Abstract
Despite the cytoreductive efforts, and the high responsiveness to standard carboplatin/pacllitaxel front-line treatment, ovarian cancer (OvCa) remains the most lethal gynaecological malignancy with a 5-yr overall survival of only 25%–30% in advanced stage disease. Among the pharmaceutical options available for treatment of OvCa, much attention has been dedicated to pegylated liposomal doxorubicin (PLD) (Doxil® in the US; Caelyx® in Canada and Europe); this drug has a unique formulation which has entrapped conventional doxorubicin in a bilayer lipidic sphere surrounded by a polyethylene glycol layer, which prolongs the persistence of the drug in the circulation and potentiates its intratumor accumulation. These properties represent the winning resource for this drug in that sustain its very favourable toxicity profile and the safe combination with other drugs.
PLD has already been approved for treatment of advanced ovarian cancer patients failing first line platinum-based treatment. Moreover, Phase III trials have been completed, which will hopefully will bring PLD to front-line treatment, and in salvage setting in combination with platinum agents.
This concise review will focus on the clinical role of PLD in the management of patients with epithelial OvCa.
Introduction
Epithelial ovarian carcinoma (OvCa) is one of the most common gynaecological malignancies, and the fifth most frequent cause of cancer death in women. Worldwide, more than 190,000 new cases of ovarian cancer are diagnosed each year accounting for around 5% of all cancers in women, and in 2009, 21,550 new cases have been estimated to be diagnosed in the US. 1 Despite the advances in surgical efforts, and the achievement of high response rates to front-line treatment, OvCa remains the most lethal gynaecological malignancy with almost 50%–75% of cases experiencing recurrence/progression of disease, and a 5-yr overall survival of 25%–30% in advanced stage disease. 1 The major determinants of clinical outcome are represented by the extent of residual tumor at primary surgery and sensitivity to platinum based therapy, defined according to the duration of the platinum-free interval. 2
Besides the extent of response rates, other issues have to be taken into account in the choice of medical treatment, be it in front-line or salvage setting, including the rate and profile of side effects especially in case of drug combinations, and their impact on patients’ quality of life.
In this context, among the pharmaceutical options currently available in the scenario of medical treatment of ovarian cancer, much emphasis has been progressively placed on pegylated liposomal doxorubicin (PLD) (Doxil® in the US; Caelyx® in Canada and Europe), which has been approved in 1999 by the FDA, and in 2000 by the European Medicines Evaluation Agency (EMEA) as single agent for treatment of advanced OvCa patients failing first line platinum-based treatment.
This concise review will focus on the clinical role of PLD in the management of patients with epithelial OvCa.
PLD: Structure, Pharmacokinetic Features, and Phase I Studies
PLD is a unique formulation of conventional doxorubicin in which the drug is encapsulated in a bilayer lipidic sphere 3 of approximately 100 nm, surrounded by a polyethylene glycol (PEG) layer: this represents a hydrophilic protective barrier and facilitates the activation of the reticulo-endothelial system, thus leading to the destruction of the liposomal structure and release of the drug. Moreover, the size of PLD reduces the extravasation of the drug in normal tissues, while allowing a facilitated uptake in tumor: these properties sustain the lower cardiotoxicity and gastrointestinal toxicity compared to the free drug, but also PLD pharmacokinetic features, such as long circulation time, minimal drug leakage from circulating liposomes, and long half-lives. 4 The pharmacokinetics of PLD is still being investigated: there seems to be a complex interaction between pharmacokinetics and pharmacodynamics that could account for some patterns of toxicity; for instance, stomatitis/mucositis is more frequently documented at higher peak dose drug level, while cutaneous toxicity is dependent from dose interval or dose intensity, as sustained by recent data showing that repeated PLD administrations result in cumulative inhibition of the clearance process. 5 The evolution of the nanoparticle technology has fuelled great enthusiasm in the possibility to further enhance the selective intratumor accumulation of PLD, and to shift the therapeutic index toward more tolerable toxicity profiles: in this context, the observations that recombinant serum albumine-conjugated PLD has longer blood circulating properties, smaller hepatic and splenic clearance, and more important, larger intratumor accumulation than PLD in preclinical models is very encouraging. 6
Different dose escalations and schedules have been explored in Phase I studies employing PLD as single agent with dose intensities ranging from 10 to 15 mg/m2/week. 7 Stomatitis was reported as the most frequent dose limiting toxicity (DLT) at PLD doses ≥60 mg/m2, while at lower doses with schedules <21 days, the most common DLT was represented by hand-foot syndrome (HFS). The current PLD dosage as single agent as indicated for ovarian cancer patients is 50 mg/m2, q28. However, PLD safety has been also investigated in combinations with two or even three agents: for combinations of PLD with cisplatin the dose limiting toxicities (DLT) were mostly represented by mucositis, and also neutropenia when CDDP was used at doses >60 mg/m2, q28. Similarly, when combining PLD with carboplatin (JM8), DLTs consisting in stomatitis/mucositis were documented at PLD doses >50 mg/m2, while thrombocytopenia represented the most frequently reported DLT for lower PLD doses.
