Abstract
Incubation of methyl 2-benzamidomethyl-3-oxobutanoate with cultured plant cells of Parthenocissus tricuspidata for 2 days afforded methyl (2R,3S)-2-benzamidomethyl-3-hydroxybutanoate with 100% de and >99% ee in 51% yield. The use of immobilized cells of P. tricuspidata in calcium alginate gel improved the yield of the product (95% yield) with 100% de and >99% ee. The immobilized cells of P. tricuspidata maintained the high potential for the reduction of methyl 2-benzamidomethyl-3-oxobutanoate (85% yield) after 5 times of usage.
Introduction
Chiral 2-benzamidomethyl-3-hydroxybutanoates are useful chiral building blocks for asymmetric synthesis of biologically active compounds; optically active 2-benzamidomethyl-3-hydroxybutanoates are chiral synthons for carbapenems.1,2 Diastereoselective reduction of 2-benzamidomethyl-3-oxobutanoates is an attractive method for the production of optically enriched 2-benzamidomethyl-3-hydroxybutanoates. Recently, microbial reduction of 2-methyl-3-oxobutanoate to give a mixture of syn- and anti- 3-hydroxy-2-methylbutanoate.3–6 However, little attention has been paid to the diastereo- and enantioselectivity in the reduction of 2-benzamidomethyl-3-oxobutanoates by cultured plant cells. Furthermore, there have been no reports on the large scale production of 2-benzamidomethyl-3-hydroxybutanoates by immobilized plant cells. 7 We report herein the high production of methyl (2R,3S)-2-benzamidomethyl-3-hydroxybutanoate by the reduction of methyl 2-benzamidomethyl-3-oxobutanoate with immobilized cells of P. tricuspidata in calcium alginate gel.
Materials and Methods
General
HPLC was carried out with a Deverosil 100-3 column (Nomura Chemical Co. Ltd.) (solvent: hexane/THF/MeOH=1000/100/1; flow rate: 1 ml/min). The 1 H and 13C NMR, H-H COSY, C-H COSY, and HMBC spectra were recorded using a Varian XL-400 spectrometer in CDCl3 solution and the chemical shifts were expressed in δ (ppm) referring to TMS. The FABMS spectra were measured using a JEOL MStation JMS-700 spectrometer.
Methyl 2-benzamidomethyl-3-oxobutanoate and methyl (2R, 3S)-2-benzamidomethyl-3-hydroxybutanoate were purchased from Aldrich or Wako Pure Chemical Co. Ltd.
Cultured P. tricuspidata cells were subcultured at 4-week intervals on solid MS medium containing 2% glucose, 1 ppm 2,4-dichlorophenoxyacetic acid, and 1% agar (adjusted to pH 5.7) under illumination (4000 lux). A suspension culture was started by transferring 20 g of the cultured cells to 300 ml of liquid MS medium in a 500 ml conical flask.
Reduction of methyl 2-benzamidomethyl-3-oxobutanoate(1) by cultured cells of P. tricuspidata
Cultured cells of P. tricuspidata were incubated in 500 ml conical flasks for 3 weeks. To a 500 ml conical flask containing 100 g of suspension cultures, 50 mg of the substrate was added. The mixture was incubated at 25 °C for 2 days on a rotary shaker (70 rpm) under illumination (4000 lux). After the incubation period, products were extracted from the medium with ethyl acetate. The chemical yields of the products were determined by HPLC analyses. The ethyl acetate fraction was subjected to chromatography on silica gel with hexane-ethyl acetate (95:5, v/v) to separate the products. The products were identified by comparisons of their HPLC, FABMS, and NMR data with those of authentic samples. The absolute configurations, diastereomeric excess (de), and enantiomeric excess (ee) of the resulting methyl 2-benzamidomethyl-3-hydroxybutanoate were determined by comparing the HPLC retention times of their (R)-MTPA esters on Deverosil 1003 column with those of the (R)-MTPA esters of authentic chiral alcohols. The retention times of (R)-MTPA esters of (2S,3R)-, (2R,3S)-, (2R,3R)-, and (2S,3S)-isomers in the HPLC were 25.1, 26.4, 31.9, and 38.5 min, respectively.
Spectral data for methyl (2R,3S)-2-benzamido-methyl-3-hydroxybutanoate
Preparation of immobilized cells of P. tricuspidata in calcium alginate gel
Sodium alginate (2%) was suspended in water (1l) by vigorous stirring at 50 °C for 1 h. The cultured cells (100 g) in the stationary growth phase have been used for experiments. Cultured P. tricuspidata cells were added to this solution and the mixture was stirred for additional 2 h until it became homogeneous. The suspension was added dropwise from a dropping funnel with a glass tube into a 10% CaCl2 solution (1l) with stirring to form pieces of spherical calcium alginate gel with 5 mm diameter immediately. Washing with water gave immobilized P. tricuspidata cells which were used for the large scale production of 2-benzamido-methyl-3-hydroxybutanoate.
Large scale synthesis of methyl (2R,3S)-2-benzamidomethyl-3-hydroxybutanoate(2) by immobilized cells of P. tricuspidata in calcium alginate gel
Substrate (50 mg) was added to the immobilized P. tricuspidata cells with 300 ml of MS medium containing 1% glucose in a 11 conical flask, and the flask was incubated for 2 days. Repetitive use of the immobilized P. tricuspidata cells was investigated. The used culture medium was changed to the freshly prepared medium containing 50 mg of substrate and continuous four batch reactions have been carried out at 2 days intervals. Products were extracted from the medium and were purified by the same method as described above. The yields of the product obtained after each batch reactions were 95, 94, 91, 89, and 85%, respectively.
Results
Methyl 2-benzamidomethyl-3-oxobutanoate

Stereoselective reduction of methyl 2-benzamidomethyl-3-oxobutanoate (1) by free and immobilized cells of P. tricuspidata.
The production of
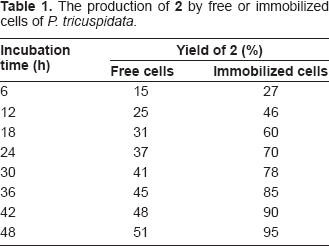
Next, reduction of
Discussion
The biological reduction of methyl 2-benzamidomethyl-3-oxobutanoate
Footnotes
Disclosure
The authors report no conflicts of interest.
