Abstract
Eosinophilic lung diseases represent a group of disorders in which lung infiltrates seen on a chest radiograph, may or may not be accompanied by peripheral blood eosinophilia. The causes are varied, and range from drug ingestion, parasite or fungal infection, or else, may be idiopathic. The clinical manifestation of these disorders may be mild or severe, depending on the underlying condition. A full history and examination, along with radiological imaging, bronchoscopy, and serological tests are useful in reaching a diagnosis. Treatment with corticosteroids usually results in a good clinical outcome, if the diagnosis is suspected and made early. Early treatment with high dose corticosteroids may be life saving in some patients, whilst other patients require prolonged and sometimes recurrent courses of corticosteroids, due to the chronic relapsing nature of their condition.
Introduction
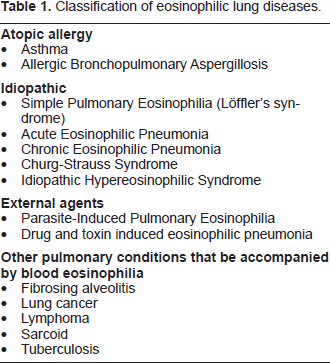
The eosinophilic lung diseases are a heterogeneous group of pulmonary disorders characterized by increased numbers of eosinophils in the airway or lung parenchyma. In the 1950's, Reeder and Goodrich introduced the term “PIE syndromes” (Pulmonary Infiltrates with [blood] Eosinophilia) to classify these syndromes in which lung infiltrates on chest radiographs (CXR) were associated with peripheral blood eosinophilia. However, as new syndromes were described, it became apparent that some disorders had evidence of increased lung eosinophils without significant blood eosinophilia, such as ‘acute eosinophilic pneumonia’. Thus the diagnosis of eosinophilic lung disease can be suspected, based on either the finding of pulmonary disease with blood eosinophilia, pulmonary disease with bronchoalveolar lavage (BAL) eosinophilia, or pulmonary disease with tissue eosinophilia on lung biopsy (Allen et al. 1994). Clinical manifestation can be varied, and their presentation can be both benign and self limiting, chronic with recurrent relapses, or potentially life threatening (Table 1). The importance of diagnosing eosino-philic lung disease is that many of these disorders are sensitive to corticosteroid treatment.
Classification of eosinophilic lung diseases.
Eosinophils
The eosinophil is the major effector cell found in this group of disorders. Eosinophils normally account for 1%-3% of peripheral blood leukocytes, and the upper limit of the normal range can be regarded as 0.4 × 109/litre of blood, depending on individual laboratory reference range. Eosinophilia can be arbitrarily defined as mild (0.41-1.5 × 109/litre), moderate (1.51-5.0 × 109/litre) and severe (>5.0 × 109/litre).
Eosinophils are polymorphonuclear leucocytes produced in the bone marrow from progenitor cells under the influence of interleukin-5 (IL-5), IL-3, and granulocyte-macrophage colony-stimulating factor (GM-CSF). Eosinophils migrate from the blood to the tissues under the chemo-attractant effects of molecules such as platelet activating factor, leukotrienes, and chemokines such as eotaxin 1 and eotaxin 2. Once in the tissues, eosinophils can persist for several weeks, provided IL-3, IL-5, and GM-CSF are present. The pathogenesis of eosinophilic lung diseases is due to the release of toxic substances stored within intracellular granules inside eosinophils. Eosinophilic granules contain a number of substances that participate in host defense against pulmonary pathogens such as parasites and fungi such as major basic protein, eosinophil cationic protein, eosinophil-derived neurotoxin, and eosinophil peroxidase. Eosinophils can also produce inflammatory cytokines and leukotrienes such as C4, D4, and E4 which have effects on vascular permeability, mucus secretion, and smooth muscle contraction within the airways. Excessive release of these granule contents by eosinophils in the lung can result in destruction of airway epithelium, interstitial destruction and oedema, or granulomatous tissue destruction, which lead to the clinical manifestations of the various eosinophilic lung diseases.
Simple Pulmonary Eosinophilia
Simple pulmonary eosinophilia was originally described by Löffler in 1932, and is often known as Löffler's syndrome. This condition is characterized by fleeting pulmonary infiltrates on CXR and is associated with a peripheral blood eosinophilia. Patients with this condition are usually asymptomatic or may have minor respiratory symptoms such as fever, cough, wheeze and malaise (Allen et al. 1994). Minor systemic symptoms such as low grade fever, arthralgias and myalgias suggesting a common cold may be present.
