Abstract
Colorectal cancer (CRC) is the third most common cancer worldwide and accounts for 10% of all new cancer diagnoses. Angiogenesis is a tightly regulated process that is mediated by a group of angiogenic factors such as vascular endothelial growth factor and its receptors. Given the widespread use of antiangiogenic agents in CRC, there has been considerable interest in the development of methods to identify novel markers that can predict outcome in the treatment of this disease with angiogenesis inhibitors. Multiple biomarkers are in various phases of development and include tissue, serum, and imaging biomarkers. The complexity of the angiogenesis pathway and the overlap between the various angiogenic factors present a significant challenge to biomarker discovery. In our review, we discuss the angiogenesis pathway and the most promising evolving concepts in biomarker discovery, as well as highlight the landmark studies that identify subgroups of patients with CRC who may preferentially benefit from angiogenesis inhibitors.
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and accounts for 10% of all new cancer diagnoses. 1 Twenty percent of patients diagnosed with CRC are, unfortunately, found to have metastatic disease at presentation. Furthermore, ~30% of patients who are diagnosed with early-stage CRC sooner or later develop metastatic disease.2,3
Although the median overall survival (OS) of patients diagnosed with metastatic CRC (mCRC) has improved from nine months to >30 months over the past decade, the five-year OS remains at 5%–15%. The poor outcome of patients with mCRC calls for the development of new therapeutic options in addition to further refinement of current treatment strategies.4–6
The growth and proliferation of mCRC depends essentially on two signaling pathways: the vascular endothelial growth factor (VEGF) and the epidermal growth factor receptor (EGFR) pathways. Fortunately, therapeutic agents have been developed to target each of these pathways; their activity is well established and they have been incorporated into routine cancer treatment worldwide. In the United States, 60%–70% of patients with mCRC will receive these agents during the course of their treatment. 7
Anti-VEGF agents, such as bevacizumab, ziv-aflibercept, regorafenib, and ramucirumab, have all shown efficacy in the treatment of mCRC and are currently approved by the United States Food and Drug Administration (FDA) for use in mCRC. Since the introduction of antiangiogenic agents, there has been significant interest in the identification of clinical and molecular markers to help predict which subgroup of patients will benefit from inhibition of the angiogenesis pathway.8–11
Herein, we review the process of angiogenesis, paying particular attention to the discovery and use of serum, tissue, and imaging biomarkers that can potentially be used to predict patient tumor response to antiangiogenic agents (Fig. 1).
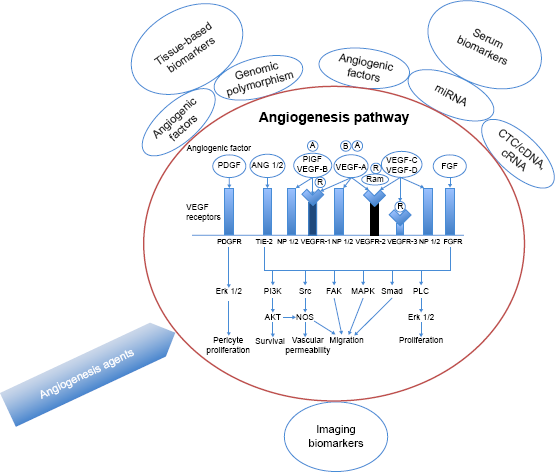
A schematic of the angiogenesis pathway, angiogenesis inhibitors, and the most promising biomarker techniques.
Angiogenesis Pathways
Angiogenesis is a complex process by which new blood vessels are formed from endothelial precursor. It is a critical step in cancer progression and is considered one of the hallmarks of cancer. This process is mediated through a group of ligands and receptors that work in tight regulation.12,13 A group of glycoproteins, including the VEGFs (VEGF-A, VEGF-B, VEGF-C, and VEGF-D) and the placental growth factor (PIGF), act as effectors of angiogenesis.14–17 These factors interact with three VEGF receptors (VEGFR-1, VEGFR-2, and VEGFR-3) and two neuropilin co-receptors (NRP1 and NRP2).18–20 The
The VEGFRs are tyrosine kinase receptors that are primarily located in the vascular endothelial cells.14,22,23 The binding of VEGF-A to VEGFR-2 is believed to be the most important activator of angiogenesis.12,24,25 This binding initiates a cascade of signals that result in endothelial cell proliferation and migration, increased vascular permeability, alteration of gene expression, and activation of the Ras pathway.14,26,27 The role of VEGFR-1 on the other hand is more complex and not fully understood. A soluble form of VEGFR-1 can act as a decoy receptor and prevent VEGF-A from binding to VEGFR-2, which, in turn, prevents signaling pathway activation. However, there is also evidence that VEGFR-1 plays an important role in the development of angiogenesis. 28 The third receptor VEGFR-3 is involved in lymphangiogenesis and does not bind to VEGF-A.
