Abstract
Hereditary angioedema (HAE) is a relatively rare disease characterized by acute episodes of swelling. These swellings can be disfiguring, painful and life-threatening. Since the symptoms occur in different areas and most patients experience a delay in their diagnosis, resulting in unnecessary suffering and dangerous situations. HAE can have a tremendous impact on the quality of life. The major genetic deficiency in this disorder is either an absent or nonfunctional C1INH which regulates the complement, fibrinolitic, kalikrein and plasmin pathways.
Introduction
Hereditary angioedema (HAE) is a relatively rare genetic disease that has an estimated prevalence of one in 10,000 to one in 50,000. 1 HAE has been reported in all races, and no gender predominance has been found. It is characterized principally by unpredictable attacks of angioedema that can be highly variable in severity, frequency, and anatomical location in each patient and in the same individual. Attacks most often affect the skin and/or mucosa of the upper airways as well as the intestinal mucosa.2,3 Zanichelli et al 4 evaluated 1532 acute HAE attacks and found the frequency of involved locations to be peripheral (45.6%), abdominal (32.8%), combined locations (15.1%), and laryngeal (6%). Approximately 22.3% of the patients had more than 12 attacks per year. 4 Typical attacks of angioedema last approximately 2-5 days before resolving spontaneously. Skin swelling may appear on the extremities, face, neck, torso, and genital area as a nonpruritic and nonerythematous lesion. Attacks may start in one location and then spread to another before resolving. Most acute attacks do not have an identifiabletriggering factor, but some are associated with local trauma, medical procedures, emotional stress, infections, menstruation, and some drugs, especially oral contraceptives, angiotensin-converting enzyme inhibitors, and sitagliptin.2,5–7 During acute attacks, patients may develop a rash misdiagnosed as urticaria. However, the skin lesions associated with HAE are erythematous, but are not warm, painful, or pruritic as in erythema marginatum.
When angioedema involves the larynx and sub-mucosal edema of the upper airways, it can lead to asphyxia. This is the most ominous and distressing symptom of HAE and in such circumstances tracheostomy can be lifesaving. Laryngeal swellings comprise a small minority (around 2%-6%) of attacks, and may cause death from airway obstruction. 8 If undiagnosed, mortality from HAE can be as high as 30%-40%. 8 The risk of one attack being fatal is low, but there is a significant lifetime mortality risk. 5
Gastrointestinal symptoms of HAE, caused by visceral edema, may result in varying degrees of intestinal obstruction. Typical symptoms are anorexia, vomiting, nausea, severe abdominal pain, and diarrhea following the attack. Ascites and hemoconcentration are possible. These symptoms can be mistaken for an acute abdomen, and this occasionally leads to unnecessary abdominal surgical exploration.
Most patients with symptomatic HAE experience disability due to the frequency and nature of their attacks, which may have a significant impact on quality of life. 7 Many patients experience more than one attack per month, and many of these are severe in nature. 9 More than 40% of patients with HAE may suffer from clinical depression. 10
There are three known types of HAE. Type I occurs in approximately 85% of cases, and is caused by a deficiency of plasma C1-INH. In most cases of type I, an autosomal dominant pattern of inheritance can be demonstrated, although spontaneous mutations can account for about 25% of these cases. Type II occurs in 15%-20% of all cases, and involves functional deficiency of C1-INH.3,11 Type III, recently identified, occurs in the setting of normal levels and function of C1-INH, and mostly affects women, particularly during pregnancy or exogenous estrogen therapy. Recently, it has been shown that a minority of type III HAE patients (15%-20%) have a gain-of-function mutation in the gene F12, which encodes factor XII. 12
Mechanisms
The major genetic deficiency in this disorder is either an absent or nonfunctional C1-INH which regulates the complement, fibrinolytic, kallikrein, and plasmin pathways (Fig. 1). The deficiency of a plasma inhibitor was discovered in the 1960s by Landerman et al, 13 who reported that patients with HAE lacked an inhibitor of serum globulin permeability factor or plasma kallikrein. Donaldson and Evans then identified the missing inhibitor as C1 inhibitor. 14

Regulation of complement activation by C1 INH occurs through inactivation of C1r and C1s in the classic pathway. It also regulates lectin pathway activation via inactivation of mannan-binding lectin-associated protease 2 (MASP2), which is activated after binding of mannan-binding lectin (MBL) to a microorganism surface. C1 INH also inhibits alternative pathway activation by reversibly binding to C3b, which inhibits factor B binding, but not involving protease inhibition.
