Abstract
Demographic and hemodynamic data from patients with idiopathic pulmonary arterial hypertension (IPAH) and chronic thromboembolic pulmonary hypertension (CTEPH) have not been systematically characterized to identify differences related to gender, age, race, disease severity, and drug response. Our goal was to define the distribution and relation of IPAH and CTEPH based on these criteria. Hemodynamic and demographic data from 242 IPAH patients and 90 CTEPH patients were collected and compared. IPAH incidence was greater in women, but men had a higher basal mean pulmonary arterial pressure (mPAP). mPAP was comparable among all IPAH ethnic groups. IPAH patients with no history of fenfluramine-phentermine use had a higher mPAP than users. Exercise-induced IPAH was apparent in 14.5% of IPAH patients. Only 9% of IPAH patients responded to inhaled nitric oxide with a ≥20% decrease in mPAP. Compared to CTEPH patients, mPAP was greater but average age of diagnosis was lower in IPAH patients. mPAP negatively correlated with age of diagnosis in IPAH patients only. These results indicate that elevated CO is not the main determinant of mPAP in both IPAH and CTEPH patients. However, the two patient groups differ in terms of their demographic and hemodynamic distributions, and according to the correlation between mPAP and other clinical hemodynamics and demographics.
Introduction
Idiopathic pulmonary arterial hypertension (IPAH) is a rare vasculopathy involving small distal pulmonary arteries that may lead to right heart failure and death (51). Multiple etiology and pathogenesis have been implicated in the development of the disease. The major causes for the elevated pulmonary vascular resistance (PVR) and pulmonary arterial pressure (PAP) in patients with IPAH include sustained pulmonary vasoconstriction, intimal lesions (e.g. the plexiform lesion), vascular remodeling characterized by severe intimal and medial hypertrophy, in situ thrombosis, and vascular endothelium injury. Demographic and hemodynamic data from patients with IPAH have not been systematically characterized and compared to determine if higher mean PAP is related to gender, age, race, fenfluramine use, and/or vasoreactivity.
Each year, up to 4% of patients with acute pulmonary embolism develop chronic thromboembolic pulmonary hypertension (CTEPH) (15, 35, 44), which accounts for 20,000-40,000 cases per year. CTEPH arises when pulmonary embolism fails to resolve (47), and occurs more frequently than IPAH. Although an initial clot has been found in most cases of CTEPH, excessive pulmonary vascular remodeling in medium-sized and small arteries may also occur and serve as a critical mechanism for the elevated PVR in these patients (5, 30, 52). Therefore, CTEPH is a vasculopathy involving coagulant occlusion of the large or central elastic proximal pulmonary arteries and thickening of proximal and distal arterial wall, rendering it accessible to surgical thromboendarterectomy.
Demographic and hemodynamic data in IPAH and CTEPH patients have not been systematically characterized and compared to establish if they are comparable in mean PAP elevation, age of onset, cardiac output, and PVR. The objectives of this study were to analyze and characterize the clinical demographic and hemodynamic data obtained from IPAH and CTEPH patients in order to a) determine if the magnitude of mean PAP elevation in IPAH patients is related to gender, age, race, fenfluramine use, and or vasoreactivity to inhaled nitric oxide (NO), and b) determine whether pulmonary hemody-namics and age at diagnosis are comparable between IPAH and CTEPH patients.
Materials and Methods
Subjects
Three hundred and thirty-two patients, who were found to have pulmonary hypertension by diagnostic right heart catheterization at the UCSD Medical Center, participated in the study. Two hundred and forty-two patients were diagnosed with IPAH and ninety were diagnosed with CTEPH (and underwent pulmonary endarterectomy at our institution). Diagnosis was based on combined hemodynamic, angiographic and clinical data. IPAH was diagnosed based on the criteria set forth by the National Institutes of Health Registry for Primary Pulmonary Hypertension. Informed consent, approved by Institutional Review Board (IRB) of the University of California, San Diego, was obtained from all patients. All patients underwent right-heart catheterization as part of the standard diagnostic procedure for pulmonary hypertension and signed an IRB approved informed consent to participate in the study.
