Abstract
Background
C1-esterase inhibitor (C1-INH) is a protein derived from fresh frozen human plasma and is widely used in the treatment of Hereditary Angioedema. C1-INH product must have high purity with preserved functionality.
Objective
The aim of the study was to perform extensive characterization of human C1-esterase inhibitor (Celestrase 1) with special emphasis on functionality of the protein by different mechanisms along with its purity and structural elucidation.
Design
Comparative study.
Methods
This study describes the purification and characterization of an economically viable, highly pure and efficient human plasma-derived C1-INH prepared from cryopoor plasma by combination of chromatography steps, capable of removing protein contaminants. The purification process includes two orthogonal virus clearance steps -solvent detergent and virus retentive filtration and further, characterized by various biochemical and functional assays along with other commercially available entities.
Results
The developed process yields 0.75 ± 0.1 vials of C1-INH /L of cryopoor plasma with 44.4 ± 3.6% overall process recovery. Celestrase 1 shows comparability with an existing market comparator, with respect to its purity by different methods. Celestrase 1 proved its functionality in binding irreversibly to the complement protein by classical pathway of the complement system and in the Kallikrein system. Additionally, the antigen-to-biologic activity ratio an indicative of the functionality for Celestrase 1 (0.94) was found comparable to market comparator (0.79). Identity of the product was confirmed by Western blot analysis. The structural analysis of Celestrase 1 was found to be similar to market comparator and exists predominantly in α-helix secondary structure by Far UV Circular Dichroism (CD).
Conclusion
For the treatment of Hereditary Angioedema (HAE), the current study presents a pure, safe, and functionally efficient product that can meet the therapeutic needs of patients deficient in C1-INH.
Introduction
C1-esterase inhibitor (C1- INH) is a single polypeptide chain of 478 amino acid residues having a molecular weight of 90–105 KDa under non-reducing and 85–93 KDa under reducing conditions. Isoelectric point of C1-INH is approximately 2.7–2.8. 1 It belongs to the serine protease inhibitor (serpin) superfamily. 2 It has a high degree of glycosylation and approximately 50% of its total weight is contributed by carbohydrates; the molecular weight of the peptide chain is ∼53 KDa.1,3
This protein is mostly produced by the liver, although it is also produced by activated monocytes and other cell types. 4 The classical complement pathway’s early activation steps are significantly regulated by C1-INH. In addition, C1-INH controls the activation of plasmin in the fibrinolytic pathway, factor XI in the coagulation cascade, activated factor XIIa, and kallikrein. In C1-INH deficiency, the classical complement pathway may be inappropriately or overactivated. Immune complexes cause activation of the first component of C1 to C1 esterase. C1 esterase then forms a C4b2a complex with its natural substrates C4 and C2. The formation of this new complex (and the associated activation of C3) results in the production of anaphylactic, chemotaxic and vasoactive peptides (C2b, C3a, C5a). C1-INH protein inhibits both the spontaneous activation of C1 and the formation of activated C1, and thus the C2 complex will not be formed. 5
C1-INH controls the conversion of prekallikrein's to kallikrein in the kinin releasing pathway. Lack of C1-INH causes an increase in kallikrein, which in turn causes an increase in bradykinin production. C1-INH has demonstrated inhibitory effect on factor XIIa, factor XIa and plasmin. C1-INH deficiency causes enormous localized uncontrolled edema and enhanced vascular permeability. There is some evidence supporting the involvement of bradykinin in angioedema but the exact component is still debatable.6‐8
The most severe and potentially fatal symptom of Hereditary Angioedema (HAE), a C1-INH deficient condition, is laryngeal edema. Also, localized swelling in the face, stomach and upper airways are symptoms of hereditary angioedema (HAE). 1 Based on the amount of C1-INH and levels of functional C1- INH present in the plasma, HAE has been classified into three types. C1-INH protein deficiency is one of the characteristics of type-I HAE. The plasma level of C1-INH in Type-II HAE is normal or high, but it is not fully functional. Type III is acquired deficiency with no abnormality found with C1-inhibitor. This condition occurs mainly in women and can be made worse with pregnancy or use of birth control medications. 2
There are several treatment options available for HAE like Ecallantide® (Dyax Corp., Cambridge, MA, USA) and Icatibant (Jerini, Berlin, Germany). Recombinant ecallantide is a protein made in yeast. It functions as a direct kallikrein inhibitor, inhibiting its activity and preventing the production of bradykinin. A synthetic drug called icatibant can be injected intravenously or subcutaneously. It shares a structural resemblance with bradykinin and functions as a selective bradykinin-2 receptor antagonist. 9 The mean half-life of functional C1-INH was 62 h after IV administration. 10
Mean elimination half-life of ecallantide at 30 mg SC dose is 2.0 ± 0.5 h 10 while the elimination half-life values of Icatibant are reported as 1.48 ± 0.35 (30 mg dose) and 2.00 ± 0.57 h (90 mg dose). 11
C1-INH concentrate is a more potent therapeutic treatment option as it replenishes the deficient protein in the patient's body. C1-INH can be prepared using recombinant DNA technology but the half-life of recombinant C1-INH was determined to be ∼3 h for a 100 U/kg dose. 12
Plasma derived C1-INH concentrate proves its superiority over recombinant C1-INH as it has a mean half-life of 32.7 h at a dose of 10 or 20 U/kg. 13 Administration of C1-INH may have a beneficial effect on clinical conditions such as sepsis, cytokine induced vascular leak syndrome, acute myocardial infarction. 2
In many cases, HAE is treated like allergy because of similar symptoms and less awareness of the disease. 14 This generates a strong need of creating awareness of the disease developing better diagnosis tools and efficient treatment options.
