Abstract
Background:
Deregulated Notch signaling is implicated in non-Hodgkin lymphoma (NHL) and chronic lymphocytic leukemia (CLL). Crenigacestat (LY3039478) prevents cleavage of Notch proteins and may benefit patients with relapsed or refractory NHL or CLL.
Objectives:
This phase I clinical trial assessed the safety and efficacy of crenigacestat in patients with relapsed or refractory NHL and CLL. The main objectives were to characterize the safety profile, to confirm the recommended phase II dose of crenigacestat in patients with hematological malignancies, and to assess preliminary antitumor activity.
Design:
A phase I trial enrolling patients with relapsed or refractory NHL and CLL, with Notch tumor alteration based on molecular or immunohistochemistry tumor pre-screening.
Methods:
Eligible patients received crenigacestat 50 mg orally three times per week, for a 28-day cycle, until disease progression or unacceptable toxicity. Tumor responses were assessed using the Revised Response Criteria for Malignant Lymphoma and the National Cancer Institute Working Group for CLL.
Results:
Overall, 62 patients (40 with NHL and 22 with CLL) were pre-screened for a Notch alteration. Notch alteration was identified in 21/62 (34%) of patients pre-screened. Nine patients (five with peripheral T-cell NHL and three with CLL) with Notch alteration were eligible for the clinical trial and treated. The most common adverse events in all grades of severity were diarrhea (56%), nausea (56%), platelet count decrease (44%), and fatigue (33%). One patient (11%) with peripheral T-cell lymphoma obtained a partial response.
Conclusion:
Crenigacestat demonstrated a modest clinical activity at the recommended dose in adult patients with relapsed or refractory NHL or CLL.
Trial registration:
NCT01695005.
Plain language summary
Background:
Some rare cancers of the blood or lymph nodes, such as non-Hodgkin’s lymphoma (NHL) and chronic cellular leukemia (CLL), are difficult to treat when they relapse and require the search for new treatments. Crenigacestat (LY3039478) is a potential new oral treatment evaluated in this clinical study.
Results:
The clinical study was a first in humans. Among 62 patients enrolled with relapsed nonHodgkin’s lymphoma (NHL) and chronic cellular leukemia (CLL), 9 patients had the molecular abnormality in the tumor compatible with the study treatment and were selected and treated with crenigacestat (LY3039478). Patients received crenigacestat orally, 3 times a week, at a dose of 50 mg. Among the 9 patients treated, 1 obtained a partial response.
Conclusion:
Crenigacestat demonstrated modest clinical activity in adult patients with relapsed/refractory NHL or CLL.
Introduction
Advances in the treatment of non-Hodgkin’s lymphoma (NHL) have been made over the past three decades using chemotherapy in combination with immunotherapies; however, about 40% of patients have a disease that relapses or is refractory to these treatments. 1 The management of relapsed or refractory NHL represents a major therapeutic challenge, particularly in patients with disease resistant to immunochemotherapy or early relapse.2,3 NHL can be classified into B and T lymphomas, and T lymphomas are more difficult to treat with a more pejorative prognosis. 4
Notch signaling is highly conserved in the human evolution pathway that plays a role in the development and tissue homeostasis.5,6 Notch signaling encompasses receptors and ligands containing single-pass transmembrane domains that are expressed on the cell surface. 5 Deregulated Notch signaling could be associated with mutation or overexpression of ligands and/or receptors and this deregulation is an oncogenic event5,7,8 and hematological malignancies including chronic lymphocytic leukemia (CLL) and lymphoma.9–14 The oncogenic functions of Notch signaling involve the inhibition of apoptosis and the promotion of cell proliferation. 7 Gamma-secretase inhibitors inhibit Notch signaling by preventing cleavage of the transmembrane domain of Notch protein. 15
Crenigacestat is an oral potent Notch inhibitor that prevents the release of the Notch intracellular domain (NICD) by inhibiting the proteolytic activity of the gamma-secretase complex. Crenigacestat decreases Notch signaling and is associated with downstream biological effects. 16 The clinical activity of the Notch inhibitor in patients with solid tumors16–18 and hematological malignancies including T-cell acute lymphoblastic leukemia 19 has been recently investigated demonstrating antitumor activity. Clinical activity was observed with tumor necrosis, metabolic response, or tumor shrinkage in patients with breast cancer, leiomyosarcoma, and adenoid cystic carcinoma.16–18 The recommended monotherapy dose was 50 mg 3 times a week (TIW), based in results from the first-in-human study of crenigacestat.16–18 Main toxicities dose-limiting were thrombocytopenia, colitis, and nausea. 16
The primary objectives of this study were to investigate the recommended phase II dose of crenigacestat in patients with hematological malignancies and to document preliminary efficacy. Secondary objectives were to define the safety profile, to estimate the Pharmaco-kinetics (PK) parameters, and to evaluate the duration of response.