Determination of pulmonary vascular hemodynamics
For both IPAH and CTEPH patients, fluoroscope-guided right heart catheterizations were performed with catheters, while pressure was continuously recorded. Briefly, a flow-directed balloon-tipped Swan-Ganz catheter was positioned into the right ventricle or pulmonary artery via the internal jugular vein. Hemodynamic measurements, such as pulmonary arterial pressure (PAP), were obtained by a pressure transducer (Namic/Boston Scientific, Natick, MA) connected to a Mac-Laboratory 7000 hemodynamic and electrocardiographic monitoring system (GE Medical System, Waukesha, WI). The PAP signals were sampled at 200 Hz and stored on a computer for later analysis. Cardiac output (CO) was measured by a thermodilution technique and pulmonary vascular resistance (PVR) was calculated according to PAP, pulmonary artery wedge pressure (Pwedge), and CO by the monitoring system. To determine vascular reactivity, PAP, PVR, and CO were recorded and compared in patients before and after inhalation of nitric oxide (NO, 20 ppm for 5 min) via a mask during pulmonary arterial catheterization. For patients with near-to-normal mPAP at rest, but still exhibiting signs of pulmonary distress with increased activity, mPAP and CO were measured during exercise (i.e. barbell lifts or cycling) while in the cath lab. Those individuals whose mPAP and CO became elevated during exercise were classified as exercise-induced pulmonary hypertensive.
Statistical analysis
The averaged values and data are expressed as mean ± SE. Statistical difference of haemodynamic changes was assessed using Students' t test. Oneway analysis of variance (ANOVA) with post-hoc analysis was also performed as indicated to compare hemodynamics between different patient groups. Differences were considered to be significant when P < 0.05.
Results
Distribution and comparison of hemodynamics in IPAH and CTEPH patients
Although initial pathogenic mechanisms may be very different in patients with IPAH and CTEPH, the distributions of mean PAP, CO, and PVR in these two groups of patients appear to be similar. The averaged mean PAP (mPAP) in IPAH patients (50.7 ± 0.9 mmHg) is slightly (but statistically significant) higher than in CTEPH patients (44.6 ± 1.3 mmHg) (Fig. 1A). The average CO was approximately 4 L/min (ranging from 2 to 10 L/min) in IPAH and CTEPH patients, well within the normal range, and there was no significant difference of averaged CO values between IPAH and CTEPH patients (Fig. 1B). Similar to the basal level of mPAP (Fig. 1A), calculated PVR was 23.7% higher in IPAH patients (913.5 ± 34.3 dyn·s−1·cm−5; n = 229) than in CTEPH patients (738.4 ± 38.3 dyn·s−1·cm−5; n = 90; P < 0.01) (Fig. 1C). These results suggest that increased CO in IPAH and CTEPH patients does not cause the elevated PAP. However, disease severity, with regard to the level of mPAP, is greater in IPAH patients than in CTEPH patients, in our patient population.
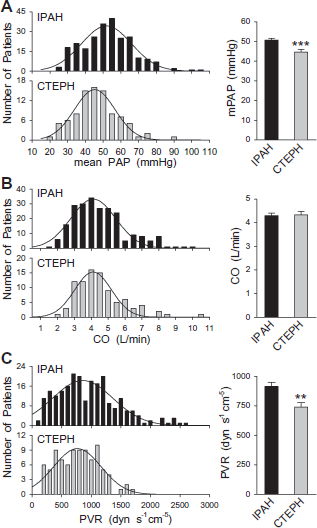
Distribution and comparison of pulmonary hemodynamics in patients with IPAH or CTEPH. A: mPAP, B: CO, C: calculated PVR. For each hemodynamic parameter, the left panel provides a histogram of the parameter distribution in IPAH (upper panel; n = 242) and CTEPH (lower panel; n = 90) patients. The bar graph (right) shows summarized data for IPAH patients (solid bar) and CTEPH patients (gray bar). ***P < 0.001 vs. IPAH, **P < 0.01 vs. IPAH.
