Abstract
Eosinophilic gastrointestinal is an uncommon disorder affecting all tissues of the digestive tract. Eosinophilic gastroenteritis presents with a complex clinical profile, lacks specificity, and is prone to misdiagnosis in patients without evident allergens. In some instances, it is mistaken for an acute abdomen or a gastrointestinal tumor, potentially leading to unnecessary surgical interventions. We present the case of a 21-year-old female with eosinophilic gastroenteritis whose primary clinical manifestations were abdominal pain and ascites.
Introduction
Eosinophilic gastroenteritis (EGE) is a rare chronic gastrointestinal disease characterized by nonspecific clinical manifestations, making accurate diagnosis challenging. A histopathologic biopsy of the gastrointestinal tract, revealing an eosinophil count of ⩾20/HPF, is pivotal for diagnosing EGE. Attention should focus on eosinophil granulocyte degranulation, eosinophilic glands or crypt abscesses, and pathological alterations like small intestinal microvillus atrophy. 1 In cases where peripheral blood eosinophils are within normal range, enhancing the multipoint biopsy during gastroenteroscopy is necessary. Repeated gastroenteroscopic biopsy may be conducted if needed to clarify the diagnosis. Treatment primarily involves dietary interventions, with steroids as the main pharmacological option. Current research investigates the potential of biologics targeting autoimmune and inflammatory pathways, requiring additional exploration. 2
Case presentation
A 21-year-old female presented to the emergency department with abdominal pain for 7 days. She had no recent travel history or any record of asthma/allergies, no family history of similar symptoms, and her living environment was comparable to that of others in her community. Physical examination revealed pain in the right lower abdomen and normal cardiopulmonary findings. Vital signs were as follows: temperature, 36.7°; heart rate, 102 beats/min; respiration, 20 breaths/min; blood pressure, 124/90 mmHg; height, 158 cm; and weight, 52.5 kg. Laboratory examination results revealed the following: white blood cells, 60.64 × 109/L; neutrophils, 10.06 × 109/L; lymphocytes, 6.84 × 109/L; eosinophils, 42.80 × 109/L; and an elevated serum total immunoglobulin E (IgE) level of 1109.30 IU/mL. A stool routine examination showed no abnormality and allergen test results were negative, 20 allergens were detected including tree mix, ambrosia artemisifolia, artemisia vulgaris, dust mite combinations, house dust, cat hair, dog epithelium, cockroaches, mold combinations, humulus japonicus, eggs, cow’s milk, peanuts, soybeans, beef, mutton, fish combinations, shrimp, crab, and cross-reacting references. Computed tomography (CT) revealed thickening of the bowel wall and ascites in the pelvic and abdominal regions (Figure 2). Paracentesis revealed significant yellowish ascites with high eosinophil counts (Figure 1). Both bacterial and tuberculosis cultures were negative, and there were no cytologic features indicative of malignancy. Bone marrow aspiration revealed active myeloproliferative bone marrow with 81% granulocytes, an abnormally high percentage of eosinophils, and 10.8% erythrocytes (Figure 3). Gastroscopy showed mild gastric mucosal erythema, and colonoscopy showed nonspecific mucosal edema, erythema, and erosions of the colon, with villous mucosa in the terminal ileum. The biopsy revealed an elevated number of eosinophils (Figures 4–12). Treatment included dietary recommendations for improved nutrition, emphasizing a low-fat, high-calorie, low-fiber, nutrient-rich diet. The patient was also advised to avoid allergenic foods like milk and seafood. Initially, the patient received methylprednisolone 40 mg for 5 days and later switched to oral prednisone at 30 mg per day. Additional treatments included abdominal puncture drainage of ascites, spasmolysis, antiemetics, and other symptomatic measures. The patient was discharged after 10 days of symptom improvement, during the 3-month telephone follow-up, the patient showed no abdominal pain or other discomfort.

Shows ascites from a diagnostic puncture of the abdominal cavity.

Shows a bone marrow aspiration biopsy.

Computed tomography of the abdomen demonstrated abdomen ascites.

Computed tomography of the abdomen demonstrates thickening of the bowel wall.

Endoscopic manifestations of esophageal mucosa.

Endoscopic pathological biopsy results of esophageal mucosa.