The neuropilins (NRP1 and NRP2) are implicated in cell guidance and increased binding of VEGF and its signaling receptors. 15
Several other factors have functions that overlap with VEGF-A, including the PIGF, fibroblast growth factor (FGF), VEGF-C, VEGF-D, angiopoietin, hypoxia-inducible factor (HIF)-1α and HIF-2α, integrin, and platelet-derived growth factor.14,17
The overlap between these factors and VEGF, and the multiple isoforms and splice variant forms of VEGF, makes assessment of individual angiogenesis pathway activation or inhibition outcomes, and thus biomarker discovery, particularly challenging.29,30
Biomarkers of Angiogenesis
Tissue-based biomarkers
Tissue vascular endothelial growth factor
The use of tissue VEGF as a predictive marker has been evaluated in several studies with conflicting results.28–34 Some studies indicate potential value for VEGF in the prediction of prognosis for patients with mCRC. For example, Tsai et al compared pre-and posttreatment VEGF expression by immunohistochemis-try in 57 patients with mCRC who underwent treatment with 5-fluorouracil (5-FU) and irinotecan (FOLFIRI regimen) combined with bevacizumab; results indicated that decreased peritherapeutic, low posttreatment, VEGF expressions were significant predictors of response to therapy and six-month progression-free survival (PFS). 35
On the other hand, Jubb et al evaluated 312 tissue samples from 813 patients enrolled in a phase III trial of irinote-can and 5-FU with or without bevacizumab. Epithelial and stromal VEGF levels, assayed by in situ hybridization, were not found to be predictive of therapy outcomes. 36
These results are confusing and highlight the need for further studies to determine the value of assaying tissue VEGF as a predictive marker.
Genetic polymorphims
At least 12 studies were conducted to evaluate the predictive value of VEGF polymorphisms in patients who were receiving treatment with bevacizumab-based regimens. 37
Formica et al conducted a prospective study to evaluate the predictive value of
To further explore the role of genetic polymorphisms in predicting response to angiogenesis inhibitors, investigators evaluated the role of multigene signatures as a predictive tool. Zhang et al analyzed the expression levels of
Furthermore, Zhang et al assessed the ability of this three-gene signature to predict response to bevacizumab in a cohort of colon cancer patients (
Other genetic polymorphisms are being investigated and include polymorphisms in
Overall this is an intriguing area of research, but the role of genetic polymorphisms in the prediction of disease outcome should be further validated in larger prospective trials.
Circulating biomarkers
Serum vascular endothelial growth factor
Analysis of bio-markers in serum represents an attractive strategy for research studies due to ease of specimen acquisition allowing for serial measurements of any biomarker of interest. Circulating VEGF levels are reported to be relevant in the prediction of outcomes of patients with solid tumors. These levels are thought to reflect VEGF-dependent tumor-mediated angiogenesis. 41 However, the predictive value of baseline VEGF and/or changes in VEGF levels during or after treatment with beva-cizumab remain a matter of debate.
An exploratory analysis by Duda et al evaluated VEGF, PIGF, and soluble VEGFR-1 in plasma from patients receiving bevacizumab combination treatment and suggested that soluble VEGFR-1 may predict response and toxicity to neo-adjuvant bevacizumab-based chemotherapy. 42
The importance of baseline levels of VEGF and soluble VEGFR-2 (sVEGFR-2) as prognostic and predictive bio-markers was evaluated in two phase III studies evaluating the role of cediranib, an experimental angiogenesis inhibitor, in mCRC: the HORIZON II study randomized 860 patients to receive FOLFOX or XELOX with (
Bates et al also evaluated the predictive value of VEGF(165)b, a VEGF splice isoform that binds bevacizumab. High VEGF(165) b appeared to predict resistance to bevacizumab therapy but this observation was not statistically significant, 44 and further work is necessary to ascertain a true link between the two.
Although of interest, the findings highlighted in this section have not been uniformly confirmed. Therefore, this area of research remains a work in progress and further studies are needed to clarify the role of circulating VEGF as a predictive marker.
