Abstract
Allergic respiratory diseases can increase serum carcinoembryonic antigen levels. We report three cases experiencing allergic symptoms that proved refractory to inhaled corticosteroids but exhibited a positive response to long-term treatment with oral corticosteroids. This response was characterized by a synchronous alteration in serum eosinophil counts and carcinoembryonic antigen levels. Immunofluorescence assays indicated localized carcinoembryonic antigen production within eosinophils. In addition, we conducted a systematic review of patients exhibiting similar characteristics on PubMed. After comprehensively reviewing this unique pathophysiological condition, we herein introduced a novel term “Allergic hyper-carcinoembryonic antigen syndrome,” defined by the following criteria: (1) recurrent asthmatic attacks; (2) eosinophilia or pulmonary eosinophilic infiltrations accompanied by elevated serum carcinoembryonic antigen levels; (3) pulmonary lesions determined by imaging or biopsy; (4) exclusion of malignancy and infections; and (5) responsive to systemic corticosteroids. Allergic hyper-carcinoembryonic antigen syndrome suggests systemic corticosteroids should be introduced early when managing allergic patients with both eosinophilia and elevated serum carcinoembryonic antigen levels.
Introduction
Allergic respiratory diseases, such as allergic asthma and allergic rhinitis, are primarily characterized by type 2 inflammation, 1 and the cells involved in this process include eosinophils, neutrophils, macrophages, and mast cells. 2 The key humoral components include immunoglobulin E (IgE), induced by T helper-2 (Th2) cells, and inflammatory mediators such as histamine, leukotrienes, and prostaglandins. 3 The serum levels of these inflammatory factors are closely associated with the severity of allergic respiratory diseases. 4
Carcinoembryonic antigen (CEA) is widely recognized as a valuable biomarker for detecting, monitoring, and post-surgical surveillance of various cancer types, including breast cancer and gastrointestinal tumors.5–7 Interestingly, in some rare cases, serum CEA levels have been found to reflect the progression of allergic respiratory diseases.8–13
Eosinophils (EOS), first described by Paul Ehrlich in 1879, play crucial roles in various pathophysiological conditions, including allergic diseases, autoimmune disorders, parasitic infections, and adverse drug reactions.14,15 They also represent a significant effector cell population in allergic reactions and chronic inflammation. Controlling eosinophil activity has been shown to mitigate the exacerbation of allergic diseases, including asthma.16,17 However, the relationship between eosinophils and CEA in allergic diseases remains unclear.
In 2015, we reported a case of a patient presenting with asthma, along with eosinophilia and an elevated serum CEA level, who exhibited poor responsiveness to inhaled corticosteroid (ICS) treatment and in whom prolonged oral corticosteroid was proven to be effective. 18 Furthermore, some other published cases also manifested similar characteristics, that is, difficult-controlled asthma, elevated blood eosinophil counts, and high serum CEA levels.10,11 Despite these observations, this pathophysiological condition and its treatment approach have not been comprehensively and systematically studied. Herein, we propose the term “Allergic hyper-carcinoembryonic antigen syndrome” (AHCEAS) to describe this condition based on our literature review and analysis of retrieved clinical cases that exhibited allergic symptoms along with synchronous changes in pulmonary imaging, elevated blood eosinophil counts, and high serum CEA levels.
Case presentation
We identified three patients hospitalized at the Sun Yat-Sen Memorial Hospital who shared similar characteristics, forming a case series with the following criteria: (1) patients diagnosed with allergic disease, excluding malignancy and infectious diseases; (2) blood elevated levels of (EOS) count >0.3 × 109/L or significant eosinophilic infiltration in tissue, as confirmed by histology accompanied with serum CEA > 5.0 ng/ml; and (3) lesions identified through imaging or biopsy. The laboratory reference ranges for the cited indices are as follows: EOS count 0.02–0.52 × 109/L, blood EOS percentage 0.4%–8.0%, and serum CEA <5.0 ng/ml.
Clinical information, including medical history, laboratory workup, imaging study, and treatment, was collected from the electronic medical records. We carefully reviewed the follow-up files of the patients for at least 5 years to exclude any latent malignancy or chronic infectious diseases. For patients with available biopsy specimens, we repeated hematoxylin and eosin (H&E) staining. In addition, immunofluorescence staining was performed to observe the distribution of CEA and EOS in the tissues. CEA-related cell adhesion molecule 5 (CEACAM5) antibody (Abcam, ab133633) and ribonuclease 3 (RNase3) antibody (Thermofisher, PA5-79927) were used to identify CEA and EOS, respectively. The details of the immunofluorescence staining process are provided in the Supplemental File. Informed consents were obtained from all three patients enrolled for the pathologic study and case report.
