Abstract
Ascites represents an infrequent sequela of hypothyroidism, manifesting in fewer than 4% of affected individuals. Herein, we delineate a case characterized by profound hypothyroidism accompanied by substantial ascites, further complicated by cardiac insufficiency. A 29-year-old female, previously diagnosed with postradiation hypothyroidism subsequent to a diagnosis of Grave’s disease 11 years prior, presented with exacerbating dyspnoea, abdominal distension, and orthopnea. In January 2024, she was admitted with massive ascites, exhibiting clinical manifestations of both hypothyroidism and cardiac failure. Thyroid function tests were markedly abnormal, with a thyroid-stimulating hormone level of 77.65 mIU/L, triiodothyronine at 2.2 nmol/L, and thyroxine levels below 3.2 pmol/L. Echocardiographic evaluation revealed dilated cardiomyopathy with a significantly reduced systolic (ejection fraction of 25.9%) and diastolic function (E/A ratio of 0.87). Analysis of the ascitic fluid demonstrated a serum-ascites albumin gradient exceeding 1.1 g/L (3 g/L). Ultrasonography of the abdomen ruled out portal hypertension, while computed tomography of the abdomen confirmed extensive ascites without evidence of malignancy. Under the supervision of a specialist, the patient was administered a high dosage of levothyroxine (300 mcg), leading to a significant amelioration in both thyroid function parameters and her ascites. Subsequent thyroid function tests demonstrated a decrease in thyroid-stimulating hormone levels to 11.7 mIU/L and an increase in thyroxine levels to 15.6 pmol/L, indicating a positive response to the thyroid hormone replacement therapy. Subsequent echocardiographic assessment showed improvement in the ejection fraction to 26.9% and diastolic function (E/A ratio of 1.27). Myxedema ascites, though infrequent, is readily amenable to treatment. The serum-ascites albumin gradient exceeding 1.1 g/L may be indicative of hypothyroidism-associated ascites, although the paucity of studies renders it uncertain whether this is a characteristic feature. Further investigation into the etiology, diagnostic criteria, and management strategies for ascites in the context of hypothyroidism is warranted.
Introduction
The manifestation of ascites as an initial symptom of hypothyroidism is a rarity, observed in a mere subset of less than 4% of individuals diagnosed with hypothyroidism. Notwithstanding its infrequency, the therapeutic intervention targeting hypothyroidism invariably results in a prompt amelioration of ascitic symptoms. 1 The preponderance of insights into the nexus between ascites and hypothyroidism is extrapolated from case reports disseminated globally.2–6
The pathophysiological underpinnings precipitating the accumulation of ascitic fluid in patients afflicted with myxedema remain nebulous. 5 Predominantly, two hypotheses have been postulated. The inaugural hypothesis conjectures that diminished concentrations of circulating thyroid hormones engender an augmented extravasation of plasma proteins, attributable to aberrant capillary permeability coupled with an insufficiency in compensatory lymphatic flow and protein reabsorption. 7 Conversely, the secondary hypothesis suggests an accumulation of hyaluronic acid within the dermal layers, culminating in edema through a direct absorptive mechanism. 8 Hyaluronic acid is only found in small quantities in patients with hypothyroidism-induced ascites and cannot be held entirely responsible for exerting the required hygroscopic effect. However, it can form hyaluronic acid-albumin complexes that prevent lymphatic drainage of extravasated albumin thus causing ascites.5,9 Ancillary research hypothesizes a diminution in renal free water clearance, ascribed to an overproduction of antidiuretic hormone in the milieu of hypothyroidism, or an escalation in capillary permeability contingent upon depleted levels of thyroxine (T4). In addition, reduced nitric oxide levels cause endothelial dysfunction through oxidative stress, leading to the activation of inflammatory cells that release substances increasing capillary permeability. Additionally, elevated Vascular Endothelial Growth Factor (VEGF) levels contribute to increased extravasation of plasma proteins due to abnormal capillary permeability. Elevated VEGF levels have been observed in hypothyroid patients, with a decline to normal values following thyroid replacement therapy.10,11
Overt hypothyroidism is prevalent in approximately 3% of the adult female demographic and is implicated in the augmentation of systemic vascular resistance, a decrement in cardiac contractility, a reduction in cardiac output, and the expedited progression of atherosclerosis and coronary artery disease. 12 Such cardiovascular detriments may be ascribed to elevated incidences of hypercholesterolemia and diastolic hypertension within this patient cohort. A scrutiny of cross-sectional studies elucidates that an estimated 30% of individuals suffering from congestive heart failure exhibit diminished levels of triiodothyronine (T3).13,14
In this case report, we delineate a scarcely encountered instance of ascites in the context of severe hypothyroidism, further complicated by cardiac failure.
