Abstract
Pegylated liposomal doxorubicin (PLD) is doxorubicin HCl encapsulated in long-circulating STEALTH® liposomes (Doxil®). PLD achieves good response rates and many patients maintain long-lasting stable disease (SD), which is one of the advantages. In addition, the clinical benefit is high in platinum-resistant disease, and PLD is thus considered to be the first option. PLD is associated with a number of adverse events, but these events are mild to moderate. PLD is safer for heavily pretreated patients than topotecan and gemcitabine due to mild bone-marrow toxicity, but that nonhematotoxity, such as PPE, stomatitis, mucositis, and other cutaneous reactions were the most common side effects attributable to PLD. Based on a review of previous studies, there are no differences in efficacy between 50 and 40 mg/m2 of PLD, therefore, a dose of 40 mg/m2 is preferable in patients with platinum-resistant disease to reduce adverse events. The 1-hour infusion schedule every 4 weeks makes PLD easy to administer. A rational approach to combine PLD with other drugs should take the slow accumulation and delayed peak of PLD in tumors into consideration. When combined with other useful agents, the lower dose of PLD (30 to 35 mg/m2) with a 3-week schedule may reduce severe PPE and stomatitis with negligible effects on the level of DI and the therapeutic efficacy.
Introduction
Epithelial ovarian cancer is sensitive to chemotherapy and approximately 75% of patients achieve complete clinical remission after the initial treatment. However, most patients have a recurrence, which results in death after a chronic course. The progression-free survival (PFS) for advanced ovarian cancer patients with optimal residual disease range from 18 to 24 months, while PFS for patients with suboptimal residual disease is 18 months.1–5 The PFS of dose-dense paclitaxel/carboplatin therapy improved to 28 months for patients with both optimal and suboptimal disease in our study (JGOG3016). 6 In the treatment of recurrent cancer, the issues to consider are the treatment-free interval (TFI), toxicity continuously observed from the initial treatment, recurrent tumor diameter and increased CA125. The TFI is the most important for selecting drugs or regimens, and the longer the TFI, the higher the response rate.7,8 When the TFI is 6 months or longer, the tumor is considered to be sensitive to chemotherapy, but when the TFI is less than 6 months, the tumor is considered resistant to chemotherapy. However, 6 to 12 months of TFI is considered to be a gray zone, and such tumors require more careful consideration when selecting drugs or regimens. Recommended therapies for tumors sensitive to drugs, based on the results of randomized controlled trials (RCT) and meta-analyses, are carboplatin-combination therapy such as carboplatin/paclitaxel, carboplatin/gemcitabine, and carboplatin/pegylated liposomal doxorubicin (PLD).9–11 For patients with TFI of 6 to 12 months, a non-platinum drug is one option for prolonging the platinum-free interval and reducing toxicity. However, patients must therefore be carefully observed to determine when the switch from non-platinum chemotherapy to platinum combination therapy should be made. A phase III trial has been recently launched comparing carboplatin/paclitaxel versus PLD in the above cited clinical setting (NCT00657878). On the other hand, for patients with a drug-resistant tumor, a drug without cross-resistance to paclitaxel and carboplatin must be selected. The goal of therapy is to delay progression, relieve symptoms and improve the QOL, and monotherapy is generally chosen for the most favorable toxicity profile. PLD, topotecan, and weekly paclitaxel are drugs that have been approved by the Food and Drug Administration (FDA), and gemcitabine (GEM), oral etoposide, and docetaxel can also be used. In Japan, weekly irinotecan (CPT-11) is widely used. When selecting drugs, the differences in toxicity must be fully understood. It is usually difficult to completely cure recurrent disease with one drug or one regimen with high efficacy and low toxicity, and the drugs must therefore be changed as required while assessing the effect and toxicity. Topotecan and GEM are highly hemotoxic, and patients should be monitored for non-hemotoxic events that reduce the QOL when PLD and irinotecan are used. There is concern about palmar-plantar erythrodysesthesia (PPE) and stomatitis, both of which can occur with PLD treatment, diarrhea due to irinotecan, and peripheral neuropathy and arthralgia during weekly paclitaxel therapy.