The tolerability of PLD (at doses ranging from 30 to 40 mg/m2) combined with paclitaxel (PTX) (135 to 200 mg/m2 q21) has been shown to be acceptable; similarly, PLD at doses of 30–40 mg/m2 mg/m2, plus docetaxel (DTX) at doses of 67.5 to 80 mg/m2, was reported as the MTD for this combination. 7 Based on the hypothesis that weekly administration could limit toxicity while keeping the dose intensity unchanged, weekly or bi-weekly administration of PLD (doses from 10 to 20 mg/m2) plus weekly PTX has been also explored.8,9 In this context, it is not surprising that PLD tolerability has been also investigated in combination with both JM8 and PTX: 10 the MTD corresponded to PLD 30 mg/m2 plus JM8 AUC 6, and PTX 175 mg/m2, q28, or, alternatively, PLD 20 mg/m2 plus JM8 AUC 5, and PTX 175 mg/m2, q21. Given the strong biological rationale of combining PLD, an inhibitor of Topoisomerase II, with Topoisomerase I inhibitors, as well as the non overlapping toxicity profile of these two classes of drugs, the safety of PLD/topotecan (TPT) combination has been the subject of active investigation.11,12 The MTD was reached at PLD doses of 30 and 40 mg/m2, q21 or q28, and at TPT doses (0.5–1.0 mg/m2/d) administered with the classic shorter courses (d1-3 or d1-5). PLD safety has been investigated also in combination with other chemotherapeutics such as vinorelbine, and gemcitabine;7,13 however, in spite of generally positive reports, only a few combinations have progressed to phase II evaluation.
PLD: Toxicity Profile
The favourable PLD toxicity profile is widely recognized as the winning resource of this drug, however some adverse side effects have to be taken into account: for instance, acute hypersensitivity reaction, characterized by flushing, facial edema, back pain, rigors, dyspnea, hypotension and chest/throat tightness can occur during drug infusion, and differently from reactions induced by other drugs which generally are documented after a previous exposure, they can be registered even during the first administration. PLD related reactions are seen in about 6.8% of patients, however, if they are not documented initially, rarely occur in subsequent cycles. 14 On the other hand, muco-cutaneous toxicity is the most frequent PLD related side effect, and represents the most important dose limiting toxicity: 15 in particular, palmar-plantar erythrodysesthesia also known as hand-foot syndrome (HFS) represents a distinctive toxic reaction to PLD administration. Its pathogenesis remains unclear although the prolonged accumulation in areas where subclinical trauma occurs might play a relevant role. 16 HFS is characterized by initial extremities paresthesia followed by patch erythema, edema, and desquamation of hands and soles. The natural history of HFS is often self-limiting, but some cases develop blistering and ulceration which can limit daily functions and negatively affect patients’ quality of life. Several factors can favour the occurrence of HFS during PLD treatment including the number of cycles, doses ≥50 mg/m2, the concomitant occurrence of peripheral neuropathy, and the number of previous chemotherapy lines; also the PLD administration according to the 3-week schedule, which may coincide with the interval of epidermal turn-over, might emphasize the potential PLD-induced keratinocyte damage. 17 Patients’ education to avoid risk factors and to precociously recognize the initial signs/symptoms of skin toxicity is relevant. More specifically, the use of ice pack cooling of hands and feet during chemotherapy, administration of corticosteroids, pyridoxine, dimethylsulfoxide, and moisturizing lotions are often used in clinical practice. 17 Although their true efficacy has been not proven in prospective trials: indeed, it is conceivable that the reduction of PLD dose intensity to 10 mg/m2/week could lead to mild or modest HFS in the vast majority of cases. 18 Recently, an international panel of experts was convened to develop recommendations for management of PLD-associated HFS according to the grade of symptoms and clinical findings, 19 however, Phase III trials are urgently needed to support the rigorous adoption of each of the previously cited intervention.
