Abstract
Background:
Bacterial peritonitis (BP) in patients with gastrointestinal (GI) cancer has been poorly described, and its prevalence is unknown.
Objectives:
This study aimed to evaluate in patients with both GI cancer and ascites the prevalence of BP, associated features, mechanisms, prognosis, and the diagnostic performance of neutrophil count in ascites.
Design:
A retrospective, multicenter, observational study.
Methods:
All patients with GI cancer and ascites who underwent at least one paracentesis sample analyzed for bacteriology over a 1-year period were included. BP was defined by a positive ascites culture combined with clinical and/or biological signs compatible with infection. Secondary BP was defined as BP related to a direct intra-abdominal infectious source.
Results:
Five hundred fifty-seven ascites from 208 patients included were analyzed. Twenty-eight patients had at least one episode of BP and the annual prevalence rate of BP was 14%. Among the 28 patients with BP, 19 (65%) patients had proven secondary BP and 17 (59%) patients had multi-microbial BP, mainly due to Enterobacterales. A neutrophil count greater than 110/mm3 in ascites had negative and positive predictive values of 96% and 39%, respectively, for the diagnosis of BP. The median survival of patients with BP was 10 days (interquartile range 6–40) after the diagnosis.
Conclusion:
BP is not rare in patients with GI cancer and is associated with a poor short-term prognosis. When a patient with GI cancer is diagnosed with BP, a secondary cause should be sought. Further studies are needed to better define the best management of these patients.
Introduction
Ascites is a common symptom in digestive diseases and therefore in gastrointestinal (GI) cancer. In Europe, the frequent causes of ascites are cirrhosis, GI and gynecologic cancers and more rarely, heart failure and peritoneal tuberculosis. 1 Up to 15% of patients with GI cancer will develop ascites, which is associated with a poor prognosis and a median survival of 2–6 months depending on the site of the primary cancer.2,3 As GI cancers are frequent (one cancer out of four), the management of ascites associated with GI cancers and its complications is a challenge for oncologists (Source: international agency for Research on Cancer, https://gco.iarc.fr/stories/gastro-intestinal/en).
The pathophysiology of malignant ascites is imperfectly understood. Several factors contribute to its development, particularly in peritoneal carcinomatosis, such as obstruction of lymphatic drainage by tumor extension, altered vascular permeability and fluid retention secondary to activation of the renin–angiotensin–aldosterone system.2,4,5 Patients with GI cancer may also develop portal hypertension, favored by portal vein thrombosis, major hepatic invasion or chemotherapy toxicity (i.e. sinusoidal obstruction syndrome or regenerative nodular hyperplasia caused by oxaliplatin), which may also contribute to the development of ascites.6–11
One of the main complications of ascites is bacterial peritonitis (BP). BP can be either spontaneous, linked to bacterial translocation from the GI tract to the mesenteric lymph nodes, or secondary to an intra-abdominal infectious source (e.g. perforation in the GI tract or abscess). 12 Spontaneous BP linked to bacterial translocation from the GI tract accounts for the vast majority of BP in patients with cirrhosis and its diagnosis can be challenging, as ascites culture is negative in over 50% of cases. 13 Therefore, the diagnosis of BP is based on a neutrophil count ⩾250/mm3 in an ascites sample in cirrhotic patients.13,14 In contrast to BP in the setting of cirrhosis, BP in patients with malignant ascites has been described in only few case reports, and little is known about its prevalence and consequences.7,15–18 ‘Most of the reported cases concerned BP in patients with GI cancer, with few cases in other cancers. The aims of this multicenter retrospective study were therefore to describe (i) the prevalence of BP in patients with GI cancer, (ii) the mechanisms of BP and the bacteria involved in patients with GI cancer, (iii) the pre-defined factors associated with the occurrence of BP, (iv) the prognostic of patients with BP, and (v) the diagnostic accuracy of ascites neutrophil count for the diagnosis of BP in patients with GI cancer.
Materials and methods
Selection of patients: Inclusion and exclusion criteria
A 1-year (2018) multicenter retrospective study was conducted in five tertiary-care hospitals affiliated with the Assistance Publique des Hôpitaux de Paris in France (Hôpital Beaujon, Hôpital Cochin, Hôpital Européen Georges Pompidou, Hôpital La Pitié Salpêtrière, Hôpital Saint-Antoine). To ensure completeness of data collection, all patients who had undergone paracentesis during the inclusion period were identified using each center’s bacteriology laboratory database.
