Abstract
Patients with relapsed/refractory T-cell acute lymphocytic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) have a dismal prognosis. Prior to the development of novel purine analogs, salvage chemotherapy was of limited efficacy. Nelarabine, and more recently, clofarabine and forodesine have demonstrated significant anti-tumor activity in relapsed/refractory T-ALL and T-LBL. As a single agent, nelarabine induces response rates in between 33% to 50% of adult or pediatric patients with T-ALL/T-LBL, respectively. On the other hand, limited activity was observed in mature T-cell neoplasms. Significant neurotoxicity has been the major obstacle for the further clinical development of nelarabine. Alternative dose-schedules for administration have been evaluated with some degree of success. Ongoing clinical studies are integrating nelarabine in the front-line chemotherapy regimens for patients with T-ALL/T-LBL in pediatric and young adult patients. The current work summarizes the pre-clinical and clinical evaluation of nelarabine, a chemotherapy agent whose development demonstrates that industry, government agencies, and academia can effectively collaborate together to improve the patient outcomes with highly resistant/refractory rare hematological malignancies.
Introduction
Acute T-cell leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) are two rare hematological malignancies, characterized by an abrupt and aggressive clinical course. 1 Although current front-line treatment results in complete remission (CR) in about 80% to 90% of the patients with T-ALL and T-LBL, half of the patients relapse within the first 24 months. 1 Patients who fail to achieve a CR or those who relapsed after initial remission have a dreadful prognosis. Highdose cytarabine–based regimens are associated with a remission rate of 50% in the salvage setting. 2 However, the duration of remission is short without consolidation therapy. Following a second remission, allogeneic hematopoietic stem cell transplant (SCT) continues to be the only treatment modality resulting in long lasting remission and improved survival. The benefit of allogeneic-SCT can be diminished by the risk involved (i.e. grade III-IV acute and chronic graft-versus-host disease).
The development and clinical evaluation of newer agents in patients with T-ALL and T-LBL is challenging given the low incidence and the speed in tumor growth of theses malignancies. Nelarabine is one of the two nucleoside analogues approved by the Food and Drug Administration (FDA) for the treatment of T-cell ALL and T-LBL. 3 Nucleoside and nucleobase anti-metabolites are the largest single class of chemotherapeutic agents. Pyrimidine derivatives have broader anti-tumor activity and have been found to be effective in patients with epithelial cancers (fluoropyrimidines, gemcitabine) or hematological malignancies (cytarabine, gemcitabine). On the other hand, purine analogs have anti-viral properties (acyclovir, ganciclovir) or anti-tumor activity in hematological malignancies (thiopurines, fludarabine, cladribine, clofarabine, nelarabine. 4
The development of more selective and less toxic nucleoside analogs in the treatment of lymphoid malignancies was crafted on the observation that patients with congenital deficiency of adenosine deaminase or purine nucleoside phosphorylase (PNP) have severe lymphopenia and selective lack of T- and/or B-cell lymphocytes.5,6
Nelarabine (also known as compound 506U78; Arranon) is a pro-drug that is demethoxyylated by adenosine deaminase to its active compound 9-β-D-arabinofuranosylguanine (Ara-G, See Fig. 1).7–11 The pre-clinical and clinical development of nelarabine demonstrated that sustained collaboration between academia, industry, and governmental agencies can result in viable therapeutic options for patients with rare malignancies who had exhausted all alternative options.
Ara-G was synthesized by Reist and Goldman in 1964. Subsequently, Elion in collaboration with Kurtzberg evaluated its anti-tumor properties in canine leukemia models. Because of her work in drug discovery, Elion was awarded the 1988 Nobel Prize in Medicine. 12 Subsequently, Cohen et al demonstrated that Ara-G was resistant to cleavage by PNP and induced cell death in T-cells at micromole concentrations. 8 Ara-G is difficult to synthesize using traditional techniques and it is poorly water soluble making it unsuitable for clinical studies. A group led by Krenitsky worked through the chemistry to overcome those obstacles and lead the invention of 506U78. Nelarabine, a compound that is 10 times more water soluble, was produced using advanced enzyme technology circumventing the initial production problems with Ara-G.