This article describes the preparation of a plasma derived Celestrase 1 and its extensive characterization. High importance was given to establish the efficacy and functionality of plasma derived C1- INH, using various analytical tools. A comparative analysis of Celestrase 1 was performed with available market comparator, by different biochemical methods which could detect differences in critical quality attributes viz. protein structure, function, quantity of protein, identity and purity of the molecule.
Materials and Methods
Materials
Freshly frozen recovered plasma was obtained from Indian blood banks approved by Drug Controller General of India (DCGI) and registered with the National Aids Control Organization under the Government of India. Antibody testing for HIV-I, II and HCV was performed using Biorad and HBV kit from Diapro. Nucleic acid testing of plasma was done using standard USFDA approved kits, from Roche, USA for Hepatitis-B virus (HBV), Hepatitis-C virus (HCV) and Human Immunodeficiency virus-I (HIV- I).
For total protein estimation, Kjeldahl equipment from Gerhardt Analytical Systems was used.
Standard protein molecular weight marker for SDS-PAGE was procured from Biorad (USA).
Primary mouse monoclonal antibodies were used for detection of C1-INH (MyBiosource). Goat anti-mouse IgG HRP (Invitrogen) was used as secondary antibody. 3, 3 –diaminobenzidine (Thermoscientific) was used for developing blot.
High performance Size-exclusion column was procured from TOSOH Biosciences, Japan. Mobile phase-phosphate buffer saline was prepared using chemicals procured from Merck (Darmstadt, Germany).
Reverse phase HPLC was performed on C4-Vydac 4.6 mm*250 mm column, 5µ.
Acetonitrile used was procured from Merck and trifluroacetic acid from Sigma Aldrich.
The TECHNOCHROM C1-INH kit was used to determine the activity of C1-INH using a chromogenic assay on ACL TOP automatic coagulometer from Instrumentation laboratories. C1-INH first WHO standard (NIBSC -08/256) was used for calibration.
The functionality of the protein was evaluated using a pre-kallikrein kit (Pathway Diagnostics, UK) with an in-house developed method.
The antigenic content of the C1-INH protein was determined using a specific C1-INH antiserum (Siemens Healthcare Diagnostics GmBH, Germany) by a nephelometer (Atellica Neph 630, Siemens).
Waters ACQUITY UPLC HClass Bio system was used to analyze N-Glycan using HILIC-FLR and glycan BEH Amide column (Waters Corporation). N-Glycan kit from Waters was used for sample processing (as per the manufacturer's instructions).
Structural elucidation was performed using J-815 CD Spectrometer (Jasco), USA.
Market comparator was procured for comparative analysis.
Methods
Antibody Testing
Every unit of plasma used for purification of C1-INH was examined for presence of antibodies against HIV-I, II, HBV and HCV. Units that were devoid of antibodies were further tested for the viral antigens by Nucleic acid testing (NAT). Plasma units which were tested positive were discarded as per standard procedures.
Nucleic Acid Testing
Every unit of Indian plasma used was additionally tested for HBV, HIV-I and HCV and all units that tested negative for the above viruses were pooled and used for the manufacturing of Celestrase 1. This testing was performed in addition to the certification provided by different blood banks.
Purification Process
Purification Process of Celestrase 1
Celestrase 1 was manufactured from cryopoor plasma obtained after separation of cold insoluble fraction of plasma. Coagulation factors present in cryopoor plasma were captured using a weak anion exchange column chromatography. The flow through of anion exchange column used for the capture of coagulation factors had protein of our interest, C1-INH, which in turn was captured on a strong anion exchange chromatography. C1-INH capture stage anion exchange column was washed with different buffers to remove the contaminating proteins and the product was finally eluted by increasing the concentration of sodium chloride. The flowthrough of capture stage anion exchange column can be used for fractionating intravenous immunoglobulin, albumin and also alpha 1 proteinase inhibitor.
Process of C1-esterase purification has been designed to ensure virus safety of the product, so anion exchange chromatography elute was treated with solvent detergent (S/D) reagents (1% v/v Tween 80 and 0.3% v/v TnBP). S/D treated anion exchange elute was then reconditioned and subjected on weak anion exchange column (for removal of solvent detergent and partial purification). This chromatography was performed in positive mode where C1-INH binds to the column while solvent detergent reagents were washed off in unbound fraction. C1-INH was eluted from this column by increasing the concentration of sodium chloride.
After removal of S/D reagents and partial purification by weak anion exchange column, the C1-INH was finally purified on hydrophobic interaction chromatography (HIC) in negative mode, wherein the contaminating proteins remain bound to the column. The bound contaminating proteins were eluted from the column using WFI. The flow through of HIC column was concentrated and buffer exchanged to remove high concentration of sodium citrate .
Adding further to the virus safety of the product; the purified C1-INH was filtered through a 20 nM virus retentive filter. Finally, the concentration was performed to achieve the desired potency of the product. The drug substance was then formulated using a formulation buffer to a targeted activity and lyophilized.
The biochemical and functional quality attributes were compared for plasma derived Celestrase 1 (3 batches) along with market comparator.