Circulating tumor cells and free nucleic acid
Circulating tumor cells (CTCs) and free nucleic acid (CTNA) detection in peripheral blood represents an attractive strategy for diagnosis and response assessment in patients with CRC, as well as to predict patient prognosis and therapeutic outcomes (Table 1). 45 The most widely used CTC enumeration platform, Cell Search™ (Veridex LLC), is currently approved by the FDA for clinical use in CRC, breast cancer, and prostate cancer. Investigators have also become increasingly interested in the detection of CTNA (DNA or RNA) to reflect the presence of CTCs. Diehl et al 46 demonstrated that for every 100 g of tumor, 3.3 g of tumor DNA could enter the blood stream. Circulating tumor DNA may give insight into genetic and epigenetic alterations, as well as being useful for diagnosis and prediction of response to therapy. Early evidence suggests that CTC may have both prognostic and predictive values in patients with mCRC. 47 High CTC counts (≥3 CTC/7.5 mL) were associated with worse PFS and OS and were predictive of a worse outcome following all treatment types administered in the study. It is worth noting that ~50% of patients in this study had received bevaci-zumab. Interestingly, Rahbari et al 48 also demonstrated that CTC detection correlated with circulating angiogenic factors and was associated with lower levels of EGF and FGF. This observation suggests that CTCs may prove to be valuable in predicting response to antiangiogenesis agents but is currently hypothesis generating at best.
Clinical utility of circulating tumor cells and tumor nucleic acid.

This technology is promising but continues to have several limitations. The median CTC detection rate is 35% 49 and CellSearch technology requires subjective CTC verification, not permitting single cell analysis. 45 Nevertheless, CTCs represent a promising biomarker for the prediction of treatment response in patients with cancer.
MicroRNA
MicroRNA (miRNA) is a class of small, single-stranded, noncoding RNA that can regulate the expression of multiple genes at the posttranscriptional level. They are thus involved in various cellular functions, including proliferation, apoptosis, regulation of embryonic stem cell development, and cancer cell invasion.
50
Recent studies have shown that miRNAs in the circulation are remarkably stable. This finding allows them to be robust and reliable biomark-ers of cancer therapy.
51
Investigators have demonstrated that miRNAs can modulate tumor angiogenesis through targeting pro-/antiangiogenic factors, including RTK signaling protein, HIF, VEGF, and EGF (Table 2). The involvement of miRNA in tumor angiogenesis has generated interest in exploring their utility as predictive biomarkers. miRNA-126 is one of the most studied miRNAs in mCRC.
50
It is thought to play an important role in the regulation of angiogenesis. High expression of miRNA-126 is associated with increased VEGF-A signaling in endothelial cells and therefore was thought to be a promising biomarker for antiangiogenic therapy. Hansen et al evaluated miRNA-126 as a predictive marker of outcomes in patients enrolled in the NORDIC ACT 1 trial.
52
This phase 3 study evaluated the use of maintenance bevacizumab and erlotinib in patients who had stable disease or a clinical response following six months of oxaliplatin- or irinotecan-based chemotherapy plus bevaci-zumab. High tumor expression of miRNA-126 was significantly related to longer PFS (HR, 0.49; 95% CI, 0.29–0.84;
Examples of miRNAS that regulate various angiogenesis proteins.

Other angiogenesis markers
Kopetz et al investigated the efficacy of FOLFIRI (fluorouracil, leucovorin, and irinotecan) and bevacizumab in 43 patients with previously untreated mCRC. In an attempt to find potential circulating biomarkers of treatment response and therapeutic resistance, this phase II study assessed the levels of 37 different cytokines and circulating angiogenic factors (CAFs) in patient plasma using multi-bead and enzyme-linked immunosorbent assays. Levels were evaluated at baseline, during treatment, and at the time of disease progression (PD).
53
Elevated interleukin 8 (IL-8) levels at baseline were associated with a shorter PFS (11 vs. 15.1 months,
Also in 2010, Goede et al 54 demonstrated that angiopietin-2, a key regulator of vascular remodeling in conjunction with VEGF, is potentially predictive of response rate, PFS, and OS.
Another potential biomarker of bevacizumab treatment outcome is CD133. Pohl et al assayed tumor tissue for germline variations on the 3′UTR-region of the CD133 gene (rs2240688, rs3130, and rs2286455) in 91 patients treated with FOLFOX or FOLFIRI plus bevacizumab. Patients who carried the CC allele in rs2286455 and rs3130 or the combination of the CT with either CT or TT experienced longer PFS (16.5 vs. 8.4 months;
In order to identify serum biomarkers that may predict the clinical outcomes of regorafenib, an analysis of samples obtained from the CORRECT trial was conducted. 56 The study, unfortunately, did not reveal any biomarkers that were predictive of clinical outcomes. In univariate analysis, high concentration of soluble TIE-1 was associated with improved OS, but not PFS, compared with a low concentration. In multivariable analysis, this association was, however, not statistically significant. Biomarkers such as IL-8 and PIGF demonstrated a potential prognostic value but neither marker seemed to play a role in predictive treatment response in patients treated with regorafenib.
