abdominal ultrasonography demonstrated, multiple nodules in the liver (the largest was 5.3 × 4.3 cm) and massive ascites (anechoic areas in left hypopharynx, right ventral, left ventral, and pelvic cavity measured 4.2 cm, 4.8 cm, 5.2 cm, 11.3 cm, respectively). A minimal left pleural effusion was also observed. There was no evidence of heart dysfunction or deep vein thrombosis.
Summary of clinical data.

WBC, white blood cell; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TB, total bilirubin; DB, direct bilirubin; γ-GT, γ-glutamyl transpeptidase; ALP, alkaline phosphatase; LDH, lactate dehydrogenase; SAAG, serum-ascites albumin gradient; APTT, Activated partial thromboplastin time; FEU, fibrinogen equivalent units.
Our provisional diagnosis was hepatitis B cirrhosis, spontaneous bacterial peritonitis, primary hepatic cancer, and acute renal failure. Treatments included: fluid infusion; potassium-lowering treatments (i.e., intravenous furosemide, calcium gluconate, sodium bicarbonate); reduced glutathione; antibiotics (i.e., cefoperazone/sulbactam combined with ornidazole). Two days later, the patient developed bilateral eyelid oedema and progressive oliguria. His hypotension (BP, 50–79/39–54 mmHg), ion-unbalance, and acidosis persisted. On the third day of hospitalization, the patient was transferred to the intensive care unit and received continuous monitoring of fluid intake and output. Fluid infusion (i.e., crystalloid solution and colloidal solution 2:1) and exogenous human albumin (20 g/day) were administered. Due to his severe distributive shock, the patient was given a dopamine infusion (10 μg/(kg/min) and noradrenaline infusion (0.4 μg/(kg/min). Oxygen was administered at 4 l/min. However, the shock failed to reverse which made us suspect that our initial diagnosis was incorrect.
On the fourth and sixth day of hospitalization, 1000 ml of ascitic fluid was aspirated by needle puncture and the sample was analysed for biochemistry, adenosine deaminase (ADA), cytokine detection, and culture. The ascites fluid was bright yellow or light red in colour (Figure 2d). The Rivalta test was positive. Repetitive cytopathology analysis of the ascites fluid showed a small number of lymphocytes, neutrophils, mesothelial cells, and many immature cells. Non-Hodgkin's lymphoma was considered (Figure 2e). Flow cytometry of the ascites fluid showed that most of the nucleated cells were lymphocytes, of which 89% were ‘normal’. Of these ‘normal’ lymphocytes, 93% were CD3+ T lymphocytes, and 2% were CD4+/CD8. Of the abnormal cells, approximately 3% were nucleated. Of these nucleated cells, 49% expressed CD19, cCD79, kappa, BCMA, and CD79b. Other abnormal cells expressed, CD19, CD79a, CD20, BCMA, and CD79b.
In the peripheral blood, inflammatory factor levels were as follows: IL-1β, <5 pg/ml; IL-2R, >7500 U/l; IL-6, 26.5 pg/ml, IL-8, 237 pg/ml; IL-10, 98 pg/ml; tumour necrosis factor alpha (TNF-α), 196pg/ml. In the ascites fluid, inflammatory factor levels were as follows: IL-1β, <5 pg/ml; IL-2R, 4983 U/l; IL-6, 1347pg/ml; IL-8, 1068 pg/ml; IL-10, 86 pg/ml; TNF-α, 210 pg/ml. Pleural fluid analysis showed high lactate dehydrogenase (LDH) levels (465 U/l) and total albumin was low (25 g/l) (Table 1).
We reviewed the patient’s circulating tumour DNA (ctDNA) results for B2M and MLL2 and noticed that there had been a marked increase in levels over the past seven months (Figure 3a). The patient had received lenalidomide (25 mg qd) and entecavir (0.5 mg qd). over this period. The latest result (12th March) displayed a marked mutation of MLL2 (50%) and B2M (21%) (Figure 3a). On reviewing the patient’s medical history, we discovered that in the previous year during chemotherapy he had experienced a similar episode of oedema in both lower limbs without an apparent cause. He had been treated successfully with the diuretics, antisterone and furosemide, for a week. Further analysis of the patient’s tumour DNA showed SOCS1 mutations.

(a) Gene mutation rate of MLL2 and B2M by liquid biopsy. (b) Serum albumin concentration during the patient’s hospital admission. Prior to Day 9 the patient received 20 g albumin iv gtt, thereafter he received 40 g albumin iv gtt. ALB, albumin, iv gtt, intravenously guttae and (c) Blood pressure following intravenous dexamethasone infusion (10 mg q12 h) and dopamine (continuous infusion, 5 ml/h).