Purification Process of Market Comparator
The manufacturing process (as reported) includes virus reduction steps, precipitation and chromatographic steps for the reduction of impurities. Post pasteurization steps include ammonium sulfate precipitation and hydrophobic interaction chromatography.
Characterization of Celestrase 1
Reconstitution of Lyophilizates and its Stability
To achieve a final concentration of 50 IU/mL, the lyophilized product was reconstituted with 10 mL of water for injection. The lyophilized powder dissolved completely within 5 min of reconstitution. The above physicochemical and functional assays were performed to assess the quality attributes of the product.
The reconstitution stability was evaluated for 3 batches at 25 ± 2 °C for 48 h. A Chromogenic assay was performed to determine the activity using Technoclone kit at each sampling time-point.
Protein Concentration
Total protein in the Celestrase 1 as well as market comparator was determined by the Kjeldahl method (Infrared Digestion Unit: TURBOTHERM TT625 and Programmable Kjeldahl distillation system: VAPODEST 20 s. The samples were diluted 10 fold using formulation buffer.
Electrophoretic Analysis of the Molecular Weight by SDS-PAGE
Sodium Dodecyl Sulphate–Polyacrylamide Gel Electrophoresis (SDS–PAGE) was performed using 8%–16% gradient separating gel under non-reducing and reducing conditions. The Biorad Miniprotean Tetra Cell Assembly was used to run a gel at a constant voltage of 120 volts for ̴45 min and was silver stained using in-house developed method.
Identification by Immunoblotting
Proteins were transferred to nitrocellulose membranes, incubated with specific antibodies, washed and developed to produce an immunoblot. Primary mouse monoclonal antibodies (1:2000 dilution fold) were used for detection of C1-INH (MyBiosource). Goat anti-mouse IgG HRP (Invitrogen) 2000 fold diluted was used as secondary antibody. 3, 3 –diaminobenzidine (Thermoscientific) was used for developing blot.
Protein Purity by Size-Exclusion High Performance Liquid Chromatography
The high and low molecular weight impurities in the final product were analysed using size-exclusion chromatograph. Chromatographic separation was performed on TOSOH G3000 SWxl column 30 cm × 7.8 mm I.D, particle size 5 µ using phosphate buffer saline pH 7.0 for isocratic elution. Samples were normalized to 0.3 mg/mL in mobile phase, 100 µL of samples were injected, flow rate was 0.5 ml/min and absorbance was measured at 280 nm.
Impurity by Reverse Phase Chromatography
Reverse phase HPLC was performed on C4-Vydac 4.6 mm*250 mm, 5µ using 10% Acetonitrile in Milli Q water + 0.1% TFA as mobile phase A and 90% Acetonitrile in Milli Q water + 0.1% TFA as mobile phase B with a flow rate of 1.0 mL/min. Samples were normalized to 0.1 mg/mL in saline solution, 100 µL of samples were injected and absorbance was measured at 280 nm.
Antigen by Nephelometry
The measurement of C1-INH antigen in the sample was performed in the presence of the N-Antiserum Human-C1 Inhibitor antibody forming an insoluble immune complexes, which are light-scattering using an automatic Nephelometer (Siemens Healthcare, Germany). When the antibody is in excess, the light-scatter is directly proportional to the concentration of antigen. A calibration curve was prepared using a series of diluted standards of known antigen concentration. Samples of unknown antigen concentration were assayed and their concentrations were determined using the calibration curve.
Functionality of C1-Esterase Protein
TECHNOCHROM C1-INH kit was used to determine the activity of Celestrase 1 using a chromogenic assay on ACL TOP automatic coagulometer from Instrumentation laboratories. C1-INH first WHO standard (NIBSC -08/256) was used for calibration.
C1-INH was titrated against an excess of C1-esterase to form an inhibitory complex and the residual C1-esterase activity was measured using a chromogenic substrate. Residual C1-esterase cleaved a chromogenic substrate & intensity of pNA was measured at 405 nm. Samples as well as standard were 2-fold serially diluted using sample buffer for combistat analysis and the absorbance's were measured at 405 nm. Statistical analysis using combistat software was used to estimate the potency as well as the confidence interval.
Another in-house developed assay was used to determine the functionality of the protein by inhibiting the pre-kalikrein activator and inhibiting the conversion of prekallikrein to kallikrein. C1-INH sample was added to a mixture of prekallikrein and prekallikrein activator. C1-INH will inhibit the activation of prekallikrein to kallikrein and will not release chromophore pNa responsible for color development at 405 nm. The control i.e. without C1-INH was included in the study to determine the pKa levels without heparin. Dose-dependent inhibition kinetics was studies for both Celestrase 1 as well as market comparator. The results were measured in terms of residual amount of prekallikrein with respect to different concentrations of C1-INH and measurements were done using a plate reader.
The functionality of the protein was also determined by calculating the ratio of the antigen to the C1-INH activity. The antigenic content of the C1-INH protein was determined by nephelometry using a Nephelometer with a specific C1-INH antiserum forming an antigen-antibody complex. The concentration was determined based on the calibration curve using N-protein standard whereas; the activity was determined using a Technoclone kit by a chromogenic assay as detailed above. The antigen-to-functional activity ratio; an indicative of the functionality of the protein for C1-INH was determined.