Case presentation
A 29-year-old female, previously diagnosed with postradiation hypothyroidism following a diagnosis of Grave’s disease 11 years prior, experienced a lapse in medical follow-up during the COVID-19 pandemic in 2020. She was placed on thyroxine; however, due to noncompliance and lost to follow-up this led to worsening of symptoms. She re-engaged with medical services at a provincial hospital in October 2023, presenting with symptoms indicative of hypothyroidism, including lethargy, dryness of the skin and hair, without any significant findings on systemic enquiry. Initiation of levothyroxine therapy at a dose of 75 mcg was undertaken, with recommendations for subsequent follow-up.
By January 2024, she presented with severe ascites and clinical manifestations consistent with congestive cardiac failure, including exacerbated dyspnoea and abdominal discomfort. She was classified as having New York Heart Association 15 Class 4 dyspnoea, experienced orthopnea requiring the support of two pillows and suffered from paroxysmal nocturnal dyspnoea.
The patient’s social history revealed unemployment with no significant occupational health exposures. Her medical history was notable solely for hypothyroidism, with no other comorbid conditions reported. She had a significant smoking history of 18 pack-years and engaged in social alcohol consumption. Her obstetric history was unremarkable, with one uncomplicated normal vaginal delivery, no history of miscarriages, and a recent year of amenorrhea without the use of hormonal contraceptives. A familial history of primary hypothyroidism was noted in her maternal grandmother, with no reported family history of cancer.
Physical examination identified a young female with pronounced ascites. The presence of bilateral nonpitting edema extending to the tibial region was suggestive of myxedema, without evidence of periorbital swelling. Bedside urinalysis did not indicate infection or proteinuria. Thyroid enlargement was not observed. Notable skin findings included dryness with desquamation, particularly on the feet and hands. Abdominal examination revealed superficial veins with gross abdominal distension. There was a positive fluid thrill and shifting dullness to suggest ascites. It was difficult to palpate for organomegaly and other masses due to abdominal distension. Overall, there were no other features of chronic liver failure and portal hypertension, apart from the ascites. Cardiovascular examination revealed an elevated jugular venous pressure, normophonic heart sounds, and a laterally displaced apex beat to the 6th intercostal space lateral to the midclavicular line, without additional heart sounds, heaves, or murmurs. Blood pressure and pulse was within normal limits. Other systemic findings were unremarkable.
Clinical photographs illustrated the patient’s hypothyroid features: dry hair with alopecic patches (Figure 1), nonpitting edema with desquamation on the feet (Figure 2), dryness of the hands (Figure 2), and extensive abdominal distension with visible superficial veins due to massive ascites (Figure 3). The extensive nature of the ascites precluded the assessment for hepatosplenomegaly. No signs of liver disease or portal hypertension were evident beyond the ascites. Neurological evaluation found the patient to be alert with normal muscle tone in all limbs with slow-relaxing reflexes (Woltman sign), further corroborating the clinical diagnosis of hypothyroidism. After 3 months of thyroid hormone replacement, there was marked improvement in signs (Figures 4–6).

Dry hair and areas of alopecia.

Brittle nails, dry skin.

Massive ascites.
