Abstract
Our previous study showed that miR-1826 was a newly identified oncogenic non-coding RNA in colorectal cancer. But the potential relationship between miR-1826 and tumor metastasis has not been fully elucidated. The purpose of this study was to evaluate the clinical significance of circulating miR-1826 and its possible associations with circulating tumor cells in colorectal cancer. Our results first found that serum miR-1826 was significantly upregulated in colorectal cancer patients, compared with that in healthy volunteers (p = 0.003). Similar results were also found in colorectal cancer with distant metastasis (p = 0.001) and advanced colorectal cancer (p < 0.001) patients, respectively. Clinicopathological analysis implied that circulating miR-1826 was positively associated with pT stage (p = 0.026), lymphatic metastasis (p = 0.034), distant metastasis (p = 0.012), and tumor–node–metastasis stage (p = 0.020). Besides, our univariate and multivariate analyses demonstrated that high serum miR-1826 expression could act as a prognostic and independent factor for overall survival of colorectal cancer patients (p < 0.05), which led to a poorer 5-year overall survival rate (p = 0.025). The area under the curve value of circulating miR-1826 was up to 0.848 ± 0.043, which strongly suggested serum miR-1826 as an effective diagnostic biomarker in colorectal cancer patients (p < 0.001). Our subsequent experiments demonstrated that patients with high level of circulating tumor cells showed a higher level of miR-1826 expression, compared with the circulating tumor cell–negative patients (p = 0.011). Similar results also showed that the amount of circulating tumor cells in high miR-1826 group was significantly higher than that in low miR-1826 group (p = 0.001). Furthermore, the relationship between serum miR-1826 and circulating tumor cells was analyzed using SPSS software and a significant logarithmic relationship was found, which meant that circulating miR-1826 closely correlated with the amount of circulating tumor cells in colorectal cancer patient serum (r = 0.283, p < 0.01). Our findings strongly suggested that serum miR-1826 could serve as an effective and non-invasive biomarker for diagnosis and prognosis of colorectal cancer. Circulating miR-1826 may be an important target in colorectal cancer therapy.
Introduction
Colorectal cancer (CRC) is the third most common diagnosed cancer in both males and females in the United States, with a total of 142,820 new cases per year. 1 Though molecular diagnostics, gene sequencing, surgical resection, and systematic adjuvant chemoradiotherapy have been successfully applied to the treatment of CRC, the long-term survival of CRC patients is extremely unsatisfactory because of recurrences and distant metastases. 2 There are few highly specific or sensitive tumor markers available for the diagnosis of CRC, especially CRC with the distant metastases (mCRC).3,4
MicroRNAs (miRNAs) are short (21–25 nt) non-coding RNAs which regulate the expression of target mRNAs by binding to the specific regions. 5 Previous studies suggested that miRNAs played crucial roles in multiple biological processes, including regulation of cell proliferation, apoptosis, and differentiation. 6 Aberrant expression of miRNAs was implicated in many diseases, and dysregulations of miRNAs also linked with many types of cancers, such as miR-124 in nasopharyngeal cancer, 7 miR-21 in breast cancer, 8 and miR-92 in gastric cancer. 9 Similarly, lots of miRNAs were proved to be potential biomarkers for early diagnosis, predicting prognosis and individual therapy in CRC patients.3,10 Emerging evidence also focused some specific miRNAs, which could be secreted into blood and called “circulating miRNAs.” These non-invasive miRNAs could be effectively detected by monitoring the blood of CRC patients. 11
Circulating tumor cells (CTCs) are rare tumor cells within the circulation and putative intermediates of metastases. Now, it is clear that CTCs play an important role in cancer metastases, in which they detach from the original tumor, survive in circulation, attach to the endothelium within the target organ, invade the parenchyma, and finally form the tumor mass at secondary sites. 12 For this reason, CTCs can be used to predict the potential of distant metastases and prognosis of cancer patients. 13 Recently, a prospective multicenter study by Cohen et al. 14 confirmed that the number of CTCs before and during treatment could be an independent predictor of progression-free survival (PFS) and overall survival (OS) in patients with mCRC. CTCs provided prognostic information in addition to that of imaging studies. Similar phenomena were also found in some other malignant tumors, including breast 15 and prostate cancers. 16 Thereby, the detection of CTCs could be a potential and effective tool to detect occult mCRC, which escaped form routine imaging methods.