The chest radiograph pattern in these circumstances may demonstrate bilateral, migratory and usually peripheral, relatively homogenous nonseg-mental infiltrates. Löffler's syndrome is an idiopathic condition and diagnosis requires exclusion of other causes of transient disease. It is thought that the original cases described by Löffler were mostly likely due to Ascaris parasitic infection or drug reaction. It is estimated however, that up to third of cases may have no identifiable cause. It is important to exclude an underlying occult parasitic infection and the possibility of a drug induced pulmonary eosinophilic syndrome, and in cases where careful evaluation reveals an extrinsic cause, the term Löffler's syndrome should not be used. Lung biopsy is unnecessary, but characteristic findings include interstitial and alveolar oedema with large numbers of eosinophils, similar to chronic eosinophilic pneumonia.
Acute Eosinophilic Pneumonia
An acute form of eosinophilic pneumonia was first described in 1989 (Allen et al. 1989; Badesch et al. 1989) after a number of cases were reported of patients admitted with acute respiratory failure associated with increased BAL or tissue eosinophilia. This condition is characterized by an acute febrile illness of usually less than 5 days duration along with cough, and dyspnoea. Most patients are hypoxic on presentation, and can progress from mild dyspnoea to life-threatening hypoxaemic respiratory failure requiring mechanical ventilation, within a matter of hours. Pleuritic chest pain and myalgia may be present in over half of all patients. Crackles are predominantly heard on chest examination, although auscultation may be unremarkable in up to 20% of patients (Pope-Harman et al. 1996). Wheezing may be heard on forced exhalation suggesting airway obstruction. Clinically it is difficult to distinguish this condition from an acute infectious process, acute respiratory distress syndrome (ARDS), and drug toxicity.
The cause of acute eosinophilic pneumonia is unknown, although it has been suggested that it is may represent an acute hypersensitivity reaction due to an unidentified inhaled antigen. The average age at presentation is 30 years (range: 14-65) and there is no gender preference. A smoking history is present in 40% of patients, and there appears to be an association between recent onset of smoking cigarettes and development of acute eosinophilic pneumonia (Shintani et al. 2000). This suggests that certain constituents of tobacco smoke may be responsible for triggering acute eosinophilic pneumonia in susceptible patients.
Unlike other eosinophilic pneumonias, patients with acute eosinophilic pneumonia usually have normal or only slightly elevated peripheral blood eosinophil counts. Raised serum IgE levels up to 2,000 IU/L can be seen in some, but not all patients. Early in the course of the disease, the CXR may show subtle interstitial infiltrates with Kerley B-lines and small pleural effusions. Over 1-2 days, these subtle infiltrates may develop into extensive mixed interstitial and alveolar infiltrates as the condition worsens. CT scans usually show diffuse parenchymal alveolar infiltrates, pleural effusion and pronounced septal markings. Unlike other causes of pulmonary eosinophilia, small to moderate pleural effusions are frequent and pleural fluid analysis usually reveals an exudates containing an increased percentage of eosinophils (Shintani et al. 2000). Pulmonary function tests are not necessary for diagnosis but in the acute phase, may show airway obstruction or a restrictive pattern, along with a low diffusing capacity. After treatment, pulmonary function studies usually return to normal values. BAL is important in excluding bacterial, fungal, and other infections, and characteristically shows more than 25% eosinophils, with an average of 42% in one series (Shintani et al. 2000). In addition to BAL eosinophilia, BAL lymphocytes and neutrophils may also be increased in many patients. Lung biopsy is unnecessary for the diagnosis of acute eosinophilic pneumonia in most cases, but may have a role in excluding other diseases such as aspergillus infection. Histological findings include infiltration of eosinophils in the interstitium and in the alveolar spaces with features of diffuse alveolar damage. Diagnostic criteria for acute eosinophilic pneumonia are given in Table 2.
Diagnostic criteria for acute eosinophilic pneumonia.