Case 1
On June 13, 2013, a 48-year-old woman presented with a persistent cough accompanied by phlegm and dyspnea for 2 months. She had a history of allergic sinusitis and nasal polyps. Her initial blood examination revealed a blood EOS count of 0.01 × 109/L and a serum CEA level of 10.5 ng/ml. Further testing revealed an IgE level of 415 IU/ml (Table 1) and an erythrocyte sedimentation rate of 38 mm/h. A chest computed tomography (CT) scan revealed a thickening of the posteriolateral bronchial wall in the left lower lung (Table 2). Spirometry showed a forced expiratory volume in the first second (FEV1) of 1.8L and a forced vital capacity (FVC) of 2.4L, with an FEV1/FVC ratio of 0.75. Sputum culture, tuberculin skin test, galactomannan test, and stool analysis showed no evidence of bacterial, mycobacterial tuberculosis, fungal or parasitic infections, and serological tests for rheumatoid factor, antinuclear antibodies, anti-extractable nuclear antigen antibodies, and antineutrophilic cytoplasmic antibodies were all negative. In addition, ultrasound scans for breasts, abdomen, and pelvis did not reveal any signs of malignancy. As the patient was initially diagnosed with asthma, she was given nebulized budesonide and salbutamol, but her symptoms were not relieved. Consequently, oral prednisone at a dose of 10 mg once daily was prescribed for 1 month, which resulted in a remarkable improvement in her symptoms.
A summary of demographic information of allergic hyper-carcinoembryonic antigen syndrome patients.

CEA: carcinoembryonic antigen; EOS: eosinophil; IgE: immunoglobulin E; LLL: left lower lobe; NA: not available; RML: right middle lobe.
A summary of radiography and biopsy information of allergic hyper-carcinoembryonic antigen syndrome patients.

CEACAM5: CEA-related cell adhesion molecule 5; CEA: carcinoembryonic antigen; CT: computer tomography; EOS: eosinophil; H&E: hematoxylin and eosin; LLL: left lower lobe; LUNG-RADS: lung imaging reporting and data system; RML: right middle lobe; RUL: right upper lobe; TBB: transbronchial biopsy; TBLB: transbronchial lung biopsy.
Two years later, the patient was readmitted with a recurring complaint of cough accompanied by phlegm and dyspnea. Her laboratory tests, this time, revealed a blood EOS count of 1.02 × 109/L, along with a notable increase in serum CEA levels, reaching 24.6 ng/ml. A thoracic CT scan demonstrated static thickening of the posteriolateral bronchial wall in the left lower lung, suggestive of chronic inflammatory changes. Drawing from our previous experience with a similar case, we recognized this as a treatable trait, and the patient was promptly administered intravenous methylprednisolone at a dosage of 40 mg every 12 h. After 4 days, her symptoms significantly improved, and both CEA and EOS levels decreased (Table 3).
A summary of treatment and follow-up information of allergic hyper-carcinoembryonic antigen syndrome patients.
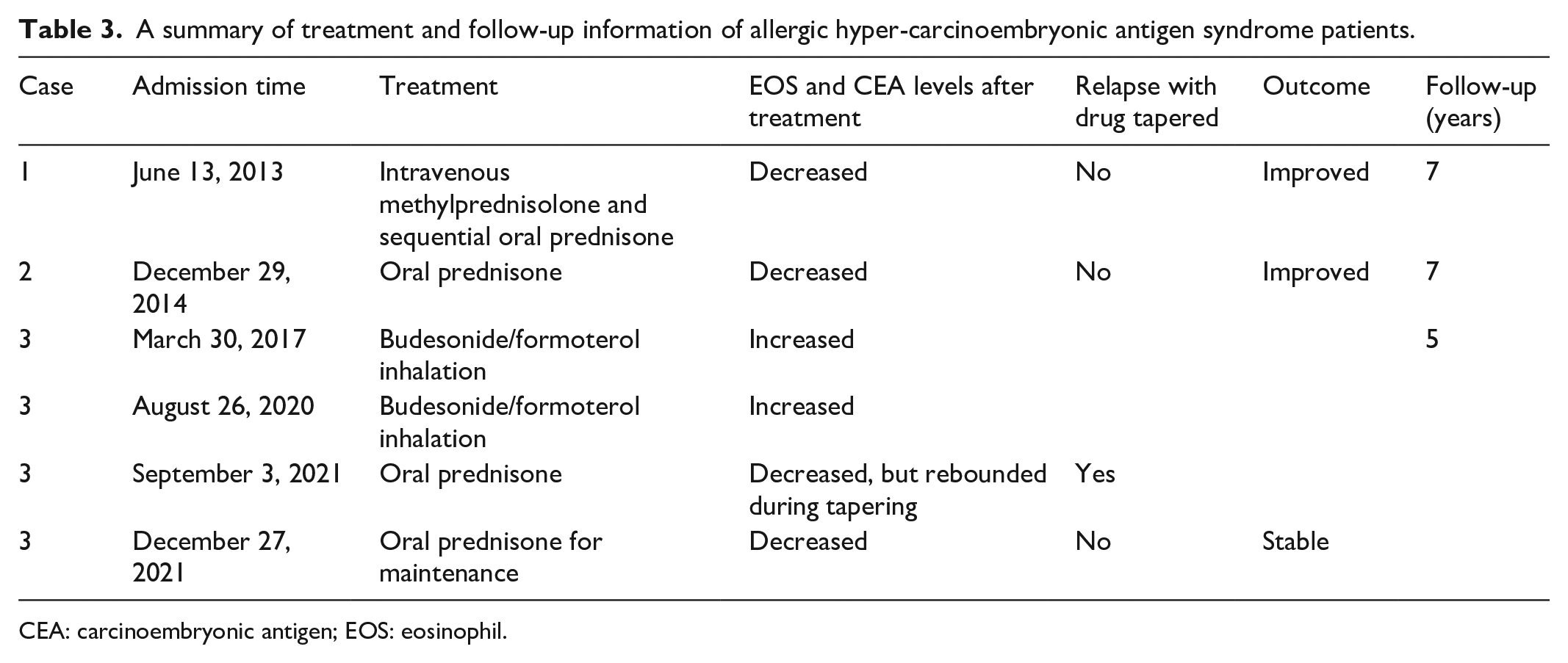
CEA: carcinoembryonic antigen; EOS: eosinophil.
