Abstract
Hereditary angioedema (HAE) is a rare but often life-threatening genetic disorder caused by either lack of or dysfunctional C1-esterase inhibitor (C1-I3NH). C1-INH is a high-cost therapy for HAE. Because plasma-derived C1-INH has a higher half-life than other therapeutics, it may be considered a suitable treatment option for HAE. However, the high cost of plasma-derived C1-INH may make the therapy unaffordable to some patients. Herein, we review the C1-INH purification processes used to date to identify the reasons for the high costs of this therapy and assess the scope of improvements in the purification processes to develop a cost-effective product. A literature search was conducted in PubMed and Google Scholar in January 2023 to identify the relevant articles. The following keywords were used: “C1 esterase inhibitor,” “hereditary angioedema,” and “C1 esterase inhibitor purification from human plasma.” All English-language articles published from 1960 to February 2021 were included. Several plasma products can be purified using the Cohn fractionation process; this decreases the cost of individual products. However, many C1-INH purification strategies developed to date fail to purify other products from the unused plasma fraction (ie, the fraction remaining after C1-INH purification) because of changes in the physical characteristics of the plasma, such as pH and ionic strength; as a result, it is unavailable for the Cohn fractionation process, leading to the high cost of C1-INH. Using cutting-edge purification technologies and more efficient virus removal steps can decrease manufacturing costs, thereby increasing the affordability and safety of the product.
Background
Hereditary angioedema (HAE) is characterized by the loss of C1-esterase inhibitor (C1-INH) protein or function.1,2 In individuals with HAE, the face, upper airways, genitalia, and gastrointestinal system are affected by recurrent bouts of nonerythematous, nonpruritic submucosal, or subcutaneous edema.1,3,4 Angioedema attacks can result in substantial morbidity. 5 Furthermore, upper airway edema can cause asphyxiation, which is life-threatening. 6 Approximately 1 in 50 000 people suffer from HAE. 7 A crucial protease inhibitor known as C1-INH is a member of the serine protease inhibitor superfamily of proteins and its deficiency is the source of HAE. 8
HAE treatment is achieved by substituting C1-INH purified from human plasma or produced via recombinant DNA technology; however, this therapy is expensive. Nevertheless, owing to the higher half-life of plasma-derived C1-INH compared with recombinant C1-INH and other therapeutics, it should be considered a suitable treatment option for HAE. To date, many purification strategies have been developed to purify therapeutic-grade C1-INH from human plasma. Globally, many plasma fractionators have successfully developed and marketed C1-INH isolated from human plasma.
Using the Cohn fractionation process, several products can be purified from plasma, thereby decreasing the cost of individual products. The Cohn fractionation process involves plasma protein precipitation at a specific pH, ethanol concentration, ionic strength, and temperature. However, many C1-INH purification strategies developed to date fail to purify other products from the unused plasma fraction (ie, the fraction remaining after C1-INH purification) because of changes in the physical characteristics of the plasma; therefore, it is unavailable for the fractionation process. Plasma fractionators and protein scientists have majorly focused on developing C1-INH as a therapeutic product; however, in the race for product development, they have overlooked the importance of appropriately using plasma for extracting all possible products, thereby increasing the demand for plasma.
In this review, we focus on the purification strategies used to isolate and purify C1-INH to date and identify the scope of using the fraction remaining after C1-INH purification for the Cohn fractionation process. Developing a C1-INH purification process that does not alter the physical characteristics of the remaining plasma, including pH, conductivity, and ionic strength, will allow more appropriate use of the plasma. Furthermore, it will not only decrease the cost of plasma products but also quench the demand for plasma in countries where recovered plasma is the major source of plasma-derived medicinal products. This review follows the recommendations of the Narrative Review reporting checklist.
A literature search was conducted in PubMed and Google Scholar in January 2023 to identify the relevant articles. The following keywords were used: “C1 esterase inhibitor,” “hereditary angioedema,” and “C1 esterase inhibitor purification from human plasma.” All English-language articles published from 1960 to February 2021 were included.