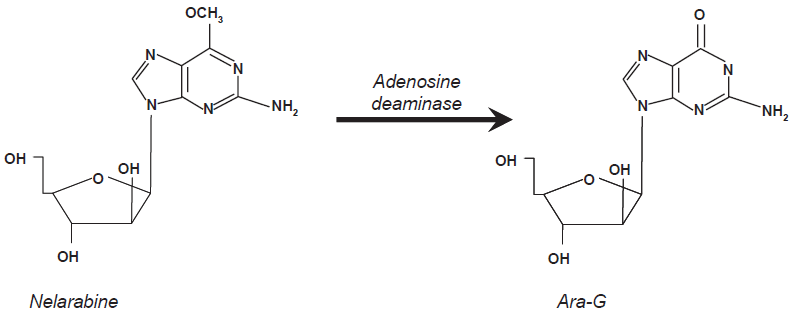
Chemical structure and activation pathway of nelarabine into Ara-G. A key-metabolic step mediated by adenosine deaminase.
Nelarabine chemically is a pro-drug of Ara-G. In vivo, it is demethoxylated by adenosine deaminase to form the active compound. Rapid intracellular phosphorylation of Ara-G by deoxyguanosine kinase and deoxycytidine kinase phosphorylate form Ara-GTP. Rodriguez et al demonstrated that Ara-G is more toxic to human malignant T-cell lines than it is to malignant B-cells, in some cells by at least a factor of 10.13,14 The accumulation of Ara-GTP is more efficient in T-cell than in B-cells which explains, to a certain degree, the pharmacological effects of nelarabine in cell lines and in T-ALL and T-LBL.
Phase I Clinical Studies with Nelarabine
Kurtzberg et al conducted the first phase I study to determine the maximum-tolerated dose (MTD), toxicity profile, and pharmacokinetics of nelarabine in pediatric and adult patients with refractory hematologic malignancies. 15 Ninety-three patients with relapsed/refractory hematological malignancies were enrolled. Nelarabine was intravenously administered daily as a one hour infusion for 5 consecutive days. Patients were enrolled at different dose levels of 5, 10, 20, 40, 60 or 75 mg/kg/day. A 90 mg/kg/day cohort was initially planned; however, as toxicity was found at the 75 mg/kg/day cohort the protocol was amended. In addition, and to facilitate subsequent clinical trials, the milligram per kilogram dosing regimen was converted to a dose per square meter of body surface area. The amended protocol evaluated the dose 1.2 g/m2/day, in 20 patients, which was the dose eventually used in the following clinical trial. This dose is equivalent to 30 mg/kg in adults and 40 mg/kg in children. The study included 59 adults and 34 children with a median number of two previous treatments, 66% had T-cell malignancies (39 patients with T-ALL/T-LBL), 22% B-cell malignancies and 12% myeloid or other hematologic malignancies. More than half of the patients received only one cycle of nelarabine. The most common non-hematological toxicity was neurotoxicity and dose-limiting toxicity was seen in one pediatric patient at the 75 mg/kg/day dose. No grade 3 or 4 toxicity was reported during the first course of therapy in any pediatric patient treated at dose levels ≤60 mg/kg/d or any adult patient treated at <30 mg/kg/day, including patients dosed at the 1.2 g/m2/day dose level. Neurologic adverse events associated with nelarabine were documented in 72% of the patients enrolled in the study, 50% and 85% of the pediatric or adult patients respectively; Most of the neurological toxicity observed was reversible. Neurological toxicity varied from somnolence, confusion, ataxia, peripheral motor or sensory neuropathies, paralysis, encephalopathy, and seizures. A syndrome similar to Guillian-Bare syndrome was observed in some patients. The severity of the neurological toxicity was dose-related. 15
Hematologic toxicities were not cumulative or dose-limiting. Grade 1 and 2 neutropenia and thrombocytopenia were observed in 50% and 76% of pediatric patients and 46% and 42% adult patients, respectively.