In July 2016, the patient underwent a nasal polypectomy, and the pathological examination revealed abundant accumulation of eosinophils beneath the nasal polyp mucosa (Figure 1(a)). Subsequently, immunofluorescence staining of the nasal polyps showed the distribution of CEA-related cell adhesion molecules 5 (CEACAM5) within the cytoplasm of some RNase3-positive cells, suggesting the potential for EOS to express CEA (Figure 2). After surgery, she had an episode of major asthmatic attack with fluctuations in the EOS count and a short course of oral steroids was immediately given. Afterward, in November 2016, her serum CEA level decreased to 4.4 ng/ml, while her EOS level returned to the normal range.

H&E staining of biopsy specimens in Cases 1 and 3. (a) Eosinophil infiltration (red arrow) and plasma cell presence (blue arrow) beneath nasal polyp mucosa (Case 1, magnification ×10). (b) Abundant eosinophilic infiltrates (red arrow) within nasal polyp tissues (Case 3, magnification ×10). (c) Evidence of lymphocyte, plasma cell, and eosinophil infiltration (red arrow) in small bronchial mucosa tissues (Case 3, magnification ×10).
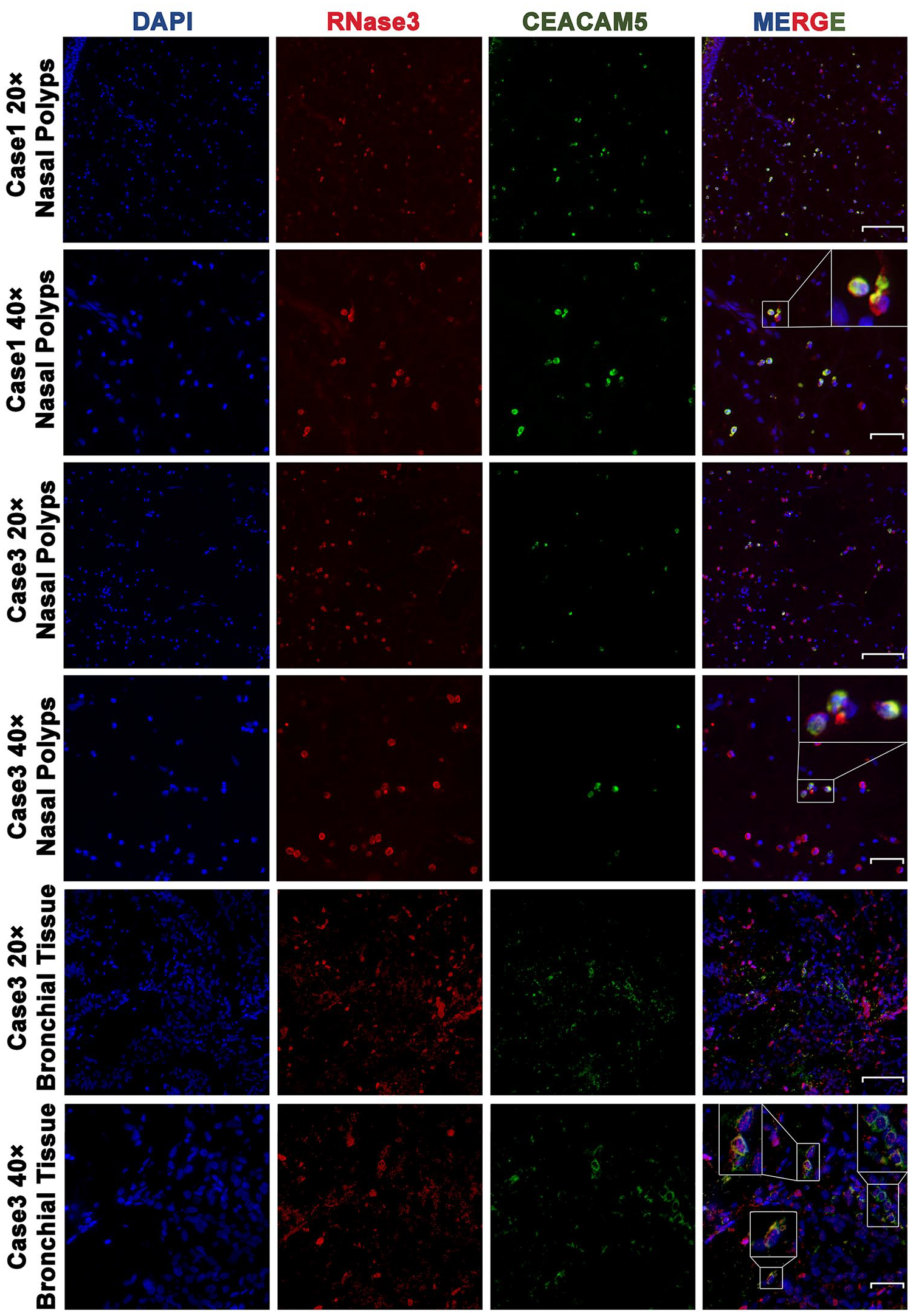
Immunofluorescence detection confirming co-localization of CEACAM5 and RNase3. CEACAM5 (green signal) is distributed within the cytoplasm of certain RNase3-positive cells (red signal) in nasal polyps (Case 1: first row, low-power magnification; second row, high-power magnification). In nasal polyps and bronchial wall tissue (Case 3: third row and fifth row, low-power magnification; fourth row and sixth row, high-power magnification), CEACAM5-positive cells (green signal) are co-localized with RNase3-positive cells (red signal). Scale bar: 100 μm in low-power magnification and 40 μm in high-power magnification.
Case 2
A 41-year-old woman with a history of allergic rhinitis and asthma had been on long-term treatment with inhaled formoterol/budesonide. On December 29, 2014, she presented to our hospital with complaints of a persistent cough accompanied by yellow sputum. Her blood EOS count was 0.81 × 109/L, serum CEA level was 8.8 ng/ml (Figure 3(a)), and IgE levels were 241 IU/ml. Chest X-ray and later CT thorax detected subsegmental atelectasis of the right middle lobe, which was not common in asthma exacerbation (Figure 4(a) and Table 2). Subsequent bronchoscopy was conducted for endobronchial screening, and stenosis of the bronchial lumen was observed in the dorsal segment of