Impact of gender of pulmonary hemodynamics in IPAH patients
Among the 242 patients diagnosed with IPAH based on the NIH Registry for IPAH, 85% were women (n = 185) and 24% were men (n = 57); the female/male ratio was 3.25. The distribution of mPAP determined by right-heart and pulmonary catheterization was comparable between women and men (Fig. 2A), although the average mPAP in male patients was slightly higher (but statistically significant, P < 0.05) than in female patients. The slightly lower mPAP in female IPAH patients may be related to the protective effect of estrogen on the vasculature. There was no statistically significant difference in the CO (Fig. 2B) and calculated PVR (Fig. 2C) distributions and means between male and female patient groups.

Comparison of pulmonary hemodynamics between female and male IPAH patients. A: mPAP, B: CO, C: calculated PVR. For each hemodynamic parameter, the left panel provides a histogram of the parameter distribution in female (top) and male (bottom) IPAH patients. The bar graph (right) shows summarized data for women (solid bars) and men (gray bars). *P < 0.05 vs. Women.
Exercise-induced IPAH patients
Exercise-induced pulmonary hypertension can occur in a fragment of the population of patients exhibiting typical pulmonary hypertension symptoms. These individuals have a relatively ‘normal’ mPAP at rest, but their cardiac output and mPAP become elevated during submaximal exercise. Exercise-induced pulmonary hypertension can be due to a number of cardiopulmonary diseases, including COPD (10, 27), systemic sclerosis (59), hypoxemia (29), and LAM (62), but could be predictive of the development of persistent pulmonary hypertension. In our patient population, we determined that 14.5% of IPAH patients whose mPAP was maintained in normal range at rest exhibited significantly increased mPAP during exercise (Figs. 3A and B); these patients were classified as exercise-induced pulmonary hypertensive patients (Ex-IPAH). As shown in Figure 3C, exercise doubled mPAP (i.e. increased by ≥100%) in approximately half (48.6%) of these patients, with an average 97.8 ± 7.1% increase in mPAP (n = 35). Most of the exercise-induced IPAH patients were female (n = 33; 94.3%); age ranged between 45 and 70 years of age for women and 47 and 55 years for men (n = 2) (Fig. 3D). The mechanisms involved in the exercise-induced pulmonary hypertension are unclear; decreased pulmonary vascular compliance or increased vascular wall stiffness has been indicated as one of the causes (34, 42, 50). It is also unclear why the female/male ratio (∼16.5) in our exercised-induced PH patients is much greater than the female/male ratio (∼3.2) in all IPAH patients. Care should be taken in interpreting our findings as the pool size of Ex-IPAH patients we evaluated was very limited and may have biased the findings.
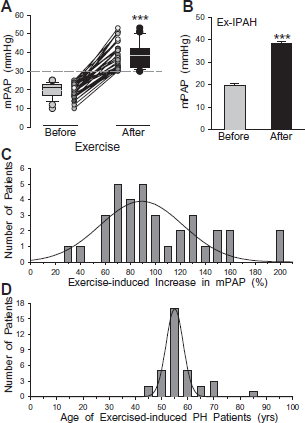
Exercise-induced change mPAP in patients diagnosed with IPAH. A: Individual and summarized data showing mPAP before and after exercise in patients with normal resting mPAP. B: Summarized data showing averaged mPAP before and after exercise in patients with exercise-induced IPAH (n = 35). ***P < 0.001 vs. Before. C: Histogram showing the distribution of exercise-induced changes in mPAP in all patients. D: Histogram showing the distribution of age in all exercised-induced IPAH patients.
Race and anorexigen use and mPAP in IPAH patients
Of the total number of IPAH patients included in this series, 83% were Caucasian, with Hispanics constituting the next largest demographic group (11%) (Fig. 4A). There was no significant difference in mPAP between any of the demographic groups represented in this study (i.e. Caucasians, Hispanics, African Americans, Asians and others) (Fig. 4B). It has been emphasized that the number of patients of African American and Asian origins was very small in our study; the data may thus not accurately reflect the severity of pulmonary hypertension in these patients.