Glycosylation
Waters ACQUITY UPLC HClass Bio system was used to analyze N-Glycan using HILIC-FLR and glycan BEH Amide column (130 Å, 1.7 μm, 2 × 150 mm; Waters Corporation) was used for separation. A column temperature of 60 °C, gradient method having run time of 55 min using mobile phase A (50 mM Ammonium formate solution pH4.4) and mobile phase B (100% acetonitrile) with an sample injection volume 10 µL. Eluting N-glycan was detected (excitation 265 nm and emission 425 nm) with 20 hz sampling rate and peak annotation was performed using LC-MS for each N-glycan species.
High Order Structure
CD spectra were captured using a J-1500 CD Spectrometer (Jasco) at 20 0.2 °C, 50 nm/min scanning speed, 1 s reaction time, and 1.0 mm bandwidth. The wavelength range of 190–250 nm was used to capture far-UV CD spectra. Far-UV CD spectra were recorded at a wavelength range of 190–250 n. For the secondary structure estimation, each spectrum was converted into molar ellipticity and secondary structure was estimated by Jasco secondary structure estimation tool present in Spectra Manager software.
To enhance the signal quality, the data presented was an average of three scanning cycles. Prior to analysis, a blank measurement using the buffer was performed to reduce the matrix interference in the protein spectra.
Results
Purification of Human Plasma Derived Celestrase 1
The purification process of C1-INH from cryopoor plasma involved a combination of chromatography steps capable of removing protein contaminants. After capturing prothrombin complex protein from cryopoor plasma, the flow through fraction of weak anion exchange column was loaded onto a strong anion exchange column which efficiently captured and partially purified C1-INH. Major contaminating proteins were removed in the flow through and subsequent washes of the strong AEX column were performed before the elution of the C1- esterase. Other two chromatography (weak anion exchange chromatography and hydrophobic interaction chromatography) used were efficient enough to remove the contaminating proteins and yielding a high product purity, desired for therapeutic use (data not shown). The average recovery of the process was 44.4 ± 3.6% yielding 0.75 ± 0.1 vials/L of C1-INH in cryopoor plasma.
Virus safety of the product was ensured by incorporating solvent detergent treatment and virus retentive filtration which will inactivate/remove potential enveloped and non-enveloped viruses.
The purified human C1-INH concentrate solution was formulated, sterile filtered and lyophilized to prepare drug product. The process flow details of the market comparator and Celestrase 1 are outlined in Figure 1. Quality attributes of this product were assessed and compared with market comparator.

Process flow of the market comparator and Celestrase 1.
Purity
Specific Activity
The specific activity is a measure of the functional C1-INH's contribution to total protein and a purity indication was comparable for both Market Comparator (8.5 IU/mg) and in-house drug product (7.2 ± 1.0 IU/mg) and results are indicated in Table 1.
Purity of Celestrase 1 showing comparable results with market comparator.

Electrophoretic Profiling
The purity of Celestrase 1 was assessed and compared by SDS-PAGE analysis with market comparator. The nature of covalent bonds can be studied by comparing the electrophoretic profiles of the proteins by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) under reducing and non-reducing conditions. As SDS disrupts interactions uniformly, noncovalent aggregates are typically not identified. 15 As a result, both reducing and non-reducing conditions were used to perform the SDS-PAGE procedure.
