Abstract
To clarify the potential ability of marine actinomycetes as biocatalysts, the stereoselective reduction of α-keto esters and α-keto amide using Salinispora arenicola and Salinispora tropica was tested. The reduction of ethyl pyruvate and ethyl 2-oxobutanoate by S. tropica gave corresponding alcohol with high conversion ratio and in high e.e. (96% e.e. (S) and 99% e.e. (S), respectively). In the presence of l-glutamate as an additive, the reduction of ethyl pyruvate by S. tropica afforded the corresponding (S)-ethyl lactate with >99% e.e. Furthermore, 2-chlorobenzoylformamide was reduced by S. tropica to the corresponding (R)-2-chloromandelamide with high conversion ratio and excellent enantioselectivity (>99% e.e.). Thus, it was found that marine actinomycetes, Salinispora strains, had high ability for the stereoselective reduction of carbonyl compounds as useful biocatalysts.
Introduction
Biotransformation of exogenous substrates have been widely studied and used in order to prepare chiral compounds.1–3 Microbial reduction of carbonyl compounds is one of the convenient and environmental-friendly methods for obtaining optically pure alcohols. For example, bakers’ yeast and fungi have often been used for the reduction to obtain optically active hydroxy esters.4–6 To date, several studies concerning the reduction of keto esters and their analogues with other microorganisms such as actinomycete and algae have been reported.7–10 However, little information is known about the reduction of keto esters using marine bacteria as biocatalysts.
Recently, marine actinomycetes were isolated, characterized, and named Salinispora arenicola and Salinispora tropica. 11 These Salinispora strains are attracting attention because useful bioactive compounds such as arenimycin (an antibiotic against methicillin-resistant Staphylococcus aureus) and salinisporamide A (a potent proteasome inhibitor) are produced by these marine actinomycetes.12,13 Thus, marine actinomycete was a species of the microorganisms expected in medicine and pharmacy field. However, the potential ability and possibility of Salinispora strains as biocatalysts for asymmetric organic synthesis has not been investigated.
This study describes the stereoselective reduction of α-keto esters and aromatic α-keto amide by marine actinomycetes, Salinispora strains, as novel biocatalysts.
Material and Methods
Instruments and chemicals
Gas chromatography was done using GL Science GC-353 (DB-Wax, J&W Scientific, USA, 0.25mm×30m;TC-1,GLScience, Japan,0.25mm x30m; CP-Chirasil-DEX CB, Chrompack, Netherlands, 0.25 mm x 25 m; Gamma DEX 225, Supelco, USA, 0.25 mm x 30 m) gas chromatographs. Ethyl pyruvate (
Microorganism and Cultivation
Salinispora arenicola NBRC105043 and Salinispora tropica NBRC105044 were purchased from the National Institute of Technology and Evaluation, Biological Resource Center, Japan (NBRC). These strains were maintained at 25 °C in the following synthetic medium solidified 1.5% agar. Medium (NBRC recommended medium 325) is comprised of 10 g polypepton, 2 g yeast extract, 0.5 g MgSO4 · 7H2O, 27 g Daigo's artificial seawater SP, per one liter of distilled water (pH 7.3). The Salinispora strains were grown in each maintained medium (500 ml) for seven days at 25 °C with aerobic shaking in baffled 2-liter flasks in dark condition. The marine actinomycete cells were harvested by filtration on a filter paper in vacuo and washed with saline (0.85% NaCl aq.).
Reduction of α-keto esters and α-keto amide with marine actinomycete whole cells
The saline-washed cell (0.5 g, corresponding 0.2 g of dry weight) was resuspended in a large test tube (φ 30 mm × 200 mm) containing 20 ml of saline, and then the substrate (0.15 mmol, corresponding substrate concentration is 7.5 mM) and additive (5.0 mmol) were added and incubated aerobically at 25 °C. A portion of the reaction mixture was filtered using a diatomaceous earth short column and extracted with ether, and then concentrated under reduced pressure.
Repetitive use of immobilized marine actinomycete cells
The Ca2+-alginate immobilized S. tropica cells (IMST) were prepared by according to the procedures in the literature.
16
Saline (20 mL), the additive (monosodium l-glutamate) and 2-chlo-robenzoylformamide (
Analysis
The conversions of products (
Results and Discussion
Reducing ability for α-keto esters
Two Salinospora strains were tested for reducing abilities toward α-keto esters (see Fig. 1). The results of the reduction of α-keto esters (
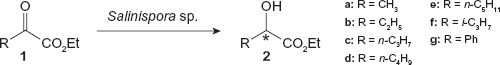
The reduction of α-keto esters (1a-g) by Salinispora strain.
The reduction of α-keto esters (1) by S. arenicola or S. tropica. a
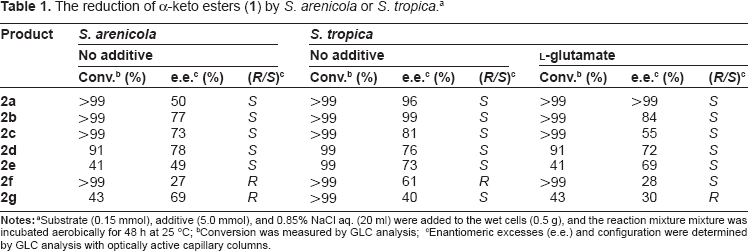
Conversion was measured by GLC analysis;
Enantiomeric excesses (e.e.) and configuration were determined by GLC analysis with optically active capillary columns.
S. arenicola reduced α-keto esters have short alkyl chains (
The reduction of ethyl pyruvate (
Reducing ability for aromatic α-keto amide
Two Salinospora strains were tested for reducing abilities toward aromatic α-keto amide (see Fig. 2). The results of the reduction of 2-chlorobenzoyl-formamide (

The reduction of 2-cholorbenzoylformamide (3) by Salinispora strain.
The reduction of 2-cholorbenzoylformamide (3) by S. arenicola or S. tropica. a
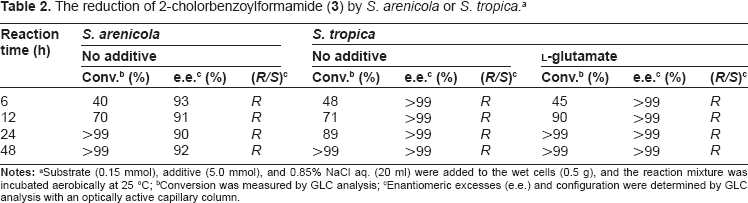
Conversion was measured by GLC analysis;
Enantiomeric excesses (e.e.) and configuration were determined by GLC analysis with an optically active capillary column.
Reduction of aromatic α-keto amide with immobilized Salinispora cells
Furthermore, the productivity of the reduction, the batch reduction of
The repetitive batch reduction of 3 to 4 by Ca2+-alginate immobilized cells of S. tropica. a

Conversion was measured by GLC analysis;
Enantiomeric excesses (e.e.) and configuration were determined by GLC analysis with an optically active capillary column.
Conclusion
We demonstrated the stereoselective reduction of α-keto esters and aromatic α-keto amide to the corresponding alcohols with marine actinomycetes. It was found that Salinispora strains were a useful tool for the preparation of chiral α-hydroxy esters and (R)-2-chloromandelamide. To gain insight into the mechanistic interpretation of the marine actinomycete reduction, further detailed studies including purification of the reductase(s), which contribute to the reduction of α-keto esters and aromatic α-keto amide, are currently under investigation.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgement
We wish to express our appreciation to Associate Prof. T. Yamamoto, Department of Applied Chemistry, Faculty of Engineering, Okayama University of Science, for his useful advice on this study.