Comparison of mPAP in IPSH patients from different races and in IPAH patients with or without fenfluramine use. A: Number of IPAH patients in different racial groups. B: Averaged mPAP in IPAH patients with different ethnical origins: Caucasian, Hispanic (Hisp), African American (African Am), Native American (Native Am), Asian, and those of “other” origins.
Only 21.6% of all IPAH patients had a history of using fenfluramine and phentermine (Fen-phen). The mPAP in these patients (43.13 ± 1.93 mmHg, n = 52) was 18.2% less than in IPAH patients who had no history of Fen-phen use (52.77 ± 0.99, n = 190; P < 0.01) (Fig. 5).
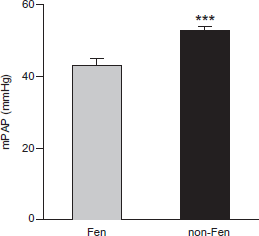
Fenfluramine use and mPAP in IPAH patients. Bar graph depicts averaged mPAP in IPAH patients (all racial groups) who had history of fenfluramine-phentermine use (Fen; n = 51) and those who did not (non-Fen; n = 191). ***P < 0.001 vs. Fen group.
Vascular reactivity in IPAH patients
During pulmonary catheterization, patients were asked to inhale NO (20-40 ppm) to determine vascular responsibility. In 9.1% of the patients with IPAH, inhalation of NO significantly reduced PAP (and PVR) by ≥20% (n = 14), whereas in >90% of the patients, inhaled NO failed to reduce mPAP (n = 139) (Fig. 6A). The basal mPAP (i.e. the PAP value before inhalation of NO), however, was not different between the “responders” (whose mPAP was reduced by ≥20% during NO inhalation) and “non-responders” (in whom NO failed to reduce mPAP) (Fig. 6B). Furthermore, the NO-mediated change in mPAP was not correlated with basal mPAP in both responders (r 2 = 5.8 × 10−6, n' = 37; P > 0.50) and non-responders (r 2 = 0.0234, n' = 113; P > 0.50) (Fig. 6C). Similarly, there was no correlation between basal mPAP and NO-induced mPAP when all patients were evaluated (r 2 = 0.0225), or in all patients who positively responded to NO treatment (r 2 = 0.0129), regardless of the % change of mPAP (Fig. 6D). These data indicate that whether our IPAH patients responds to acutely applied nitric oxide appears to be independent of the basal mPAP or the severity of pulmonary hypertension.
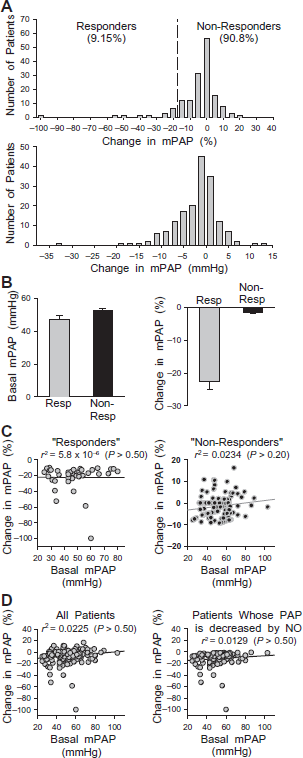
Response of IPAH patients to acute inhalation of nitric oxide (NO). A: Histogram showing distribution of changes in mPAP (in % of the basal level before NO inhalation, upper panel; and in mmHg, lower panel) in patients with IPAH after inhalation of NO (20 ppm for 5 min). “Responder” is defined as any patient who demonstrated a 20% reduction or greater in mPAP during NO inhalation. B: Summarized data showing the averaged basal mPAP (left panel) and the averaged change in mPAP (%) (right panel) in “responders” (gray bars) and “non-responders” (solid bars). C: Relationship between the change in mPAP (%) and the basal level of mPAP (mmHg) in “responders” (left panel) and “non-responders” (right panel). D: Relationship between the change in mPAP (%) and basal mPAP in all IPAH patients (left panel) and all IPAH whose PAP was decreased by NO (right panel).