All patients over 18 years of age who had at least one paracentesis with an ascites sample sent to the bacteriology laboratory were identified, and those with histologically proven GI cancer (colorectal, pancreatic, biliary tract, metastatic neuroendocrine tumor, stomach, adenocarcinoma of unknown origin with histology compatible with a digestive origin) were included.
Patients were excluded from the study if they had (i) confirmed or histologically proven or a suspected cirrhosis on imaging (i.e. liver morphologic alterations and features of portal hypertension), (ii) hepatocellular carcinoma, (iii) another cause of ascites [heart failure, tuberculosis, postoperative complications, benign pancreatic disease (such as fistulae or recent acute pancreatitis)], (iv) abdominal surgery during the month prior to paracentesis, (v) non-GI cancer, (vi) unavailable clinico-biological data.
Definitions of BP in GI cancers
Currently, there is no gold standard for the diagnosis of BP in patients with GI cancer, and the neutrophil count in ascites has not been validated or even evaluated in this population. Based on the princeps studies defining BP in patients with cirrhosis and the indication for antibiotics in patients with cirrhosis who have an ascites sample with a neutrophil count below 250/mm3 and a positive ascites culture associated with signs of systemic inflammation (European Association for the Study of the Liver guidelines 13 ), the diagnosis of BP was retained in case of positive culture in ascites associated with clinical (fever, abdominal pain) and/or biological (biological inflammatory syndrome) signs compatible with infection in patients with GI cancer.13,19
Secondary BP was defined as BP related to a direct intra-abdominal infectious source such as an abscess, GI tract perforation or a systemic one (e.g. cholangitis). 12 As it is difficult to conclude that BP is spontaneous (i.e. related to bacterial translocation from the GI tract) in malignant ascites, when a PB was not clearly identified as secondary, it was considered a BP of unknown mechanism. ‘Contaminated’ ascites was defined as the presence of commensal skin bacteria in ascites in the absence of clinical and/or biological signs consistent with ongoing infection. Persistent BP was defined as persistent bacterial culture on several consecutive paracentesis.
Data collection
Patients’ data were collected from medical records and stored in a protected anonymous database. Data included: inclusion center, general data (age, gender, medical, and surgical history), cancer data (date of diagnosis, current treatment, primary site), and laboratory data (including ascites leukocyte and neutrophil counts). The appearance of ascites was defined by the first mention in the patient’s record of clinical ascites and/or the diagnosis of ascites on computed tomography (CT) scan. Imaging data were collected from the most recent computed tomographic scan (CT scan) in relation to the ascites paracentesis. A second centralized imaging assessment of CT scans was performed by an experienced abdominal radiologist to assess signs of portal hypertension (i.e. portosystemic shunts and splenomegaly), peritoneal carcinomatosis, liver dysmorphia, and signs of intra-abdominal infection, when CT-scan images were available. Major liver involvement was considered invasion of both hepatic lobes by cohesive metastases. Patients were considered malnourished if they had a weight loss of >5% in 1 month or >10% in 6 months or a plasma albumin level <3.5 g/dL.
Outcomes
The primary outcome was to determine the annual prevalence of BP in patients with GI cancer with at least one ascites paracentesis.
Secondary outcomes were the evaluation of the mechanisms and bacteria involved in BP, factors associated with BP, performance of ascites neutrophil count for diagnosis of BP in GI cancer ascites and survival time after diagnosis of BP.
Statistical analysis
Qualitative variables were expressed as absolute and relative (percentage) frequencies and compared using the Chi-square or the Fisher’s test, as appropriate. Quantitative variables were expressed as median [interquartile range (IQR)] and compared using the Mann–Whitney test or Kruskal–Wallis tests, as appropriate. The Youden index for determining the best threshold for neutrophil count in ascites to diagnose BP was calculated using the coordinates of the receiver operator characteristic (ROC) curve. The positive and negative predictive values of the neutrophil count threshold were calculated using the prevalence rate of BP determined in the study.
Follow-up time was defined as the period of time between the first ascites paracentesis and death or last news. When patients were still alive 30 days after the first paracentesis, they were censored. The cumulative incidence of death was assessed using the Kaplan–Meier method and compared using the log-rank test. All tests were two-tailed and performed with a first-species risk of 0.05. Statistical analyses were performed using SPSS version 27.0 software (SPSS Inc., Chicago, IL, USA) and Prism® (Version 9, Graphpad™, Boston, USA).