Phase II Studies with PLD as Single Agent or in Combination
When considering the Phase II studies with PLD as single agent, the response rates in the subset of platinum resistant OvCa patients ranged from 9.1 to 40.0%,20–22 and did not seem to present a schedule or dose dependence. In particular, several studies have shown that a more acceptable toxicity profile in terms of decreased rates of HFS and stomatitis/mucositis can be obtained with a PLD dose of 40 mg/m2 q28, with comparable response rates and outcome. 20 More recently, also bi-weekly schedules have been investigated in the same clinical setting in order to further improve the toxicity pattern. 23 Although the direct comparison across non randomized Phase II studies is difficult, it seems that the biweekly regimen represents a good therapeutic option, since it does not worsen clinical outcome while preventing severe haematological and non-hematological side effects.
One of the most frequently studied partners in PLD-based combination is represented by gemcitabine (GEM): in the subset of platinum resistant disease the response rate ranged from 22% to 33%, with median PFS from 2.7 to 6.0 months regardless of the schedule administered.24–26 Considering that the percentage of patients who already received >1 lines of chemotherapy was high in some studies, 26 the regimen was relatively well tolerated: indeed, Grade 3/4 anemia was documented in the range of 3%–9% in studies utilizing the classical 3-week and 4-week schedules; Grade 3/4 neutropenia was negligible in case of bi-weekly regimens but raised up to 30% in the 3-week schedule. 26
The investigation of PLD/platinum doublets in platinum sensitive disease in which the currently administered partners of platinum agents are generally associated with elevated neurologic and hematological toxicity is clinically relevant: for PLD/JM8 combinations results are consistent across the studies with response rates ranging between 46.0% to 68.0%, and median PFS of approximately 9–10 months.27–30 In the so-called partially platinum sensitive patients (i.e. recurring/progressing within 7–12 months from completion of primary treatment) the rate of response is lower (around 50%) and also median PFS is shorter with an average of 9 months versus 14 months in totally platinum sensitive cases.28–31 In this context, the MITO-8 multicentric Phase III study has been recently launched comparing PLD (40 mg/m2, q28) versus JM8/PTX (AUC 5, 175 mg/m2) in OvCa patients recurring within 6–12 months from the completion of primary treatment.
Overall, the toxicity rate/severity related to combinations of PLD and JM8 were consistent across the studies with and Grade 3/4 neutropenia occurring in around 50% of cases with the exception of two studies.28,30 The rate of severe thrombocytopenia seems closely related to PLD dosage: indeed, in studies utilizing PLD at 30–40 mg/m2, Grade 3/4 thrombocytopenia remain within 14% and 26%, while at PLD doses of 50 mg/m2 it rose up to approximately 40%. As expected, severe HFS was negligible in studies utilizing the dosage of 30 mg/m2 PLD.27,30,31
PLD: Phase III Studies