Patients and methods
Study design and treatment
I6F-MC-JJCD is a multicenter, non-randomized, open-label, dose-escalation (part A) followed by cohort expansions in patients with advanced or metastatic cancer or relapsed or refractory NHL or CLL (part D) (ClinicalTrials.gov identifier NCT01695005). Results of part A of the study were reported previously 16 and the recommended phase II dose was established for crenigacestat orally at 50 mg 3 TIW, on a 28-day cycle. The results dose expansion cohort in patients with NHL or CLL, are presented in this article. All adult patients must have histological evidence of advanced NHL or CLL and pre-screened alterations in the Notch pathway such as mutations, amplification, or gene expressions related to Notch as described in Table S1. Overall, 62 patients with relapsed or refractory NHL or CLL were pre-screened for Notch alterations, 10 patients with relapsed or refractory NHL or CLL were enrolled in the clinical trial and 9 patients with relapsed or refractory NHL or CLL received at least one dose of crenigacestat (Figure 1). Recruitment periods for patients with hematological cancers were between January 2016 and October 2017.

Flowchart of the study.
All eligible treated patients received orally 50 mg of crenigacestat 3 TIW on a 28-day cycle, monotherapy, at phase II recommended dose defined in a previous study. 16 Crenigacestat was administered until symptomatic or confirmed progressive disease, unacceptable toxicity, or other reasons for the study drug discontinuation.
Ethics
The study was conducted in compliance with the Declaration of Helsinki, the Council for International Organizations of Medical Sciences International Ethical Guidelines, and the International Conference on Harmonization Guidelines for Good Clinical Practice and applicable local regulations. The ethics committees of all participating centers approved the protocol, and all patients provided written informed consent before study entry. The name of the ethics committee that approved the protocol in France was the Committee for the Protection of People Sud-Est IV, Center Léon Bérard, Lyon Cedex 08, with approval number CPP 16-037.
Patients
Key inclusion criteria were having histological evidence of advanced or metastatic cancer with preselected alterations in the Notch pathway. Participants were required to have relapsed or refractory CLL, mature T cell, B cell, or natural killer (NK) cell non-Hodgkin’s neoplasm. All eligible patients were at least 18 years old, must have at study entry an Eastern Cooperative Oncology Group performance status (ECOG PS) score of 0 or 1, must have adequate organ and hematologic functions, were 18 years or older, and had measurable disease according to disease international criteria for lymphoma 20 or CLL. 21 Key exclusion criteria were having a symptomatic or unstable central nervous system malignancy; have an active bacterial, fungal, and/or viral infection; have malabsorption syndromes, enteropathies, gastroenteritis (acute or chronic), or diarrhea (acute or chronic).
Study assessments
Efficacy assessments of tumor responses were measured using the Revised Response Criteria for Malignant Lymphoma, 20 the Guidelines from the National Cancer Institute (NCI) Working Group for CLL.19,21 Objective response rate was defined as the proportion of patients who achieved a complete response or partial response (PR) in evaluable patients who received at least two cycles of treatment. A PET scan was used to evaluate the treatment effect of crenigacestat in this study. Pharmacokinetic assessments intensive PK samples were obtained in part A during the dose escalation phase, 17 and sparse PK samples were obtained in patients in part D.