Results
Characteristics of patients with GI cancer and ascites
During the inclusion period, 890 patients had a paracentesis with the ascites sample sent to the laboratory for bacteriological analysis. Of these, 646 were excluded because the cause of ascites was unrelated to a GI cancer, and 36 because of an underlying cirrhosis, recent abdominal surgery, or missing data (Figure 1: flow chart). A total of 208 patients with GI cancer and ascites with 557 ascites samples were included. One hundred six (51%) patients were male, and median age was 64 years (IQR 57–71). The most frequent cancer was pancreatic adenocarcinoma (55% of patients). The median Charlson score was 8 (IQR 7–9). Eighty (39%) patients were still receiving systemic chemotherapy at the time of paracentesis. The median number of paracenteses per patient was 2 (IQR 1–3) and during follow up, 125 patients (60%) had two or more paracenteses.

Flow chart.
Among the 198 patients who had recent CT-scan assessment prior to ascites paracentesis, 118 (60%) had liver metastases, of whom 38 (19%) had major liver involvement, and 109 (55%) and 146 (74%) had evidence of portal hypertension and visible peritoneal carcinomatosis, respectively (Table 1). Regarding the indication for paracentesis, 457 (82%) were performed for therapeutic reasons (i.e. large volume paracentesis), 62 (11%) for diagnostic reasons, including 34 (6%) to explore the cause of ascites, 18 (3%) for suspected BP, and 10 (2%) to control ascites after the treatment of BP. The indication for paracentesis was not available in 38 (7%) procedures. Median albumin and C-reactive protein (CRP) concentrations were 2.5 g/dL (IQR 2.1–2.8) and 83 mg/L (IQR 43–148), respectively. Median ascites leukocyte and neutrophil count were 160/mm3 (IQR 70–405) and 25/mm3 (IQR 7–127), respectively (Table 2). Neutrophil count in ascites exceeded 250/mm3 in 13% of ascites. Protein concentration in ascites was 1.9 g/dL (IQR 1.2–2.9) and exceeded 2.5 g/dL in 37% of ascites samples. Protein concentration in ascites was significantly lower in patients with signs of portal hypertension at CT-scan evaluation than in those without [1.5 g/dL (IQR 1.1–2.4) versus 2.7 g/dL (IQR 1.4–3.8), p < 0.0001].
Patients’ characteristics.
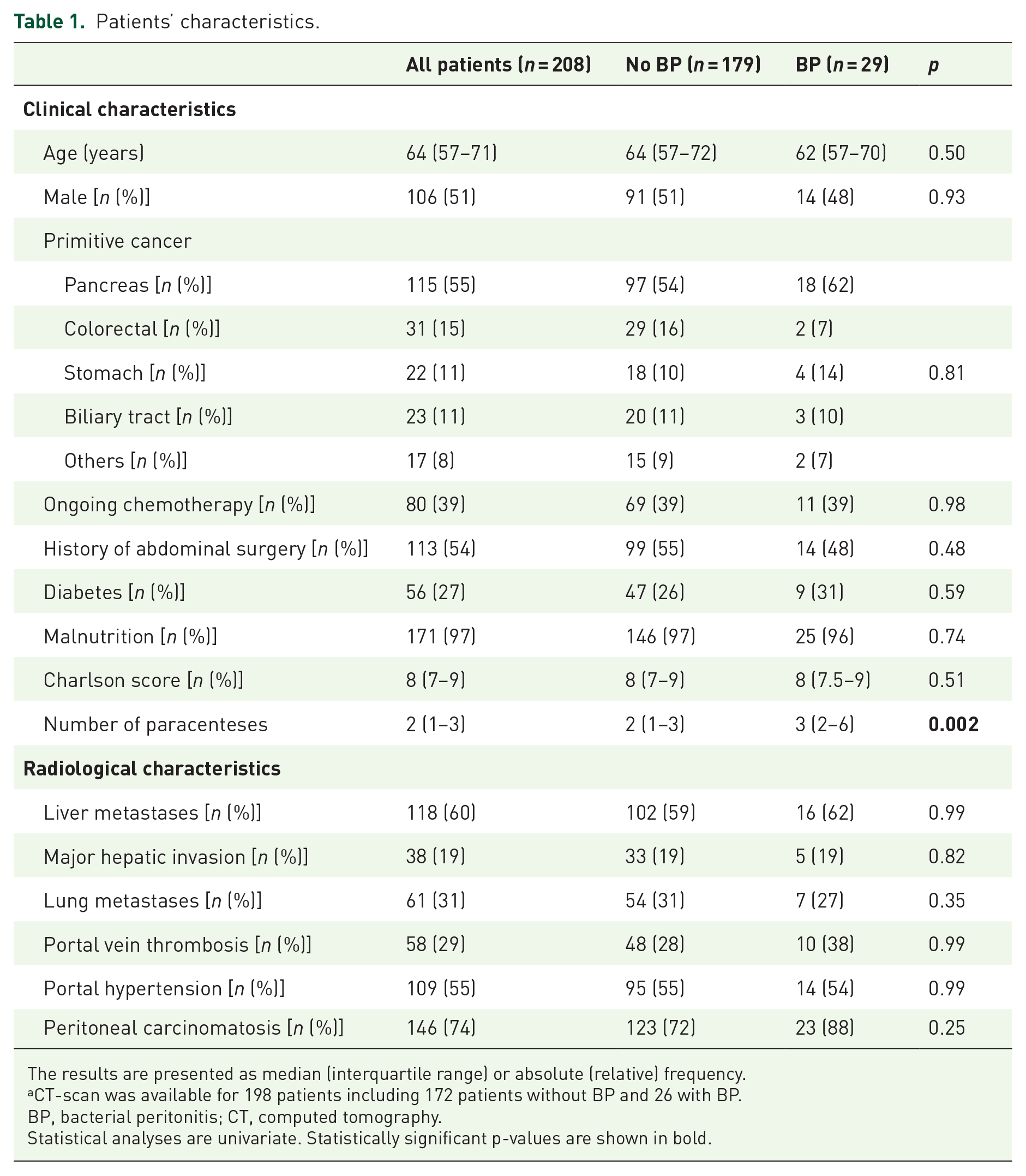
The results are presented as median (interquartile range) or absolute (relative) frequency.
CT-scan was available for 198 patients including 172 patients without BP and 26 with BP.
BP, bacterial peritonitis; CT, computed tomography.