Chronic Eosinophilic Pneumonia
Chronic eosinophilic pneumonia is characterized by typical pulmonary infiltrates accompanied by lung and/or blood eosinophilia. The first defining series of patients with chronic eosinophilic pneumonia were first described in 1969 (Carrington et al. 1969). It can occur at any age, but the peak incidence is in the fifth decade and women are affected twice as often as men. Approximately half of patients will have pre-existing asthma or atopic disease, usually of recent onset. Onset of the condition is insidious, with progressive respiratory and systemic symptoms being present for an average of 8 months before diagnosis. Patients may have had recurrent courses of antibiotics for non resolving pneumonia before the diagnosis is reached. Patients typically present with some combination of cough (90%), fever (87%), dyspnoea (57%), and weight loss (57%). In addition, patients may complain of malaise, night sweats, wheezing, pleuritic chest pain, hemoptysis, and myalgia. The physical examination is usually normal, although wheeze, and/or crackles may be heard.
Investigations show mild to moderate peripheral eosinophilia in more than 90% of patients, and IgE levels are elevated in two-thirds of patients and usually less than 2,000 IU/L. The erythrocyte sedimentation rate (ESR) and C-reactive protein are usually markedly elevated. Anaemia and peripheral blood thrombocytosis may be present. The CXR shows characteristic peripheral infiltrates mostly in the upper and mid zones, which have been described as being the “photographic negative” of pulmonary edema, and is considered to be virtually diagnostic of the disease (Gaensler et al. 1977). However, less than 50% of patients demonstrate this classic radiographic pattern. Occasionally, the infiltrates may be patchy or even unilateral. Pleural effusions are uncommon. HRCT scans show peripheral consolidation, ground glass infiltrates and reticular opacities. Approximately 50% of patients have mediastinal adenopathy on CT scan which is not apparent on CXR. Pulmonary function test results are variable and can be normal in one third of patients, show obstruction in one third, and a restrictive pattern in one third of patients. The diffusing capacity will be low in 50% of patients. Hypoxemia or an increased A-a gradient is present in nearly all patients. Eosinophils can be found in sputum samples in 50% of patients and BAL fluid shows gross eosinophilia (>25%) in nearly all patients. Unlike acute eosinophilic pneumonia, BAL lymphocyte and neutrophil percentages are usually normal or only minimally elevated. Surgical lung biopsy is usually not necessary for the diagnosis of chronic eosinophilic pneumonia, although transbronchial biopsy should be undertaken. Histology shows infiltration of the interstitium and alveoli with eosinophils. Occasionally, areas of bronchiolitis obliterans can be seen.
Churg-Strauss Syndrome
Churg-Strauss syndrome, also known as allergic granulomatous and angiitis, is a rare disorder that affects men and women equally with an average age of onset of 38-48 (Guillevin et al. 1999). It can be thought of as having 3 phases. Patients generally have a history of asthma and/or allergic rhinitis for 8-10 years before presentation. The second phase is characterized by a dramatic peripheral blood eosinophilia and eosinophilic tissue infiltrates resembling Löffler's syndrome or chronic pulmonary eosinophilia, which may recur over a number of years, and most commonly affecting the lungs, heart and liver. Finally, a vasculitic phase occurs, and affects the nerves, lung, heart, gastrointestinal tract, and uncommonly, the kidneys. These phases may not be clinically identifiable in all patients and may not occur in sequence.
Clinical features of Churg-Strauss syndrome include asthma, a history of atopic disease, fever, malaise and weight loss. Multi systemic involvement is commonly seen. Upper airway findings commonly include sinusitis, rhinitis, and nasal polyps. The skin is affected in 70% of patients and can show nodules, purpura, or urticaria. Peripheral neuropathy and mononeuritis multiplex are common and central nervous system involvement occurs in 27%. Cardiac involvement can include congestive cardiac failure, pericarditis, and hypertension. In the gastrointestinal tract, abdominal pain, diarrhea, and bleeding can occur. Renal impairment occurs in half of patients, as do symptoms of arthralgia or myalgia. Churg-Strauss syndrome has been associated with use of leukotriene antagonists in patients with pre-existing asthma (Wechsler et al. 1998). However, it is likely that the use of these medications permitted reduction of corticosteroids, thus unmasking the syndrome in patients previously mis-diagnosed as having simple asthma.