About HAE
HAE is inherited in an autosomal dominant manner and has two primary subtypes. Approximately 85% of HAE cases are Type I, which is caused by a quantitative deficiency of C1-INH. The remaining 15% of HAE cases are type II, which is caused by a defective C1-INH protein. Additionally, a third type of HAE has been identified. This condition is predominant in females, and most individuals have normal quantitative and functional C1-INH levels. This condition may be linked to higher estrogen levels. In some cases, a gain-of-function mutation in factor XII, a protein implicated in the coagulation cascade, has been linked to HAE. 9
Bradykinin, a consequence of the plasma contact system, is the main mediator of HAE during episodes of edema. The regulation of the complement cascade, coagulation, and contact systems depends on C1-INH. It inhibits important proteases in these pathways, including plasmin, kallikrein, and activated Hageman factor (Figure 1). Ultimately, C1-INH is responsible for controlling bradykinin synthesis. Stressful or traumatic events may activate the complement and contact pathways. Increased bradykinin levels in the presence of C1-INH deficiency (type I HAE) or C1-INH dysfunction (type II HAE) cause recurring episodes of angioedema. 10
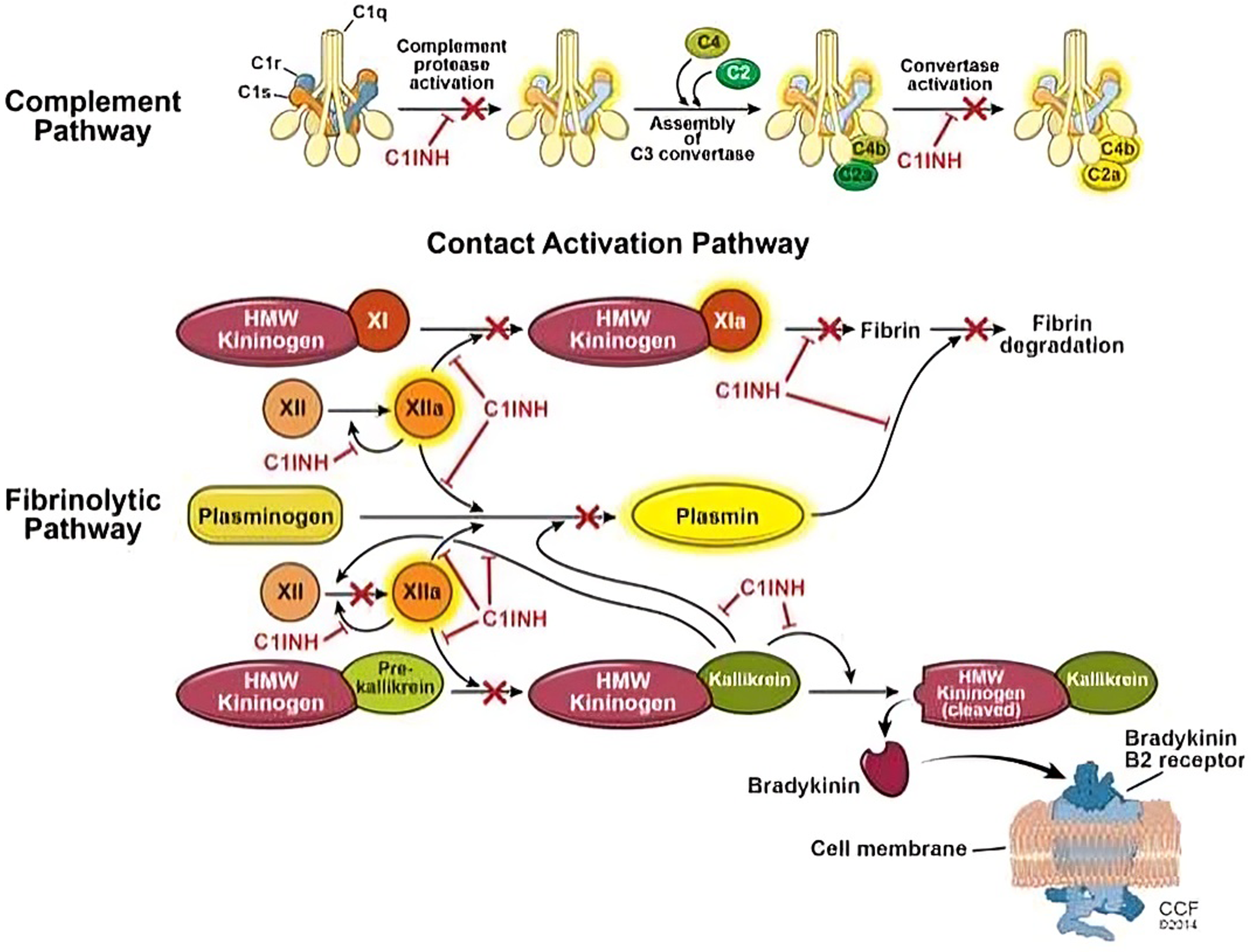
Role of C1-INH in inhibiting the contact activation and fibrinolytic pathways. The image has been adopted from Siles R. Hereditary angioedema. C1-INH, C1-esterase inhibitor.