Safety assessments
As defined in Part A of the study and previously reported,17,18 all adverse events (AEs) were coded according to the Medical Dictionary for Regulatory Activities, version 20.0, and graded by the NCI’s Common Terminology Criteria for Adverse Events (CTCAE) 4.03. A dose-limiting equivalent toxicity was defined as an AE during cycle 1 that is related to crenigacestat and fulfills any one of the following criteria using the NCI CTCAE v 4.03 CTCAE grade III non-hematological toxicity (exceptions made for nausea, vomiting, or constipation that lasts <72 h and can be controlled with treatment; transient grade III elevations of alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST)), CTCAE grade IV hematological toxicity of >5 days duration, any febrile neutropenia, grade III thrombocytopenia with bleeding or grade IV thrombocytopenia and other significant toxicity deemed to be dose limiting by the investigator.
Pre-screening selection of patients
The pre-screening for Notch signaling alteration in the tumor was performed in a pretreatment archival diagnostic biopsy as formalin-fixed, paraffin-embedded (FFPE) tissue blocks or unstained at 4–5 µm sections. The tumor tissue was stained by immunohistochemistry with the cleaved Notch1 (Val1744; D3B8) NICD rabbit monoclonal antibody (Cell Signaling Technology, Danvers, MA, USA). Results were interpreted and scored by a board-certified pathologist (J.-Y.S). Specimens were scrutinized for the level of endogenous background signal by examining additional sections using an isotype control. Control tissues were processed in parallel with tissues exposed to the primary antibody. Immunohistochemical results were scored by a qualitative method based on an assessment of the immunoreactivity observed in the specimen using a scale of 0–3+, translating to no (0), weak (+), moderate (+), or intense (+) staining, respectively. A cutoff of ⩾10% tumor cells with specific nuclear staining with ⩾1+ immunoreactivity was used to determine positive immunoreactivity.
Patients with CLL with circulating tumor lymphocytes were pre-screened from a blood sample. The lymphocytes were cell-sorted, and the tumor lymphocyte cells were isolated. DNA extraction from the tumor lymphocyte cells was performed. For patients with CLL, a next-generation sequencing (NGS) panel of 31 genes was performed including a search for mutations of NOTCH1 and FBXW7. The details of all the genes analyzed by the NGS panel are indicated in Table S2.
Statistical analyses
Data from all patients who received at least one dose of crenigacestat treatment were included in analyses of safety and efficacy. A sample size of 10 patients enrolled in the clinical trials was estimated to be enough to estimate the preliminary response rates for this stage of clinical development and to explore the efficacy in the subsets of interest. Change in the tumor size was assessed in each patient with NHL using computed tomography (CT) imaging and based on Revised Response Criteria for Malignant Lymphoma. 20 Changes in the tumor size were evaluated in each patient with CLL using circulating tumor lymphocyte count according to the Guidelines from the NCI Working Group for CLL. 21 The reporting of this study conforms to the CONSORT statement. 22 Roles and responsibilities of the study sponsor and funders were study design; data collection, management, analysis, and interpretation of data collected; and writing the study report. The investigators’ roles and responsibilities were all activities in patient management and site activities regarding the protocol, on-site data collection, and writing the manuscript.
Results
Pre-screening of patients with hematological malignancies for alteration in Notch signaling
Overall, 62 patients with relapsed or refractory NHL or CLL (40 patients with NHL and 22 patients with CLL) were pre-screened for alteration in the Notch pathway. Patients with NHL were pre-screened by immunohistochemistry on NICD and the overall positivity rate was 35% (14 patients found positive in 40 patients tested). According to lymphoma subtypes, the positivity rate was 52% in patients with peripheral T-cell lymphoma (PTCL), 20% in patients with cutaneous T-cell lymphoma (CTCL), and 14% in patients with Diffuse Large B- Cell Lymphoma (DLBCL) (Figure S1). Twenty-two patients with relapsed or refractory CLL were pre-screened in sorted tumor circulating lymphocytes in blood by NGS. Seven (32%) of the 22 tested patients were found with molecular alteration in the Notch pathway (6 patients with NOTCH1 mutation and 1 patient with FBXW7 mutation). The most frequent mutation in CLL-mutated patients involved NOTCH1 and was c.7541_7542delCT implicating at the protein level p.Pro2514fs. The detailed molecular abnormalities in CLL-mutated patients are indicated in Table S3. The results of screening on tumor tissue by NICD (immunohistochemistry) for T-cell lymphomas are detailed by subtypes in Table S4. Five CTCLs were tested and 21 PTCLs were tested. Among the different subtypes, angioimmunoblastic T-cell lymphoma (AITL) was positive in 7/12 (58%) patients. Among other T-cell lymphomas, they were positive in 4/9 (44%).