Statistical analyses are univariate. Statistically significant p-values are shown in bold.
Laboratory characteristics of patients at the time of paracentesis.

The results are presented as median (interquartile range) or absolute (relative) frequency.
BP, bacterial peritonitis; CRP, C-reactive protein; LDH, lactate dehydrogenase.
Statistically significant p-values are shown in bold.
Primary outcome: Prevalence of BP in patients with GI cancer
Twenty-nine patients had at least one BP, corresponding to an annual prevalence rate of BP in patients with GI cancer of 14%. Among the 557 ascites samples, 42 (8%) showed a BP, and 19 (3%) were contaminated by skin germs.
Secondary outcomes
Mechanisms of BP and bacteria involved in patients with GI cancer
The mechanism of BP was considered to be secondary in 19 (65%) patients and unknown in 10 (35%) patients. Eight (28%) patients had persistent BP on consecutive paracenteses. The causes of secondary BP were cholangitis (n = 5; 26%), liver abscesses (n = 4; 21%), GI obstruction (n = 5; 26%), and other (acute pancreatitis, appendicitis, mesenteric ischemia, or digestive perforation) (n = 5; 26%).
Considering only the first episode of BP in patients with recurrent ascites, 17/29 (59%), patients had multimicrobial infection. Eleven (58%) secondary BP and 6 (60%) unknown BP were multimicrobial (p > 0.999). The most frequently identified types of bacteria were Enterobacterales (Escherichia coli and cephalosporinase-producing Enterobacterales; 66%), Streptococci spp. (41%), and Enterococcus spp. (21%) (Table 3). Information on antibiotic therapy was available for 24 (83%) patients, all but one of whom received antibiotics. Of the 29 patients who had BP, 8 had concomitant bacteremia. Of these eight patients, four had bacteremia with a germ identical to that identified in ascites, while four had bacteremia with a germ different from that identified in ascites. The most frequently administered antibiotics were third-generation cephalosporins in nine (38%) patients, piperacillin–tazobactam in five (21%) patients, and vancomycin in four (17%) patients, either alone or in combination. Data on clinico-biological course after the start of treatment were available in 18/24 patients; only 6 patients improved (clinically and/or on biological signs). A follow-up paracentesis was performed 48 h after the first one in five patients and showed a negative culture and a drop in neutrophil count >50%. All five patients recovered from their BP.