Typical HAE symptoms and indicators generally appear during childhood or puberty and persist till death. The prodromal tingling feeling that typically precedes an episode may be followed by a nonpruritic wavy rash. Thereafter, swelling gradually develops and progressively disappears over 48-72 h. Steroids and/or antihistamines neither affect the type of swelling observed in HAE nor are they connected with urticaria occurrence. Oropharyngeal edema should be carefully treated because, if left untreated, it can lead to suffocation and even death. Although these attacks are few, more than 50% of patients with HAE experience at least one oropharyngeal attack in their lifetime. 11
Treatment of HAE
The treatment regimens for HAE include short- and long-term prophylaxis and the management of acute HAE attacks. The long-term objective is to decrease the frequency and severity of angioedema episodes. Studies have reported that individuals with HAE often experience 1-2 swelling attacks per month. 12
Long-term Prophylaxis
Both long-term prevention and acute therapy are possible with C1-INH concentrate. This method of care was used to treat the first patient with HAE-C1-INH in 1989, with good results. Additional research revealed positive outcomes in terms of efficacy and safety. The recommended dosage of C1-INH concentrate is two 500 IU doses each week. The amount of HAE attacks dropped from 12.7 to 6.3 in a randomized, controlled cross-over study including 22 patients over two 12-week periods using the nanofiltered C1-INH concentrate Cinryze® at a dose of 2 1000 IU/week. The frequency and length of the remaining assaults were markedly reduced. In the US and Europe (Shire, UK), Cinryze® was approved for long-term prevention in October 2008 and June 2011, respectively. Since 2017, the EU has approved Cinryze® for preventing HAE-C1-INH in children starting at age 6. 13
Berinert® is also used in long-term intravenous prophylaxis. Case reports and patient series provide evidence of its strong efficacy. Fourteen individuals were administered one or more Berinert® injections weekly for an average of 9 years in an uncontrolled research. As a result, only 3.8% of the attacks during prophylaxis were severe compared with 93.3% of attacks before therapy. 13
Even in patients on long-term prophylaxis and receiving frequent injections of C1-INH concentrate, an increase in disease activity is occasionally observed, as evidenced by an increase in the number of attacks, higher C1-INH concentrate dosage requirements, and/or the occurrence of rapidly progressing multilocular episodes. 13
Most patients who receive long-term prophylaxis with C1-INH concentrate either administer the medication themselves or permit a family member or worker from a for-profit home-care business to do so (see above, “Self-treatment at home”). Multiple intravenous injections are administered in this treatment method; thus, patients should be prepared for it (see “Self-treatment at home” above). Furthermore, regular follow-up are needed at least once each year to review efficacy and determine whether long-term prophylaxis should be continued or if switching to on-demand medication is an option. 13
Long-term prophylaxis with the Berinert derivative Haegarda®, which has been approved in the US since 2017, has demonstrated considerable success. As a result, the subcutaneous forms of “Berinert 2000 IU” and “Berinert 3000 IU” will also be offered in Europe. 13
Short-term Prophylaxis
Trauma or stress may induce angioedema episodes. To decrease the risk of oropharyngeal edema, individuals with HAE must be extremely precautious during the perioperative period, particularly when undergoing dental treatment. Fresh frozen plasma, high-dose anabolic androgens, and C1-INH replacement therapy are effective for this purpose.14,15 When using Cinryze®, the manufacturer advises short-term prophylaxis with 1000 IU within 24 h preoperatively, and when using Berinert®, the manufacturer advises administration of 1000 IU for adults within 6 h preoperatively and 15-30 IU/kg BW for children and adolescents. Because not all attacks can be prevented despite this type of short-term prophylaxis, additional medication should be accessible during the course of the next 24 h. 13
Acute Attacks
Berinert was initially approved by the FDA in 2009 for managing acute facial, laryngeal, or gastrointestinal attacks in adolescents and adults. Berinert is a human, pasteurized, plasma-derived C1-INH medication. In July 2016, the FDA approved it for use in children. In a double-blind, placebo-controlled study involving 125 patients with HAE, Berinert decreased the median time of onset of relief from an acute attack from 1.5 to 0.5 h (P = .0025 vs placebo). The dosage used was 20 U/kg IV. In general, symptoms disappear within 30-60 min, and long-term prophylaxis is not needed with this C1-INH concentrate composition.