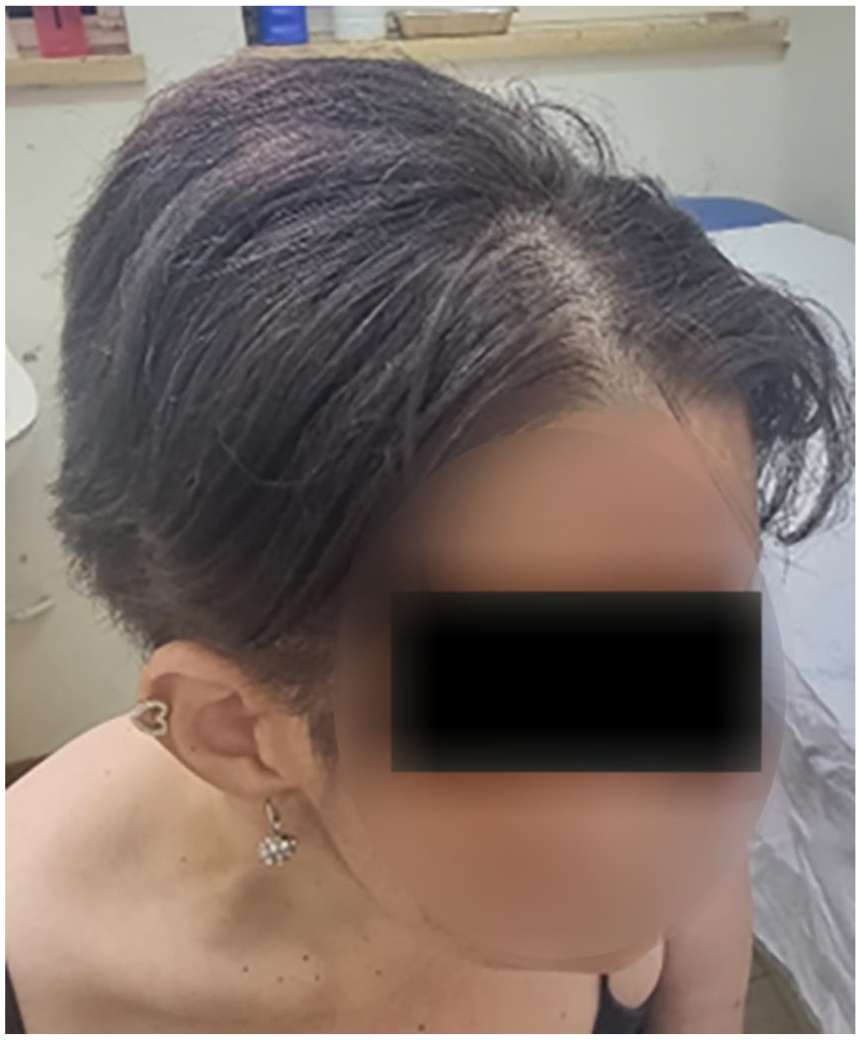
Less brittle hair.
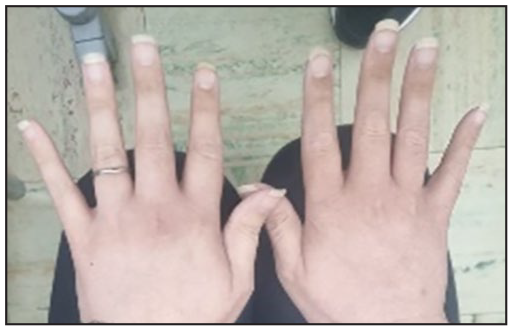
Nails grown, less dry skin.

Resolution of ascites.
An abdominal ultrasound examination of the patient disclosed the presence of significant complex ascites, with an absence of hepatosplenomegaly or signs indicative of portal hypertension or Budd–Chiari syndrome, although the extensive ascites impeded optimal visualization. Subsequent computer tomography scanning of the abdomen corroborated the absence of neoplastic masses or malignancy-associated features, with the notable presence of massive ascites documented. A liver span of 14 cm was noted with normal echotexture and no evidence of cirrhosis (Figure 7).

Computer tomography abdomen depicting ascitic fluid in abdomen on thyroxine treatment.
The patient’s diagnostic workup included comprehensive thyroid function tests and ascitic fluid analysis, the results of which are consolidated in Tables 1 and 2, respectively. Additional laboratory investigations encompassing urea and electrolytes, liver function tests, full blood count, international normalized ratio, and an autoimmune screen, including anti-nuclear antibodies were all reported within normal limits. Screening for hepatitis A, B, and C yielded negative results, indicating no viral hepatitis infection.
Thyroid function test results.

Results not performed.
Ascitic fluid results and SAAG analysis.

Echocardiographic evaluation provided further insights into the patient’s cardiac status, with findings summarized in Table 3.
Echocardiogram report.