Noncovalent aggregates are usually not detected because the SDS disturbs the interactions alike. 15 Thus, the SDS-PAGE technique was carried out under non-reducing and reducing conditions (Figure 2). The silver-stained gel of SDS-PAGE analysis of the C1-INH purified drug product showed one major band for all the samples and market comparator at approximately 105KDa under non-reducing condition (Figure 2A) whereas ̴85–93 Kda in reducing condition (Figure 2B)
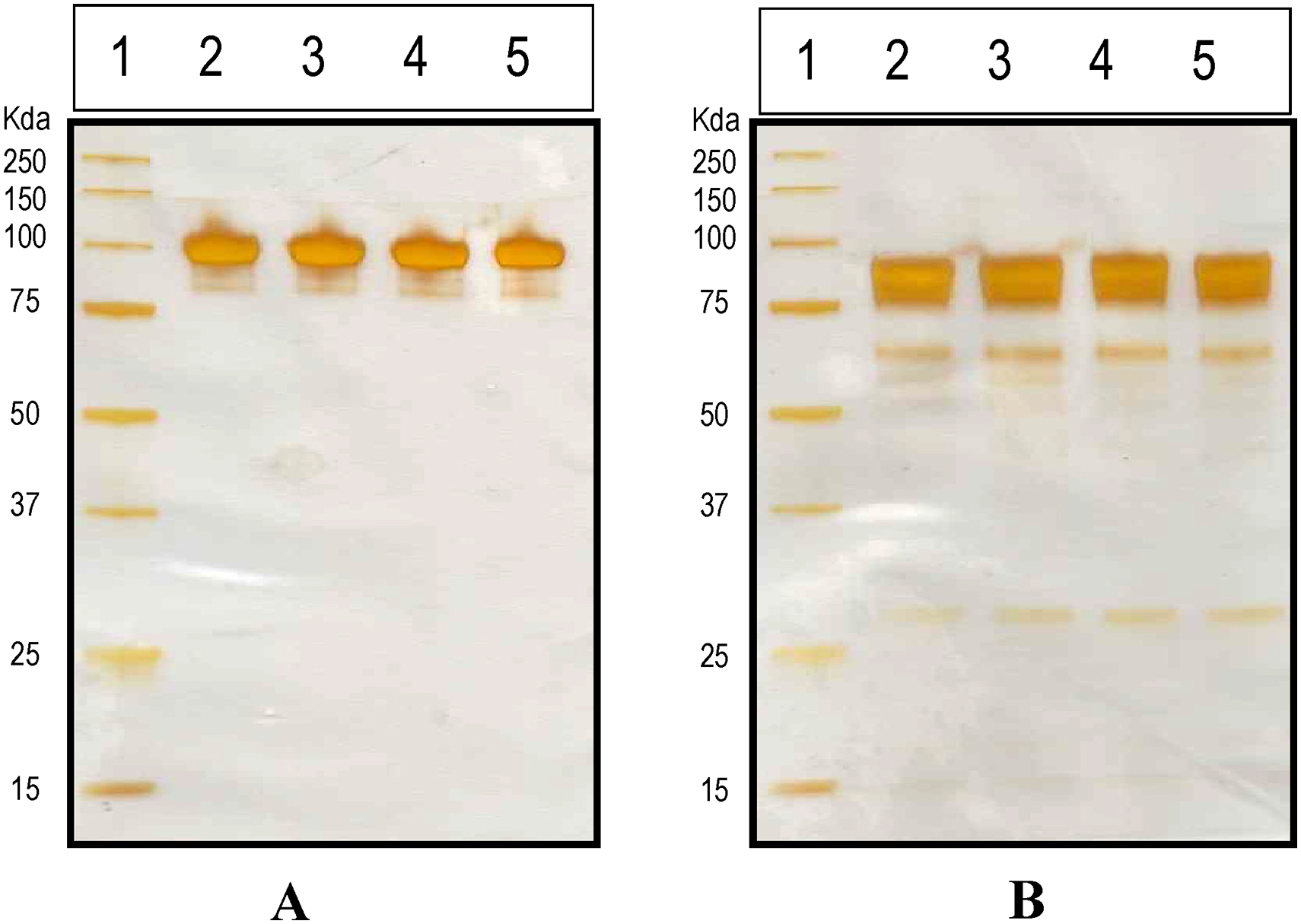
Composition and purity of Celestrase 1 by 10% silver-stained electrophoresis under non-reducing and reducing conditions. A: Lane 1: Standard Molecular weight Markers in Kda (3 µl), Lane 2: Market comparator, Lane 3: Batch-1, Lane 4: Batch-2, Lane 5: Batch-3 under non-reducing conditions. B: Lane 1: Standard Molecular weight Markers in Kda (3 µl), Lane 2: Market comparator, Lane 3: Batch-1, Lane 4: Batch-2, Lane 5: Batch-3 under reducing conditions.
Additionally, one minor band was observed below the main band at approx. 100 KDa under non-reducing conditions. Identical band profiling was evident in the reducing condition between 25 and 75 kDa for both drug product as well as market comparator.
By immunoblotting using a monoclonal antibody against C1-INH, the identity of the bands belonging to the complement 1 inhibitor protein was confirmed. Targeted protein; C1-INH was identified in Celestrase 1 as well as market comparator (Figure 3).

Identification of Celestrase 1 by immunoblotting.
Size-Exclusion Chromatography
In order to compare the quantities of protein aggregates, monomers, and fragments in Celestrase 1 to market comparator, we performed size exclusion chromatography. This is because protein aggregates of recognized biologic immunomodulators have been shown to trigger an immune response. 16 The mean retention time of the three batches of Celestrase 1 (14.226) closely matched with that of the market comparator (14.213).
The purity profile of drug product batches was comparable to the market comparator, with one major monomer peak along with aggregate peak eluting before the main peak and low molecular weight impurities/ fragments just after the main peak (Figure 4). Table 1 shows the purity of the C1-INH product was 96.749% ± 0.7%, aggregate levels were 2.507% ± 0.8% and low molecular weight fragments 0.744% ± 0.4% whereas market comparator exhibited 95.605% purity with aggregates 3.803% and low molecular weight impurities 0.592%.

Overlay of size exclusion chromatograms of Celestrase 1 and market comparator.
Reverse Phase Chromatography
Reverse-phase chromatography (RP-HPLC) allows for the separation and measurement of hydrophobic protein variations including chain chipping and oxidation. 17 The overlaid chromatogram's of C1-INH product was comparable with market comparator (Figure 5). The retention time of the major peak in the drug product (21.401 ± 0.02 min) was in close proximity with market comparator 21.427 min. The purity of C1-esterase drug product was 99.226% ± 0.1% while for market comparator was characterized by 99.366% purity. In the purified therapeutic product, the level of minor proteoform impurities with varied hydrophobicity that correspond to forms of C1-esterase was 0.774% 0.1%, which was comparable to that in the market comparator (0.635%) and results are indicated in Table 1.

Overlay of reverse-phase chromatograms of Celestrase 1 and market comparator.
Functional Activity
C1-INH inhibits several proteases and thus plays an important role in the down-regulation of major plasmatic cascade systems. It is the only regulator of the early proteases of the classical complement pathway activation (C1s and C1r).18‐20 The inhibitory effect of C1-INH on prekallikrien and conversion of high molecular weight kininogens was used to develop an endogenous analytical tool to estimate the functionality of purified C1-INH.