IgE levels are elevated in 75% of patients and appear to correlate with disease activity. Inflammatory markers such as ESR and CRP may be elevated. There may be marked peripheral eosinophilia and anaemia. Patients can have low titers of rheumatoid factor and anti-nuclear antibodies. The anti-neutrophil cytoplasmic antibody (ANCA) is present in approximately 50% of patients, usually in the perinuclear (P-ANCA) form (Goeken et al. 1991). BAL fluid typically shows very high percentages of eosinophils, averaging 33%. Chest x-rays and CT scans typically show transient patchy peripheral pulmonary infiltrates in about 30% patients, and are indistinguishable from lesions seen in chronic eosinophilic pneumonia. Although uncommon, nodular lesions have also been reported. Exudative pleural effusions are present in a third of patients, and contain increased numbers of eosinophils. Alveolar haemmorhage is rare in Churg Strauss syndrome, in contrast to other pulmonary vasculitides such Wegener's granulomatosis and microscopic polyangiitis, which should be considered in the differential diagnosis. Pulmonary function testing is usually consistent with asthma. A restrictive pattern may be seen with extensive parenchymal infiltrates.
The diagnosis can be established by biopsy of an involved organ. In the lung, surgical biopsy is often necessary since transbronchial biopsy generally provides limited vascular material. The earliest or prevasculitic phase is characterized by eosinophilic tissue infiltration without vasculitis. This is followed by a vasculitic phase with eosinophilic vasculitis of the small arteries and veins. Interstitial and perivascular granulomas are common.
Allergic Bronchopulmonary Aspergillosis
Allergic bronchopulmonary aspergillosis (ABPA) refers to the colonization of the lower respiratory tract in susceptible allergic asthmatics, by the fungus Aspergillus fumigatus. It found in soil and decaying vegetation, and can grow at body temperature following inhalation of spores. It is more common in the autumn and winter when spore counts are higher. Fungal hyphae develop in the bronchial wall, lumen or lung parenchyma, and release antigens which trigger an immunological response resulting in tissue damage, including proximal bronchiectasis and irreversible airway damage, if episodes are recurrent or left untreated. ABPA can be seen in approximately 2%-28% of asthmatic patients, and up to 15% of patients with cystic fibrosis. Both genders and all age groups can be affected. The diagnostic criteria for ABPA are listed in Table 3 although some patients may not have all of these findings.
Diagnostic criteria for allergic bronchopulmonary aspergillosis.

ABPA can be divided into 5 clinical stages (Patterson et al. 1982) to help characterize and manage patients appropriately, but patients do not necessarily progress through these stages. Stage 1 (acute), may be clinically silent, or patients may present with fever, malaise, and worsening asthma. Investigations show elevated IgE levels usually greater than 1000 IU/ml, peripheral eosinophilia, and pulmonary eosinophilic infiltrates on CXR. Organizing exudates may cause bronchial obstruction and segmental/lobar collapse, and expectoration of hard sputum plugs containing Aspergillus hyphae, eosinophils and mucus. Stage 2 (remission) occurs when there is resolution of symptoms, CXR abnormalities, peripheral eosinophilia and a reduction in total IgE level towards normal. During exacerbation (stage 3), there is recurrence of asthma symptoms, at least a doubling in the stage 2 baseline IgE level, and new chest x-ray infiltrates. Corticosteroids are generally required to treat these patients. In stage 4 disease, (corticosteroid-dependent asthma), continuous oral corticosteroids are required to control asthma symptoms or to prevent new infiltrates from developing during attempts at tapering steroid therapy. Patients with long standing disease can develop irreversible fibrotic lung disease (stage 5), characterized by dyspnoea, cyanosis, and cor pulmonale.
The IgE level correlates with disease activity and a normal IgE level in a symptomatic patient virtually excludes the diagnosis (Patterson et al. 1986). In most patients, the level exceeds 1000 IU/ml. In patients with lower total IgE levels, elevated Aspergillus fumigatus specific IgE or IgG antibody may be present. Chest radiographs and CT scans usually show flitting patchy infiltrates with segmental/lobar collapse. Proximal bronchiectasis of the saccular type involving the upper lobes with signs of mucus impaction may be seen in more advanced disease. About 15% of patients will present early in the disease before bronchiectasis develops. In early stages, pulmonary function tests show obstruction due to underlying airway obstruction, which progresses to chronic fixed airflow obstruction as a consequence of lung fibro-sis. As the disease advances, patients develop a restrictive pattern with a low diffusing capacity. Lung biopsy is generally unnecessary to make the diagnosis, but if performed will show a bronchocentric inflammatory infiltrate composed of eosinophils, lymphocytes, plasma cells, and monocytes. Aspergillus hyphae can be identified using fungal stains, and may also be present in sputum samples.