Recombinant C1-INH (Ruconest), which was approved by the FDA in July 2014 to treat acute attacks in patients with HAE, is the most recent therapeutic option. This preparation falls under pregnancy category B (ie, studies that have failed to demonstrate a risk to the fetus, and there are no adequate and well-controlled studies in pregnant women). However, this medication is contraindicated for patients with known or suspected rabbit hypersensitivity reactions because it is purified from the milk of genetically engineered rabbits. The medication is self-administered at a dose of 50 IU/kg for individuals less than 84 kg and at 4200 IU for those more than 84 kg. 16
Table 1 describes the C1-INH-based therapeutic treatment options for HAE.
C1-INH-based therapeutic options for hereditary angioedema.
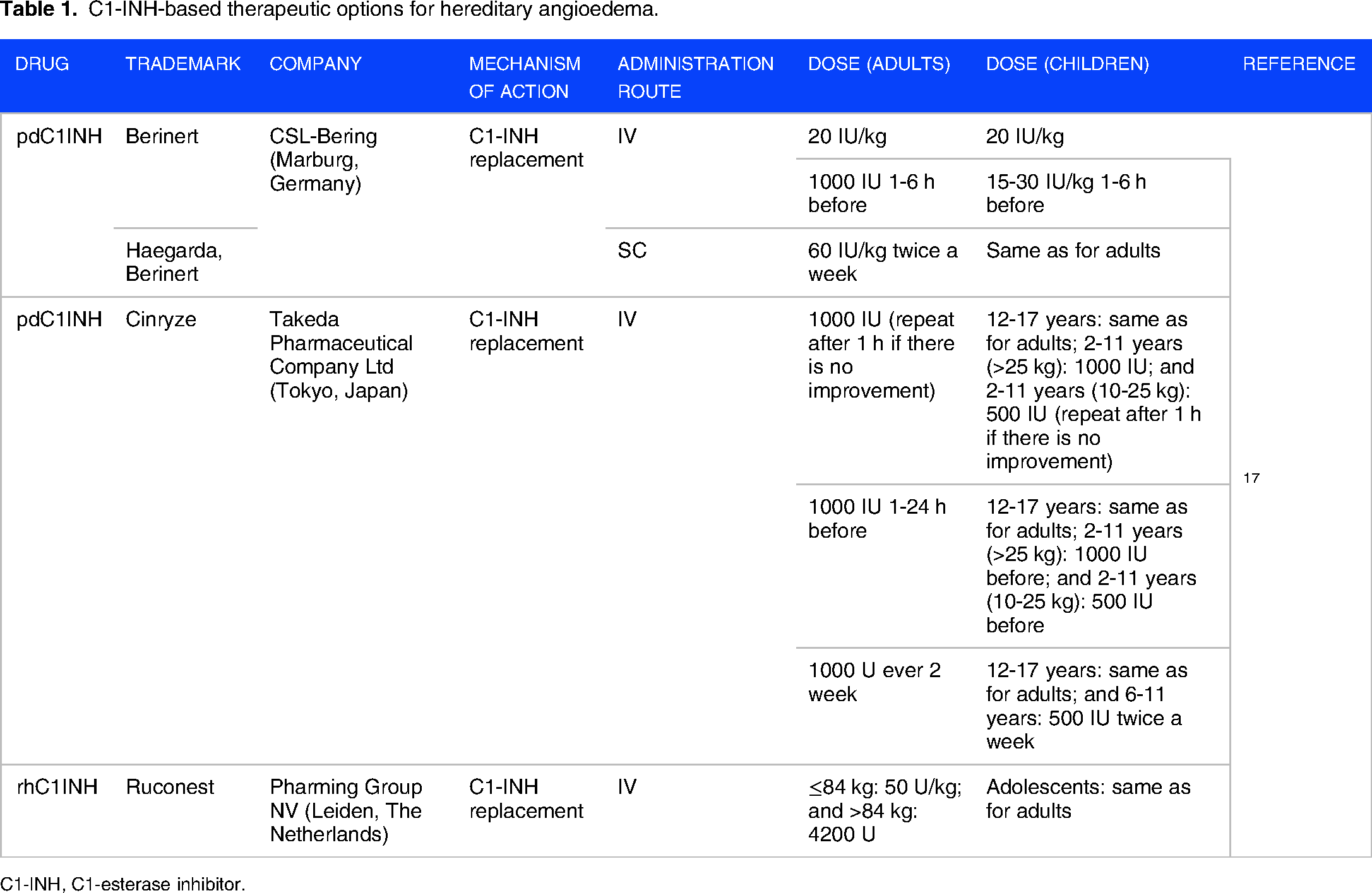
C1-INH, C1-esterase inhibitor.
Plasma-derived C1-INH is a Suitable Treatment Option
Table 2 demonstrates that plasma-derived C1-INH has a longer half-life compared with the recombinant version; therefore, it should be considered a suitable treatment option for HAE.
Comparison of the pharmacological data of various C1-INH products.

C1-INH, C1-esterase inhibitor.
Donor Selection Criteria
Prospective donor screening is the first major step in the manufacture of plasma-derived proteins. Then, plasma is collected from eligible donors and used for testing. Table 3 summarizes the donor eligibility criteria by combining FDA requirements, European requirements, and industry-adopted regulations. 22 In case of conflicting rules, the most restrictive rule is used. Plasma samples are aseptically collected in a closed, sterile system using an anticoagulant solution containing 4% sodium citrate. Each sample is labeled with a unique identification number that is connected to the individual donor. Therefore, each sample can be uniquely traced and connected to laboratory test results as it is transported through the inventory hold, logistic centers, and plasma pool. Source plasma is stored at −20 °C even during transport.