C1 inhibitor is a broad-spectrum serine protease inhibitor that is a member of the serpin (serine protease inhibitor) superfamily, which has significant homology with α1-antitrypsin. It is the major inhibitor of several complement proteases (C1r, C1s, MASP-1, and MASP-2) and contact system proteases (plasma kallikrein and activated Hageman factor [coagulation factors XIIa and XIIf]). 15 C1 inhibitor is also an inhibitor of plasmin (a fibrinolytic protease) and factor XIa (a coagulation protease).
C1 inhibitor is a 110 kD single-chain glycoprotein consisting of 478 amino acids and a 22 residue signal peptide. The protein is organized into two domains, ie, an N-terminal of 100 amino acids containing N-linked and O-linked carbohydrates, and a C-terminal of 378 amino acids containing the active site in a stressed loop conformation typical of the serpins. 16 The sequence of the gene was identified in 1991 and is now known as SERPING1. 17 It is located on chromosome 11 (p11.2-q13), contains eight exons and seven introns, does not contain a 5-TATA box, and is distinguished by 17 A1u repeats in introns 3-7. More recently, the crystal structure has been solved. 18
The C1 inhibitor deficiency in HAE has been shown to result from mutations of the SERPING1 gene. A large number of SERPING1 mutations have been identified, with additional mutations still being reported.19,20 In type II, most of the SERPING1 mutations involve residues at or near the active site on the reactive mobile loop that result in a mutant C1 inhibitor protein that is secreted but is not functional. 21 In both type I and type II HAE, the low functional level of C1 inhibitor results in diminished regulation of the complement and contact systems.
Bradykinin is the main mediator for an attack of HAE. This potent vasodilatory peptide results from activation of factor XII and the kallikrein system. CINH is the main regulator of this system 22 (Figs. 1 and 2). Although the triggering factors are mostly unknown, excess kallikrein is episodically released, which cleaves high molecular weight kininogen to release the vasoactive peptide, bradykinin. Bradykinin binds to specific G2 protein-coupled receptors on endothelial cells (bradykinin B2 receptors), which leads to vasodilatation and increased microvessel permeability. The edema is caused by plasma leakage from postcapillary venules due to bradykinin-induced changes. In addition, bradykinin causes tissue smooth muscle contraction, leading to pain and cramping. 23
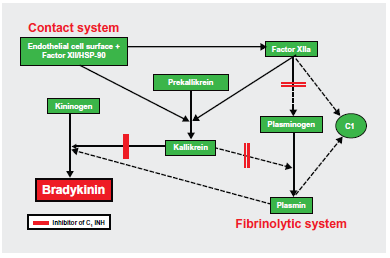
Interactions among the complement, contact, and fibrinolytic systems. Solid lines with arrows indicate reactions of defined biologic relevance, and dotted lines with arrows indicate reactions demonstrated in vitro but with unproven biological importance.
The exact biochemical events responsible for an attack have not been fully elucidated. Because quantitative or functional deficiency of C1-INH is the defect in HAE, replacement of this missing physiological protein for the treatment of acute attacks of HAE has been recommended in countries where the product is available.
Indications for C1 Inhibitor
A report that patients with HAE were deficient in plasma protein C1 inhibitor, 14 along with the gradual development of better techniques for purifying proteins from blood donor plasma, has enabled replacement of this missing plasma protein. Fresh frozen plasma has been reported to be used widely in the treatment of acute attacks. 24 Fresh frozen plasma is usually efficacious, curing attacks in the vast majority of patients by supplying the necessary inhibitor, but in some patients it may prove dangerous. An infusion of fresh frozen plasma and high molecular weight kininogen, present in normal plasma, is administered along with C1 inhibitor. The same companies that developed immunoglobulin G for intravenous use began to purify C1 inhibitor protein. Approximately 30 years ago, acute attacks of HAE were first treated with partly purified C1 inhibitor.25,26
Therapeutic options for patients with HAE have been limited, and the approach to treating acute attacks has been problematic, mainly because of the restricted availability of the necessary agents. Human C1 inhibitor concentrates have been found to be highly effective and safe in the treatment of acute attacks of HAE caused by C1 inhibitor deficiency. 26 C1-INH concentrate has several advantages, in that it has a reduced risk of causing blood-borne infections, it can be self-infused, and its effect is much faster than that of fresh frozen plasma. Thus, this review will focus on this specific therapy, which recently became available in the US and has been in use for a long time in Europe, but is still unavailable in most developing countries.