In our previous study, miR-1826 was found to be upregulated in CRC tissues, compared with adjacent non-tumor tissues. High tissue miR-1826 expression might serve as a novel prognostic marker of CRC. Besides, miR-1826 could effectively increase the abilities of cell invasion and migration in human CRC cells, which implied that miR-1826 might participate in the CRC metastasis. 17 This study will determine the circulating miR-1826 expression and CTC level in CRC patients and elucidate the potential correlations between circulating miR-1826 and CTCs. Moreover, the diagnostic and prognostic roles of circulating miR-1826 will also be discussed.
Materials and methods
Clinical specimens
A volume of 10 mL of peripheral blood samples was drawn from 62 preoperative CRC patients (38 males, 63.5 ± 11.1 years old; 24 females, 61.2 ± 11.9 years old) from January 2010 to July 2014 at the Department of General Surgery, the First Affiliated Hospital of Soochow University. All CRC patients had a clear clinicopathological diagnosis based on the tumor–node–metastasis (TNM) staging of the International Union Against Cancer. 18 No treatment modalities for CRC, including chemotherapy, radiation therapy, or immunotherapy were done before surgical operations. As controls, 10 mL of peripheral blood samples was collected from 25 healthy volunteers with no history of cancerous disease; 2.5 mL was for RNA isolation for polymerase chain reaction (PCR) assays. These blood samples were collected in ethylenediaminetetraacetic acid (EDTA) blood bottles, centrifuged at 3000 r/min in 4°C for 10 min within 2 h and stored at −80°C for further experiments. Another 7.5 mL was used for CTCs detections, which was added into CellSave Preservative Tubes (Immunicon Inc., Huntingdon Valley, PA, USA) containing EDTA and optimized cell preservative. We blended the blood upside down gently, which ensured that there were no blood clots. Violent vibration was forbidden which would lead to cell rupture. The blood samples were stored at room temperature and would be detected within 96 h. Written informed consents were obtained from all patients and volunteers. The Ethics Committee of Soochow University approved the study. All CRC patients in this study were followed up to 5 years, and the survival rate was calculated from the date of post-operation to the date of death or last date of follow-up.
Quantitative real-time polymerase chain reaction
According to the manufacturer’s instructions, total RNA was extracted from the 2.5 mL of peripheral blood of CRC patients or controls by TRIzol reagent (Invitrogen, Carlsbad, CA, USA). MiR-1826 and U6 (as an internal control) specific complementary DNA (cDNA) were reversely transcribed from total RNA using the Reverse Transcription System (Promega, Madison, WI, USA). The primer sequences of miR-1826 for quantitative real-time polymerase chain reaction (qRT-PCR) were purchased and synthesized from GenePharma (Shanghai, China). PCR assay was run in a 7900HT Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). SDS2.2.2 software (Applied Biosystems) was used for the comparative ΔCt analysis.
Detection of CTCs
Cell Search System was used to identify CTCs from 7.5 mL of peripheral blood samples from CRC patients. Cell Search System included CellTracks AutoPrep, CellTracks Analyzer II, and matching CellSave Preservative Tubes/reagent kits. The Cell Search Circulating Tumor Cell Kit contained ferrofluid particles which were coated with anti-EpCAM antibodies, phycoerythrin (PE)-conjugated cytokeratin (CK) antibodies recognizing CKs (8, 18, and/or 19), and allophycocyanin-conjugated CD45 antibodies in order to identify white blood cells. The 7.5 mL of blood samples were collected in CellSave Preservative Tubes spiked with 6.5 mL of buffer fluid. After centrifugation, we took the lower cell precipitation into CellTracks AutoPrep System. The system would automatically processed the samples, enriched EpCAM-positive cells by magnetic beads, and stained the nucleus using anti-EpCAM, anti-pan-CK-PE (CK8, 18, and 19), and the DNA stain 4′,6-diamidino-2-phenylindole (DAPI) after cell fixation. The processed samples were automatically transferred to MagNest. Then, lucifugal incubation was performed more than 20 min. After that, these samples were put into Analyzer system for fluorescence scanning analysis and restored to image. The images were presented in a gallery format which classified the CTCs according to Veridex’s predetermined criteria. The CD45−CK+DAPI+ cells, which were confirmed to the criteria of tumor morphology (karyoplasmic ratio greater than 1, cell length greater than 10 µm, uniform texture, and outline complete), were finally considered as CTCs. Most studies used CTCs ⩾3/7.5 mL of blood as a normative cut-off value in CRC, 19 and our study also employed this cut-off value of CTCs for our subsequent analyses. A CTC count <3 was considered negative, while a CTC level ⩾3 was positive.