The overall response rate was 31%, including 10 patients with complete responses (CR) and 19 with partial responses (PR). Major responses were seen in patients with T-ALL and T-LBL. Complete remission and PRs were observed in 23% and 31% of the patients with T-ALL, respectively. Pharmacokinetic parameters were similar in children and adults. Accumulation of Ara-GTP in leukemic blasts was correlated with cytotoxic activity. 15
A second phase I study was conducted in an effort to minimize neurologic toxicity and optimize the anti-tumor activity of nelarabine. Adult patients with refractory hematological malignancies (n = 39), including 28 patients with relapsed/refractory T-ALL/T-LBL received nelarabine at a dose of 1.5 g/m2/day on days 1, 3 and 5 infused over 2 hrs. Dose escalation was planed, however the MTD was found to be 2.2 g/m2/day. Of the 28 T-ALL or T-LBL patients, 5 achieved a CR. Based on the phase I studies, the dose of 1.5 g/m2/days on days 1, 3 and 5 was selected for further phase II studies in adults. 3
Phase II Clinical Trials
The first phase II study in pediatric patients with relapsed/refractory T-ALL/T-LBL was conducted by the Pediatric Oncology Group (POG) and reported by Berg et al Pediatric patients were stratified in four cohorts: 1) >25% bone marrow blasts in first relapse; 2) >25% bone marrow blasts in second or subsequent relapse; 3) central nerve system (CNS) relapse or 4) extramedullary (non-CNS) relapse. The planned nelarabine dose was 1.2 g/m2 daily for 5 consecutive days every 3 weeks for all cohorts of patients. Due to neurotoxicity encountered early in the study, the dose was de-escalated and reduced to 650 mg/m2/day for cohorts 1 and 2 and 400 mg/m2/day for cohorts 3 and four. 16
A total of 153 patients were registered into the study and several patients were removed for the efficacy analysis (17 patients) or toxicity analysis (2 patients). Response rates varied between the four cohorts of patients studied. Patients with T-ALL/T-LBL at first relapse (cohort 1) had a response rate of 55%, including 16 CR's and 2 PR's. The response rate for patients with T-ALL/T-LBL in second or greater relapse dropped to 27%, including 7 CR's and 1 PR. Similarly, the response rate for relapsed T-ALL/T-LBL with CNS disease was 33% including 5 CR's and 2P R's. Finally, patients with etramedullary relapse had the lowest overall response rate (14%), without a single patient achieving a CR. It is important to note that patients in cohorts 3 and 4 received a lower dose of nelarabine (400 mg/m2/day) than patients in the first two cohorts (650 mg/m2/day).
Non-hematological toxicity was primarily CNS related. Grade 3 or 4 neurological adverse events occurred in 18% of the patients. The type of CNS toxicity seen was similar to that observed in phase I studies. The severity appeared to be, once again, dose dependent. This study confirmed the anti-tumor activity of nelarabine in patients with relapsed/refractory T-ALL and T-LBL. In addition, it stressed the need for more clinical studies to evaluate this agent in other T-cell malignancies and to explore alternative dose-schedules to diminish the significant neurological toxicity encountered with nelarabine. 16
DeAngelo et al evaluated the anti-tumor activity of nelarabine single agent in patients with relapsed/refractory T-ALL/T-LBL using a different dose-schedule of administration. The study was conducted by the Cancer and Leukemia Group B (CALGB) and enrolled a total of 39 patients who were either refractory or relapsed to at least one multi-agent regimen T-ALL (26 patients) or T-LBL (13 patients). Patients were treated with an alternate day schedule (days 1, 3, and 5) of nelarabine at 1.5 g/m2/day. Cycles were repeated every 21 days. Clinical responses were observed in 41% of the patients, with 31% CR. The median disease-free survival (DFS) was 20 weeks and the overall survival was also 20 weeks. The 1-year survival rate for all treated patients was 28% and 7 patients subsequently underwent an allogeneic bone marrow transplant. 17
With this schedule of administration, the principal toxicity was grade 3 or 4 neutropenia (37%) and thrombocytopenia (26%). Of interest, and in contrast to what was observed in previous clinical trials, CNS toxicity was mild (grade 1 or 2). Peripheral neuropathy (Grade 1-2) was reported in 14 patients (37%) and peripheral motor neuropathy was seen in 21% of the patients (one grade 3). Grade 4 neurological toxicity was seen in one patient (reversible depressed level of consciousness). 17 The results of this study demonstrated comparable anti-tumor activity to the previous dose-schedule tested and a better toxicity profile. Subsequently, nelarabine was evaluated in mature T-cell lymphomas.