the right lower lobe and the right middle lobe, respectively, due to bronchial mucosa edema. Transbronchial biopsy revealed inflammation of the bronchial mucosa but did not identify any malignant cells. Furthermore, bronchial lavage fluid workup for pathogens indicated no presence of fungi or bacteria, particularly Mycobacterium tuberculosis. Empirical antibiotic therapy, consisting of ceftriaxone and moxifloxacin, was initiated, and continued for 2 weeks; however, there was minimal improvement in lung lesions as traced by chest X-ray. Considering the possibility of a unique allergic disease, a trial of oral prednisone at 30 mg once daily was initiated. The patient showed significant improvement in both symptoms and imaging within a week. The prednisone dosage was then gradually tapered by 5 mg weekly until withdrawal. Subsequent chest X-rays demonstrated complete resolution of consolidation in the right middle lobe following prednisone therapy (Figure 4(b)). After discontinuation of prednisone, the patient’s CEA and EOS levels showed fluctuating changes, but her symptoms did not reoccur. Until now, she has been followed up for 8 years, during which time her CEA and EOS levels keep normal.
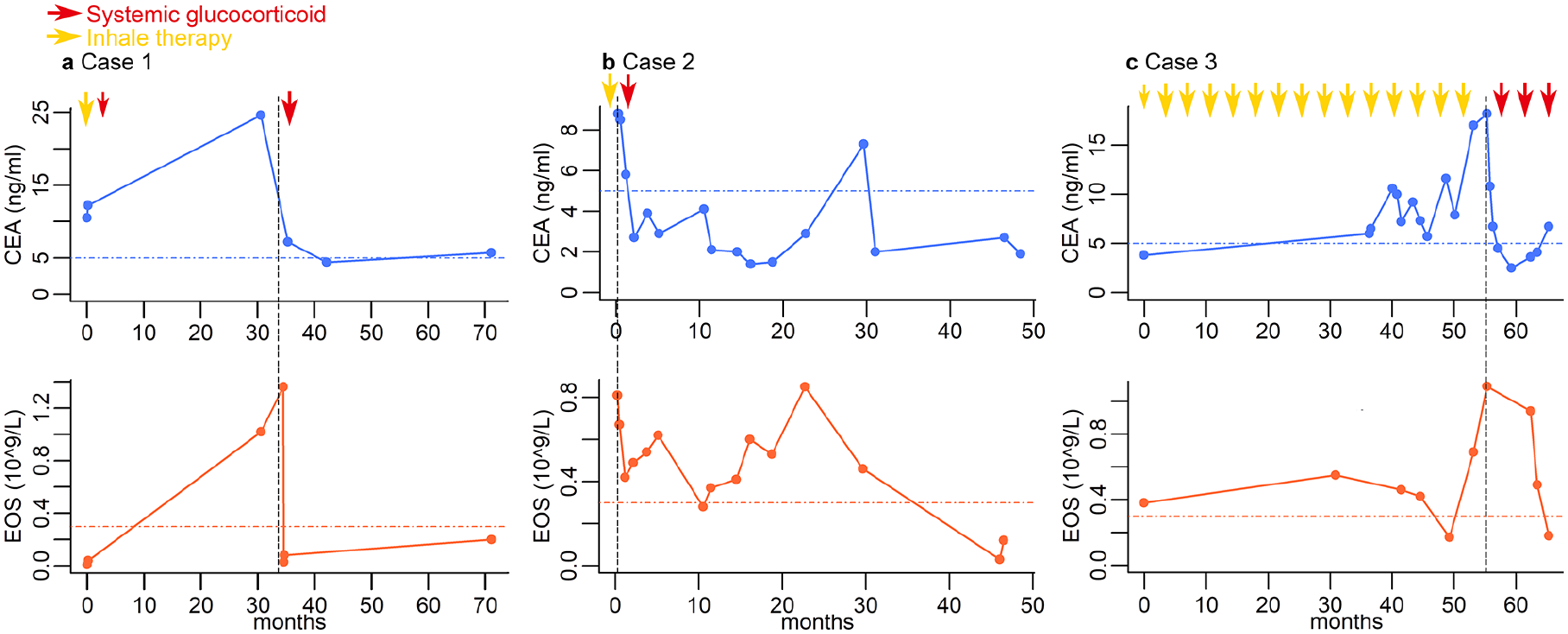
Variations in CEA and EOS in the case series from the initial admission. Line charts (a)–(c) illustrate the variations in EOS (orange line) and CEA (blue line) levels for Cases 1–3, respectively, from their initial admission. Serum CEA levels consistently mirrored the trends in blood EOS counts, and both parameters decreased following systemic corticosteroid therapy. By contrast, prior to systemic corticosteroid treatment (red arrow), the use of inhaled corticosteroids with or without bronchodilators (yellow arrow) showed less effectiveness. The size of the arrow indicates the dosage of corticosteroids administered.

Comparison of pulmonary imaging in Cases 2 and 3 before and after treatment. (a) Chest X-ray of Case 2 before prednisone therapy, illustrating inflammation in the middle lobe of the right lung with incomplete bronchial occlusion of the middle lobe. (b) Chest X-ray of Case 2 after 6 weeks of prednisone therapy shows the substantial resolution of the original inflammation in the right middle lobe. (c) Chest computed tomography (CT) scan of Case 3 before prednisone therapy reveals the bronchial occlusion in the dorsal segment of the left lower lung and complete consolidation with atelectasis in the left lower lung. (d) Chest CT scan of Case 3 after 3 months of prednisone therapy, indicating alleviation of atelectasis in the left lower lung.