Parasitic Infection
Parasites can cause pulmonary infiltrates with peripheral blood and/or alveolar eosinophilia. This occurs as a result of the tissue inflammation caused by the presence of the parasite in the lung at a certain stage in its life cycle, and the host's immunological response which includes a local accumulation of eosinophils, and an accumulation of eosinophils in the peripheral blood. The travel history is therefore important to exclude this as a potential cause of pulmonary infiltrates with eosinophilia. In the United States, Strongyloides, Ascaris, Toxocara, and Ancylostoma are the most common parasitic causes of eosinophilic lung disease. Other parasites causing pulmonary eosinophilia around the world include Schistosoma, Trichinella spiralis, Paragonimus westermani, Echinococcus granulosus, and Dirofilaria immitis.
Tropical pulmonary eosinophilia refers to a severe illness caused by the filarial worms, Wuchereria bancrofti and Brugia malayi which are endemic in certain parts of the world such as India, Pakistan, Sri-Lanka, Thailand, Malaysia, and the Philippines. (Ong et al. 1998). Symptoms usually relate to the gastrointestinal system, but patients may present with fatigue, malaise, fever, dyspnoea, nocturnal cough, wheeze and weight loss. Findings may include basal crepitations, and scattered wheeze. Blood eosinophil counts are usually greater than 3.0 × 109/litre and total IgE levels are greater than 2000 IU/ml. The ESR is usually raised and titres of anti-filarial antibody can be detected. CXR usually shows a diffuse, finely nodular infiltrate, whilst CT scans may, in addition, reveal hilar adenopathy. In shistosomiasis infestation, ova lodge in pulmonary vessels, eventually leading to restriction of the pulmonary vascular bed. In this situation, CXR may reveal signs of pulmonary hypertension and diffuse reticulonodular infiltrates. Lung function tests in the early stages of tropical pulmonary eosinophilia may show an obstructive pattern in one third, but if patients present late or remain untreated, long term sequelae such as pulmonary fibrosis may ensue, leading to a restrictive pattern.
Diagnosis of tropical pulmonary eosinophilia is based on a history of residence in an endemic area in association with marked total IgE levels, peripheral blood eosinophilia, diffuse pulmonary infiltrates, and high antifilarial antibody titres. Diagnosis of other parasite infestations can be made by identification of ova, larvae or adult worms in stool samples. This however is not possible with certain parasites such as Trichinella, Paragonimus, Ancylostoma, Toxocara, and filarial infestation, and detection of serum antibodies against the parasite is required.
Treatment of tropical pulmonary eosinophilia is with diethylcarbamazine, 2 mg/kg, three times daily for 7-10 days. Thiabendazole can be used to treat Strongyloides and ankylostoma braziliense. Oral mebendazole 100 mg twice daily for 5 days can be used to treat Ascaris lumbricoides.
Drug and Toxin Associated Eosinophilic Pneumonia
Drugs and chemicals are a common cause of pulmonary infiltrates with peripheral blood eosinophilia (Allen, 2004). The commonly used drugs associated with pulmonary eosinophilic disorders are listed below.

Patients with drug-induced eosinophilic lung disease can have variable presentations, depending on the drug involved. They may present with an urticarial or papular rash as the first presentation of an adverse reaction, and later develop symptoms such as fever, cough, and dyspnoea with continued use. A raised peripheral eosinophil count can be found, along with an elevated ESR, but IgE levels are usually normal. CXR appearances can be varied, and may include ill-defined, soft, patchy, or linear /reticular infiltrates, occasionally associated with a pleural effusion. Diagnosis rests on having a high index of suspicion and a thorough drug history should be taken. Many patients will improve by simply discontinuing the offending drug, which represents the best diagnostic tool. In severe or persistent cases corticosteroids may be necessary.