The clinical role of PLD in recurrent setting has been actively investigated in Phase III randomized studies (Table 1): in particular, in the Gordon study, 33 whose updated findings have been presented in 2004, 34 OvCa patients recurring/progressing after front-line chemotherapy were randomized to receive PLD versus TPT: in platinum resistant disease no significant difference was seen in response rate, PFS, or OS between the two treatment arms, while in platinum sensitive patient median PFS and OS were significantly prolonged in PLD versus TPT treated patients. Moreover, for partially platinum sensitive disease (n = 122), the HR favoured PLD versus TPT (HR = 1.58, p value = 0.021). The toxicity profiles of the two drugs were completely different with Grade 3/4 hematological toxicity occurring more frequently and more severely in TPT versus PLD arm. Two more recent Phase III trials addressed the comparison of PLD versus GEM in recurrent platinum resistant OvCa: In the study by Mutch et al 35 no difference in any of the studied survival outcomes was documented between the two treatment arms, while the MITO-3 trial, 36 although failing to show any difference in PFS, however, documented a more favourable OS in PLD versus GEM treated arm (median OS = 14 months versus 12.7 months, respectively; p value = 0.048). Higher global QoL scores especially for physical and emotional findings and fatigue were found in PLD treated patients at the first and second post-baseline assessments, and this is likely to reflect the profile of toxicity: indeed, hematological toxicity was negligible with only 6% of Grade 3/4 neutropenia compared to 23% in GEM arm. Moreover, severe HFS was documented in only 5% of cases. Recently, the activity of canfosfamide (CAN) (Telcyta®), a glutathione analogue prodrug, has been tested toward a control arm represented by PLD or TPT in platinum resistant OvCa patients who had already progressed on 2nd line treatment with PLD or TPT 37 (ASSIST-1, NCT00057720). The response rate was higher in the control arm versus CAN (10.9% versus 4.3%, respectively), as was also median PFS (4.3 months versus 2.3 months, p value = 0.01). A Phase III trial (ASSIST-5, NCT00350948) comparing the combination PLD/CAN versus PLD alone had been planned based on the demonstration that PLD might favour glutathione S-transferase expression, thus potentially increasing cancer cell susceptibility to CAN: The study was temporarily suspended while waiting for the data from the ASSIST-1 study, so that, at time the enrolment was re-started only 125 patients had been enrolled. 38 No difference emerged between the two treatment arms in terms of response rate and PFS; however, when considering the subgroup of platinum refractory and primary platinum resistant patients, a statistically significant advantage in favour of the combination was observed in terms of response rate (15.0% versus 5.7%) and PFS (median 5.6 months versus 2.9 months, HR = 0.55, p value = 0.042). While haematological toxicity was generally higher in the combination arm versus PLD alone, the rate of Grade 2/3 HFS was lower in PLD/CAN than PLD alone (9% versus 21%, respectively). At this time, the available data support the Phase II derived suggestions that in platinum resistant disease none of the currently most frequently adopted drugs, such as PLD, GEM, or TPT shows superiority over the others in terms of response rate and survival; in this context the 3-week schedule administration of PLD at 40 mg/m2 seem to offer the most favourable toxicity profile which is likely to sustain the achievement of better QoL scores. 36
Randomized studies with PLD alone or in combination in salvage setting.
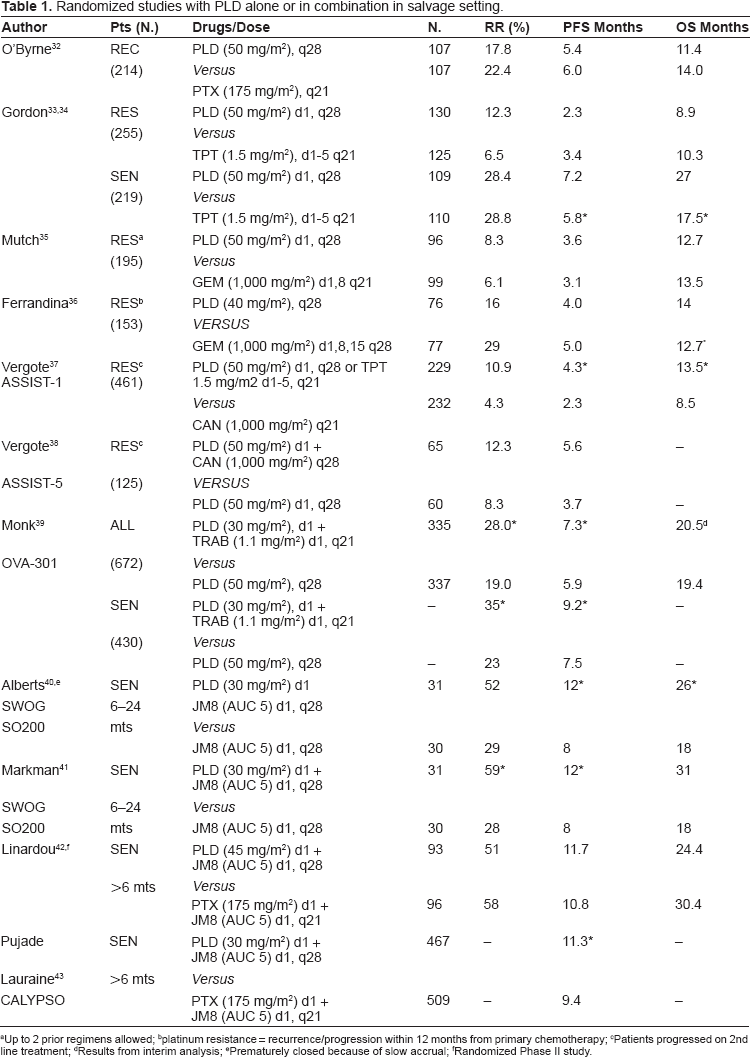
Up to 2 prior regimens allowed
platinum resistance = recurrence/progression within 12 months from primary chemotherapy
Patients progressed on 2nd line treatment
Results from interim analysis
Prematurely closed because of slow accrual
Randomized Phase II study.