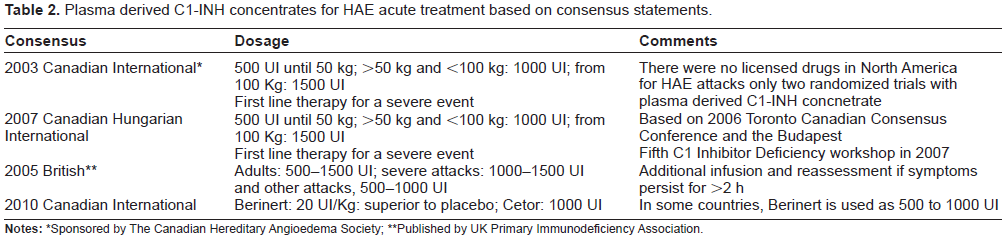
Anabolic androgens and antifibrinolytic drugs are used for long-term prophylactic management of HAE, but these agents may be associated with clinically significant adverse effects.27,28 C1 inhibitor concentrates have been shown to be effective for the treatment of acute HAE attacks and their use is supported by the results of randomized trials and published consensus statements (Tables 1 and 2).
Human C1 INH preparations available for HAE attacks.

Plasma derived C1-INH concentrates for HAE acute treatment based on consensus statements.
Notes:
Sponsored by The Canadian Hereditary Angioedema Society;
Published by UK Primary Immunodeficiency Association.
Short-term prophylaxis with plasma-derived C1-INH (pdC1-INH) has been suggested. According to the treatment recommendations, attacks to be treated are those involving the face, neck, and oral/ upper airways mucosa, as well as abdominal attacks with moderate to severe pain. For peripheral and mild abdominal attacks, the recommendation is to start tranexamic acid 1 g for adults and 20 mg/kg for children every four hours orally or to use pdC1-INH based on attack-related disability. Zanichelli et al observed that the use of tranexamic acid in patients compared with those not treated did not reduce the duration of attacks (1.79 versus 1.85 days, respectively). 4 Minor procedures, such as dental treatment, can be done without prior administration of pdC1-INH, unless the patient has a history of attacks in the same circumstances. It is advisable that pdC1-INH is on hand for application anyway. For major procedures, pdC1-INH should be used at least one hour prior to surgery, and less than six hours before a procedure requiring endotracheal intubation. A second dose of pdC1-INH should be available at the time of surgery, the recommendation being 10-20 U/kg.29,31 Previous reports suggest that it is essential to achieve a C1-INH level of at least 39% of normal.32,33
Interestingly, Zanichelli et al report that the majority of attacks (61.6%) are untreated, 4 and there is a perception that physicians are not always aware of the correct treatment for HAE. With pdC1-INH, there is no risk of infection acquired from blood products but the fear of using these products frequently limits its use to acute attacks of angioedema.
Products Available
There are three human pdC1-INH products available, all of which are pasteurized. The preparations available contain high levels of C1-INH, ie, 100 U/mL, given as one unit, which is equivalent to the amount of C1-INH activity in one mL of plasma. 34 A human, plasma-derived, nanofiltered, lyophilized C1-INH product (Cinryze®, Viropharma, Exton, PA) was approved recently in the US. Another human, plasma-derived, non-nanofiltered C1-INH product (Cetor®, Sanquin, Amsterdam, The Netherlands) has been used for attacks and routine prophylaxis in Europe.
A previous Phase III trial has demonstrated the efficacy of both prophylactic treatment (P < 0.0001) and treatment of acute attacks (P < 0.005) of HAE. 35 Seventy-one patients were treated within four hours of the onset of an attack, with relief of symptoms within a median two hours in comparison with four hours in the placebo group. The treatment was administered to all patients if laryngeal edema occurred. This protocol failed to reach statistical significance (P = 0.026), so the indication for use in acute attacks was not approved in US. 3 However, the same product was then given twice a week in a randomized, double-blind, placebo-controlled, crossover study, and the results showed highly significant improvement in frequency and severity of attacks, leading to its approval in this indication by the US Food and Drug Administration.