Statistical analysis
Statistical analyses were performed with SPSS 19.0 software (SPSS Inc, Chicago, IL, USA). Data were expressed as the mean ± SD. Survival curves were performed using the Kaplan–Meier analysis using the log-rank test. Differences between groups were estimated by the chi-squared test, Student’s t-test, and repeated-measures analysis of variance (ANOVA) test; p < 0.05 was considered statistically significant. The receiver operating characteristic (ROC) curves and the area under the ROC curve (AUC) were used to assess the feasibility of using serum miR-1826 concentration as a clinical diagnostic tool for CRC patients.
Results
Circulating miR-1826 was upregulated in CTC-positive patients
First, we detected the serum expressing level of miR-1826 by qRT-PCR. As shown in Figure 1(a), serum miR-1826 expression in CRC patients (89.67 ± 6.064) was significantly upregulated, compared to that in healthy volunteers (negative control (NC) group, 25.99 ± 6.183, p = 0.003). Moreover, the miR-1826 expression in mCRC patients (M+ group, 144.3 ± 8.271) was higher than that in CRC patients without metastases (M0 group, 79.16 ± 6.058, p = 0.001, Figure 1(b)). Similarly, our data also indicated that advanced CRC (ACRC) patients (III/IV stage) presented a relative higher level of serum miR-1826 (119.6 ± 7.857) than that in the early CRC (ECRC) patients (I/II stage, 68.07 ± 6.838, p < 0.001, Figure 1(c)). All these findings implied that circulating miR-1826 might participate in the development and metastases of CRC. Besides, we also determined the serum miR-1826 expressions in CTC-positive and negative patients. Our results confirmed that patients with high level of CTCs (CTCs ⩾ 3, 16.57 ± 2.01) showed a higher level of miR-1826 expression, compared with the CTC-negative patients (CTCs < 3, 14.92 ± 2.61, p = 0.011, Figure 1(d)).

Circulating miR-1826 was upregulated in CTC-positive patients: (a) serum miR-1826 expression in CRC patients; (b) serum miR-1826 expression in mCRC patients; (c) serum miR-1826 expression in ACRC patients; and (d) serum miR-1826 expression in CTC-positive CRC patients (*p < 0.05).
Circulating miR-1826 correlated with CTCs in CRC patients
We divided 62 CRC patients into the low miR-1826 (n = 31) and high miR-1826 groups (n = 31) according to the median of serum miR-1826 expressions in CRC patients. Our results showed that the amount of CTCs in high miR-1826 group (3.903 ± 0.633) was significantly higher than that in low miR-1826 group (1.581 ± 0.226) (p = 0.001, Figure 2(a)). Meanwhile, the amount of CTCs in mCRC patients (6.300 ± 1.375) was also higher than that in non-metastatic CRC patients (2.058 ± 0.263, p < 0.001, Figure 2(b)). Similar results were also found between ACRC group (4.500 ± 0.676) and ECRC group (1.472 ± 0.234, p = 0.002, Figure 2(c)). Furthermore, the relationship between serum miR-1826 and CTCs was analyzed using SPSS software, and a significant logarithmic relationship was found (r = 0.283, p < 0.01, Figure 2(d)), which meant that circulating miR-1826 statistically correlated with the amount of CTCs in CRC patient serum.

Circulating miR-1826 closely correlated with CTCs in CRC patients: (a) the amount of CTCs in high miR-1826 group; (b) the amount of CTCs in mCRC patients; (c) the amount of CTCs in ACRC patients; and (d) a significant logarithmic relationship was found between circulating miR-1826 and the amount of CTCs in CRC patient serum (*p < 0.05).