Plasma pools are tested by using nucleic acid testing and serology. For manufacturing CI-INH, the plasma pools must be negative for HBV, HCV, HIV, and HAV and contain ≤10 000 IU/mL of B19 V DNA. A threshold concentration of B19 V is allowed in plasma pools because during the viremic period for B19 V, which can be very long, low levels of B19 V coexist with B19 V antibodies (potentially complexing with and neutralizing the virus). Therefore, removing plasma units with low levels of B19 V DNA is undesirable because it will decrease the B19 V antibody levels in plasma pools and some of the resulting plasma-derived products (immunoglobulins). 25 Furthermore, from the perspective of pathogen safety, the presence of B19 V DNA does not represent a risk because the C1-INH manufacturing process contains a dedicated virus reduction step. This step can efficiently inactivate and remove any B19 viruses that may be present in the pool.
Cohn Fractionation Process and Its Importance in the Plasma Industry
Cohn and his colleagues developed the first plasma fractionation process in the 1940s to prepare albumin and immunoglobulins. This process relies on sequential precipitation steps at negative temperatures, different ethanol concentrations, and various pH values to segregate bulk plasma proteins. Table 4 describes the conditions for the various fractionation steps.
Conditions for plasma fractionation.

To date, the Cohn fractionation process has undergone several modifications to optimize the yields of various plasma-derived products. In modern plasma fractionation approaches, the cold insoluble fraction of plasma (cryoprecipitate) is separated via centrifugation, which is rich in factor VIII, fibrinogen, and von Willebrand factor. The remaining plasma (cryopoor plasma/cryosupernatant) can further be fractionated using the Cohn fractionation scheme. 27 Table 5 summarizes the products that can be isolated from an individual plasma fraction.
List of products from various plasma fractions.