To prove the functionality of C1-INH in inhibiting the complement protein in classical complement pathway, the chromogenic assay was performed for both in-house drug product as well as market comparator.
The functionally active C1-INH protein in purified product exhibited 56.06 ± 1.7 IU/mL activity whereas 60.87 IU/mL in market comparator (Table 2).
Functionality of Celestrase 1 as compared to market comparator.

C1-INH activity converted from IU/ml to mg/ml, 1IU corresponds to 220ug.
Additionally, comparative dose dependent analysis for Celestrase 1 and market comparator was performed to estimate the concentration of C1-INH which inhibited the conversion of fixed quantity of prekallikrein to kallikrein. The results indicated in Figure 6 showed that the purified product at a concentration of 0.031 IU/mL could inhibit 8 IU/mL of prekallikrein whereas market comparator exhibited its functionality at an inhibitory C1-INH concentration of 0.062 IU/mL (Table 2). Thus, it is evident that lower concentration of Celestrase 1 was more effective is inhibiting 8 IU/mL prekallikrein than market comparator.

Dose dependent kinetics of Celestrase 1 and market comparator.
The functionality was also proved by determining the ratio of antigen-to-biologic activity. For this purpose, the antigenic content in the Celestrase 1 as well as market comparator was estimated. The antigenic content of Celestrase 1 was 11.62 ± 0.2 mg/mL whereas market comparator exhibited 10.6 mg/mL (Table 2). The estimated biologic activity was used for calculating the antigen-to-biologic ratio. This ratio for Celestrase 1 (0.94) was found comparable to market comparator (0.79) and the results are indicated in Table 2.
Glycosylation
C1-INH is highly glycosylated single chain polypeptide protein with 6 N linked and more than 12 O linked glycosylation sites. The N-glycan profiling of both Celestrase 1 and market comparator was studied.
Each N-glycan profile typically showed one major peak accompanied by some minor species. The major peak in Celestrase 1 and market comparator were Biantennary, disialated, galactosylated, non-fucosylated (A2G2S2) structures which contributed to 57%–60% of total glycan species (Table 3). These structures are associated with sialic acids, which are not essential for biological activity but enhance in vivo half-life and in vitro protein stability.21‐23
The relative % area of glycan species in Market comparator and Celestrase 1.
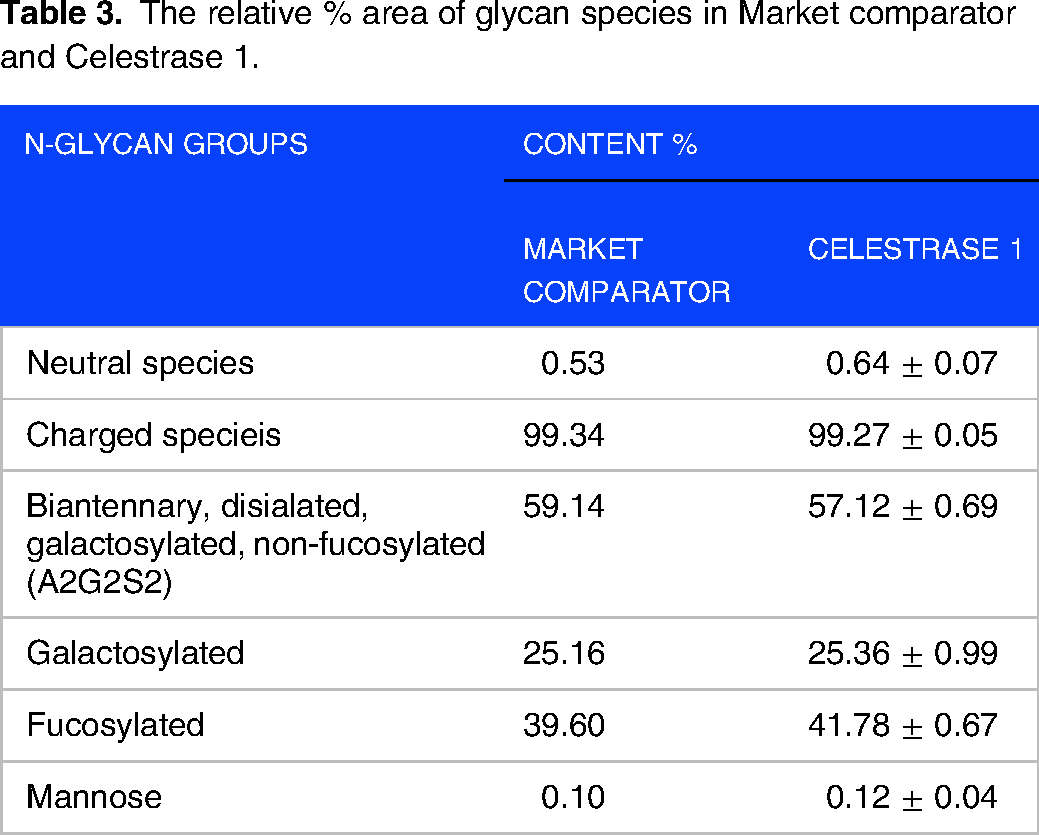
The entire pattern of market comparator & Celestrase 1 product was represented by charged species in abundance (99.34%, 99.27 ± 0.05%), Biantennary, disialated, galactosylated, non-fucosylated species (A2G2S2- 59.14%, 57.12 ± 0.69%), Galactosylated (25.16%, 25.36 ± 0.99%), Fucosylated (39.60%, 41.78 ± 0.67%), Neutral species (0.53%, 0.64 ± 0.07%) and mannose species (0.10%, 0.12 ± 0.04%). Less mature type such as high-mannose are present in relatively small amounts.