Among the most intriguing novel drugs, trabectedin (TRAB) (ET743) (Yondelis®), has gained much attention for treatment of OvCa patients. TRAB has a unique mechanism of action, in that it binds to the minor groove of DNA unlike most other agents, thus affecting tumor cell proliferation, and the nucleotide excision repair system. A Phase III trial (OVA-301) has been planned comparing PLD/TRAB versus the PLD in ovarian, peritoneal, and tubal cancer recurring/progressing after first line chemotherapy: 39 in the whole series the response rate, as assessed by independent radiology review, was significantly higher in PLD/TRAB than PLD alone group, as was also median PFS (HR = 0.79, p value = 0.019); a clear advantage favouring the combination was evident only in platinum sensitive disease (response rate 35.3% versus 22.6%, p value = 0.0042; median PFS 9.2 months versus 7.5 months; HR = 0.73, p vale = 0.017). These findings were also evident in the subset of partially platinum sensitive disease with median PFS of 7.4 versus 5.5 months in PLD/TRAB versus PLD arm (HR = 0.65, p value = 0.0152). As far as toxicity issues are concerned, Grade 3/4 anemia, neutropenia and thrombocytopenia were significantly more frequent compared to PLD alone. Grade 3/4 elevation of sGOT, and sGPT was reported in 38% of cases in the combination arm, but these side effects were described as of short duration. Based on these results the combination PLD (30 mg/m2) and TRAB (1.1 mg/m2) on a 3-week basis, has been submitted to the USA FDA approval for treatment of patients with relapsed platinum sensitive OvCa.
In platinum sensitive recurrent disease, apart from the SWOG SO200 trial40,41 which compared PLD/JM8 combination to JM8 alone, and showed a statistically significant improvement of PFS with the combination, other randomized trials testing PLD/JM8 regimen against PTX/JM8 doublet deserve mention: in particular, in particular, the Phase II randomized study by Linardou et al 42 documented no difference between the two arms in terms of response rate and PFS, while confirming the very favourable toxicity profile for the combination which resulted in lower rate of severe thrombocytopenia and, more important, in no case of severe neurotoxicity. On the other hand, much attention has to be reserved to the CALYPSO Phase III study, 43 which randomized 976 patients to PLD/JM8 versus PTX/JM8 regimen. The trial showed the statistically significant superiority of PLD/JM8 over PTX/JM8 combination in terms of PFS, while data on OS were too early to be reported. Grade ≥ 2 HFS was documented in 13% of PLD/JM8 versus 2% of PTX/JM8 arm, whereas lower rates of Grade 2/3 neurotoxicity were reported in the experimental than in the standard arm (5% versus 28%, respectively). While waiting for the mature OS data, it can be reasonably stated that PLD/JM8 combination represents a valid alternative to other platinum-based regimens in recurrent platinum sensitive OvCa especially for patients whose QoL can be heavily compromised by alopecia, or who had experienced or had not yet rescued from taxane-induced neurotoxicity.
Attempts to include PLD in front-line treatment have been also investigated: the GOG182/ICON5 randomized trial, 44 which really represents the largest cooperative effort attempted worldwide, enrolled 4,312 stage III–IV patients who were randomized to five arms including the standard treatment, as follows 1) JM8 AUC 6, PTX 175 mg/m2 q21, for 8 cycles, versus 2 triplets: 2) JM8 AUC 5, PTX 175 mg/m2, GEM 800 mg/m2 d1,8, q21; 8 cycles; 3) JM8 AUC 5, PTX 175 mg/m2, PLD 30 mg/m2, q21; 8 cycles, or 2 sequential doublets: 4) JM8 AUC 6, TPT 1.25 mg/m2 d1–3, 4 cycles, followed by JM8 AUC 6, PTX 175 mg/m2 4 cycles; 5) JM8 AUC 6, GEM 800 mg/m2 d1,8 q21, 4 cycles followed by JM8 AUC 6, PTX 175 mg/m2 4 cycles. Despite the documentation of increased hematological and non hematological toxicity in the triplet regimens, neither was there any PFS or OS advantage with sequential doublets nor with triplets compared with the control arm.
In front-line setting, mature results from the MITO-2 45 trial are eagerly awaited; preliminary data from this study which is the first to investigate the combination PLD/JM8 (30 mg/m2, AUC 5, q21) versus the standard treatment documented the equivalence of the two treatment arms in terms of response rate (59.0% in the standard versus 57.0% in the experimental arm, p value = 0.70). As of March 2009, with a median follow up of 35 months 530 events for PFS were documented, therefore survival data cannot be considered mature enough for final analysis which will be hopefully available in 2010. As expected, the pattern of toxicity was largely different in the two groups: in PLD/JM8 arm haematological toxicity was more frequent while Grade ≥ 3 neurotoxicity and alopecia was lower than in the standard treatment arm.