Czuczman et al reported on behalf of the CALGB, the results of the first phase II study evaluating the anti-tumor activity of nelarabine in patients with untreated cutaneous T-cell lymphoma (CTCL) and refractory/relapsed systemic mature T-cell lymphoma. Nelarabine was administered at 1.5 gr/m2/day over 2 hrs on days 1, 3, 5 and cycles were repeated every 21 days. Cycles were continued up to two cycles beyond CR up to a maximum of eight cycles. Between 2000 and 2004 nineteen patients were enrolled in the study, subsequently, the study was closed due to neurological toxicity. The final analysis included 19 patients, 11 with CTCL and eight systemic mature T-cell lymphoma patients. The limited activity observed in this cohort of patients was offset by the significant toxicity encountered. There were only 2 patients who achieved PR's, no CR's were documented. The duration of the remission was short lived (3 and 5.5 months, respectively). Grade 3 or 4 adverse events were documented in 50% and 28%, respectively. 18 Median event-free survival was 1.2 months and median overall survival was 3 months. In particular, 33% of patients experienced Grade 3 or 4 neurologic toxicities. In contrast to the favorable results observed in patients with T-ALL/T-LBL, nelarabine single agent had limited efficacy in adult patients with CTCL and mature T-cell lymphomas.
Pharmacokinetics of Nelarabine
The pharmacokinetics of nelarabine had been studied in both, adults and pediatric patients. Data is available from 101 adult patients and 25 pediatric patients with refractory hematological malignancies.2,19,20 These patients had received different doses of nelarabine raging from 104 to 2,900 mg/m2/day. Nelarabine is rapidly converted into Ara-G in the circulation and the plasma concentrations of both compounds decline rapidly in a monoexponential fashion following a 2-hour intravenous infusion. The area under the curve (AUC) of Ara-G is 36 times higher than nelarabine, implying that Ara-G drug exposure in the circulation is higher than nelarabine (162 μg/ml/hr vs. 4.4 μg/ml/hr, respectively. 3 Both molecules exert liner pharmacokinetics and are similar for pediatric and adult patients.
The maximum concentration (Cmax) of nelarabine was observed at the end of the infusion and the Cmax increased linearly with the nelarabine dose. 19 The median Cmax was not different between pediatric or adult patients, 0.51+/-0.60 μmol/Lvs.0.49+/-0.42 μmol/L, respectively (P = 0.47). The harmonic mean half-life (t1/2) of nelarabine in pediatric and adult patients was 14.1 minutes and 16.5 minutes, respectively.
In vivo conversion into Ara-G occurred rapidly during nelarabine infusions. The Cmax of Ara-G typically occurred at the end of the nelarabine infusion and was proportional to the nelarabine dose. 19 The Ara-G Cmax values ranged from 11.6 μmol/L to 308.7 μmol/L and were not different between pediatric or adult patients. Of interest, the plasma clearance (CL) of Ara-G was 46% higher in pediatric patients as compared with adult patients (P = 0.0001). Ara-G AUC in adults patients was significantly higher (5.28 +/-9.93 μmol/L/hr) than in pediatric patients (3.59 +/-1.21 μmol/L/hr) (P < 0.0001). The median t1/2 of Ara-G was 30% shorter in pediatric patients (2.1 hrs) than in adult patients (3 hrs). 19 A similar Ara-G median t1/2 (ranging between 3 to 4 hrs) was observed in a separate cohort that included mostly adult patients. 20 It is uncertain if these differences play a role in the severity of neurotoxicity's observed in pediatric versus adult patients treated with nelarabine.