On the sixth day of hospitalization, immunostaining of the ascites fluid biopsy showed positive results for CD20, CD19, BCL2, BCL6, MUM1, MLL2 and the KI-67 index was nearly 100%. Based on the patient’s medical history, results of the liquid biopsy, B-mode ultrasound, ascites cytology, hypovolemic shock, and hypoalbuminemia (Figure 3b) we modified our diagnosis on the sixth day of hospitalization, as ‘abdominal lymphoma recurrence complicated with CLS’. Intravenous dexamethasone (10 mg q12 h), continuous veno-venous hemodiafiltration (CVVHDF) continuous veno-venous hemofiltration (CVVH) and plasmapheresis were used from the seventh day of hospitalization. 15 Surprisingly, about an hour after the dexamethasone infusion, the patient’s BP increased dramatically from 70/40 mm Hg to 180/97∼220/100 mm Hg accompanied by persistent chest tightness. His high BP was only maintained for about an hour before it fell back to 70/40 mmHg (Figure 3c).
On the eighth day of hospitalization, the patient was transferred to our haematology department to receive chemotherapy (i.e., rituximab 400 mg for two days and etoposide 50 mg for one day followed by 100 mg for three days). The dexamethasone, and hemodiafiltration were continued and dopamine (continuous infusion, 5 ml/h) was also administered. However, his treatment did not correct his low BP or bulk ascites. He developed mild oedema in his neck and lower limbs and his condition deteriorated dramatically with accompanying dyspnoea and myocardial injury. Subsequently, the patient developed multiple organ dysfunction syndrome (heart, renal, liver, pancreas, lung) and died on the fifteenth day of hospitalization.
The reporting of this study conforms to CARE guidelines. 16 Written informed consent was obtained from the patient’s family before publishing his anonymised data. All studies involving human participants are reviewed and approved by Medical Ethics Committee of Wuhan Tong Ji Hospital (TJ-IRB20210605).
Discussion
The syndrome was first described by Clarkson et al. in 1960; 17 they reported a case of a woman who experienced unexplained periodic attacks of marked anasarca and distributive shock for no apparent reason. The researchers concluded that extreme generalized increase in the permeability of her capillaries accounted for the damage to her vital organs and her sudden death. Importantly, the diagnosis of CLS is based on a triad of haemoconcentration, hypotension, and hypoalbuminemia in the absence of other causes of distributive shock. 1
Three phases have been described in the development of CLS (prodromal, leak and post-leak). 18 In the prodromal phase, patients have atypical symptoms such as fatigue, weight gain, and diarrhoea. 18 The leak phase is characterized by a sudden generalized or segmental oedema in the trunk or extremities. 12 The brain, heart, and lungs are not involved in this early stage and so patients are able to preserve consciousness. 7 With the rapid development of vascular leakage, vital organs can be involved and life-threatening complications ensue. The post-leak phase usually starts two to four days after the leak phase; fluid and molecules mobilize from the peripheral cavity into the intravascular space and as a consequence the patient’s BP increases. Organ failure will persist because of their consistent hypoperfusion. 1
In our present case study, we observed that the ascites-serum cytokine gradient was elevated. Therefore, we tried to purify the patient's blood and extract the ascites fluid, in an attempt to reduce the volume of ascites while reducing the content of cytokines in the abdominal cavity. Initially, the treatment proved to be effective; the patient became more lucid than he had been, and his abdominal swelling subsided. However, his tumour recurred and progressed rapidly, and his ascites-serum cytokine gradient continued to increase.
Symptoms of the disease, as well as haematological examination results, manifested themselves gradually. In the first week of admission, the patient’s symptoms were consistent with secondary CLS with hypotension, hypoalbuminemia, coagulation dysfunction, rapid irreversible kidney injury and hypovolemic shock. Mild diffuse oedema and massive ascites were observed. Critical indicators of lymphoma increased rapidly, including LDH, ferritin and β-2 macroglobulin. 19 Importantly, lymphoma cells were discovered among the ascites. Therefore, on the fifth day of admission, following failure of anti-cirrhosis therapy, lymphoma-associated complications were suspected. In addition, the patient had a strong and short-time response to corticosteroid therapy, which was supportive for a diagnosis of CLS. 20 Of the haematologic malignancies associated with CLS, non-Hodgkin lymphoma is the most common (23%), and has been reported to have a high mortality rate from this complication. 8
In this present case, diagnosis and treatment were delayed due to nonspecific symptoms of CLS especially the absence of diffuse oedema in the early stage. Initially, the nephrologist we consulted suggested that the patient's ascites was possibly caused by pre-renal factors and so did not advise dialysis. However, as the patient's condition deteriorated, routine haemodialysis was initiated. On the seventh day of admission, following diagnosis of abdominal lymphoma recurrence complicated with CLS, the patient was treated with corticosteroids, CVVHDF and CVVH. Since there is no standard first-line treatment for CLS, patients with CLS and cancer may recei multiply types of therapy. including corticosteroids and volume replacement. Plasmapheresis was also performed to eliminate inflammatory factors and other harmful substances from the patient's body in the advanced stages of the illness.