Management and outcome
Therapeutic paracentesis was performed to alleviate severe discomfort associated with the ascites, with no more than 2 L of fluid being evacuated at any one session to mitigate the risk of hypotension. We drained 2-L of peritoneal fluid on two separate occasions over a 2-week period. Pharmacological management was initiated, comprising an angiotensin-converting enzyme inhibitor (Enalapril, 5 mg bi-daily), a mineralocorticoid receptor antagonist (Spironolactone, 25 mg daily), and a loop diuretic (Furosemide, 40 mg bi-daily). This was the highest patient tolerated doses. Despite these interventions, there was no appreciable resolution of the ascites through diuretic therapy. No albumin was administered to patient.
Subsequently, the patient was administered the maximal therapeutic dosage of Levothyroxine, 200 mcg daily, which resulted in only marginal amelioration of thyroid hormone levels after 1 month of treatment. Following consultation with specialist endocrinology services, the levothyroxine dosage was escalated to 300 mcg daily. This adjustment led to notable clinical improvement in both the ascitic accumulation and thyroid function parameters by the February 16, 2024. By April 2024, she displayed resolution of ascites and other symptoms of hypothyroidism. Subsequent echocardiographic assessment indicated a more pronounced enhancement in diastolic compared to systolic ventricular functions, with an increase in the ejection fraction to 26.9% and an E/A ratio of 1.27.
Discussion
A subset of up to 4% of individuals with significantly uncontrolled hypothyroidism may manifest with abdominal ascites. 16 Ascitic fluid associated with portal hypertension is characterized by a low total albumin level, with the Serum-Ascites Albumin Gradient (SAAG) exceeding 1.1 g/dL, indicative of a high gradient. 17 This high SAAG is commonly observed in patients with liver cirrhosis and congestive heart failure. Conversely, a SAAG of less than 1.1 g/dL, denoting a low gradient, typically suggests that the ascites is not attributable to portal hypertension. Low SAAG values are observed in conditions such as peritoneal malignancies, tuberculous peritonitis, pyogenic peritonitis, and pancreatic ascites. It has been posited, based on a review of eight patients, that the SAAG may surpass 1.1 g/dL in cases of myxedema ascites. 18 In the case under discussion, the SAAG was greater than 1.1 g/dL, aligning with documented instances of myxedema ascites. However, the assertion that a high SAAG is a characteristic feature of this disease remains tentative due to the scarcity of data. Our patient had a total fluid protein of 4.8 g/dL, which is considered from studies to be a prominent feature of myxedema ascites (>2.5 g/dL). 5
Hypothyroidism’s capacity to induce ascites, frequently associated with a high SAAG, diverges from the typical etiology linked to portal hypertension. Instead, it results in a transudative type of ascites, with the high SAAG reflecting the systemic influence of hypothyroidism on vascular permeability and fluid homeostasis, rather than direct portal hypertension. 19 The pathogenesis of ascites in hypothyroidism is multifactorial, encompassing increased capillary permeability, reduced lymphatic drainage, and a disrupted balance between hydrostatic and oncotic pressures, cumulatively leading to fluid accumulation within the peritoneal cavity. Despite the transudative nature of the ascites as indicated by the high SAAG, the primary etiology does not involve increased portal pressure but rather the metabolic and physiological repercussions of hypothyroidism on the body’s fluid management mechanisms. 20
Heart failure and hypothyroidism
The causal relationship between hypothyroidism and the development of dilated cardiomyopathy (DCM) in this case remains ambiguous. There is substantial evidence indicating that diastolic dysfunction is a prevalent abnormality in hypothyroidism.21,22 The most consistently observed cardiac anomaly in patients with overt hypothyroidism is the impairment of left ventricular (LV) diastolic function, which is characterized by delayed myocardial relaxation and compromised early ventricular filling.23–25 Nonetheless, the manifestation of hypothyroidism as cardiomyopathy with reduced LV systolic function is relatively rare. To date, only a limited number of case reports have documented hypothyroidism-induced DCM characterized by decreased LV systolic function.