Case 3
On March 27, 2017, a 52-year-old woman presented with exhalation wheezing and recurrent cough for 1 month. She had a history of allergic rhinitis and chronic rhinosinusitis with polyps. One week prior to this admission, she had experienced a transient fever of 39°C, which spontaneously subsided within a day. Her initial blood EOS count was 0.38 × 109/L, serum CEA level was 3.8 ng/ml, and IgE levels were 42 IU/ml. A CT scan revealed minimal infiltration and fibrotic changes in the posteriolateral basal segment of the left lower lobe. Lung function test results are shown in Table 4, and a bronchoprovocation test by histamine was positive. She was initially diagnosed with asthma and was treated on inhaled formoterol/budesonide, which partially alleviated her wheezing, though her cough persisted.
A summary of pulmonary function information of AHCEAS patients.

AHCEAS: allergic hyper-carcinoembryonic antigen syndrome; FEV1: forced expiratory volume in the first second; FVC: forced vital capacity.
In 2018, the patient underwent pansinusectomy at our hospital, and the pathology results revealed EOS infiltration in the resected tissues (Figure 1(b)). Subsequent immunofluorescence double labeling demonstrated that some CEACAM5-positive cells were co-localized with RNase3-positive cells in nasal polyps (Figure 2), and at that time, her IgE level was 142 IU/ml.
On August 25, 2020, the patient was admitted for incidentally finding lung nodules and segmental atelectasis of the left lower lobe by CT thorax (Figure 4(c)). Her serum CEA level was elevated to 10.6 ng/ml at that juncture while her blood EOS count was 0.46 × 109/L. Spirometry indicated an FEV1/FVC ratio of <0.70, and the bronchodilation test was negative. Bronchoscopy was then performed, and transbronchial lung biopsy from the left lower lobe demonstrated infiltrations of lymphocytes, plasma cells, and EOS in small bronchial mucosa tissues (Figure 1(c)). Immunofluorescence staining revealed the colocalization of RNase3-positive cells and CEACAM5-positive cells in bronchial tissue (Figure 2). Workup including bronchoalveolar lavage fluid (BALF) cultures and bronchial brushing for cytology showed no evidence of infections or malignancy. In addition, gastroscopy and colonoscopy revealed multiple polyps, while ultrasonogram detected no signs of malignancy.