Berinert® (CSL Behring, King of Prussia, PA) is a human, plasma-derived, pasteurized C1-INH licensed in European countries, Japan, Argentina, Canada, and the US. It is a lyophilized product approved for short-term prophylaxis and acute attacks in all countries except for the US. The US Food and Drug Administration indication is only for abdominal or facial attacks in adults and adolescents. Although the efficacy of this product for acute laryngeal attacks has not been tested in a double-blind protocol, the prompt relief (median 16 minutes) observed by Levy et al suggests its use is beneficial. 36
Although not within the scope of this review, a recombinant human C1-INH (Rhucin®, Pharming group, Leiden, The Netherlands) obtained from transgenic rabbits has been tested in several studies, with good results.37,38 The human gene for C1 inhibitor has been cloned and introduced into animals, in this case rabbits, under the regulatory control of a bovine a S1-caseine promoter. 39 Under these conditions, the human protein is secreted in the milk of the lactating rabbit, and the C1 inhibitor is isolated from the milk. It has been observed that the different glycosylation pathway used by the rabbit-produced C1 inhibitor leads to a shorter half-life in the recipient comparing with the native protein.39,40
Pharmacokinetics
Bernstein et al evaluated the pharmacokinetics of pdC1-INH infused during the IMPACT 1 (International Multicenter Prospective Angioedema C1-INH Trial 1) study. The product was administered as two doses of 10 U/kg or 20 U/kg for the treatment of acute facial or abdominal attacks and, if needed, could be repeated after four hours. The population estimate for the half-life of pdC1-INH was 32.7 hours, which is longer than for new drugs such as icatibant (a bradykinin receptor inhibitor), ecalantide (a kallikrein inhibitor), or recombinant C1-INH, and thus reduces the risk of a rebound effect. Martinez-Saguer et al treated patients with a single 500 U or 1000 U dose of the same pdC1-INH concentrate (ie, Berinert) during an attack-free and substitution-free interval of at least three days. The mean half-life of pdC1-INH in adults with a high frequency of severe attacks at least one attack per week, treated with individualized replacement therapy) was 33.3 hours, and the mean half-life in adults with a lower frequency of severe attacks (maximum of 1-2 attacks per month, treated with on-demand therapy) was 47.8 hours. This estimate was comparable with the mean half-life of 37.9 hours reported for patients with symptomatic HAE treated with a different pdC1-INH concentrate. 41 The pdC1-INH concentrate recently approved in the US for prophylactic treatment of HAE attacks (Cinryze) was reported to have a half-life of 56 hours in adults with asymptomatic HAE (n = 7). 42
Brackertz et al studied the half-life of 125-iodide-labeled C1-INH in three HAE patients and three normal subjects, and found no apparent differences (67.7 hours for HAE patients, 64 hours for healthy controls). 43 One potential effect on the half-life of C1-INH in plasma is the temporal relationship between an HAE attack and the administration of pdC1-INH concentrate, on which the pharmacokinetic analysis is based. It has been reported that the half-life of pdC1-INH after infusion is about 4.5 days in normal subjects, about 46.5 hours in subjects with mild HAE, and about 31.75 hours patients with severe attacks.44,45 Furthermore, Martinez-Saguer et al using the same mean U/kg body weight doses of pasteurized C1-INH in pediatric and adult populations (15.2 U/kg on demand in adults, 16.2 U/kg in children) showed that pediatric patients had a shorter half-life of functional plasma C1-INH for on demand therapy (31.5 hours versus 47.8 hours in adults) but the number of children evaluated was small. 46
One possible explanation for these findings is that, at a half-normal serum concentration of C1-INH, there is activation of the early classical complement pathway and/or other systems in which this protein acts as an inhibitor. In addition to increased catabolism, decreased synthesis of normal C1-INH contributes to the low serum concentration of this protein in all forms of HAE. 47
Dosage
Injection of 500-1000 U of pasteurized C1-INH has proved to be life-saving during the course of acute laryngeal attacks.48,49 Kunschak et al found doses of 23-39 U/kg of pasteurized C1-INH to be effective in a double-blind, placebo-controlled study. 41 Craig et al showed that a dose of 20 U/kg was more effective than 10 U/kg in a double-blind protocol. 50 The IMPACT 1 study had previously established 20 U/kg as an effective dose of C1-INH concentrate in the treatment of acute HAE attacks. Craig et al evaluated the clinical response in HAE patients with laryngeal edema (IMPACT 2) who did not undergo placebo-controlled investigation for ethical reasons, and the median time to symptomatic relief was 15 minutes. Farkas et al treated their patients with an initial dose of 500 U regardless of age, and an additional dose was given in only a few cases (Tables 1 and 2). 31