Baseline levels of IL-8 and PlGF were found to have prognostic value for OS, although only IL-8 was also prognostic for PFS. Neither factor was predictive of OS or PFS.
In summary, the CORRECT trial biomarker analysis did not reveal any biomarkers that were predictive of clinical outcomes.
There is growing evidence to suggest that baseline and posttreatment circulating endothelial cell (CEC) and endo-thelial progenitor cell number and viability can predict response to antiangiogenic treatment. Willet et al demonstrated a decrease in the blood concentration of viable CECs on day 12 of bevacizumab administration, 57 compared with baseline, suggesting a possible response to treatment.
Also, in another study, a reduced percentage of viable CECs posttreatment significantly correlated with pathologic complete response (
Taken together, these data indicate that select biomarkers appear promising, but their potential utility has to be further evaluated in well-designed prospective studies.
Imaging biomarkers
Recent developments in imaging technologies have led to significant improvements in the management of patients with CRC. Diagnostic techniques, such as diffusion-weighted imaging (DWI), fluorodeoxyglucose positron emission tomography (FDG-PET), and dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI), are transitioning from bench-to-bedside and appear to be useful in providing insights into tumor biology and possibly response to treatment.59–61
Functional imaging allows investigators to map the distribution of tumor mass and surrounding tissues corrected for injected dose and patient weight. FDG-PET provides information on tumor cell viability after treatment, and it has been shown that metabolic changes in response to treatment occur before any structurally detectable change (eg, tumor shrinkage). In the neoadjuvant setting, serial FDG-PET examinations may assist treatment planning. It is thought to be an important marker in rectal cancer due to the fact that sequential FDG-PET after neoadjuvant chemoradiation can predict response to therapy and has been shown to be an independent predictor of DFS and OS.62,63
Whole-body DWI is being explored in mCRC but there is currently no evidence to suggest that this technique can replace PET/CT. 64 DWI has been shown to be feasible as an early marker of treatment response because cell death and vascular alterations typically occur before tumor size changes. 65
Another potential prognostic biomarker in mCRC is the dynamic contrast-enhanced CT (DCE-CT) or perfusion CT. This technology uses quantitative vascular parameters such as blood flow (BF), blood volume, and mean transient time. Goh et al reported that tumor BF was significantly lower in primary tumors of patients who ultimately developed metastatic disease, 66 whereas Hayano et al 67 found that low BF of tumors correlated with poorer outcomes and tumor progression. In terms of the role of DCE-CT as a predictive biomarker for chemoradiation, the data are controversial; Bellomi et al 68 suggested that low perfusion values at baseline were associated with a poorer response, whereas Sahani et al 69 reported the contrary.
Similarly, DCE-MRI is a functional imaging modality that is being investigated as an imaging biomarker. This is an imaging modality that may be useful as a predictive marker in patients receiving angiogenesis inhibitors. 70 It evaluates the extravasation of paramagnetic contrast agents, follows their uptake, and changes in signal intensity over time. Preliminary evidence suggests that contrast-enhanced imaging may be a useful tool to predict patient treatment response to angiogen-esis inhibitors.
Although none of these imaging technologies have entered routine clinical practice yet, they highlight important scientific concepts. It is conceivable that one or more imaging markers will enter clinical practice in the not too distant future.
Conclusion
The poor outcome of mCRC, as well as the widespread use of antiangiogenic agents, has prompted the need for reliable measures to predict response to treatment. Given the complexity of the angiogenesis pathway, the discovery of biomark-ers that predict angiogenesis inhibitor-related outcomes has been challenging.
Circulating and tissue biomarkers show promising potential as predictive biomarkers. Additionally, functional imaging is emerging as a superior tool with to predict response to treatment with antiangiogenic agents. Although several studies have produced encouraging results, definitive data is lacking. Large-scale studies are needed to confirm that promising biomarkers are truly predictive rather than just prognostic in patients with mCRC. In the absence of biomarkers, the decision to treat patients with angiogenesis inhibitors remains a clinical decision based on the perceived balance between the benefit and toxicities of antiangiogenic agents.
Author Contributions
Conceived the concepts: SM and LM. Analyzed the data: SM and LM. Wrote the first draft of the manuscript: SM, MS and LM. Contributed to the writing of the manuscript: SM, MS and LM. Agree with manuscript results and conclusions: SM, LM, MS and MH. Jointly developed the structure and arguments for the paper: SM, LM. Made critical revisions and approved final version: SM, LM and MS. All authors reviewed and approved of the final manuscript. We thank Marion Hartley for her assistance in editing the manuscript.