Characteristics of patients eligible and treated with crenigacestat
The flow chart of patients with relapsed or refractory NHL or CLL pre-screened for Notch alterations and treated with crenigacestat in the clinical trial is summarized in Figure 1. In total, 10 patients were included in the I6F-MC-JJCD clinical trial and 9 patients received at least one dose of crenigacestat; 1 patient was screened for failure due to not measurable lymphoma disease on CT baseline scan.
The nine patients (one woman and eight men) with NHL or CLL eligible and treated with crenigacestat monotherapy have peripheral T-cell NHL (n = 5), CLL (n = 3), and CTCL (n = 1) (Table 1). The first treated patient was included on March 9, 2016. Median age of patients was 64 years (range 39–81). The median of prior lines of systemic therapy was 4 (range 2–10). Two (22%) patients received prior study entry auto-stem cell transplantation. Seven out of the nine patients (78%) of patients had ECOG PS at 1 at study entry. All six patients with T-cell lymphoma included and treated in the study had extensive disease at study entry with Ann Arbor stage III or IV. The three patients with CLL included and treated in the study were previously exposed to a BTK inhibitor and had failed this treatment.
Patient’s characteristics, with non-Hodgkin lymphoma or CLL, included in the clinical trial I6F-MC-JJCD.

The auto-stem cell transplant was not counted as a systemic treatment line.
CLL, chronic lymphocytic leukemia; ECOG, Eastern Cooperative Oncology Group.
Safety and tolerability
Most events in the nine patients receiving crenigacestat were mild to moderate in CTCAE-evaluated severity. The most frequent treatment-related AEs were diarrhea and nausea experienced by five patients (56%). Other AEs were platelet count decrease (n = 4; 44%), fatigue (n = 3; 33%) patients, and rashes (n = 2; 22%). One-third of patients had grade 3 or higher diarrhea. The AE profile detailed in Table 2 provides additional insight into the safety of Notch inhibitors (Table 2). No patient with NHL or CLL had a defined dose-limiting toxicity. Two patients had a crenigacestat dose reduction beyond the first cycle due to AE: one for grade 3 diarrhea and another for grade 3 nausea. Serious AEs were summarized in Table 3. The treatment drug discontinuation reasons were reported in Table 4. The main reason for discontinuation of the study was disease progression (6/9 patients; 67% of patients). Two patients withdrew from the study on the investigator’s decision. One patient died during treatment due to cardiac causes (unrelated to the study treatment).
All TEAEs with crenigacestat 50 mg were given orally three times a week in patients with non-Hodgkin lymphoma or CLL (extension part of the I6F-MC-JJCD study).