Afterward, the patient was regularly followed up and kept compliance to inhaled formoterol/budesonide. In September 2021, a deterioration of cough was noted along with the progression of lung lesions, as CT scans revealed nearly complete atelectasis of the left lower lobe (Figure 4(c)). Her blood EOS count had risen to 1.09 × 109/L, and her serum CEA level had increased to 18.2 ng/ml. Given the allergic presentation, she was initiated on oral prednisone at a dosage of 10 mg, three times daily, for 1 month. Remarkably, her symptoms improved, and her CEA level decreased to 6.7 ng/ml. Subsequently, the prednisone dose was gradually tapered by 5 mg every week until her symptoms deteriorated and the CEA level rebounded when on a daily dose of 15 mg. As a result, the tapering strategy was slowed down. After 3 months of steroid therapy, CT thorax showed significant improvement as well as resuming lung volume of the left lower lobe (Figure 4(d)). Currently, she continues to receive prednisone with a maintenance dose of 10 mg daily.
Case series summary
All patients included in this case series presented with a common complaint of cough, and each had a history of allergic rhinitis or sinusitis. All cases had elevated levels of CEA, EOS, and IgE and were initially diagnosed with asthma. Case 1 had initially normal EOS levels, but as CEA levels increased, her EOS levels also increased (Table 1). Chest CT findings consistently exhibited features such as panbronchiolitis, bronchial wall thickening, bronchial occlusion, and, in some cases, atelectasis. Bronchoscopy examination revealed stenosis of one or more lung segments due to bronchial mucosa swelling. Two patients presented with nasal polyps, and biopsy specimens from these polyps demonstrated EOS infiltration. Immunofluorescence detection in both cases confirmed the co-localization of CEACAM5 and RNase3, suggesting that CEA was expressed in EOS in AHCEAS patients (Figure 2). Despite initial attempts with ICS and bronchodilators, the treatment response was poor. However, systemic corticosteroid therapy effectively alleviated their symptoms, eosinophilia, elevated CEA levels, and even atelectasis. The relationship between serum CEA and blood EOS levels, along with the timeline of corticosteroid use, is shown in Figure 3. In the case of patient 3, the symptoms relapsed after drug withdrawal, but the following maintenance treatment with prednisone successfully controlled their symptoms (Table 3). We conducted thorough follow-up examinations for all patients, extending for at least 5 years, to rule out any latent malignancies or infectious diseases.
Discussion and literature review
The three cases reported and the 14 patients reviewed in this series showed a common clinical profile characterized by pulmonary lesions, elevated blood eosinophil levels, and increased serum CEA levels, which remarkably had a favorable response to systematic steroid treatment. Based on these, we propose the term AHCEAS, which can be defined as a clinical syndrome characterized by (1) recurrent asthma symptoms such as cough, wheezing, and dyspnea; (2) elevated blood EOS count exceeding 0.3 × 109/L or extensive eosinophil infiltration confirmed by pathology, in conjunction with a serum CEA level surpassing 5.0 ng/ml; (3) identification of respiratory lesions through imaging or biopsy; (4) absence of malignancy and infectious diseases; and (5) effective clinical symptom control achieved through intravenous or oral corticosteroid therapy, accompanied by reduced serum CEA and blood eosinophil levels.
The proposal of this syndrome offers valuable guidance for clinical physicians in addressing the complex clinical presentations of allergic symptoms combined with elevated EOS and CEA levels. For instance, when a patient exhibits asthma-like symptoms unresponsive to conventional treatments such as ICSs and bronchodilators, and also has abnormal lung imaging findings and elevated CEA levels without malignancy, this syndrome could be of diagnostic significance for differentiation. In such cases, considering systemic steroids as an initial treatment option may result in remarkably positive therapeutic effects. Still, further investigations into the potential underlying mechanism of this syndrome are warranted to validate our findings.
Literature review
A search on PubMed using the specific search terms yielded 162 papers of interest according to the inclusion criteria in the Supplemental File. Among these, 13 articles discussed eosinophil-related allergic diseases associated with elevated serum CEA. We excluded three articles related to malignancy or infection diseases and another three articles due to the absence of treatment information. One of the articles was a case we had previously reported. 18 Eventually 6 individual case reports comprising 14 patients were selected for our study analysis. Notably, one of these articles documented nine eligible patients, 12 and herein, we compiled these clinical records and analyzed them, presenting it as a review series (Supplemental material Table S1).
Baseline characteristics and risk factors
Among the 14 patients included, one case was from Germany, and the rest were from Japan. The onset age of the disease can be described as a median of 57.5 years old, with a lower quantile and upper quantile of 47.8 and 67.0 years old, respectively. The age range of the patients was from 37 to 70 years. Females (10/14) were more prevalent in the disease group. All patients were in an atopic state or had a history of allergies, with three patients having sinusitis. Few of them were smokers (3/14).