Recent estimates by the Marketing Research Bureau place the current volume of plasma segregated globally between 42 and 45 million liters. Since the start of the plasma fractionation industry, this volume has been gradually increasing over time. Plasmapheresis is a specialized method for isolating only plasma (commonly referred to as “source plasma”), while blood cells, particularly red blood cells, are returned to the donor. This method is used to acquire the majority (approximately 80%) of the fractionated plasma. As a by-product of whole blood collection that is motivated by the clinical need for red blood cells for transfusion, a small portion of the plasma used for fractionation (referred to as “recovered plasma”) is retrieved.
As long as the collection procedure complies with national blood policies, the quality requirements from the fractionator, and the GMP specifications from the pertinent regulatory authorities who oversee the blood collection system and are accountable for the marketed plasma products, both source and recovered plasma are suitable for fractionation.
The volume collected at each donation (approximately 600-880 vs 100-250 mL, respectively), maximum frequency (two times/week or 24 times/year vs 4-5 times/year, depending on jurisdictions), and chemical make-up of the citrate-based anticoagulant solution are the main differences between source and recovered plasma.
Source plasma can normally be frozen within minutes after completing collection; however, the freezing process of recovered plasma is longer because whole blood must first be processed into its component parts. Furthermore, source plasma typically contains 10%-30% more labile coagulation factor VIII than recovered plasma but 10%-20% less IgG in response to the frequency of plasma donation.29,30 In a public context, the price of recovered plasma, for which cost allocation with red blood cells and platelets is conceivable, is higher than that of apheresis plasma because recovered plasma does not have the same strict cost-effectiveness requirements as in private for-profit organizations. 31
This is significant because plasma costs account for 20%-40% of the cost of producing plasma products, which affects the dynamics of the plasma fractionation market.32,33
The amount of plasma required for fractionation to meet clinical needs is predicted to continue increasing in the coming years, 33 considering that any potential advances in plasma fractionation technology, particularly in the recovery of the driving proteins (IgG), are not anticipated to significantly affect fractionation outcomes in the near future.
The Cohn fractionation process allows the efficient use of the plasma for isolating maximum plasma-derived medicinal products from a single lot of plasma. Furthermore, it provides the liberty of storing plasma fractions and their processing as per requirements.
Therefore, fractionation is a key factor in the decision-making of various products on a campaign basis while maintaining the economics of the process, particularly in countries where recovered plasma is the major source of plasma-derived medicinal products.
Figure 2 indicates the stage of plasma fractionation process at which C1-INH is isolated and further purified. During C1-INH isolation, if physical conditions such as pH and conductivity of the remaining fraction are altered, the Cohn fractionation process cannot be performed. This makes the remaining plasma unavailable for the purification of other products. Therefore, the capture stage of the C1-INH purification process is an important factor in controlling the cost of C1-INH product.

Detailed process flow for the C1-INH capture stage and fate of remaining plasma fraction. C1-INH, C1-esterase inhibitor.
C1-INH Purification Process
Numerous purification techniques that rely on anion exchange, affinity, and hydrophobic interaction chromatography and precipitation (either using polyethylene glycol [PEG] or ammonium sulfate), have been documented to date (Table 6). Protein denaturation during the precipitation step often results in a lower process yield. 34 Table 6 summarizes the various purification methods documented to date. Each strategy has combined the chromatographic method with at least one precipitation step. C1-INH was captured using an anion exchange column with a plasma preparation step using PEG precipitation, according to a technique described by Nilsson et al in 1982. Furthermore, two processes using PEG precipitation and chromatographic methods were reported in 1987 and 1989; these processes also used PEG precipitation and chromatographic methods. In 1990, Pelzer et al reported that coagulation factor capture before C1-INH collection can enhance the efficiency of plasma usage. Recently, other procedures where C1-INH was purified from residual plasma after the removal or capture of coagulation factors have been reported (Table 6 and Figure 3).

Purification flow chart of commercially available C1-INH 43 . C1-INH, C1-esterase inhibitor.
Processes developed for purifying C1-INH from human plasma.

Abbreviations: Yield, Amount of C1-INH recovered in IU, with respect to initial amount of C1-INH in plasma (IU); C1-INH, C1-esterase inhibitor; DEAE, Diethylethanolamine; QAE, Quaternary ammonium; CM, Carboxymethyl; TMAE, Trimethylammoniumethyl; PEG, Polyethylene glycol; AEX, Anion exchange chromatography; CEX, Cation exchange chromatography; HIC, Hydrophobic interaction chromatography; NA, Not available.