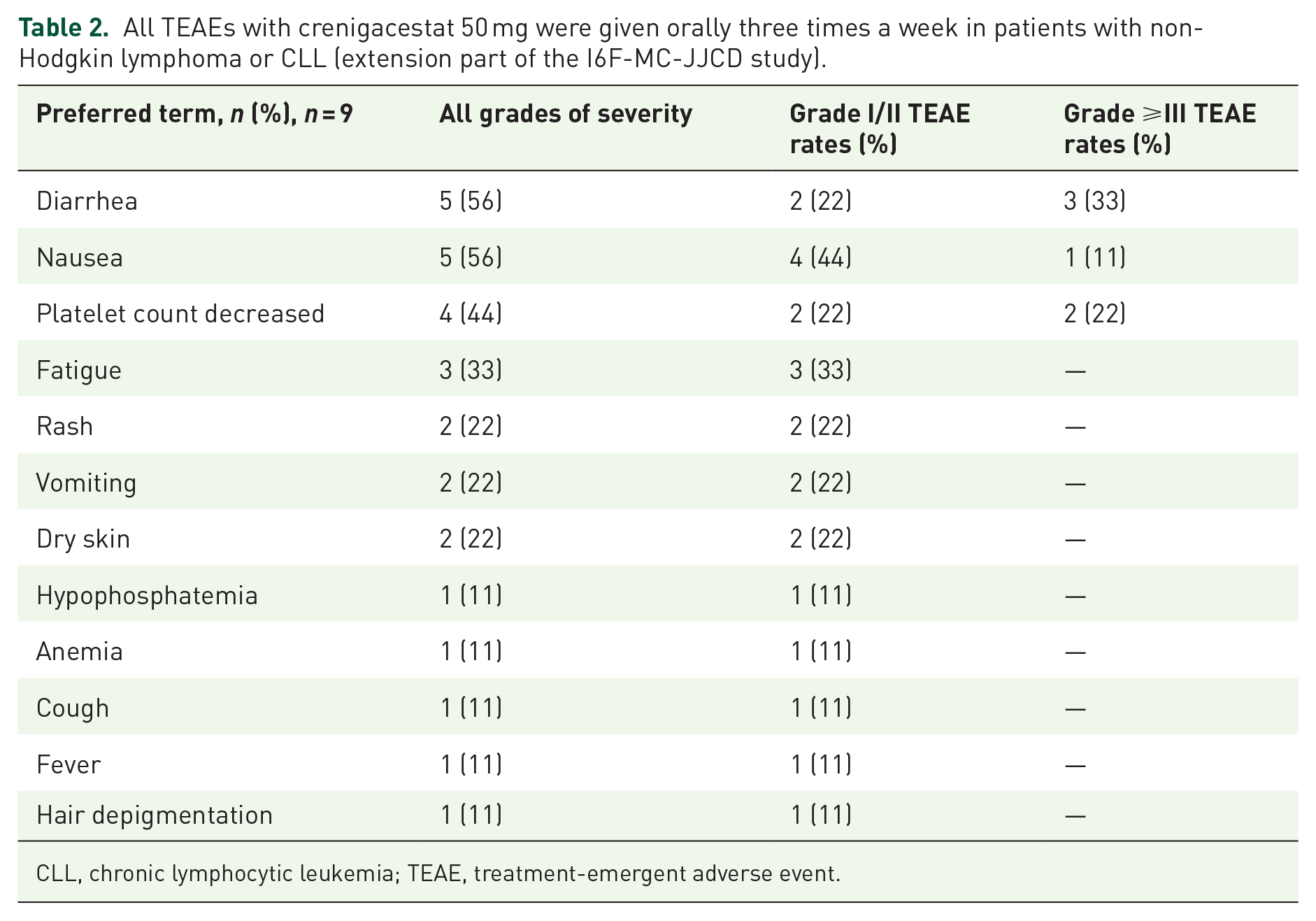
CLL, chronic lymphocytic leukemia; TEAE, treatment-emergent adverse event.
Treatment-emergent SAEs in a safety population of patients with relapsed or refractory non-Hodgkin lymphoma or CLL (n = 9) treated with crenigacestat.

This table summarizes all the SAEs that occurred in the study in the nine patients in the population with lymphoma or CLL. The SAEs are all reported in the table regardless of their relationship with the crenigacestat or the procedures of the protocol. SAE terms were summarized according to the preferred term of the Medical Dictionary for Regulatory Activities.
CLL, chronic lymphocytic leukemia; CTCAE, Common Terminology Criteria for Adverse Events; NCI, National Cancer Institute; SAE, serious adverse event.
Treatment/study drug discontinuation reasons in the safety population of patients with relapsed or refractory non-Hodgkin lymphoma or CLL (n = 9) treated with crenigacestat.