Endoscopic manifestations of gastric mucosa.

Endoscopic pathological biopsy results of gastric mucosa.

Endoscopic manifestations of terminal ileal mucosa.
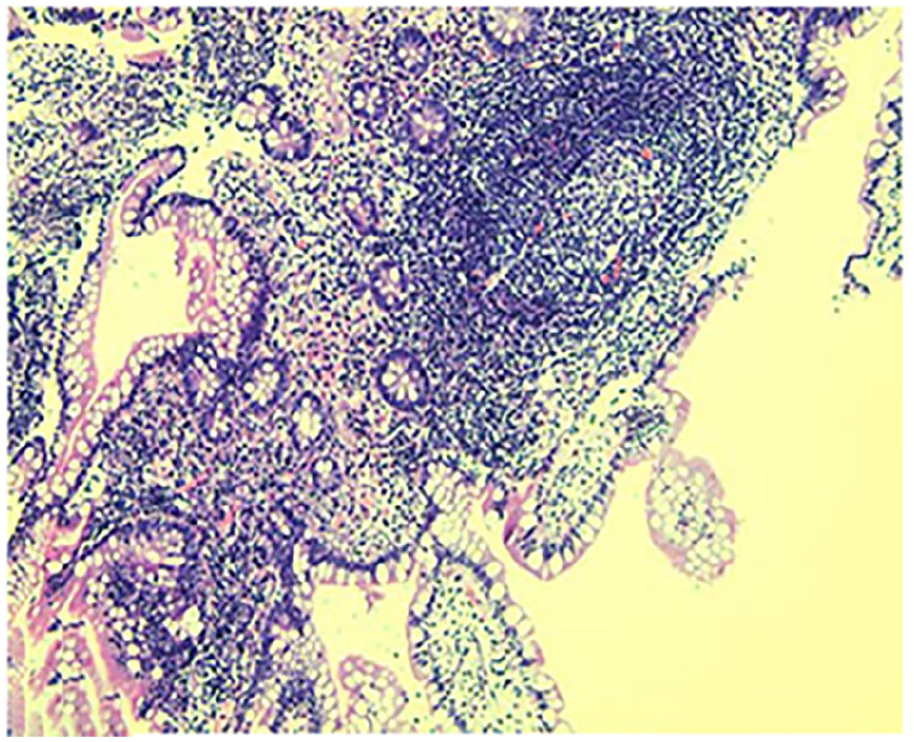
Endoscopic pathological biopsy results of terminal ileal mucosa.

Endoscopic manifestations of transverse colon mucosa.