Organisms (n = 58) isolated from 29 patients with bacterial peritonitis.

The results are presented as absolute (relative) frequency.
Enterobacter cloacae (5), Morganella morganii (2), Klebsiella aerogenes (1).
Klebsiella pneumoniae (2), Klebsiella variicola (1), Raoultella spp. (1), Proteus vulgaris (1).
Streptococcus oralis (2), Streptococcus parasanguinis (2), Streptococcus salivarius (2), Streptococcus mitis (1), Streptococcus sanguinis (1), Streptococcus vestibularis (1).
Streptococcus anginosus (3), Streptococcus constellatus (2).
Clinical features associated with BP and GI cancer
Patients with BP had significantly more paracenteses [3 (IQR 2–6) versus 2 (IQR 1–3); p = 0.002] (Table 1). At the time of paracentesis, diarrhea was more frequently observed in BP (8/18 versus 42/263; p = 0.003) while fever and abdominal pain were not significantly more frequent (data not shown). Blood albumin concentration was lower at the time of paracentesis in patients with BP (p = 0.0001) despite these patients were not more malnourished (p = 0.74). Primary cancer site, history of abdominal surgery, presence of liver metastases or major liver invasion, presence of portal hypertension or peritoneal carcinomatosis were not different between patients with and without BP.
Biological features associated with BP and GI cancer
Leukocytes, neutrophils, and lymphocytes count, and the lactate dehydrogenase concentration in ascites were significantly higher in presence of BP (p < 0.01) (Table 2). CRP level was higher at the time of paracentesis in BP [154 mg/L (IQR 74–221) versus 76 (IQR 42–131); p = 0.001], while leukocyte count was not [7.7 G/L (IQR 3.4–16) versus 8.5 (IQR 5.3–13.5); p = 0.41]. BP was not more frequent in cases of protein concentration <1 g/dL in ascites (3/30 versus 51/394; p = 0.64). Hemorrhagic ascites was more frequent in case of BP [7 (18%) versus 38 (9%); p = 0.035].
Diagnostic performance of neutrophil count in ascites for the diagnosis of BP in malignant ascites
The neutrophil count in ascites was significantly higher in patients with BP than in those without [969/mm3 (IQR 134–3563) versus 22/mm3 (IQR 7–83); p < 0.0001] (Table 2). Neutrophil count was >250/mm3 in 20 (61%) ascites with BP versus 41 (10%) ascites without BP (p < 0.0001). Calculating the Youden index from ROC curve coordinates, the best threshold for neutrophil count in ascites to discriminate BP from ascites without BP was 110/mm3 (sensibility 81%, specificity 79%, area under the curve 0.851) (Figure 2). Using a prevalence of 14% for BP, a threshold of 110/mm3 for neutrophil count in ascites had a positive predictive value and negative predictive value of 39% and 96%, respectively, for the diagnosis of BP. There was no significant difference in survival time after the first paracentesis for ascites between patients who had more or less than 110 neutrophils/mm3 in ascites, either in the overall cohort (p = 0.3) or in the cohort of patients without BP (p = 0.58) (Supplemental Figures 1 and 2).