The death event in this patient was related to myocardial infarction not related to the study treatment.
CLL, chronic lymphocytic leukemia.
Pharmacokinetics
Pharmacokinetics were evaluated in the nine treated patients with hematological malignancies. As observed in previous studies,17,18 we observe a peak plasma concentration (Cmax) at approximately 2 h after administration of 50 mg oral doses of crenigacestat three times daily. Pharmacokinetic parameters were similar to those calculated from patients who underwent intensive pharmacokinetic sampling in the dose-escalation portion of study I6F-MC-JJCA. 18
Efficacy
A summary of the response rates is shown in Table 5 and tumor changes are shown in Figure 2. As per Revised Response Criteria for Malignant Lymphoma, one patient (11%) out of the nine patients had confirmed partial remission, and the duration of anti-tumoral response reached 5.1 months (Figures 2 and 3). This patient had PTCL with AITL subtype and the PR was illustrated by PET-CT imaging in Figure 3, with cervical lymph nodes and axillary lymph nodes regression. As only one patient achieved an objective therapeutic response, the median duration of response did not apply to the results of the study.
Summary of response rates.

CLL, chronic lymphocytic leukemia.

Spaghetti plot of antitumor activity of crenigacestat in patients with relapsed or refractory hematologic malignancies. Patients treated for lymphoma (represented by full lines) were monitored for target lesion size according to the CHESON International Criteria for the Evaluation of Malignant Lymphoma by CT scan. Patients with chronic lymphocytic leukemia (represented by dashed lines) were monitored for circulating tumor lymphocyte counts.
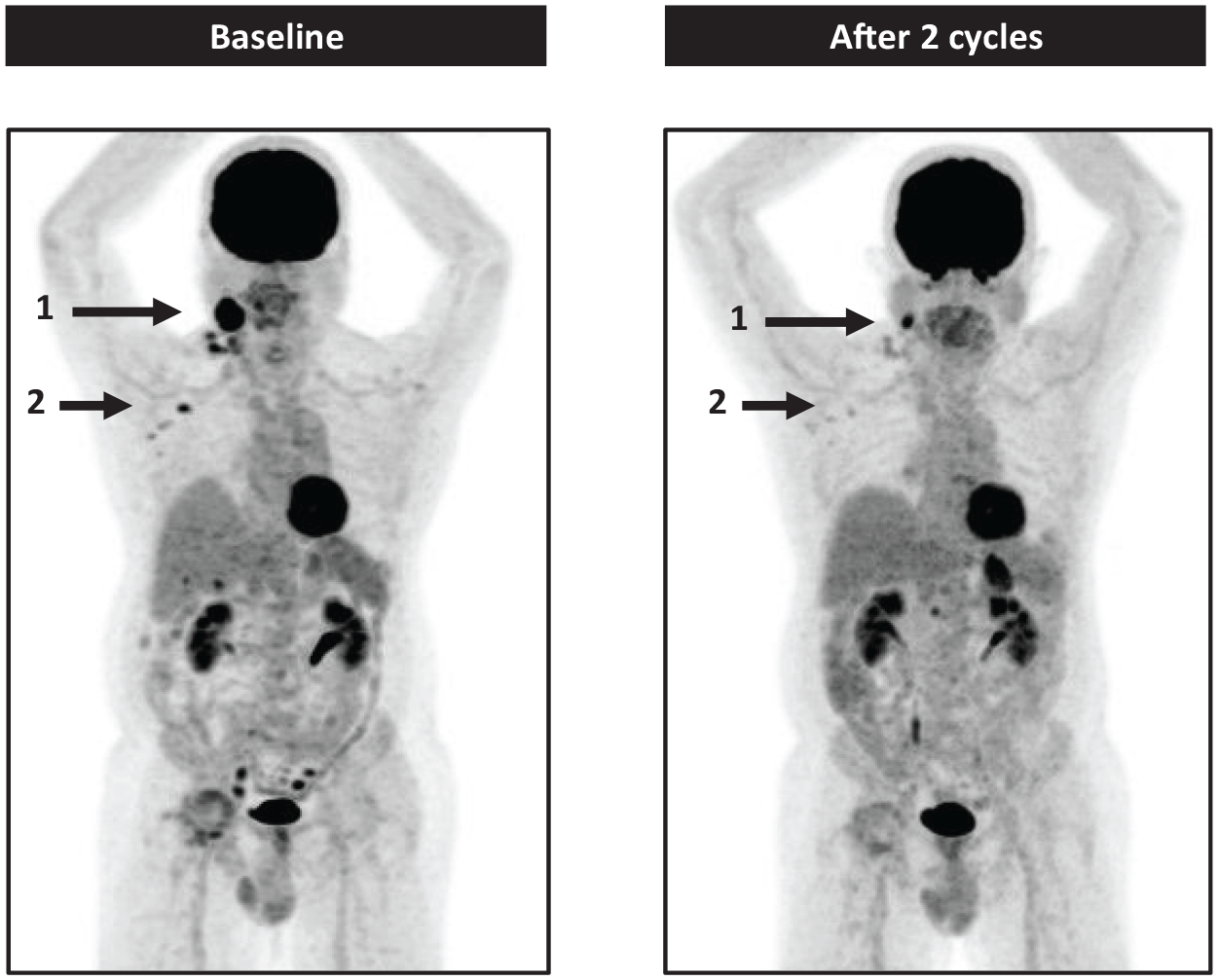
Partial response in a patient treated with crenigacestat for a refractory peripheral T-cell lymphoma AITL subtype. Arrow 1 shows a partial regression of right cervical lymphadenopathy. Arrow 2 shows complete regression of right axillary lymphadenopathy.