Correlations of circulating miR-1826 (or CTCs level) with clinicopathological characteristics in CRC patients
As shown in Table 1, the serum miR-1826 expression was positively associated with pT stage (p = 0.026), lymphatic metastasis (p = 0.034), distant metastasis (p = 0.012), and TNM stage (p = 0.020). However, no significant differences were found in other clinicopathological features, including the age, gender, tumor location, tumor size, differentiation, and peritoneal dissemination (all p > 0.05). Meanwhile, we also analyzed the relationships between CTCs level and clinicopathological characteristics in CRC patients. Our data demonstrated that CTCs in serum did not closely link with these factors, such as the age, gender, tumor size, tumor location, differentiation, pT stage, lymphatic metastasis, and peritoneal dissemination of CRC patients (all p > 0.05). However, the number of CTCs in patient serum significantly correlated with distant metastasis (p = 0.014) and TNM stage (p = 0.018, Table 1).
Correlations of miR-1826 or CTC levels with clinicopathological characteristics in CRC patients.

TNM: tumor–node–metastasis; CTCs: circulating tumor cells.
Correlations of circulating miR-1826 (or CTCs level) with the prognosis of CRC patients
In all, 62 CRC patients with radical resections in our study were followed up and the Kaplan–Meier survival curves were performed to predict the correlations between the serum miR-1826 expressions (or CTCs level) and 5-year overall survival (5-year OS) rate. Our data showed that high expression of serum miR-1826 led to a poorer 5-year OS rate than low expressions (p = 0.025, Figure 3(a)). Similarly, positive CTCs also cause a decreased 5-year OS rate in CRC patients than that of CTC-negative patients (p = 0.015, Figure 3(b)). Besides, univariate analysis for OS showed that lymphatic metastasis, distant metastasis, CTCs level, and the expression of serum miR-1826 were all prognostic factors for poor prognosis (p < 0.05). Multivariate analysis also demonstrated that these four features (lymphatic metastasis, distant metastasis, CTCs level, and the expression of serum miR-1826) were retained as independent and significant prognostic factors for OS of CRC patients (p < 0.05, Table 2).
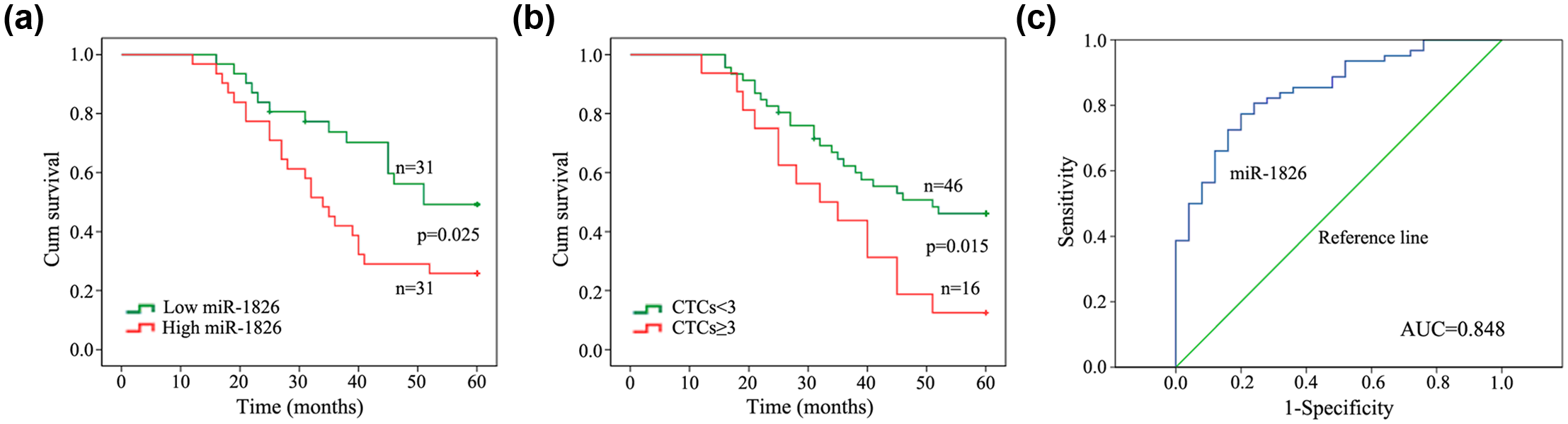
Diagnostic and prognostic efficiencies of circulating miR-1826 in CRC patients: (a) high serum miR-1826 expression led to a poorer 5-year OS rate than low expressions; (b) positive CTCs caused a decreased 5-year OS rate in CRC patients; and (c) the AUC value of circulating miR-1826 was up to 0.848 ± 0.043, which strongly suggested serum miR-1826 as an effective diagnostic biomarker in CRC patients (*p < 0.05).