The N-linked glycan profile for three batches of Celestrase 1 was found comparable to market comparator (Figure 7).

Comparative N-glycan profiles of the Celestrase 1 and market comparator.
The analysis of O-linked glycosylation remains a challenge due to the lack of equivalent enzymes and the inherent structural heterogeneity of O-glycans. Fortunately, in N-glycan analysis, a universal enzyme exists to deglycosylate a variety of common-core structures from proteins for analysis using mass spectrometric and fluorescence techniques. Unfortunately, for their O-linked counterparts, no such enzyme exists. Furthermore, O-glycan heterogeneity is vast due to the lack of a common glycan core, making analysis challenging. As a result, O-glycans are released via chemical techniques; nevertheless, these techniques frequently harm the glycan's structure because of “peeling” processes.
Far-UV CD Spectroscopy
The secondary structure (α-helixes, β-sheets, and “random” structures) of the Celestrase 1 was determined by circular dichroism (CD) spectroscopy in the far UV region (Figure 8).
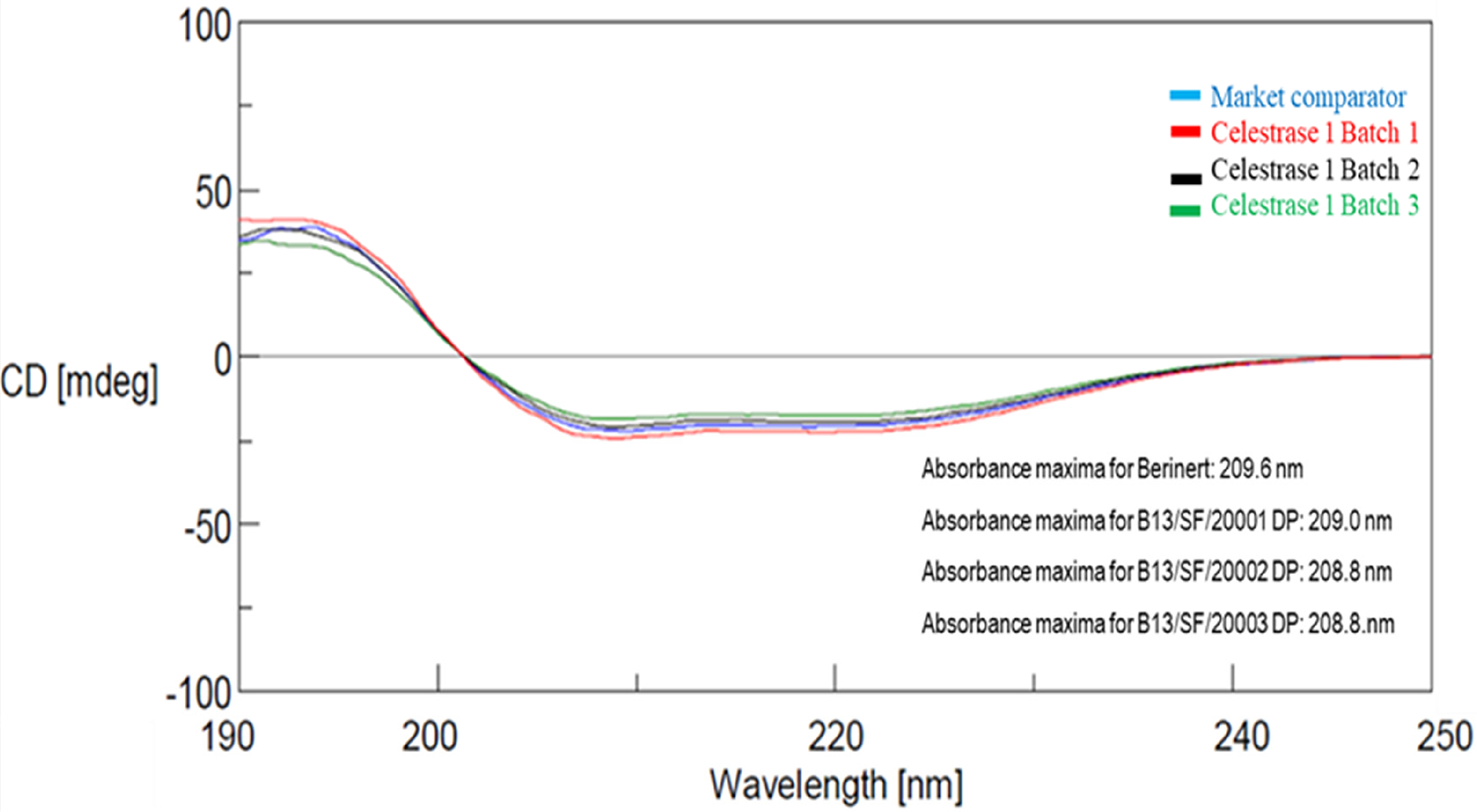
Far UV CD spectra - overlay of Celestrase 1 with market comparator.
The spectra in the far-U.V region showed peak maxima at 191.1–193.7 nm and peak minima at 203.8–209.6 nm; characteristic of α-helical proteins. Far-UV CD spectroscopy results depict that the indigenously developed Celestrase 1 product shared a similar structure with market comparator (Figure 7) with absorbance minimum (∼209 nm).