PLD was approved in 1999 by the FDA and in 2000 by the European Medicines Evaluation Agency as a treatment for chemorefractory and chemoresistant epithelial ovarian cancer, and has been used worldwide as the first option for patients with chemorefractory and chemoresistant epithelial ovarian cancer.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
PLD consists of doxorubicin encapsulated in N-(carbonyl- methoxypolyethylene glycol 2000)-1, 2-distearoyl-sn-glycero-3-phosphoethanolamine sodium salt (MPEG-DSPE) coated liposome (STEALTH® liposome) (Fig. 1). Liposomes have the advantage of biocompatibility and versatility of formulation for intravenous use. However, the disadvantage of liposomes are rapid uptake by the reticuloendothelial system (RES) and removal from the circulatory system, thus reducing the amount of drug that reaches the tumor. MPEG-DSPE is a hydrophilic material and characteristically decreases RES uptake. Therefore, the STEALTH® liposome achieves prolonged circulation time without rapid uptake by RES, and PLD has made prolonged delivery of doxorubicin and prolonged circulation time possible. Gabzon et al undertook a pilot clinical study about the pharmacokinetics compared with PLD and free (unencapsulated) doxorubicin (DOX), and reported that the AUC of PLD in plasma was approximately 250-fold higher than that of DOX. 12 Furthermore, the diameter of PLD of approximately 100 μm makes it generally difficult to be absorbed into the capillaries. However, tumor tissues have a hypervascular environment compared to normal tissues, and the absence of the basement membrane and of the tight conjunction in the tumor neovessels have to be recognized as possible causes facilitating the extravasation of PLD in tumor tissue. Vaage et al examined the tissue distribution of DOX and PLD in mice with carcinoma, and reported that the AUC of PLD in tumor tissue was approximately 25-fold higher than that of DOX. 13 PLD enables prolonged circulation time and accumulates selectively in carcinoma tissues by intravenous administration, STEALTH® liposomes gradually disintegrate, and doxorubicin is released and metabolized therefore, the metabolic pathway of doxorubicin is the same whether or not the doxorubicin is encapsulated with STEALTH® liposomes. Doxorubicin hydrochloride is metabolized in the liver, and doxorubicinol, the main metabolite, is formed by cytosolic carbonyl reductase. Finally, doxorubicin is formed by deglycosidation. This product is metabolized in the liver and excreted in the urine and feces (bile). The main enzymes that are involved in doxorubicin metabolism are NADPH-dependent aldo-keto reductase and microsomal glycosidase.
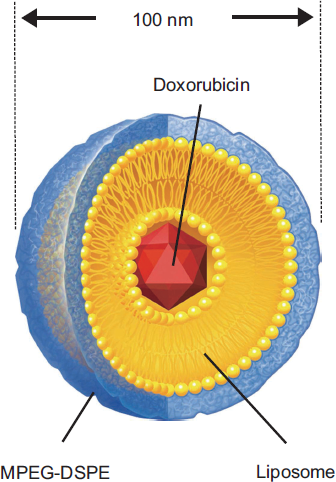
The structure of pegylated liposomal doxorubicin.
Clinical Studies of PLD
Efficacy
The results of phase II and III studies of PLD monotherapy in patients with platinum- and/or taxane-resistant disease are shown in Table 1.14–26 The response rates of PLD administration at a dose of 50 mg/m2 every four weeks in 4 phase II studies ranged from 7.7% to 21.0% and the ranges of stable disease (SD) rate and clinical benefit were 40.3% to 51.3% and 57.3% to 61.3%, respectively.14–23 The response rates in 2 phase III studies were 8.3% and 12.3% with SD rates of 27.7% and 38.5%, respectively, and the clinical benefit was 40.0% and 46.9%.24–26 The response rates of PLD administration at a dose of 40 mg/m2 every four weeks in 4 phase II studies ranged from 9.1% to 31.4%, and the ranges of SD rate and clinical benefit were 7.8% to 48.6% and 39.2% to 62.2%, respectively.16–18,21 The response rate in a phase III study was 15.7% with an SD rate of 42.9%, and the clinical benefit was 58.6%. 26 The time to progression (TTP) ranged from 4 to 19 months with both doses of 40 and 50 mg/m2. The efficacy of PLD at a dose of 50 mg/m2 was similar to that at a dose of 40 mg/m2 (Table 1). The results of a phase III comparative study with topotecan indicated that PLD was more effective in improving survival than topotecan. 24 To be specific, in platinum-resistant disease, the progression-free survival (PFS) and over all survival (OS) were higher with topotecan than with PLD, while in platinum-sensitive disease, in contrast, the PFS and OS were significantly higher with PLD than with topotecan. In 2004, Gordon et al reported long-term follow-up outcomes of patients who were registered in the comparative study that had been done previously. 27 At the time of this evaluation, 87% had died. There was a significant survival advantage for all patients enrolled in this study that the patients who had taken PLD had an 18% reduction in the risk of death. On the other hand, Mutch et al conducted a phase III comparative study with gemcitabine (GEM) that showed no significant differences in PFS and OS between PLD and gemcitabine. 25 Ferrandina et al reported no differences in PFS between PLD and GEM in relapsed patients with a treatment-free interval (TFI) of 12 months or less, but a significant efficacy in OS with PLD compared to GEM (Table 2). 26 From the results of the above phase III studies, PLD was considered to have similar efficacy as other novel drugs on platinum-resistant disease; however, it was more effective at improving the survival rate in patients with recurrent ovarian cancer, including platinum-sensitive cancer, than other drugs.