Since the inaugural documentation of DCM in four hypothyroid patients in 1918, 26 only a handful of similar cases have been reported in the literature. Bezdah et al. 27 described a patient with severe hypothyroidism accompanied by DCM complicated by heart failure, which showed improvement following levothyroxine therapy. They advocated for the systematic consideration of hypothyroidism as a potential underlying cause upon the diagnosis of DCM. Ladenson et al. 28 identified reversible alterations in myocardial gene expression in a young man with DCM and hypothyroidism, further supporting the link between these conditions. Similarly, Khochtali et al. 29 presented two case studies that demonstrated reversible DCM caused by hypothyroidism, underscoring the potential for cardiac recovery with appropriate thyroid hormone replacement therapy. These findings collectively suggest that hypothyroidism, though an uncommon etiology for DCM, should be systematically evaluated as a contributing factor in patients diagnosed with this form of cardiomyopathy. Noteworthy, diastolic heart failure was strongly associated with hypothyroidism and was more prevalent in women as depicted in this case report. 30
There is limited data on treatment outcomes with thyroxine in patients with hypothyroidism related systolic dysfunction; however, diastolic dysfunction has been well reported on. An impairment of diastolic function is a common finding in many cardiac diseases, and it often precedes and causes systolic dysfunction. It has been documented that 30%–40% of heart failure syndromes are secondary to impaired diastolic function. Patients with overt hypothyroidism have bradycardia, decreased ventricular filling, and decreased cardiac contractility, which leads to decreased cardiac output leading to systolic compromise. 31 A study found in patients with subclinical hypothyroidism diastolic function were normalized by L-T4 therapy, whereas systolic function improved minimally after 6 months of therapy showing the lag time in recovery in systolic dysfunction. 32 A study in patients with hypothyroidism in 2001 showed that LV diastolic function is enhanced by thyroxine therapy during a rather short follow-up period; however, systolic dysfunction was not analyzed. 33 A case report on a patient with hypothyroid cardiomyopathy in 2018 found marked improvement in ejection fraction (EF) after 5 months of thyroid hormone replacement. 23 Evidence is conflicting regarding outcomes with systolic function; however, the improvement of diastolic function after thyroid hormone replacement in our patient is well established in literature. We postulate that the limited improvement of systolic function in our patient could be attributed to duration of illness and pathophysiology associated with quicker improvement in diastolic function rather than systolic function.
Differential diagnosis
The differential diagnosis for ascites encompasses a wide array of conditions, among which myxedema ascites should be contemplated in patients presenting with ascites alongside a documented history of hypothyroidism. The definitive diagnosis of myxedema ascites is substantiated through the clinical amelioration of ascites following thyroid hormone replacement therapy, culminating in the complete resolution of the ascitic accumulation. In the case under discussion, cardiac failure was initially considered a contributing factor to the ascites; however, the patient’s lack of response to diuretic therapy alone posited hypothyroidism as a more plausible etiology for the massive ascites observed.
The potential for malignancy and infectious peritonitis as underlying causes was methodically excluded following negative cytological and culture results from the ascitic fluid analysis. Furthermore, abdominal ultrasonography failed to demonstrate any indications of portal hypertension, reinforcing the absence of this common ascites etiology. This comprehensive diagnostic approach underscores the importance of considering hypothyroidism, specifically myxedema ascites, in the differential diagnosis of ascites, particularly when conventional causes such as cardiac failure, malignancy, and infectious peritonitis have been ruled out and when the patient exhibits a history of hypothyroidism.
Conclusion
Myxedema ascites, while infrequent, is acknowledged for its responsiveness to treatment. The administration of thyroid hormone replacement therapy has been documented to result in the complete remission of ascites. Despite the paucity of comprehensive data on management protocols for these instances, anecdotal evidence suggests a consensus on the efficacy of treating the underlying hypothyroidism, which consequentially leads to the resolution of ascites. In the presented case, the implementation of high-dose thyroid hormone replacement therapy, as advised by a specialist, yielded a positive outcome.
Given the limited understanding and the rarity of myxedema ascites, there is a pressing need for further research into its etiology, diagnostic criteria, and management strategies. Such investigations are essential to enhance the clinical approach toward patients presenting with ascites in the context of hypothyroidism, ensuring timely and effective treatment modalities.
Footnotes
Author contributions
K.D.K. acquired and interpreted the data and drafted the manuscript. S.P. and N.S. critically revised the manuscript. S.P. provided final review and approval of the manuscript.
Data availability
All the data supporting our findings are contained within the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this article are of the authors scientific opinion and not an official position of the institution or funder.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