Age and the severity of pulmonary hypertension in IPAH and CTEPH patients
Although it is largely dependent of the diagnosis time, IPAH patients tended to be younger at age of diagnosis than CTEPH patients (46.8 ± 14.3 vs. 54.4 ± 14.5 years; P < 0.001) (Fig. 7A). Furthermore, there was a significant negative correlation (r 2 = 0.1040, n' = 240; P < 0.001) between age and the severity of pulmonary hypertension in our IPAH patients population (Fig. 7B, left panel), while there was no correlation at all (r 2 = 0.0019, n' = 88; P > 0.50) in the CTEPH patients we evaluated (Fig. 7B, right panel). The slope of the linear regression curve is -0.31888 for IPAH patients and -0.03779 for CTEPH patients, respectively (Fig. 7C), an 8.44 fold greater in IPAH patients. These data indicate that age of the disease onset in IPAH patients is an indicator of the severity of pulmonary hypertension in these patients, i.e. the younger the patient starts to have the disease, the more severe the disease will manifest.

Age distribution of patients with IPAH and CTEPH. A: Histogram showing distribution of age at the time of diagnosis in IPAH (top) and CTEPH (bottom) patients. B: Relationship between age and mPAP in IPAH (left panel) and CTEPH (right panel) patients. mPAP is negatively correlated with age in IPAH patients (P < 0.001). C: comparison of the linear regression curves between IPAH and CTEPH patients. The slope of the curves is -0.31888 for IPAH patients and -0.03779 for CTEPH patients.
Discussion
With an incidence of 1-2 cases per million in the generation population, IPAH is rare and fatal disease. Patients have an average survival of ≤3 years after diagnosis; with drug therapy, survival is improved, but the disease does not regress completely. Surgical intervention (i.e. lung transplantation) is only used when drug therapy is proven ineffective in IPAH patients. The underlying cellular and molecular mechanisms of IPAH are not completely understood, but many mechanisms have been proposed (4, 24, 66). These studies have led to the development of a number of therapeutic agents which have been used with limited success to treat IPAH patients: anticoagulants (e.g. warfarin), voltage-dependent calcium channel blockers (e.g. nifedipine, amlodipine, diltiazem), infusion or inhalation of prostacyclin (e.g. epoprostenol, iloprost, treprostinil) or prostacyclin analogues (e.g. beraprost), inhaled NO, endothelin receptor antagonists (e.g. bosentan, sitaxsentan, ambrisentan, ABT-306552), and phosphodiesterase inhibitors (e.g. sildenafil, tadalafil, UK-369003, vardenafil) (25, 48). Novel therapies with limited clinical trials and still under investigation for suitability as PAH treatments include: i) 3-HMGCoA reductase inhibitors (e.g. simvastatin) (26), ii) KATP channel openers (e.g. levosimentan) (36), iii) vasointestinal active peptide (e.g. aviptadil) (45), iv) Rho-kinase inhibitors (e.g. fasudil) (16), v) angiotensin II receptor I antagonists (e.g. losartan) (28, 40), vi) natriuretic peptides (31), vii) selectin antagonists (e.g. bimosiamose, which is being tested in asthma and COPD patients).