Non-Industrial process.
Commercial process.
Plasma processing steps such as PEG precipitation and pH/conductivity adjustment before chromatography in the capture stage will alter the physical conditions of plasma, making it unavailable for the Cohn fractionation process. This will lead to the inappropriate use of plasma as other potential products cannot be purified from the remaining plasma.
Additionally, lower process yields may be associated with precipitation steps. There is a wide scope of process development, and yields may be considerably increased by applying cutting-edge chromatographic techniques such as tentacle resin, which provide better binding capacity for large protein molecules. 45 Furthermore, the flexibility of C1-INH capture at higher conductivity (near plasma conductivity) may be possible by using next-generation chromatographic resins such as affinity and mixed-mode resins, allowing the remaining fraction to be diverted to the fractionation process and producing other plasma-derived medicinal products from the same lot of plasma.
Longer process time also increases product costs because it involves the cost of running a GMP facility used for product manufacturing. Moreover, steps such as precipitation are time-consuming because they involve the separation of precipitated proteins by centrifugation and can be easily replaced by using advanced purification techniques such as affinity chromatography.
Below are two flow charts for the purification of C1-INH (Figure 3), where ammonium sulfate and PEG precipitation has been used. These steps can be omitted by using the most recent purification technologies.
Similarly, pasteurization, the virus inactivation step can be replaced with other virus inactivation methods, such as solvent detergent treatment, which take lesser time, have less impact on product quality, and inactivate enveloped viruses. 46
Dialysis, a lengthy procedure performed for buffer exchange, has also been performed in some processes 38 ; this step can easily be replaced with tangential flow filtration.
Efficiently optimized chromatography steps may lead to the successful purification of C1-INH from plasma with better recovery and purity. Furthermore, solvent detergent treatment and virus retentive filtration during purification may lead to a virus-safe product. Buffer exchange using tangential flow filtration may decrease the process time. Finally, carefully using optimized unit operations will result in an efficient manufacturing process.
Virus Reduction and Purity in Available Market Products
The manufacturing process for Berinert or Haegarda & Cinryze includes multiple steps that reduce the risk of virus transmission. The virus inactivation/reduction capacity consists of three steps (Table 7).
This was evaluated in a series of in vitro spiking experiments. The total mean cumulative virus inactivation/reduction is shown in Table 8.
Abbreviations: HIV, Human immunodeficiency virus type 1, a model for HIV-1 and HIV-2; BVDV, Bovine viral diarrhea virus, a model for HCV; PRV, Pseudorabies virus, a model for large enveloped DNA viruses; WNV, West Nile virus; HAV, Hepatitis A virus; CPV, Canine parvovirus; B19V, Human Parvovirus B19; ND, Not determined; NA, Not applicable.