Response rate in patients with platinum- and/or taxane-resistant disease: Pegylated Liposomal Doxorubicin.
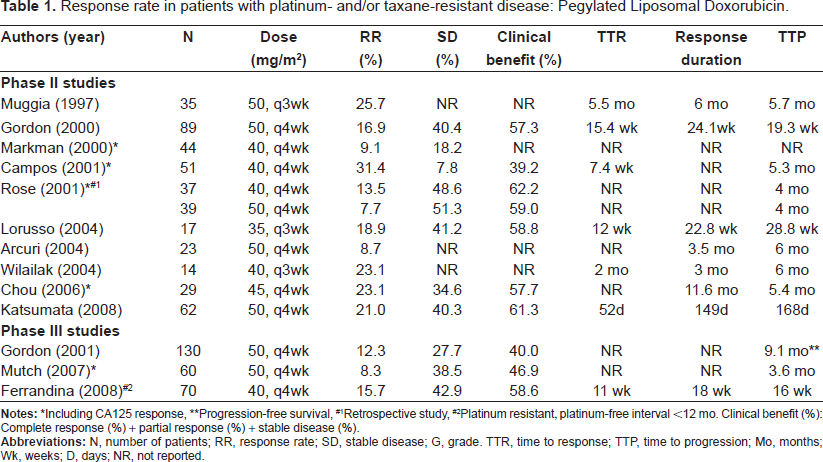
*Including CA125 response,
**Progression-free survival,
#1Retrospective study,
#2Platinum resistant, platinum-free interval <12 mo. Clinical benefit (%): Complete response (%) + partial response (%) + stable disease (%).
Survival in randomized studies.
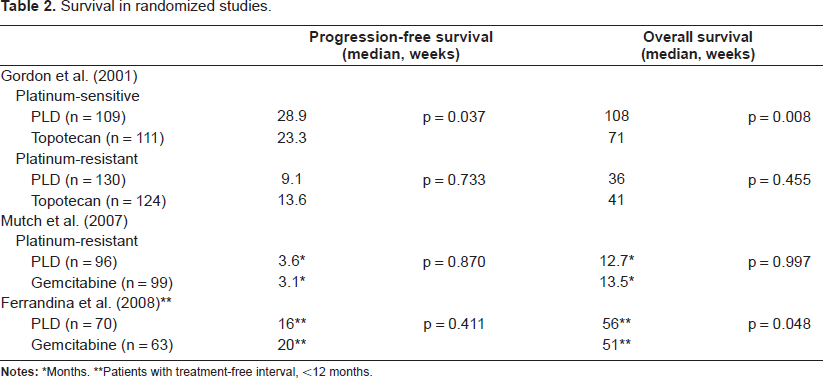
*Months.
**Patients with treatment-free interval, <12 months.
Safety
PLD is associated with several adverse events, but these events are mild to moderate (Table 3).14–17,19–26 PLD required fewer dose modifications compared with topotecan and gemcitabine. The superiority of PLD in terms of QOL has been shown.24–26 PPE, stomatitis, mucositis, and other cutaneous reactions were the most common side effects attributable to PLD. PPE and stomatitis developed in approximately 40% of patients but, grade 2 or higher PPE and stomatitis were found in 19.8% to 31.5% of patients treated with 50 mg/m2 of PLD (every 4 weeks), and 2.8% to 15.5% (less than half) of those treated with 40 mg/m2 or less of PLD (every 3 to 4 weeks). Grade 2 or higher stomatitis developed in 14% to 38% or more of patients treated with 50 mg/m2 of PLD14,15,23 in contrast, it developed in 8.0% of those treated with 40 mg/m2. 16 The results of phase III comparative studies of PLD with topotecan or GEM confirmed that PPE and stomatitis developed significantly in patients treated with 50 mg/m2 of PLD compared with those treated with topotecan and GEM, while the incidence of PPE was slightly higher in patients treated with 40 mg/m2 of PLD compared with those treated with GEM, and no differences were found in the incidence of mucositis between PLD and GEM. 24–26 These toxicities were usually handled by increasing the cycle length or reducing the dose. On the other hand, PLD induced less hematotoxicity than topotecan and GEM. One quarter of patients who received 50 mg/m2 of PLD developed grade 3 or 4 neutropenia, but febrile neutropenia was rarely found. The incidence of thrombocytopenia was even less. Cardiac toxicity was also uncommon. The results of a phase II study in Japan showed PPE in 78% (grade 2 or higher: 51%) and grade 3 or higher neutropenia in 68%, suggesting racial differences. 23
Adverse events: Pegylated Liposomal Doxorubicin.