In addition to standard pharmacokinetic studies, Gandhi et al evaluated the intracellular accumulation of nelarabine and its metabolites in patients with hematological malignancies enrolled in one of the nelarabine phase I studies. A total of 26 patients participated in this analysis, who received nelarabine at doses ranging from 20 to 60 mg/kg/day on days 1 to 5. In this patient population, T-lymphoblast isolated from the peripheral blood of patients with T-ALL/T-LBL accumulated higher levels of Ara-GTP, nelarabine active metabolite, (median, 140 μmol/L; n = 7) compared with other diagnoses (median, 50 μmol/L; n = 9) and normal mononuclear cells (n = 3) (P = 0.0008). In addition, patients responding to nelarabine accumulated significantly higher levels of Ara-GTP (median, 157 μmol/L) compared with patients who failed to respond (median 44 μmol/L) (P = 0.0005). 20
The results of this work provide some insightful information regarding nelarabine anti-tumor activity in T-ALL/T-LBL. It is uncertain if the same principles can be applied to explain the selective CNS toxicity observed in clinical trials.
Effects of Nelarabine on the Central Nervous System
It is possible that Ara-G is accumulated at higher concentrations in neurons, as it has been demonstrated in T-cell lymphocytes, explaining the neurotoxicity observed in pre-clinical studies and clinical trials. In dose finding studies in non-human primates, the predominant toxicities were neurologic, with tremors, weakness, and/or ataxia observed especially when nelarabine was administered while animals were under ketamine anesthesia. The toxicity was different between animals studied. In mouse models the administration of nelarabine at 500 mg/m2 as associated with shallow breathing, tremors, and decrease activity. On the other hand, dogs tolerated much higher doses of nelarabine without significant CNS toxicity. The toxicity observed in non-human primates (i.e. monkeys) was very similar to that observed in clinical trials. When nelarabine was administered at doses higher than 3,600 mg/m2 to monkeys, death occurred from acute CNS toxicity.3,21
Acute CNS toxicity was more noticeable when monkeys were anesthetized with ketamine prior to nelarabine treatment and it was characterized by paralysis, tremors, seizures, and death. Autopsy revealed significant damage in the brain parenchyma such as cerebellar degeneration and perivascular cuffing in the cortex and/or cerebellum. Similar findings were found in the spinal cord. A postulated mechanism to explain the acute neurotoxicity observed with nelarabine, is an extremely high intracellular accumulation of Ara-G and its subsequent phosphorilation to Ara-GTP in neurons, leading to disruption in RNA synthesis and signal transmission. It has been demonstrated that brain and nerve tissue express high levels of deoxyguanosine kinase activity the enzyme that convert Ara-G into its active and lethal metabolite Ara-GTP.3,21
In addition to acute CNS toxicities, nelarabine administration is associated with delayed neurological manifestations including a clinical form of ascending paralysis similar to Guillian-Barre syndrome (GBS). Monkey studies suggest that nelarabine active metabolite, Ara-GTP disrupts the transport of neurotransmitters affecting the function of GTP-dependent proteins located in the long axons and leads to generalized degradation in neuronal function. 3
Summary
Nelarabine is an effective chemotherapy agent with a narrow therapeutic spectrum and dose-dependent significant non-hematological toxicity profile. The role of nelarabine in the treatment of relapsed/refractory T-ALL/T-LBL is well supported by previous phase I/II clinical trials. It appears to be that responses are higher when used in patients in first relapse and the toxicity is acceptable when used at daily doses of 400-650 mg/m2/day for five days or alternated schedule (days 1, 3, 5) at 1 gr/m2/day. Pharmacological studies have provided insightful information in defining not only the group of patients most likely to benefit from nelarabine treatment (i.e. patients who concentrate higher intracellular levels of Ara-G in malignant T-cells) but also the mechanisms responsible for the neurotoxicity observed in clinical trials.
The clinical approval of nelarabine for the treatment of T-ALL/T-LBL has paved the road for the further development of other potent purine analogs such as clofarabine or forodesine with a safer toxicity profile for patients with relapsed/refractory immature T-cell malignancies. Ongoing clinical studies are seeking to define the role of nelarabine in combination with other chemotherapy agents in the front-line setting.
Disclosure
The authors report no conflicts of interest.