Laboratory characteristics
The median of blood EOS count in baseline was 1.265 × 109/L (interquartile 0.683–3.015 × 109/L; range 0.403–8.308 × 109/L). The median serum CEA level before treatment was 13.5 ng/ml (interquartile 7.4–28.1 ng/ml; range 5.4–102.5 ng/ml), showing a trend compatible with the variation in blood EOS count. A decreasing trend of serum CEA level along with blood EOS count after corticosteroid therapy was noted in all the patients. Among the patients with available information, a significant majority had elevated serum total IgE levels (11/12).
Radiographic characteristics
Abnormalities including patchy ground glass opacities, bronchiectasis, and consolidations were observed on chest X-rays or thorax CT scans (Supplemental material Table S2). Atelectasis was observed in patient 3, 4 and central bronchiectasis and consolidations were described in all the patients with allergic bronchopulmonary aspergillosis (ABPA) (patients 6–14). 12 The above signs suggest airway remodeling in the background of long-term uncontrolled inflammation.
Pathological characteristics
Biopsy data were available for five patients (Supplemental Table S3). EOS infiltration, characterized by the accumulation of EOS, was observed in all the specimens. Not only did the CEA levels increase in the blood, but immunohistochemistry also revealed positive CEA stains in the biopsied specimens (3/5). In patient 2, CEA secretion was identified in goblet cells. Immunohistochemical analysis revealed CEA expression in the bronchiolar epithelium and on the mucous surface of nasal polyps in patient 5.
Treatment and prognosis
Patient 1 was not given steroids and recovered after radiotherapy for extensive polypoid sinusitis and bony destruction of the middle and posterior skull base (Supplemental Table S3). All the other patients were relieved in both symptoms and laboratory workup after intravenous or oral corticosteroid treatment. Patient 4 exhibited steroid dependence while the EOS and CEA levels increased, and clinical symptoms relapsed during tapering. These findings suggest that allergic diseases with elevated CEA levels could be related to a steroid-dependent phenotype. Furthermore, it was intriguingly found that systemic corticosteroid was still effective even though symptoms relapsed. No mortality related to this condition was reported. To determine the relationship with long-term survival, a larger population and an extended follow-up period are still required.
Role of CEA in diseases
CEA was initially identified as a tumor antigen by Gold and Freedman. 19 The CEA gene family can be categorized into two branches, known as CEACAMs and pregnancy-specific glycoproteins. Among the CEACAMs, there are 12 human CEACAM genes, including CEACAM1, CEACAM3 through CEACAM8, CEACAM16, and CEACAM18 through CEACAM21.20,21 Serum CEA levels have long been recognized as potential biomarkers for diagnosing tumors, monitoring tumor progression, and post-surgical surveillance in various cancer types, such as breast cancer and gastrointestinal tumors.5–7 Ohnishi et al. 22 reported a patient exhibited elevated CEA and EOS levels, which led to the detection of malignancy, and was subsequently diagnosed as allergic bronchopulmonary mycosis complicated with lung adenocarcinoma. However, elevated CEA levels have also been reported in benign conditions. A retrospective study involving 4796 patients with benign lung diseases, including chronic obstructive pulmonary disease, pneumonitis, and interstitial lung disease, found that approximately 3.1% of cases exhibited elevated CEA levels. 23 Moreover, elevated serum CEA has been detected in half of the patients with idiopathic pulmonary fibrosis. 24 It is worth noting that even patients with the coronavirus disease 2019 (COVID-19) demonstrate elevated CEA levels. 25 In certain allergic conditions, an increase in EOS count often coincides with elevated CEA levels, thereby serving as an indicator of disease progression.8–13 However, the origin of elevated CEA in allergic diseases remains unclear. In this study, the asthmatic patients with hypereosinophilia and elevated serum CEA levels were thoroughly ruled out for potential causes related to infections, tumors, autoimmune disorders, and monoclonal gammopathy.
Potential relationship between CEA and allergic respiratory disease
The secretion of CEA in AHCEAS might be related to eosinophils or other cells in allergic respiratory diseases, such as hypereosinophilic syndrome and ABPA.13,26 In this study, immunofluorescence indicated that CEACAM5-positive cells were co-localized with EOS in bronchial tissues and nasal polyps (Figure 2). In ABPA patients, EOS can secrete CEA, the serum level of which was significantly higher in patients with pulmonary consolidation than those without consolidation. 12 Yang et al. 26 confirmed CEA positivity in eosinophils via immunostaining in lung tissues from an ABPA patient who also had elevated serum CEA and eosinophil levels, which was consistent with the result of immunofluorescence staining in bronchial tissues and nasal polyps in our present study. A previous report indicated that interleukin-5, produced by activated lymphocytes or eosinophils, could stimulate eosinophils to enhance CEACAM expression on lipid rafts. 27 Furthermore, severe allergic reactions and inflammatory responses may lead to airway epithelial cell damage, potentially up-regulating CEACAM expression. Over time, chronic allergies can impact the growth and differentiation processes of cells, such as goblet cell hyperplasia and epithelial metaplasia, potentially promoting CEACAM production by altering cell expression patterns.28,29 Moreover, long-term allergic response-induced stimulation on the respiratory tract can lead to symptoms such as cough, asthma, and nasal polyps, suggesting a correlation between CEA levels and the severity of these symptoms, 30 indicating that elevated CEA levels might not only serve as a marker for certain types of cancer but could also reflect the intensity of allergic and inflammatory conditions in the respiratory system.