Idiopathic Hypereosinophilic Syndrome
This is a rare condition of unknown aetilogy. Originally termed ‘eosinophilic leukemia’, it is characterized by blood and bone-marrow eosinophilia associated with eosinophilic infiltration of two or more organs. It may occur as a result of a primary eosinophilic myeloproliferative disorder, or else an abnormal clonal T-helper lymphocyte proliferative disorder producing IL-5 with secondary eosinophilia (Simon et al. 1999; Chang et al. 1999).
Patients usually present in their third or fourth decade and there is a 7:1 male predominance (Spry et al. 1983). Typical symptoms include night sweats, anorexia, weight loss, pruritis, cough, and fever. The illness may be mild or fatal. Cardiac involvement occurs in more than 50% of patients, including endocardial fibrosis, restrictive cardio-myopathy, valvular damage, and mural thrombus formation is the most serious complication and is the major cause of morbidity and mortality. Pulmonary involvement occurs in up to 40% of patients and typically presents as a dry cough, and dyspnoea. Long-standing disease can result in pulmonary fibrosis. Arterial and venous thromboembolic disease occurs in approximately two-thirds of patients. Symptoms depend on the organ affected, and can include renal or splenic infarcts, deep vein thrombosis, cerebrovascular accident, and retinal arteriolar embolism. Other findings can include peripheral neuropathy, gastrointestinal symptoms, renal insufficiency, arthralgias, and skin rash (Spry et al. 1982).
Diagnosis is made by [1] demonstrating peripheral blood eosinophilia of greater than 1.5 × 109/litre for more than 6 months, [2] absence of parasitic, allergic, vasculitic or other cause of eosinophilia, and [3] signs or symptoms of end organ damage related to the increased eosinophils. Eosinophil counts are very high and can reach 30%-70% of the total white cell count, or more than 20 × 109/litre absolute. In addition, eosino-philic metamyelocytes and myelocytes may be seen in peripheral blood, and large numbers of eosinophils are seen in the bone marrow. BAL fluid can show significantly increased numbers of eosinophils when there is lung involvement. The CXR may be normal, or shows diffuse, interstitial non-segmental infiltrates in up to one third of cases. Approximately 50% of affected patients have pleural effusions. Chest CT shows small pulmonary nodules and focal areas of ground glass infiltrates, predominately in the lung periphery.
Miscellaneous Lung Diseases Sometimes Associated with Eosinophils
Several interstitial lung diseases may have increased BAL or tissue eosinophils, usually as part of a mixed inflammatory cell infiltrate. Bron-choalveolar lavage specimens in Idiopathic pulmonary fibrosis demonstrate increased BAL eosinophils, but usually less than 20%. Similarly, pulmonary fibrosis associated with collagen vascular disease can be associated with mild BAL eosinophilia in up to a quarter of patients. An increased BAL eosinophilia is seen in about a quarter of patients with bronchiolitis obliterans organizing pneumonia, usually amounting to less than 20% of the total BAL cells. Other interstitial lung diseases which can have mildly elevated BAL eosinophil percentage include hypersensitivity pneumonitis and sarcoidosis. Langerhan's cell granulomatosis (eosinophilic granuloma) is characterized by abnormal proliferation and activation of Langerhan's cells, a histiocyte normally found in the skin and bronchiolar epithelium, leading to uncontrolled inflammation in the lung. With progression of disease, progressive inflammation and fibrosis is seen, with honeycombing in more advanced disease. An increase in tissue eosinophils is noted on open lung biopsy in most patients, but an increase in BAL eosinophils is seen in only a small percentage of patients.
Several malignancies can be associated with increased lung eosinophils including non-small cell lung cancer, Hodgkin's disease, non-Hodgkin's lymphoma, lymphocytic leukemia, and eosino-philic leukemia. Bronchogenic carcinoma and Hodgkin's disease are associated with BAL eosinophilia. Eosinophils have been shown to invade specimens of lung cancer suggesting a possible role in host defence against tumours. Many malignancies which metastasize to the lungs can be associated with peripheral blood eosinophilia.