Univariate and multivariate analyses of overall survival in CRC patients (Cox’s regression model).

RR: relative ratio; CI: confidence interval; CTCs: circulating tumor cells.
p < 0.05.
Diagnostic efficiency of serum miR-1826 in CRC patients
ROC curves were used to evaluate the potential usage of serum miR-1826 as a non-invasive biomarker for diagnosis of CRC. As shown in Figure 3(c), the AUC value of circulating miR-1826 was up to 0.848 ± 0.043 (p < 0.001), and the 95% confidence interval was 0.765–0.932. This finding confirmed circulating miR-1826 as an effective diagnostic biomarker in CRC patients.
Discussion
Circulating miRNAs are a novel class of stable, minimally invasive disease biomarkers, which have been proved to be significantly valuable in diagnosis, prognosis, and treatment response monitoring. 20 In 2008, the significance of circulating miRNAs was first highlighted by Lawrie et al., 21 who explicitly elucidated the relationships between elevated serum miR-21 and a longer relapse-free survival in diffuse large B-cell lymphoma (DLBCL). To date, many circulating miRNA biomarkers have been employed for diagnosis and prognosis of different types of cancers.13,22 For CRC, several related circulating miRNAs, such as miR-223, miR-92a, miR-194, and miR-29b have been reported.23,24 In 2015, our group showed that miR-592 might function as a potential carcinogen-initiated and metastasis-related biomarker in CRC. 25 Recently, similar circulating biomarker, serum miR-372, was also found to be a novel and non-invasive tumor biomarker for ECRC diagnosis. 26 Moreover, using peripheral plasma to determine miRNAs expressing levels was relatively cheap, time saving, and easy to perform.
Tumor tissue miRNAs can be released into the circulation. Though plasma from peripheral blood may not be exactly the same from the microenvironment of tumor, these cell-free miRNAs from tissues to some extent reflect the general characteristics of patients’ tumors. This phenomenon suggests that the serum level of circulating miRNAs may be synchronous with the results in tumor tissues. In 2013, Yong et al. found that dysregulations in circulating blood miRNAs were reflective of those in colorectal tissues. They suggested that the triple miRNA classifier of miR-193a-3p, miR-23a, and miR-338-5p appeared to be a potential blood biomarker for early detection of CRC. 27 Similar results were also found in our previous study in 2015. Our data proved that both serum and tissue miR-592 expression maintained a high level in CRC patients. 25 Recently, our published study demonstrated that the expressing level of miR-1826 was upregulated in CRC tissues compared with noncancerous tissues. And the high level of miR-1826 was closely associated with lymph node metastasis and advanced TNM clinical stage. 17 This led us to suspect the possible role of circulating miR-1826 in CRC. What is the potential relationship between circulating miR-1826 and tumor metastasis? Fortunately, our data in this study confirmed that serum miR-1826 was also significantly upregulated in CRC patients, compared with that in healthy volunteers. Moreover, the miR-1826 expression in mCRC patients was higher than that in CRC patients without metastases. Similar data also indicated that ACRC patients presented a relatively higher level of serum miR-1826 than ECRC patients. Subsequent clinicopathological analysis implied that circulating miR-1826 was positively associated with pT stage, lymphatic metastasis, distant metastasis, and TNM stage. Besides, our univariate and multivariate analyses demonstrated that high serum miR-1826 expression could act as a prognostic and independent factor for OS of CRC patients, which led to a poorer 5-year OS rate. All these findings verified that circulating miR-1826 might closely correlate with the occurrence and development of CRC. In addition to the prognostic role of circulating miR-1826, ROC curves were also used to evaluate the potential usage of serum miR-1826 as a non-invasive biomarker for CRC diagnosis. The AUC value of circulating miR-1826 was up to 0.848 ± 0.043, which strongly suggested serum miR-1826 as an effective diagnostic biomarker in CRC patients. However, little is known about the potential mechanisms of miR-1826 in the blood of CRC.