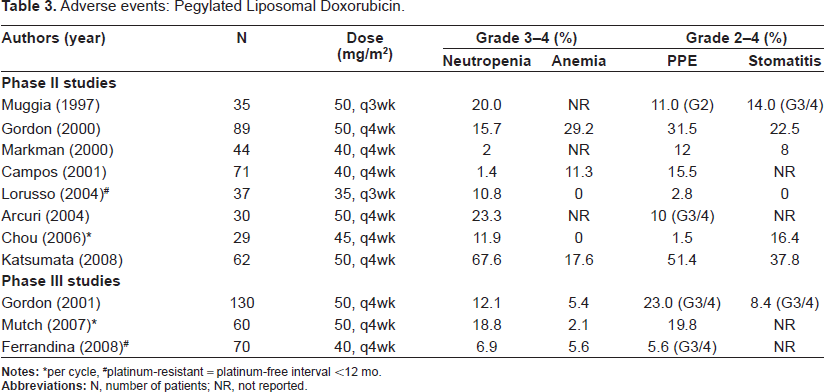
*per cycle, #platinum-resistant = platinum-free interval <12 mo.
The first adverse effect seen is an infusion-related reaction that is characterized by flushing, facial edema, headache, back pain, rigors, hypotension, chest/throat tightness and dyspnea. These reactions are seen in 7% to 19% of patients during the first cycle, and are all resolved on the day of onset or the following day.23,28,29 It has been reported that decreasing the infusion rate reduces the risk of reaction.
There were no treatment-related deaths reported in the 657 patients summarized in Table 3.
Clinical Study of PLD Combined with other Novel Agents
There may be the potential for synergism between topoisomerase II inhibitors PLD and topoisomerase I inhibitors such as topotecan in platinum-resistant disease,30,31 and in vitro data suggested a potential synergistic interaction between PLD and GEM.32,33 A median total response rate of 28% and clinical benefit of 72% were demonstrated, with a median TTP of 30+ weeks in the combination of PLD and topotecan for platinum-resistant disease (Table 4). 34 These data compare favorably to the data of both drugs administered as a single agent. Combination chemotherapy of PLD and GEM achieved good response rates ranging from 22% to 33%; however, the clinical benefit was between 28% and 61%, which was similar to PLD monotherapy. 35–38 As for hematotoxicity, grade 3/4 neutropenia was slightly higher and grade 2/3 PPE was slightly less (Table 5). The combination of PLD and GEM is an active and acceptably tolerated option for the treatment of patients with platinum-resistant ovarian cancer. These combinations at the chosen dosages seem suitable for this patient population. In a comparison of two studies of combination chemotherapy of PLD and oxaliplatin, the response rates of platinum-resistant disease were 28.6% and 38.5% and the clinical benefit was 71.4% and 76.9%, suggesting higher efficacy compared with PLD monotherapy.39,40 Furthermore, the response rates for platinum-sensitive disease were 66.7% and 81.5% and the clinical benefit was 82.8% and 100%, showing similar or better efficacy than other platinum combination chemotherapies. The incidence of PPE was low and no marked increase in hematotoxicity was found; consequently, toxicity was considered to be acceptable (Table 4). In patients with platinum-sensitive disease, the combination of PLD and trabectedin significantly improved PFS compared to PLD alone (9.2 months vs. 7.5 months, HR = 0.73, p = 0.0170), on the other hand, no significant difference in PFS was found in patients with platinum-resistant disease. 41 A RCT of the combination of PLD and canfosfamide and PLD alone was conducted in patients with platinum-resistant disease. Consequently, the median PFS was significantly longer for canfosfamide/PLD than PLD alone. Canfosfamide may ameliorate the adverse events of stomatitis and PPE associated with PLD. 42
Phase II studies of PLD-combination in platinum- and taxane-pretreated patients.