PLD and Target Based Agents
Novel perspectives related to PLD use in ovarian cancer are represented by PLD combination with target based agents: an ongoing Phase II study has recently reported the preliminary results of the combination PLD plus bevacizumab (BEV) (Avastin®), the monoclonal antibody against VEGF, in 2nd line treatment of OvCa patients: 46 48 patients received PLD (30 mg/m2, q21) alone at the first cycle, and then associated with BEV (15 mg/kg, q21) for the following 6 cycles; in the 21 patients available for analysis, response was documented in 14.3% of cases. Interestingly enough, the addition of BEV-did not induce any modification of PLD pharmacokinetics. Grade 3 HFS was registered in 5% of cases, while Grade 3 hypertension and FEV reduction >10% were reported in 15% of cases. With the aim to reduce the rate/severity of side effects, a weekly regimen of PLD/BEV (PLD 10 mg/m2 and BEV 2 mg/kg d1,8,15 q28, for at least 3 cycles) was investigated in 30 recurrent heavily pre-treated OvCa patients. 47 Response was achieved in 38.4% of cases with disease stabilization in 34.6%. Overall, the weekly regimen seems well tolerated although the potential cumulative cardiovascular side effects of the two drugs need to be explored in a larger series. A large Phase II randomized study (AURELIA, NCT00976911) is recruiting patients with recurrent platinum resistant disease, who are randomized to PTX, or TPT, or PLD versus the experimental arm in which BEV 10 mg/m2 bi-weekly or 15 mg/m2 q21 is added to the same drugs. The interest to angiogenesis inhibitors is testified by the efforts to study molecules acting on different targets, such as Volociximab (VOL): this is a chimeric monoclonal antibody able to directly target α5/β1 integrin, a protein characterizing the activated endothelial cells, thus preventing its interaction with the extracellular fibronectin and disrupting tumor neoangiogenesis; a Phase II study by Vergote et al, 48 has been carried out in recurrent OvCa patients administered up to 2 lines of chemotherapy; 66 patients received PLD or PLD plus VOL 15 mg/m2 biweekly (n = 34) or PLD plus VOL 15 mg/m2 weekly (n = 27). Median PFS was 27.5 weeks in the first arm, 18 weeks in the second one and 31.6 weeks in the last group, thus suggesting no superiority of the combination versus PLD alone. Based on preclinical studies and encouraging Phase I data showing the absence of any interference of bortezomib (BOR) (a proteasome inhibitor) (Velcade®) with PLD pharmacokinetics, 49 a Phase II study combining the two drugs has been planned: in particular,; recurrent platinum resistant (n = 15), and sensitive (n = 15) OvCa patients were administered PLD 30 mg/m2 and BOR 1.3 mg/m2 d1,4,8,11 q21. Responses were seen only in the group platinum sensitive disease which proceeded to the second step of enrolment. 50 Among the other novel biological targets considered for designing PLD/target based therapy, attention has been focused on Farletuzumab (MORAB-003), an inhibitor of folate receptor-α under investigation in combination with PLD and JM8 in recurrent platinum sensitive OvCa. A list of the ongoing trials investigating the combination of PLD with other target based drugs such as IMC-3G3, panitumumab, pazopanib, and vandetanib can be found at the www.clinicaltrials.gov
Conclusions
The pegylated liposomal formulation of doxorubicin, because of its unique and favourable toxicity profile, has enlarged the scenario of the clinical applications of the parent compound: indeed, whether used alone and in combination PLD has been introduced in the management of almost all clinical settings in OvCa patients. Results from Phase II and Phase III randomized trials have led to the FDA approval of PLD in the salvage treatment of recurrent disease; moreover, the results of the CALYPSO trial suggest that PLD/JM8 combination is a very valid option in recurrent platinum sensitive disease especially in patients who had experienced or had not yet rescued from taxane-induced neurotoxicity or just refuse alopecia.
While awaiting for the MITO-2 final results which might lead to replace paclitaxel from the front-line doublet, or at least provide a useful alternative to JM8/PTX depending on patients’ performance status and preference, future perspectives come from the combination of PLD with target-based agents which do not seem to alter PLD pharmacokinetics which remain the winning resource of this agent.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