Efficacy
There have been several reports of improvement of laryngeal edema and abdominal attacks of HAE after treatment with C1 inhibitor34,51,52 but doubleblind clinical trials have only recently been published showing improvement using C1 inhibitor in comparison with placebo. Waytes et al published the first double-blind study of a vapor-heated C1 inhibitor concentrate. 53 In recent years, some studies have evaluated self-administration of C1 inhibitor, reporting that early treatment results in more rapid relief of symptoms (Table 2).54,55
Safety
Transmission of hepatitis C virus was a problem before 1986 (more than 80% of patients were infected), when products were not virus-inactivated. No report of patients infected with human immunodeficiency virus has been published so far.30,34 Nowadays, intravenous C1-INH is derived from pooled human plasma that is subjected to rigorous safety measures, including donor selection, sensitive and specific nucleic acid testing/polymerase chain reaction assays (for hepatitis A, B, and C virus, human immunodeficiency virus Type 1, and human parvovirus B19). Over the years, several procedures have been developed in order to reduce the possibility of contamination, including use of detergents (polyethylene glycol precipitation), heat, nanofiltration, and chromatography. The plasma used has been tested for a variety of viruses and antibodies to viruses. In addition, the plasma is stored for several months so the donor can be retested later for the development of antibodies to some viruses. No virus transmission has been reported since the introduction of C1-INH in January 1985. 56 Nanofiltration along with pasteurization of the preparation provides safety based on exclusion by size, which may be particularly important for protection against nonenveloped viruses and newer infectious agents, such as prions. 42
The clinical safety of pasteurized C1-INH concentrate has been established over the last 25 years in more than 500,000 well tolerated administrations (data on file, CSL Behring). In this period of time, only eight allergic or anaphylactic reactions, including four episodes in one HAE type I patient from Frankfurt, have been observed. This corresponds to an incidence rate of 1/50,000 administrations and thus to classification of allergic reactions as “very rare” (< 1/10,000) according to the Council for International Organizations of Medical Sciences III standard categories for classification of adverse drug reaction frequency. 57 No proven cases of viral transmission from pasteurized C1-INH concentrate have been reported in the last 25 years. 56 Pasteurized pdC1-INH concentrate can be used in pediatric and adult patients. It is also well tolerated in pregnant and breastfeeding women. 31 Minor adverse events have been reported, including nausea, shortness of breath, dizziness, faintness, vertigo, headache, paresthesia, fever, and chills.41,52
Home-Based Therapy
Although oral prophylaxis reduces the frequency and severity of HAE attacks, it does not always abolish them, so patients require immediate access to effective acute treatment. The requirement to travel to a medical facility for acute treatment implies at least half a day away from home, school, or work. 55
Self-administration of C1-INH concentrate has been introduced in a number of countries to improve access to acute HAE treatment.54,58 Extensive education of patients should be undertaken on how to prepare the lyophilized medication with sterile water, perform self-venipuncture and, although the administration of C1-inhibitor does not cause allergic reactions, patients should be instructed to handle such a situation. 54
Home therapy with pasteurized C1-INH concentrate in HAE patients was developed in line with subcutaneous gammaglobulin, whereby the missing protein is administered by the patient at home on an as-needed basis. Aygören-Pürsün et al reported that home therapy using pasteurized C1-INH concentrate on demand has been implemented for a period of 28 years in Frankfurt, 57 where a total of 274 patients (49% of all patients with C1-INH deficiency) have been treated at home. Of these, 248 were adults (55% of all adult patients) and 26 were children (24% of all pediatric patients). The age of the adult and pediatric patient groups was 18-81 years and 6-17 years, respectively. In young pediatric patients, parents administer home therapy. With pasteurized home C1-INH therapy, faster symptomatic relief and complete resolution of the attack has been observed.54,57 Adverse events recorded were skin irritation at the injection site, minor hematoma at the puncture site, dizziness at the time of injection, mild pain in the extremities after injection, and a sub febrile increase in temperature. 54