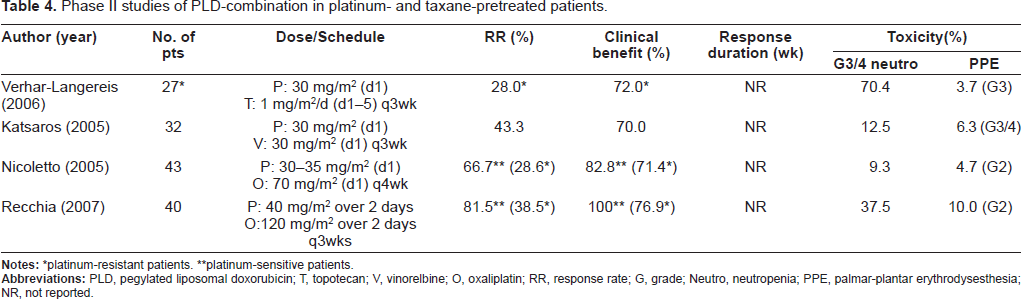
*platinum-resistant patients.
**platinum-sensitive patients.
Phase II studies of PLD + GEM in patients with platinum-resistant disease.
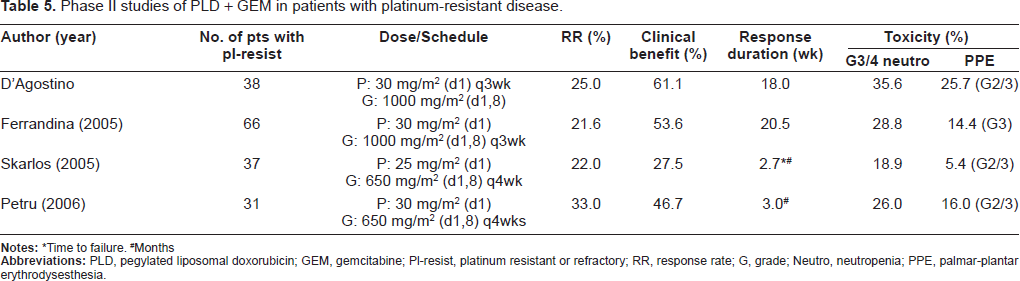
*Time to failure.
#Months
While these initial phase II trials are encouraging, they contain a relatively small number of patients, and no RCTs have been performed to date to confirm the benefit of combination therapy with PLD over mono therapy with PLD in the platinum-resistant population.
Patient Preference and Place in Therapy
Patients with platinum-resistant disease have a poor outcome, and most would like to prolong their survival with relieved symptoms and improved QOL. The effects of drugs on these patients are similar, and it is usually difficult to cure recurrent disease with one drug. Based on the performance status (PS), toxicity persisting from the initial treatment, and the bone-marrow function of an individual patient, the drugs that can be administered should be discussed with the patient. Since the results of phase II studies of PLD showed good outcomes,14–23 a large-scale study of initial chemotherapy with PLD was performed. 6 However, polychemotherapy of PLD associated with paclitaxel/carboplatin (TC) therapy and sequential doublet combination with carboplatin/PLD and TC therapies did not improve survival compared with TC therapy. Consequently, at present, PLD is regarded as a key drug for TC-refractory and resistant ovarian cancer with no cross-resistance to paclitaxel or carboplatin. PLD achieves good response rates, and many patients maintain long-lasting SD, which is one of the advantages. In addition, the clinical benefit is high in platinum-resistant disease and PLD is thus considered to be the first option. Since PLD has mild hematotoxicity, it is appropriate for patients with early recurrence after the completion of TC therapy, as well as heavily pretreated patients. It has also been reported that PLD did not induce multidrug resistance,43,44 which is one of the reasons for selecting PLD as the first option for patients with platinum-resistant disease. On the other hand, in a crossover study of PLD and GEM, PPE redeveloped when GEM was administered after patients had been treated with PLD. 25 This result suggests that much of the PPE observed in the PLD/GEM cross-over group may be the result of latent or delayed toxicity secondary to initial PLD treatment.