Tumor cells that have passed the basal membrane are highly mobile and quickly penetrate the vascular endotheliums or lymphatic vessels. This process is called “intravasation,” where these tumor cells circulate as CTCs and may extravasate at secondary sites.28,29 Since their introduction more than 50 years by Engell, CTCs have been evaluated in cancer patients, and their detections significantly correlated with the clinical outcome of many types of solid tumors, including breast cancer, 15 non-small-cell lung cancer, 30 rectal cancer, 31 prostate cancer, 16 and cholangiocarcinoma. 32 Emerging evidence suggested that the prognosis of CRC was closely linked to the occurrence of distant metastases, which putatively develop from CTCs shed into circulation by the tumor. CTC-positive rate seemed to be associated with an unfavorable outcome in metastatic CRC.29,33 In addition, some other studies proved that CTCs, which could provide additional prognostic information to tumor radiographic imaging, might be used as a surrogate and ideal predictive marker for the response to chemotherapy. For example, in 2015, Abdallah et al. 34 demonstrated that CRC patients who had CTC counts presented more thymidylate synthase (TYMS) expression and had tumor progression after treatment with 5-FU. In 2016, they also pointed that patients treated with irinotecan-based chemotherapy and multidrug resistance–associated protein 1 (MRP1)-positive CTCs showed a worse PFS in comparison to those with MRP1-negative CTCs. 35 Mohamed Suhaimi et al. 36 observed that CRC patients with KRAS mutation in their primary tumor had also KRAS mutation in CTCs. Therefore, CTCs played a primary role in tumor recurrences and metastases, and the existence of CTCs could be an independent predictor of overall and PFS in CRC. Our results of this study showed that the amount of CTCs in mCRC patients was significantly higher than that in non-metastatic CRC patients. Similar results were also found between ACRC and ECRC groups. Our data also demonstrated that the number of CTCs in patient serum significantly correlated with distant metastasis and TNM stage in CRC patients. CTCs level, similar to serum miR-1826, was retained as an independent and significant prognostic factor for OS of CRC patients. These results re-confirmed the clinical role of CTCs in CRC. But what was the relation of circulating miR-1826 and CTCs? Our subsequent experiments demonstrated that patients with high level of CTCs showed a higher level of miR-1826 expression, compared with the CTC-negative patients. Similar results also showed that the amount of CTCs in high miR-1826 group was significantly higher than that in low miR-1826 group. Furthermore, the relationship between serum miR-1826 and CTCs was analyzed using SPSS software and a significant logarithmic relationship was found, which meant that circulating miR-1826 closely correlated with the amount of CTCs in CRC patient serum. These findings could help us better understand the possible metastatic mechanisms of circulating miR-1826 via the elevated level of CTCs.
In summary, on the basis of the previous article, this study re-proved the clinical value of miR-1826 in CRC with our reliable and explicated data. Our results showed that serum miR-1826 was significantly upregulated in CRC, especially in mCRC and ACRC patients. The clinicopathological analysis implied that circulating miR-1826 was positively associated with pT stage, lymphatic metastasis, distant metastasis, and TNM stage. High serum miR-1826 expression could act as a prognostic and independent factor for OS of CRC patients, which led to a poorer 5-year OS rate. ROC curves also confirmed miR-1826 as a non-invasive biomarker for diagnosis of CRC. Our subsequent experiments well explained the metastatic significance of miR-1826 via the elevated level of CTCs. These findings strongly suggested that serum miR-1826 could serve as an effective and non-invasive biomarker for diagnosis and prognosis of CRC.
Footnotes
Acknowledgements
Z.X., T.X., and Y.H. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (Nos 81672379 and 81672348).