In the sixth C1 Inhibitor Deficiency Workshop held in May 2009, an approach for home therapy was established. Any HAE patient could opt for home therapy, but training is necessary and it can take place with other treatments. Treatment had been recommended as soon as patients identify symptoms, which, if untreated, would likely develop into a moderate or severe attack. 55
For airway events, the dose of pdC1-INH should be 20 U/kg rounded up to the next highest vial. 50 For treatment of other attacks, the best dosage and timing of administration may differ from that reported before, and a dose-finding trial for home therapy with pdC1-INH is needed. Venipuncture using a small butterfly needle for infusion is recommended, and venous ports should be avoided considering the higher risk of complications, such as infection and occlusion. It is likely that home therapy would improve quality of life, although there is a concern that such availability of the product could increase its unnecessary use.54,58
Monitoring
Although no severe adverse event has ever been reported with human C1-INH products, some safety issues should be evaluated before administration of any plasma-derived product.49,59 An ongoing potential safety concern with any biological medicine derived from blood or plasma is the transmission of blood-borne pathogens. This concern has lessened significantly in the last decade as a result of the implementation of more effective predonation and postdonation screening tests for potential pathogens, and implementation of pathogen reduction strategies to which many plasma-derived biological medicines are now routinely subjected. It is suggested to perform liver and renal function tests, serology for human immunodeficiency virus, and hepatitis C and hepatitis B surface antigens. If the patient was not immunized for hepatitis B, it is recommended to give the vaccine. 49 After the three doses of the vaccine, the antibody response should be checked. Liver and renal function should be checked on an annual basis. It is also recommended to record all batch data, including product number, date of delivery, and expiry date.
Pregnancy
Treatment of HAE during pregnancy is particularly difficult. All prophylactic medications should be stopped during pregnancy. Severe attacks should be treated with the concentrate as in nonpregnant women. There is little evidence that vaginal delivery could result in severe edema attack. pdC1-INH prophylaxis is safe during pregnancy, and it is proposed that C1-INH should be available in the delivery room. 60 In case of operative delivery, regional analgesia is preferable to endotracheal intubation in order to avoid laryngeal edema.
Use in Children
Intravenous administration of C1-INH concentrate is the most appropriate therapy for significant acute HAE attacks (laryngeal or diffuse facial edema and severe abdominal attacks). The indications for pdC1-INH infusion are the same in children as in adults, and Farkas et al described the use of similar doses (ie, 500-1000 U) with high efficacy. Doses could be calculated as 10-20 U/kg (500 U up to 50 kg, 500-1000 U for weights of 50-100 kg, 1500 U if weight is > 100 kg). 31
Surgical or diagnostic interventions in the head and neck region may require short-term prophylaxis with pdC1-INH, 63 although short-term prophylaxis is less frequently required in children than in adults. The clinician must remain especially vigilant, because small children can suffocate quickly because of their small airway diameters. 33
Conclusion
The ideal HAE treatment offers good quality of life. C1 inhibitor is a plasma protein which can be purified from pools of human blood plasma, and adequate techniques have been used to remove or inactivate blood-borne viruses. Infusion of human C1-INH has been demonstrated to relieve attacks. In addition to improving the clinical condition of patients with HAE, therapy avoids potentially laryngeal edema. Cicardi and Zingale commented that after introduction of pdC1-INH to the market, the number of patients with access to the drug who died due to laryngeal edema decreased to almost zero. 34 Given the side effects related to prophylaxis with androgens and antifibrinolytic agents, and their limited effectiveness in some patients, C1-INH concentrate administered as a weekly course has emerged as a possible long-term treatment. There are still some controversial issues surrounding this therapy, so prospective studies with larger samples could be worthwhile.6,61 Although the intravenous C1-INH concentrate is strongly recommended, unfortunately its availability is still limited, particularly in developing countries.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