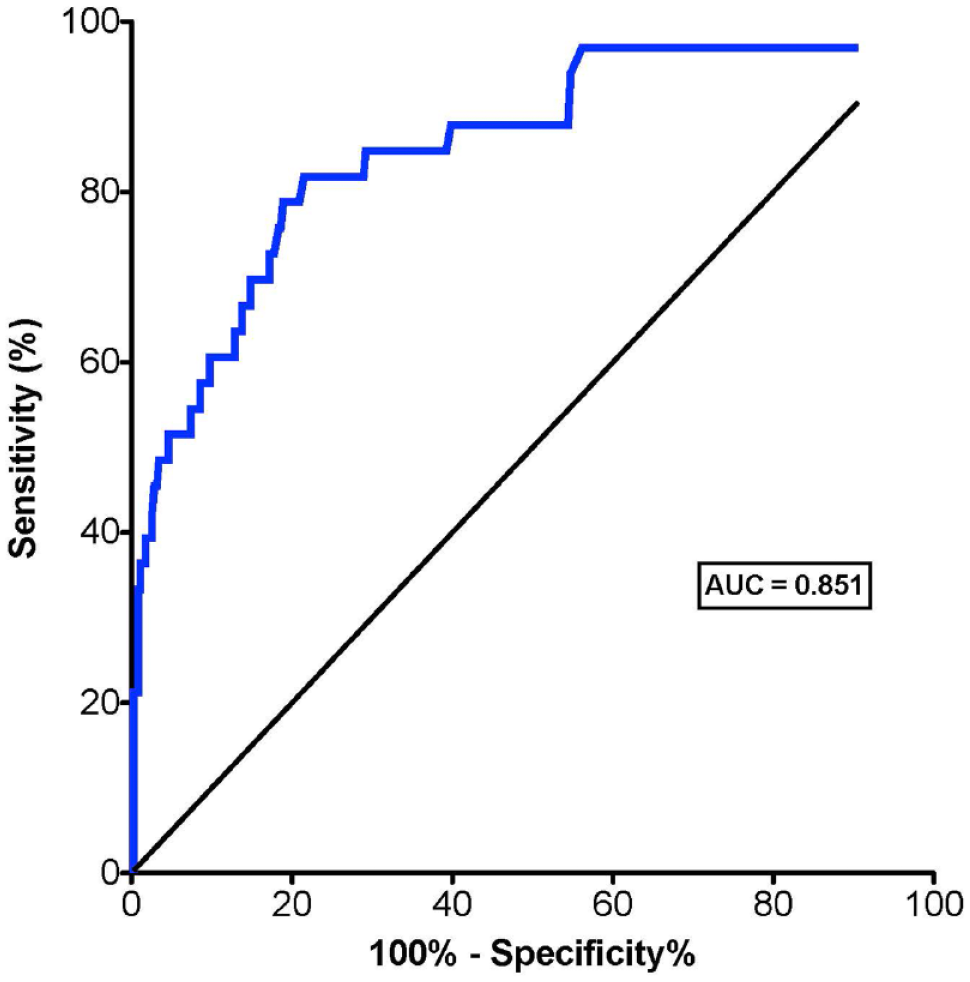
ROC curve of neutrophil count in ascites for the diagnosis of bacterial peritonitis in patients with gastrointestinal cancer.
Prognosis of patients with GI cancer and BP
The median time from cancer diagnosis to first paracentesis and from onset of ascites to death was 14 (IQR 4–26) months and 2.6 (IQR 1.4–4.4) months, respectively. After the first episode of BP, median survival was 10 days (IQR 6–40). Survival at 30 days after the first paracentesis was not different in patients with and without BP (Figure 3, p = 0.10). The mortality rates at 30 and 90 days after the first episode of BP were 50% and 79%, respectively.

Survival at 30 days after the first paracentesis in patients with gastrointestinal cancer with and without BP.
Discussion
BP in GI cancers and its diagnostic and therapeutic management have been little described in the literature. This retrospective multicenter study aimed to fill this knowledge gap in order to help oncologists better manage this disease. The first major finding of this study is that this event was not an uncommon one, with a 1-year prevalence rate of 14%, a figure similar to the prevalence of spontaneous BP in patients with cirrhosis in Europe (18.4%). 20 In addition, 8% of ascites samples over 1 year in patients with GI cancer revealed BP. The occurrence of BP is a very pejorative sign, as it was associated with a median survival of only 10 days.
The second important finding of our study is that BP in patients with GI cancer often has a secondary origin (65% of cases), unlike SBP in patients with cirrhosis, which is rarely secondary (5% of cases). The most frequent causes of BP were cholangitis, liver abscesses, and obstruction of the digestive tract. Therefore, when a bacterium is found in an ascites sample in patients with GI cancer without any argument in favor of contamination, a secondary cause of BP should be evoked, and an abdominal CT-scan should be considered. As this exam was not systematically performed at the diagnosis of BP, it is possible that secondary BP has been overlooked in patients with ‘unknown’ BP and not treated accordingly. Furthermore, some patients whose BP was classified as ‘unknown’ in our study had a multimicrobial or persistent BP at follow-up paracentesis despite antibiotherapy, suggesting an unidentified secondary cause.