IL-6, is often elevated in autoimmune diseases, and low grade-chronic inflammation is believed to hold a key role in the pathogenic mechanisms of cytokine release syndrome (CRS).21,22 Moreover, it has been reported that IL-6 is elevated in CLS.23,24 In an anaplastic lymphoma kinase (ALK) positive large cell lymphoma case complicated with CLS, IL-6 was elevated in the serum and was suggested to account for the fever in the patient. 13 However, these investigators did not test the cytokine level in the serous cavity effusion.
By testing cytokine levels in both serum and ascites, we found an interesting correlation between local IL-6 levels and the extent of oedema; we observed a high IL-6 level in the extremely oedematous abdomen cavity. We hypothesised that due to the short half-life and high variability of IL-6 in serum, the high serum-ascites IL-6 gradient forms following a rapid diffusion between ascites and plasma. 25 The high concentration of inflammatory cytokines in the abdominal cavity will induce activation of vascular endothelial cells and increases capillary permeability. This will be the trigger for CLS. 26 In addition, our patient was found to have SOCS1 gene mutations. The intracellular protein SOCS1 is known to downregulate cytokine signalling by inhibiting the JAK-STAT pathway and SOCS1 gene mutation is capable of facilitating malignant lymphocytes to produce IL-6. 27
Following the rapid progression of the lymphoma and tumour lysis syndrome, cytokine concentrations in the abdominal cavity will continue to increase. As a consequence, bloody ascites will keep on increasing and so the fall in BP will be difficult to correct. Compared with high concentration of IL-6 in the abdominal cavity, other parts of the body will have lower IL-6 concentrations, which may explain why only mild oedema was observed in the chest, neck, and lower limbs. Interestingly, although IL-6 concentrations in serum and ascites are high in cirrhosis and hepatic cancer, they were found to be much higher in this present case study.28,29
Our patient showed several features that are similar to ascites seen in CLS. 1 The unique cytokine microenvironment may provide a new understanding of the mechanism of CLS, particularly for organ-specific cases. For cases of CLS that present with organ specificity, it may be necessary to examine the concentrations of cytokines in the serous cavity effusion to explore potential patterns. Anti-IL-6 therapy has demonstrated effectiveness in treating cytokine release syndrome, 22 but its potential efficacy in treating CLS remains uncertain and requires further investigation. 9 For lymphoma patients, chemotherapy and immunotherapy tend to reach a limit. It may be difficult to differentiate tumour-induced CLS from drug-induced CLS without specific examination. 8 In our case, marked ascites supported the diagnosis of secondary CLS.
In conclusion, we report here, the first case of relapsed lymphoma with CLS presenting as marked ascites. We suggest that the high cytokine gradient between serum and ascitic fluid may have played an important role in its aetiology. With marked progress in the field of lymphoma, differential diagnosis is becoming complex. Our case highlights the importance of evaluating the possibility and pathogenesis of lymphoma recurrence and providing timely therapy in the early stages for patients with refractory ascites and a history of lymphoma. Cytokine examination and gene testing may be a useful tool for early diagnosis and treatment selection. Blood purification should be performed at an early stage if CLS is suspected.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231174313 - Supplemental material for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231174313 for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report by Xu Han, Huanjun Huang, Mingyu Zhang, Yue Wen, Shuhui Wang, Zheng Huang, Dean Tian and Yan Yu in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231174313 - Supplemental material for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605231174313 for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report by Xu Han, Huanjun Huang, Mingyu Zhang, Yue Wen, Shuhui Wang, Zheng Huang, Dean Tian and Yan Yu in Journal of International Medical Research
Research Data
sj-pdf-3-imr-10.1177_03000605231174313 - Research Data for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report
Research Data, sj-pdf-3-imr-10.1177_03000605231174313 for Capillary leak syndrome presenting as refractory ascites in a patient with lymphoma: A rare case report by Xu Han, Huanjun Huang, Mingyu Zhang, Yue Wen, Shuhui Wang, Zheng Huang, Dean Tian and Yan Yu in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank Professor Zhou Jianfeng, chief director of our haematology department and other members of the study team for their assistance throughout the therapy. We would also like to thank the patient and his family for providing consent to use the data in this case report.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This study was supported by research funding from the National Natural Science Foundation of China (81900476 to YY).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