In addition to eosinophils, CEACAM can also be produced by other cells in allergic respiratory diseases. Among the CEA gene family members, CEACAM6 is noteworthy as it targets the apical membranes of polarized epithelial cells. 31 Severe asthma often presents with characteristics of elevated CEACAM6 levels in both neutrophils and epithelial cells within the airway. 29 Furthermore, an increase in CEACAM6 has been observed in intubated infants with lung diseases. 32 Shikotra et al. 29 revealed that allergens and neutrophils can promote CEACAM6 expression in the epithelium through mechanisms involving epithelial wounding and the action of neutrophil elastase, respectively. Intriguingly, peptides of CEACAM6, when bound by another N-domain of CEACAM6, can generate superoxide, a molecule capable of activating neutrophils. 33 This may exacerbate the disease.
Given the limited sample size of the cases presented, it was not possible to conduct an in-depth statistical analysis. To further consolidate the concept of our proposed syndrome, a larger sample size from retrospective studies or additional reported cases with similar characteristics is required. Moreover, in-depth basic research is essential to uncover the specific conditions under which allergic reaction tissues involved in allergic reactions produce CEA.
Conclusion
In conclusion, patients diagnosed with AHCEAS displayed consistent changes in serum CEA levels and blood EOS counts, which correlated with their disease severity. In clinical practice, systemic corticosteroids could be introduced in the early stage for those allergic patients presenting with both hypereosinophilia and elevated CEA levels. Nevertheless, more comprehensive research is warranted to further understand the underlying mechanisms of this condition.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X241261152 – Supplemental material for Allergic hyper-carcinoembryonic antigen syndrome: A syndrome summarized by case series
Supplemental material, sj-docx-1-sco-10.1177_2050313X241261152 for Allergic hyper-carcinoembryonic antigen syndrome: A syndrome summarized by case series by Zhaolin Li, Luna Hong, Yuewei Li, Xiaoling Lin, Ming Chen, Shiyi Bu, Shuwan Xu, Qiaojun Zeng, Yijiao Huang, Lijuan Bian, Jianming Zheng, Boying Gao, Guirong Liu, Wenman He, Xueming Song, Fengzhan Ye, Linjie Huang, Shanping Jiang, Jianting Shi and Tiantian Tang in SAGE Open Medical Case Reports
Supplemental Material
sj-docx-2-sco-10.1177_2050313X241261152 – Supplemental material for Allergic hyper-carcinoembryonic antigen syndrome: A syndrome summarized by case series
Supplemental material, sj-docx-2-sco-10.1177_2050313X241261152 for Allergic hyper-carcinoembryonic antigen syndrome: A syndrome summarized by case series by Zhaolin Li, Luna Hong, Yuewei Li, Xiaoling Lin, Ming Chen, Shiyi Bu, Shuwan Xu, Qiaojun Zeng, Yijiao Huang, Lijuan Bian, Jianming Zheng, Boying Gao, Guirong Liu, Wenman He, Xueming Song, Fengzhan Ye, Linjie Huang, Shanping Jiang, Jianting Shi and Tiantian Tang in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
We are grateful for the approval from the patients for the useful comments and suggestions from the anonymous referees. We appreciate the technical support provided by Huajian Xu, Darong Lu from Sun Yat-Sen University, Zhimin Zhu from Peking University, and Lijian Zhu from Yangchun.
Author contributions
S.J. and T.T. designed the study. L.H., T.T., S.B., Z.L., Y.L., L.H., Q.Z., and G.L. contributed to patient recruitment and data collection. Z.L., S.X., J.Z., B.G., W.H., X.S., S.X., and F.Y. collected and analyzed the clinical data. Z.L. performed the literature review. Z.L. wrote the manuscript. S.J., T.T., J.S., and M.C. revised the manuscript. Y.L., L.H., Y.H., and L.B. performed pathological and immunofluorescence analyses. All authors contributed to the paper.
Data sharing statement
The data sets and images supporting the conclusions of this article are included in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (No. 82071804 and No. 81700033) and the Guangzhou Science and Technology Program Key Project (No. 202102020429).
Ethics approval
The procedures performed in this study adhered to the tenets of the Declaration of Helsinki and were approved by the Ethics Review Board of Sun Yat-sen Memorial Hospital (Guangzhou, China, IRB number SYSKY-2022-425-01).
Informed consent
Written informed consent was provided by all three patients for the publication of the case details and images. Details of the case are published with the approval of the Ethics Committee of our hospital.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