The bacteria most frequently found in our study were Enterobacterales. Some germs were common to those encountered in cirrhotic patients with BP (i.e. E. coli, Streptococcus viridans) but here BP was more often multimicrobial. Some germs (Streptococcus milleri, Enterococcus spp., and Clostridium spp.) found in ascites samples in our study are more frequently found in cases of secondary BP. Around 25% of patients had BP caused by cephalosporinase-producing Enterobacterales. In contrast, most patients were treated with third generation cephalosporins or piperacillin–tazobactam as probabilistic therapy, which was retrospectively unsuitable for this type of bacteria and could explain the frequent persistence of positive culture and a high rate of rapid unfavorable outcome. A probabilistic therapy in this context could therefore be cefepime, which acts on cephalosporinase-producing Enterobacterales. Of note, the pattern of antibiotic resistance observed in this study in ascites from patients with digestive cancer is consistent with the epidemiology of hospital-acquired infections.
The third finding of this study is that a neutrophil count threshold in ascites of 110/mm3 had a negative predictive value of 96% and could be propose to exclude the diagnosis of BP, in the absence of bacteria. In contrast, its positive predictive value was poor (39%), thus not ideal for diagnosing BP in patients with GI cancer. Nevertheless, given the severity of BP and its poor prognosis, if the neutrophil count in ascites exceeds 110/mm3 or if bacteria are present on direct examination without argument for a contamination, an abdominal CT-scan and laboratory workup (including blood cultures and liver tests) should be carried out to exclude a cause of secondary BP, and probabilistic antibiotic therapy targeting cephalosporinase-producing Enterobacterales should be administered in case of likely systemic infection until definitive bacterial culture results are available. When the neutrophil count is below 110/mm3 and no bacteria are found, the diagnosis of BP is less likely. It should be noted that it takes at least 48 h to obtain the results of bacterial cultures. However, it seems suitable to rapidly start an antibiotherapy at the slightest doubt given the seriousness of this infection. A diagnostic algorithm is suggested in Figure 4.

Diagnostic algorithm of bacterial peritonitis in patients with gastrointestinal cancer.
This study has some limitations. Firstly, some data were missing due to the retrospective design. In addition, of all ascites samples taken during the inclusion period, it is likely that some were not systematically sent to the bacteriology laboratory. To limit this bias, all clinical departments were informed in advance to ensure that all samples would be sent for bacteriology analysis during the inclusion period. Moreover, the proportion of secondary BP in our study may have been underestimated because not all patients had a CT-scan. It should also be noted that the prevalence reported here only takes into account patients admitted to hospital who have had at least one paracentesis; patients with ascites but who have not had a paracentesis were not included, and the prevalence reported here may therefore have been overestimated. This study also has several strengths: it is the first to describe the prevalence of BP in patients with GI cancer and to discuss its mechanisms. A large number of patients and ascites samples were included in this multicenter study. In addition, the completeness of the ascites collection was ensured by using bacteriology laboratories software to identify all the ascites collected during the inclusion period. These results have enabled us to propose a simple algorithm for everyday practice. A prospective study to confirm our proposal and to better define the therapeutic management of BP early is needed in order to improve the comfort and prognosis of patients.
In conclusion, BP in patients with GI cancer is not an uncommon event, with an annual prevalence of 14%. The main mechanism is secondary, mainly complicating a cholangitis, intrahepatic abscess, or digestive obstruction. BP is associated with a poor short-term prognosis that could be potentially improved by early recognition and treatment of the infection. Further studies are needed to define the optimal therapeutic management of these patients and its impact on prognosis.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241258440 – Supplemental material for Diagnosis and treatment of bacterial peritonitis in patients with gastrointestinal cancer: an observational multicenter study
Supplemental material, sj-docx-1-tam-10.1177_17588359241258440 for Diagnosis and treatment of bacterial peritonitis in patients with gastrointestinal cancer: an observational multicenter study by Alix Riescher-Tuczkiewicz, Jules Grégory, Frederic Bert, Magaly Zappa, Anna Pellat, Valerie Lalande, Claire Gallois, Jean-Luc Mainardi, Jean-Baptiste Bachet, Jérôme Robert, Anne Sophie Bourrel, Romain Coriat, Dominique Thabut, Pascal Hammel, Vinciane Rebours and Diane Lorenzo in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