Table 9 indicates the size exclusion chromatographic determination of purity of two commercially available products.
Purity evaluation of C1-INH concentrates. 44
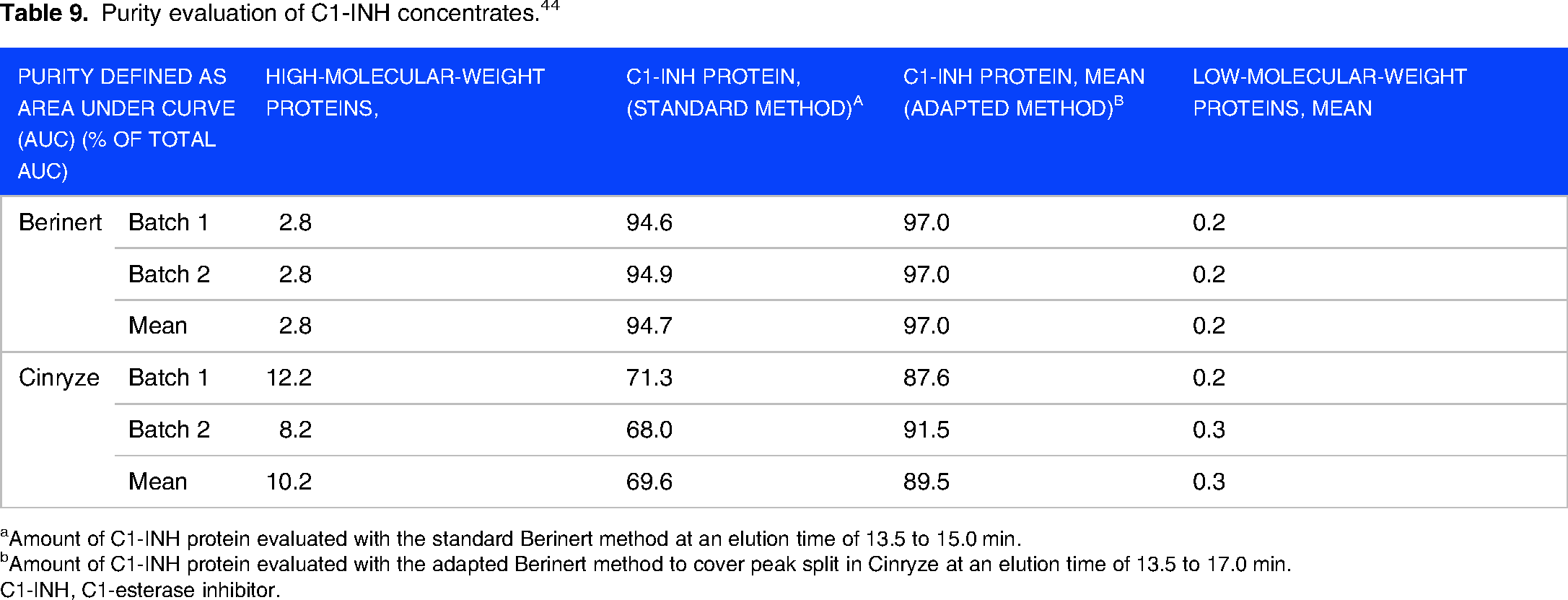
Amount of C1-INH protein evaluated with the standard Berinert method at an elution time of 13.5 to 15.0 min.
Amount of C1-INH protein evaluated with the adapted Berinert method to cover peak split in Cinryze at an elution time of 13.5 to 17.0 min.
C1-INH, C1-esterase inhibitor.
All plasma-derived C1-INH concentrates showed main protein bands at approximately 105 kDa, whereas the transgenic product showed main protein bands at approximately 98 kDa (picture not shown). Additional band patterns varied between items but were the same among batches of a same product. A band at approximately 64 kDa was discovered in the plasma-derived C1-INH preparations; it was more pronounced for Berinert than for Cinryze and was recognized as 1-antichymotrypsin by PMF analysis. Complement factor C3 in Cinryze, immunoglobulin heavy constant mu in Berinert, and ceruloplasmin in all plasma-derived C1-INH concentrate samples were additional non-therapeutic proteins. None of the samples contained any coagulation factors. 44
Conclusion
C1-INH concentrate has been available for treating acute HAE attacks for more than 20 years in parts of Europe; at present, it is considered the standard of care in many countries. 49 In this review, we discussed the importance of the beneficial effect of plasma-derived products over other therapeutic options. In India, volunteer plasma donation is enforced; in such scenarios, the most appropriate use of plasma would be isolating maximum possible products from each lot. In many purification strategies, the physical conditions of plasma, including pH and conductivity, are manipulated to isolate C1-INH; this makes plasma unavailable for the Cohn fractionation process. However, chromatographic techniques can still be used to isolate other products from the remaining plasma fraction after isolating C1-INH. Manufacturers using the Cohn fractionation platform for isolating multiple plasma-derived medicinal products may not be able to use the plasma if the physical conditions are modified. Therefore, there is a strong need to develop a process that can isolate C1-INH under the physical conditions of plasma without affecting its availability for the fractionation process, thereby making the treatment affordable.
Footnotes
DECLARATION OF CONFLICTING INTERESTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
FUNDING
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Not required as the study is review of available data.
Consent to Participate
Study does not involve participation of any subjects or patients.
Consent for Publication
Study did not involve any participants or patients.
Author contribution(s)
Acknowledgments
We would like to thank the management of Intas Pharmaceuticals Ltd and the Institute of Advanced Research for providing the opportunity to complete this project.
Availability of Data and Materials
Not applicable.