The spaghetti plot demonstrated the reduction in tumor size for the patient with T-cell lymphoma who achieved PR (Figure 2). Interestingly, Figure 2 also showed the evolution of circulating B-cell lymphocyte count for patients with CLL (dashed lines), and the three patients with CLL treated with crenigacestat had a reduction in the number of circulating tumor cells. Despite these reductions, CLL patients in the study were not evaluable among protocol NCI Working Group for CLL criteria and they discontinued the study before the completion of complete CLL evaluation after two cycles for response criteria evaluation. In the three patients with CLL, the tumor lymphocyte level after one cycle of treatment decreased, without reaching a decrease of more than 50%. Reasons for discontinuation in these patients with CLL were based on investigator decisions with clinical tumor progression in two patients and death in one patient (Figure 2).
Discussion
This study describes crenigacestat, a highly potent and selective Notch inhibitor in a cohort expansion of patients with NHL or CLL. Crenigacestat was previously investigated in a phase I study, and the recommended dose was 50 mg 3 TIW. 16 Notch pathway inhibition for target genes was observed at 50 mg TIW according to study parts results reported elsewhere, 15 and supporting this dose as a recommended dose. We report here a cohort-expansion phase in patients with NHL or CLL. During the pre-screening phases of the study, we found Notch alterations by detecting positive NICD staining in tumor cells were mainly found in patients with PTCL; 52% of patients with PTCL were positive. It was also found that patients with relapsed or refractory CLL had mutations in the Notch pathway in almost one-third of cases, as described elsewhere. 23
We found crenigacestat treatment confirmed to have antitumor clinical activity in patients with relapsed or refractory lymphoma. One patient out of the 9 (11%) patients treated achieved an objective antitumor response. This patient had PTCL with AITL subtype and he achieved a partial remission. Interestingly, a PR to treatment was documented in a patient with AITL, since this subset of PTCL exhibited a near 100% prevalence of non-mutational Notch activation.24,25 More in-depth studies of NOTCH pathway inhibition in the subgroup of patients with AITL would deserve to be undertaken.