Endoscopic pathological biopsy results of transverse colon mucosa.
Discussion
The pathogenesis of EGE remains incompletely understood; however, there is a prevailing consensus that an exaggerated Th2-type immune response, triggered primarily by exogenous antigens such as food and airborne particles, plays a pivotal role. 3 Diagnosing EGE necessitates the presence of gastrointestinal symptoms, substantial eosinophil accumulation in the gastrointestinal tract, and the exclusion of secondary causes for eosinophil buildup. 4 Despite the similarity of the pain in the right lower abdomen to that of appendicitis, computed tomography (CT) scans did not reveal any acute appendiceal manifestations, such as appendiceal thickening or appendiceal fecaliths, and the absence of significant changes in leukocytes and neutrophils but a markedly elevated level of eosinophils in the peripheral blood was crucial for the diagnosis of EGE. CT has limited diagnostic value in EG but is useful in classifying EGE and ruling out other abdominal pathologies. Typically, CT shows features such as ascites, bowel wall thickening, localized lymph node enlargement, and bowel rotation and perforation. 5 The patient’s CT revealed notable abdominal effusion and peritoneal puncture demonstrated yellowish ascites with numerous eosinophils on the smear. Diagnostic peritoneal puncture aids EG diagnosis and rules out acute abdominal conditions like hemorrhage and perforation. In addition, EGE needs to be differentiated from a number of disorders that can cause eosinophilia. In addition, EGE needs to be differentiated from a number of disorders that can cause eosinophilia: (1) Parasitic infections constitute a prevalent cause of eosinophilia, characterized by detectable parasitic pathogens (eggs or worms), potentially elevated serum IgE levels, approximately normal myeloproliferation with no augmentation in myeloid progenitor cells, and a mild to moderate eosinophilia predominantly in peripheral blood. 5 Organ damage manifestations may also arise, exemplified by Strongyloides stercoralis-induced intestinal wall inflammation and ulceration, leading to symptoms such as abdominal pain, diarrhea, and hematochezia. 5 Notably, anthelmintic therapy is generally efficacious, and chromosomal evaluations frequently yield normal results. (2) Drug reactions, particularly drug reactions with eosinophilia and systemic symptoms (DRESS), represent a rare yet potentially fatal adverse drug reaction. Characterized by a prolonged incubation period, DRESS is accompanied by rash, hematological abnormalities, and visceral damage. 6 Anticonvulsants, antibiotics, sulfonamides, and antirheumatic drugs are among the medications capable of inducing eosinophilia. In some patients, eosinophilic changes may be insignificant initially and may manifest only 1 to 2 weeks post-disease onset. DRESS syndrome should be strongly considered in patients experiencing fever, characteristic edematous erythema, lymphadenopathy, visceral organ involvement, and eosinophilia following drug administration. 6 (3) Connective tissue disorders, which are autoimmune in nature, can also be associated with eosinophilia. Conditions indicative of possible connective tissue diseases include positive antinuclear antibody tests, increased serum IgG and IgM levels, positive C-reactive protein, accelerated erythrocyte sedimentation rates, and elevated anti-streptococcal “O” titers. Bone marrow examination typically reveals no specific changes, with mild eosinophilia and no increase in myeloid progenitor cells. 7 The clinical presentation can definitively identify the corresponding connective tissue disease, such as systemic lupus erythematosus, polyarteritis nodosa, rheumatoid arthritis, or scleroderma. During the differential diagnosis, physicians should obtain a detailed patient history, including family and travel histories, and repeat screenings for parasite eggs and parasite-related antibodies. Based on the clinical presentation, appropriate tests should be selected to exclude infections, allergic diseases, drug reactions, rheumatologic immune diseases, vasculitis, or other tumors. In cases of diagnostic difficulty, specialists should be promptly consulted for collaborative diagnosis and treatment. For patients presenting with atypical abdominal symptoms and non-diagnostic laboratory findings, laparoscopy should be swiftly performed to ascertain the underlying cause, especially in the presence of serious gastrointestinal complications like obstruction or perforation. 8
Endoscopy and biopsy are pivotal in EGE diagnosis. The endoscopic examination of this patient revealed pronounced mucosal swelling, diffuse punctual erosion, and a substantial eosinophil presence in the biopsy, providing crucial evidence for EGE diagnosis. 9 Fujiwara et al validated significant endoscopic findings in 287 patients, with erythema being the most prevalent (72%), followed by ulceration (39%), discoloration (33%), erosion (28%), nodules (28%), and polyps (28%). 10 Nevertheless, specificity is lacking. Japanese scholars analyzing endoscopic features in six EGE patients postulated that microvillous flattening in the small intestine could be a distinctive marker for diagnosing EGE. 4
EGE treatment should focus on suppressing allergic reactions and eliminating allergens.9,10 Research indicates that corticosteroids form the cornerstone of EGE treatment. Therapeutic options encompass mast cell stabilizers, leukotriene receptor antagonists, antihistamines, dietary adjustments, immunomodulators, tumor necrosis factor inhibitors, and surgical intervention for patients with obstruction from muscle layer involvement.11-13
Conclusion
EGE is a rare disease of the gastrointestinal tract, which is difficult to diagnose due to the lack of specificity of clinical and endoscopic manifestations. Endoscopic biopsy is particularly important, and in addition, elevated blood eosinophils percentage combined with pathology helps in the diagnosis of the disease. Computed tomography (CT) has limited diagnostic value in EGE, but it is effective in identifying other abdominal diseases. The treatment of EGE should be based on suppression of allergic reactions and elimination of allergens, and corticosteroids are the most effective, but not the only, treatment option.
Footnotes
Data availability statement
The materials described in the manuscript, including all relevant raw data, will be freely available to any scientist wishing to use them for non-commercial purposes.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Patient privacy is fully protected in this article, and the patient’s detailed information has been removed. Written informed consent was obtained from the patient for her anonymized information to be published in this article.