Other therapies which to date have only been tested in experimental models of pulmonary hypertension or using isolated disease tissues include: i) PDGF receptor inhibitors (e.g. imatinib) (58), ii) 2-methoxyestradiol analogues (e.g. pulmoLAR) (64), iii) ghrelin (an endogenous vaso-dilatory peptide that stimulates the release of growth hormone) (22), iv) bradykinin (61), v) serotonin transporter inhibitors (escitalopram, fluoxetine) (14, 20), vi) serotonin receptor blockers (e.g. ketanserin, PRX-08066) (12, 37), vii) κ-opioid receptor antagonists (e.g. U50,488H) (43), viii) nonspecific phosphodiesterase inhibitors (pentoxyfilline, theophylline, dipyramidole) (53, 54), ix) voltage-gated potassium channel therapies (e.g. adenoviral Kv1.5, dichloroacetate) (39, 46), x) peroxisome proliferator-activated receptor ligands (e.g. pioglitazone, troglitazone) (21), xi) pro-inflammatory cytokine (IL-1, IL-6, IL-10) regulators (19, 65), xii) immunosuppressive agents (e.g. mycophenolate mofetil) (60), xiii) elastase inhibitors (13), xiv) novel prostacyclin receptor antagonists (e.g. NS-304), and xv) natriuretic peptides (32).
Although the incidence of CTEPH (∼20,000-40,000 cases/year in the United States) is greater for IPAH, the cellular and molecular mechanisms which underlie CTEPH are not clearly understood (52). Surgical removal of the fibrotic clot and thickened intima of downstream pulmonary arteries via pulmonary endarterectomy is an effective treatment (i.e. decreased PAP and PVR post-operatively) for CTEPH patients (63). However, a subset of patients is considered inoperable, while others have sustained elevation of PVR or mPAP after surgery. The post-operative persistent or residual pulmonary hypertension in some CTEPH patients suggests that there is a component of CTEPH which is due to distal (i.e. downstream of the fibrotic clot) pulmonary artery abnormalities which contribute to remodeling of the vessels, much like that observed in IPAH patients. As a result, some IPAH drugs are now being tested for their efficacy in treating residual high PAP/PVR in CTEPH patients (7-9, 18, 33).
The advent of new therapies for pulmonary arterial hypertension has been associated with a proliferation of clinical trials targeting pulmonary hypertension patient populations. Demographic and clinical characteristics in these patient groups are usually described within the context of individual studies. There is a paucity of cellular and molecular data proposing links between IPAH and CTEPH, and the increasing use of “conventional” IPAH therapies are being used to treat CTEPH patients suggests that the patient populations may share similar hemodynamics. While many therapeutic studies have documented “baseline” clinical characteristics of participants, a rigorous, methodical and descriptive comparison of the clinical similarities and differences between CTEPH and IPAH is needed. In our study, we compared the basic demographic, clinical, and hemodynamic characteristics between IPAH and CTEPH patients.
Comparison between IPAH and CTEPH: demographic and hemodynamic correlations
We found that the average mPAP was higher in IPAH patients than in CTEPH patients, suggesting that IPAH patients have more severe pulmonary hypertension in terms of PAP and PVR. Among the patients referred to our institution, there are more IPAH patients whose mPAP is ≥60 mmHg (23%) and calculated PVR ≥1500 dyn·sec−1·cm−5 (17%) than CTEPH patients (10% and 4%, respectively). In addition, IPAH patients tend to be younger than CTEPH patients (46.8 vs. 54.4 years, respectively). However, the level of mPAP negatively correlated with age only in IPAH patients. This finding also indicates that the severity of IPAH is related to the onset of the disease; if a patient develops the disease at younger age, he/she would have more severe hemodynamic abnormalities than an individual who develops the disease later in life. This correlation between age and mPAP severity, however, was not apparent in our pool of CTEPH patients. Possible explanations as to why age and severity did not correlate in CTEPH patients include: i) early surgical treatment with pulmonary endarterectomy “conceals” the progression and the severity; ii) a dramatic difference in pulmonary hemodynamics between CTEPH patients whose PVR is increased mainly by central vascular occlusion and patients whose PVR is increased by proximal vascular occlusion and distal vascular remodeling; and iii) early diagnosis of CTEPH in patients with acute pulmonary embolism.
As with age, mPAP was negatively correlated with CO in IPAH patients (data not shown), indicating that severe IPAH patients might have developed mild right heart failure by the time they were diagnosed with IPAH. However, the trend for a negative correlation of mPAP with CO is not statistically significant (data not shown) in CTEPH patients.