Apart from Aspergillus lung infection, peripheral blood eosinophilia is often seen in patients with coccidioidomycosis fungal disease but is usually absent in other fungal infections such as his-toplasmosis, blastomycosis, candidiasis, and cryptococcosis. In addition, eosinophils can also be recovered from BAL or biopsy specimens in patients with primary coccidiomycosis. A travel history to areas endemic for coccidioidomycosis (the American Southwest) is important because administration of corticosteroids can result in acceleration of the infection with fatal dissemination. Pneumocystis jerovecci pneumonia associated with HIV infection can also present with moderately increased BAL eosinophil percentages, with 15% of patients having a BAL eosinophil count of greater than 5%. (Allen et al. 1990).
Bronchocentric granulomatosis represents a necrotizing granulomatous inflammation of the medium or small bronchi or bronchioles, resulting in destruction of the airway walls, surrounding lung tissue, and occlusion of the airway lumen. In asthmatics, it may represent a hypersensitivity reaction to inhaled antigens, particularly, aspergillus species, and may represent one tissue manifestation of ABPA. In non-asthmatic patients, the causative agent is unknown, although cases have been associated with ankylosing spondylitis, glomerulonephritis or rheumatoid arthritis. Certain infections such as tuberculosis, histoplasmosis, blastomycosis, and coccidiomycosis need to be excluded before the diagnosis of bronchocentric grunulomatosis is made. Patients present with dyspnoea or wheezing due to airway obstruction. Chest radiographs show nodules or infiltrates. Lung biopsies show necrotizing granulomatous infiltration of the airways and frequently have eosinophils as a major inflammatory component.
Clinical Approach
The history and physical examination are the most important means of determining a specific etiology in patients presenting with eosinophilic lung disease. The presence of asthma in the setting of pulmonary infiltrates and peripheral eosino-philia suggests the possibility of Churg-Strauss syndrome or ABPA, and further investigations should be directed at excluding these. A careful drug history should be taken for use of prescription, non-prescription, and illicit drugs as a potential cause of drug related eosinophilic pneumonia. A detailed travel history is important as this may suggest parasitic infection such as tropical pulmonary eosinophilia, Schistosoma, or coccidioidomycosis. Exposure to dogs or cats may suggest Ancylostoma and Toxocara infection. The finding of multi-system involvement affecting, for example, the skin, cardiac, CNS and renal systems should suggest Churg-Strauss syndrome. Thorough clinical evaluation should exclude primary or secondary malignancies, and other interstitial lung diseases.
High numbers of peripheral blood eosinophils may be seen in certain conditions such as simple pulmonary eosinophilia, chronic eosinophilic pneumonia, parasitic and fungal infections, some drug-induced lung diseases, ABPA, Churg-Strauss syndrome, and the idiopathic hypereosinophilic syndrome. Increased BAL eosinophils without blood eosinophilia suggests, in the appropriate context, acute eosinophilic pneumonia, P. jerovecci pneumonia, some cases of drug-induced lung disease, and Langerhan's cell granulomatosis. Stool samples and serology are useful in excluding parasitic infections. Very high levels of IgE suggest ABPA and the presence of anti-neutrophil cytoplasmic antibodies or anti-myeloperoxidase (MPO) antibodies suggests Churg-Strauss syndrome. Bronchoalveolar lavage is useful in the evaluation of the patients with eosinophilic lung disease, both to verify the presence of increased eosinophils in the lung and also to exclude parasitic or fungal infection. Lung biopsy is generally required for the diagnosis of Churg-Strauss syndrome, malignancy, bronchocentric granulomatosis, and some cases of interstitial lung disease.
Eosinophilic pneumonias can vary in their clinic manifestations from benign and self limiting at one end of the spectrum, to potentially life threatening at the other. Most are very responsive to oral cor-ticosteroids, although patients will need to be monitored for signs of relapse when therapy is stopped. In some instances, prolonged therapy with corticosteroids may be required, for example, in ABPA, chronic eosinophilic pneumonia and Churg-Strauss syndrome, and may require the addition of further immunosuppressant drugs as steroid sparing agents. High doses of intravenous corticosteroids may be life saving in patients with acute eosinophilic pneumonia. Drug induced eosinophilic pneumonia usually responds simply to withdrawing the offending drug. Parasitic infections and certain fungal infections such as coccidioidomycosis respond to specific anti-parasite and anti-fungal agents, and the administration of steroids in this situation may be potentially harmful, and thus should be excluded by appropriate investigations.
Disclosure
The author reports no conflicts of interest.