The three patients with CLL treated were not formally evaluable according to international criteria, although a slightly decreased circulating lymphocyte count was observed. The limited efficacy of crenigacestat in this study could be due to several factors, including dose interruptions due to gastrointestinal toxicities. Future studies with larger populations and specific histological types are required probably in targeting a specific subgroup of patients with subtype lymphoma for example with AITL or with NHL known to be more frequently altered in Notch pathway such as marginal zone lymphoma26–28 or mantle cell lymphoma.29–31
The safety profile in the present study is consistent with the prior results of crenigacestat in solid tumors16–18 and previously reported clinical safety data for Notch pathway inhibitors.32–37 Gastrointestinal toxicity (diarrhea and nausea) was the most frequent drug toxicity observed in this study, as reported in other patients treated with Notch inhibitors for solid tumors. 38 This gastrointestinal toxicity is linked to the mechanism of action of Notch inhibitors that turn proliferative cells in intestinal crypts into goblet cells.35,38 Information on larger patient groups and longer treatment exposure durations is needed to accurately define the benefits and harms of NOTCH inhibitors in hematologic cancer patient populations.
Moreover, recent data demonstrate that the two paralogs Notch1 and Notch2 facilitate immune evasion and strongly downregulate the expression of CD19, possibly limiting the efficacy of immunotherapies targeting CD19. 31 Thereby, another potential use of Notch inhibitors could be to block the Notch pathway to restore the expression of the CD19 tumor target and to increase the efficacy of immunotherapies targeting CD19 B tumor cells such as monoclonal antibodies, immunoconjugates agents, or even CAR-T-cells.
In summary, Crenigacestat demonstrated a modest clinical activity at the recommended dose in adult patients with relapsed/refractory NHL or CLL. Better knowledge of the Notch pathway biology and detailed characterization of Notch activating mutations sensitive to crenigacestat will be required to identify the patients most likely to benefit from Notch inhibition therapy in the context of relapsed or refractory NHL or CLL. In addition, this study provides meaningful information for the potential use of Notch inhibitors in patients with relapsed or refractory lymphomas, on the way to potentially restoring tumor antigens such as CD19, CD20, or CD22 at the cell surface to potentialize effect of immunotherapies.
Limitations
The main limitation of this study in patients with hematological cancer is the small number of patients included in this part of the study. Since dysregulated Notch signaling is implicated in several malignancies, this part of the study was not statistically powered as was exploratory in nature.
Conclusion
Crenigacestat demonstrated in patients with hematological malignancies a safety profile consistent with the prior results of crenigacestat in solid tumors. Crenigacestat demonstrated a modest clinical activity at the recommended dose in adult patients with relapsed or refractory NHL or CLL.
Supplemental Material
sj-doc-1-taw-10.1177_20420986241311461 – Supplemental material for Notch pathway inhibition with crenigacestat (LY3039478) in a phase I first-in-human clinical trial for patients with relapsed or refractory non-Hodgkin lymphoma and B-cell chronic lymphocytic leukemia
Supplemental material, sj-doc-1-taw-10.1177_20420986241311461 for Notch pathway inhibition with crenigacestat (LY3039478) in a phase I first-in-human clinical trial for patients with relapsed or refractory non-Hodgkin lymphoma and B-cell chronic lymphocytic leukemia by Jean-Marie Michot, Zsofia Balogh, Jennifer R. Brown, Vincent Ribrag, Antoine Hollebecque, Rastislav Bahleda, Cyril Quivoron, Samy Ammari, Jean-Yves Scoazec, Karim A. Benhadji and Christophe Massard in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986241311461 – Supplemental material for Notch pathway inhibition with crenigacestat (LY3039478) in a phase I first-in-human clinical trial for patients with relapsed or refractory non-Hodgkin lymphoma and B-cell chronic lymphocytic leukemia
Supplemental material, sj-docx-2-taw-10.1177_20420986241311461 for Notch pathway inhibition with crenigacestat (LY3039478) in a phase I first-in-human clinical trial for patients with relapsed or refractory non-Hodgkin lymphoma and B-cell chronic lymphocytic leukemia by Jean-Marie Michot, Zsofia Balogh, Jennifer R. Brown, Vincent Ribrag, Antoine Hollebecque, Rastislav Bahleda, Cyril Quivoron, Samy Ammari, Jean-Yves Scoazec, Karim A. Benhadji and Christophe Massard in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