After explaining the characteristics of PLD, that PLD is safer for heavily pretreated patients than topotecan and GEM due to mild bone-marrow toxicity, but that PPE, stomatitis and mucositis frequently develop, the patient's wishes are considered, and the drugs are finally selected. The 1-hour infusion schedule every 4 weeks makes PLD easy to administer.
Conclusions
PLD (Doxil®) is doxorubicin HCl encapsulated in long-circulating STEALTH® liposomes. Although doxorubicin has been associated with a poor response in recurrent ovarian cancer, PLD is active, and is an emerging option for patients with platinum-refractory and -resistant disease. A phase II study has recently been completed in Japan, and the response rate for recurrent platinum-resistant ovarian cancer was 21.0%, the SD rate was 40.3% and the clinical benefit was 61.3%, which were similar to those in studies in Europe and the United States. 23
One of the toxicity characteristics of PLD is a low incidence and severity of hematotoxicity such as neutropenia.14–17,19–23 Also, Growth factor support are not frequently required in PLD, leading to advantages in healthcare costs. Overall, PLD proved to be preferable to topotecan and gemcitabine due to negligible hematologic toxicities.24–26 On the other hand, another characteristic of PLD is that the incidence of nonhematotoxity, including PPE, stomatitis and mucositis, is high at FDA-approved doses and schedules. PPE, also referred to as hand-foot syndrome, is a cutaneous reaction typically involving the palms of the hands and the soles of the feet. Although the cause of PPE is unknown, it is theorized that the long half-life and small size of the liposomes result in localization of the drug in areas of skin trauma. The incidence of grade 2 or higher PPE, which reduces the QOL, ranged from 20% to 50%; therefore, these adverse events should be appropriately treated during PLD administration. The severity of PPE can be decreased by dose modification, either decreasing the dose or lengthening the dosing interval. Dose modification often allows continued treatment without recurrence of PPE. Various pharmacologic approaches have been used, including topical dimethyl sulfoxide, pyridoxine (B6), and topical or systemic steroids. 45 We are examining the effects of cooling the wrists and ankles during infusion to prevent PPE. 46
Based on a review of previous studies,14–26 there are no differences in efficacy between 50 and 40 mg/m2 of PLD therefore, a dose of 40 mg/m2 is preferable in patients with platinum-resistant disease to reduce adverse events. To scientifically confirm the dosage, the Japanese Gynecologic Oncology Group (JGOG) plans to conduct a randomized comparative study of 50 and 40 mg/m2 of PLD in patients with recurrent platinum-resistant ovarian cancer (TFI < 6 months) (accrual of 350 patients, primary endpoint: PFS, secondary endpoints: OS, adverse events).
A rational approach to combining PLD with other drugs should take the slow accumulation and delayed peak of PLD in tumors into consideration. When combined with other useful agents, the lower dose of PLD (30 to 35 mg/m2) with a 3-week schedule may reduce severe PPE and stomatitis with negligible effects on the level of DI and the therapeutic efficacy. The response rate of combination chemotherapy of 30 mg/m2 of PLD and 650 to 1000 mg/m2 of GEM exceeded 20%, and the clinical benefit was similar to that of PLD monotherapy at 50 mg/m2.35–38 As for toxicity, hematotoxicity slightly increased but was easy to control, and nonhematotoxicity, such as PPE, slightly decreased. Furthermore, it was confirmed in a study of combination chemotherapy of topotecan, 34 vinorelbine, 47 oxaliplatin,39,40 trabectedin, 41 or canfosfamide 42 with PLD (30 mg/m2) that the response rate and clinical benefit were improved with reduced PPE. Drugs such as PLD with specific nonhematotoxicity and low hematotoxicity should be administered by combination chemotherapy rather than monotherapy to reduce the specific toxicity. In patients with platinum-sensitive disease with a TFI of 6 to 12 months (gray zone), PLD was more effective than topotecan or GEM. 24 In addition, in patients with platinum-sensitive disease, the response rate of PLD + GEM was 53.7%, the clinical benefit was 90.2% and the platinum free interval was simultaneously prolonged. Recently, in patients with platinum-sensitive relapsing ovarian cancer, the combination of PLD-carboplatin was not inferior to paclitaxel-carboplatin in terms of PFS, and was even found to be significantly superior. 48 The results of two other studies showed that combination chemotherapy of PLD and oxaliplatin achieved similar efficacy and safety as those of paclitaxel/ carboplatin or more. Therefore, the combination chemotherapy of PLD and platinum is one of the options for patients with platinum-sensitive disease with a TFI of 12 months or less.
Disclosures
The authors report no conflicts of interest.