The reconstituted product was found to be stable for 36 h at 25° ± 3 °C (Figure 9).

Reconstitution stability study of Celestrase 1 at 25° ± 3 °C.
Discussion
This study illustrates the biochemical and functional characterization of Celestrase 1. The product quality attributes were determined by employing different analytical methods.
The manufacturing process involved three chromatographic purification steps to yield a high purity product. Two dedicated orthogonal virus clearance steps – solvent detergent treatment and virus retentive filtration were incorporated into the process to ensure the virus safety of the Celestrase 1.
The purity of Celestrase 1 was determined to assess the amount of high molecular weight impurities by SE-HPLC and number of impurities with different hydrophobicity by RP-HPLC. Purity of the Celestrase 1 was comparable with market comparator by both the methods.
Additionally, electrophoretic purity profile of the Celestrase 1 matched with the purity profile obtained with market comparator, used for reference. The results of the characterization studies indicated the comparability of the protein with an existing market comparator with respect to its purity by SDS-PAGE, RP-HPLC, SE-HPLC and specific activity. This demonstrates the effectiveness of the manufacturing process steps for removal of contaminating proteins. As a result, the side-effects are minimized with products having high purity. 24
Since it is known that reduce functional activity of the protein is attributed to protein aggregation, degradation and can induce immunogenicity, study of functional activity by different mechanisms was thus emphasized upon. 25 C1-INH plays an important role in the regulation of the classical pathway of the complement system, kallikrein system and fibrinolytic system.6‐8 The C1 INH protein blocks both spontaneous activation of C1 and the formation of activated C1, thereby disallowing the generation of C2 and C4 complexes leading to the production of anaphylactic, chemotactic and vasoactive peptides.
Likewise, Celestrase 1 was found to be functional in blocking the activation of complement 1 protein and thereby proving its potential to regulate the classical pathway.
Also, functionality was determined in the kinin releasing system wherein C1 INH regulates conversion of prekallikrein to kallikrein. C1 INH insufficiency leads to increased kallikrein, which in turn increases bradykinin product. This results in increased vascular permeability and massive local uncontrolled oedema.6‐8 The purified product was shown to inhibit the activation of prekallikrein to kallikreinin in dose dependent manner, thereby regulating the kinin pathway. This confirms the great similarity in biological activity of C1-INH-based therapeutics. 26
The biochemical and functional analysis of pure Celestrase 1 showed that the manufacturing process resulted in a consistent product composition with preserved functionality, as evidenced by functional activity and by ratio of antigen to activity (by chromogenic assay).
C1-INH is a highly glycosylated protein. Its glycosylation pattern significantly affects the proteolytic specificity, activity and potency.27‐29 It is known that N-glycans can influence protein folding, immune regulation, cellular homeostasis, potency, efficiency and the biological half-life of proteins. 30 The N-glycans are considered a critical quality attribute contributing to the conformational stability and kinetics of protein binding to the target proteases. The highly glycosylated N-terminal domain of the C1-INH plays an important role in binding to the substrate.28,31‐33 Recent research has shown that a high level of sialylation can increase the serum half-life of a protein, while desialylation of the protein can lead to faster elimination from the blood, possible due to the affinity of penultimate galactosyl residues to the asialoglycoprotein receptor in the liver. 34
The structural analysis of Celestrase 1 was found to be comparable to market comparator and exists predominantly in α- helix secondary structure with absorbance minimum (∼209 nm) according to far-UV CD spectroscopy findings.
Limitations
India annually collects about 10–11 million units of blood which is about 0.9% rate of blood donation. This may not be sufficient and therefore many times, especially in the situations of crisis, blood is in short supply in urban and rural areas. 35
Conclusion
Despite of the difference in the purification methods of Celestrase 1 and market comparator, both the products showed similar quality attributes. A pure, consistent and functionally efficient protein reflects a controlled and robust manufacturing process. Thus, the plasma derived Celestrase 1 can be used as a therapeutic option for patients with hereditary angioedema (HAE). C1-INH can also be explored for its clinical efficacy in other therapeutic applications like clinical amniotic fluid embolism. 36
Footnotes
Ethics Approval and Consent to Participate
Indian Blood banks provided freshly frozen recovered plasma in polyvinyl bags. These blood banks had been approved by the Drug Controller General of India (DCGI) -NOC No. X11026/269/2018-BD and registered with the government of India's National Aids Control Organization. Blood was collected after receiving written consent from the blood donors and after verification by medical practitioner.
Author Contributions
Sheetal Dolia: Formal analysis, Project administration, Supervision, Writing – original draft. Sachin Verma: Methodology, Supervision, Writing – original draft. Suma Ray: Formal analysis, Project administration, Supervision, Writing – review & editing.
Authorship Contributions
The Celestrase 1 process development mentioned in this manuscript was performed by Sachin Verma. Analytical assays were developed under the guidance of Dr. Sheetal Dolia. Dr. Suma Ray has provided technical input and guidance for this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the management of Intas and did not receive any financial grant from other funding agencies in the public or commercial sectors.
Acknowledgments
We are grateful to the management of Intas Pharmaceuticals Ltd for providing the opportunity and resources to complete this project. We are thankful to all donors who have contributed to the project by donating their blood/plasma.