Although the reason for the difference between IPAH and CTEPH patients is unclear, one of the possibilities is that some CTEPH patients, especially those with clear central or proximal vascular occlusion, still have one to two lobes of the lungs to tolerate (or to receive) entire CO. Therefore, it doest not significantly increase the after-load of the right ventricle, and therefore is less prone to inducing right heart failure. Nonetheless, based on observations that CO values were similar in IPAH and CTEPH patients, we determined that elevated CO is not main determinant of mPAP in both IPAH and CTEPH patients. This suggests that the increased mPAP in IPAH and CTEPH patients may be highly influenced by sustained pulmonary vasoconstriction and excessive vascular remodeling in proximal, medium-sized and small vessels are involved in the development of pulmonary hypertension, in addition to the coagulant tissues occluded in central or proximal vessels.
IPAH patient characterization: demographics, hemodynamics, and vasoreactivity
Among the IPAH patients referred to our institution, 76.4% are women and 23.6% men (3.24:1), which is similar to the data reported by other investigators in American, European and Asian patient populations (2, 11, 23, 38, 41, 56, 57). However, our study revealed a slightly higher mPAP in male IPAH patients (54.1 ± 2.0 mmHg) than in female patients (49.6 ± 1.0 mmHg; P < 0.05), a 9.1% difference that may be related to the vascular protective effect of estrogen in female patients (3, 6, 17). The use of the appetite suppressants has been linked to the incidence of IPAH (1, 55). In our patient cohort, individuals with no history of Fen-phen use had a significantly higher basal mPAP, suggesting that the use of these agents does not enhance the severity of IPAH.
There appeared to be no significant difference in the incidence or severity (basal mPAP) of IPAH based on ethnicity. However, caution is warranted in making this conclusion because of the limited number of Hispanic, African American and Asian patients with IPAH in our study. More patients of non-Caucasian ethnicity would be needed to study potential racial differences in the severity of the disease.
Our data also show that approximately 14.5% of IPAH patients had a mPAP within the normal range at rest, but significantly increased to the level greater than 30 mmHg during exercise (in almost half of the patients, PAP was doubled during exercise). Most of the exercised-induced IPAH patients were female (94.3%) and the average age was 55 ± 1 years. The predominant incidence in women and occurrence at the age ranging from 50 to 60 years (77.1%) of exercised-induced IPAH suggest that decreased compliance (or increased stiffness) and high sensitivity to acute hypoxia in pulmonary arteries may be an important cause for the elevated PVR and PAP in female patients. However, we cannot rule out the possibility that extremely high female/male ratio and very restricted age group in exercise-induced IPAH patients is related to other environmental factors such as diet, stress, and hormones.
Finally, we determined that 9% of IPAH patients could be defined as “responders” based on the ≥20% reduction in mPAP in response to acute application of a vasodilator (49, 56). In all our IPAH patients, the NO-induced percentage decrease in mPAP and PVR was not correlated with the basal level of PAP before NO inhalation (an indicator of disease severity), suggesting that vascular reactivity is an intrinsic property of the pulmonary vasculature and is not controlled by or related to disease severity (or to the basal level of PVR and mPAP). The patients with mild pulmonary hypertension may not respond to acute intake of vasodilator (e.g. inhalation of NO), whereas the patients with severe pulmonary may be a good responder (although very few) to vasodilators. The cellular and molecular mechanisms responsible for different vascular reactivity between “responders” and “non-responders” are still unclear.
In summary, our study found that IPAH and CTEPH patients share many similarities in terms of demographics and hemodynamics. However, the two groups of patients also show some profound differences in terms of female/male ratio of incidence, severity of the disease in age/gender-differentiated groups, and potential correlation of the severity with cardiac output or right heart function. In addition, we identified a couple of important factors that affect the severity of hemodynamics in IPAH patients: age of the onset of the disease and basal mPAP.
Footnotes
Acknowledgments
This work was supported by grants from the National Heart, Lung, and Blood Institute of the National Institutes of Health (HL054043, HL064945, and HL066012). We thank Ms. Diane Conger and Ms. Ann Nicholson for assistance.
