Abstract
The developing retina is an excellent model to study cellular fate determination and differentiation in the context of a complex tissue. Over the last decade, many basic principles and key genes that underlie these processes have been experimentally identified. In this review, we construct network models to summarize known gene interactions that underlie determination and fundamentally affect differentiation of each retinal cell type. These networks can act as a scaffold to assemble subsequent discoveries. In addition, these summary networks provide a rational segue to systems biology approaches necessary to understand the many events leading to appropriate cellular determination and differentiation in the developing retina and other complex tissues.
Introduction
Multicellular organisms are made of tissues with multiple specialized cell types. Understanding the determination and differentiation of heterogeneous cell types within the context of complex tissues is fundamental to many areas of biology. This knowledge will have widespread application in treatment of developmental disorders and disease states such as cancer and will be critical for successful bioengineering and transplantation of tissue types to replace damaged or degenerate structures. The determination and differentiation of a given cell within a tissue is the culmination of the expression of many gene products and their subsequent intra- and intercellular signaling events. To address the challenge of understanding cell fate determination and differentiation we must adopt a broad systems biology approach to adequately take into account the activities of large numbers of genes and signaling pathways.
One emerging systems-based strategy to analyze and integrate large datasets is to generate network models, in which genes or proteins are represented by nodes and their relationships by edges in the graph (network). However, most large expression datasets are too sparse to infer high statistical confidence gene relationships which are based on the estimate of a covariance matrix. 1 In addition, the networks generated de novo are often large, and do not facilitate prioritization of candidate genes and gene relationships for hypothesis based validation. To address this problem, we have previously described a heuristic approach that uses a seed network to summarize prior knowledge of a small part of the gene network involved in cellular development.2,3 The seed network can then be used to query large datasets in order to identify additional molecules with putative relationships to seed genes. These candidate molecules can then be used to expand the network and are the basis for generating testable hypotheses to validate their functional role.
Cell fate determination and differentiation in the vertebrate retina provides many opportunities to generate and utilize systems-based tools and approaches to understand development of cells within complex tissues. First, development of the retina is well-characterized4–6 and the sequence of cell genesis and differentiation is well-documented and largely conserved among vertebrates.7–11 Thus, activity of gene networks that underlie the fate determination and differentiation in a particular retinal cell type will take place in known cells with known birthdates and known locations within the tissue. Second, the retina is highly accessible and is very amenable to in vivo hypothesis testing, 12 thus the role of hypothesized gene candidates and network interactions in cell fate determination and differentiation can be readily assessed. Third, we can build on the foundational system-based approaches developed through the study of single cell organisms like yeast, 13 diffuse systems like the immune system, 14 or cultured tissue systems, 15 and extend these methods to examine the development of more complex tissues that comprise living organisms.
Here we review what is presently known about the genetic networks that underlie cell fate determination and differentiation in the developing retina and present the seed networks that we have constructed based on our examination of published literature. The developing retina is an extensively reviewed16–20 system regarding cell fate determination during retinogenesis, but a summary of literature-curated gene networks underlying differentiation of each retinal cell type has not been previously presented. In order to demonstrate its potential as a model to study determination and differentiation of multiple cell types within the context of a complex tissue, we have assembled seed networks to summarize what is known about the genes and their relationships that underlie cell fate determination and largely influence the differentiation of each of the basic retinal cell types. Finally, we demonstrate that the experimentally-based summary network for photoreceptors can be extracted from an independent gene expression data set.
Retinal Cell Types
The mature mouse retina is composed of seven basic cell types, six neuronal and one glial (Fig. 1). While this review focuses on only the differentiation of the basic cell types, many retinal cells can be further subdivided morphologically, biochemically and functionally.21––31 Photoreceptors (rods and cones) reside in the outer nuclear layer (ONL) and are responsible for phototransduction and necessary for vision. 32 Photoreceptors synapse with bipolar cells, neurons that reside in the inner nuclear layer (INL). Bipolar cells relay visual stimulus to retinal ganglion cells in the ganglion cell layer either directly or indirectly via amacrine cells, which also reside in the INL. Other cells present in the INL are horizontal cells, which mediate lateral interactions between photoreceptors and Müller glia that play a critical role in retinal homeostasis. 33 Axons of the retinal ganglion cells project into the visual centers in the brain, thereby relaying the visual information detected by the retina. While appropriate processing of visual stimuli requires the function of all retinal cell types, most blinding retinal diseases are the result of the degeneration of photoreceptors or ganglion cells.34,35 Interestingly, the seven cell types that comprise the retina are derived from a common pool of retinal progenitor cells. 17 Thus, the developing retina provides a relatively simple, yet elegant system to study the generation and maturation of a complex tissue. We know that the cell fate decisions made by retinal progenitor cells are governed by an intrinsic genetic program that determines their response to extrinsic cues from their environment.16,17 The sequence of retinal cell genesis is highly conserved in vertebrates,8,11,36––39following a general progression of retinal ganglion cells (RGCs), horizontal cells (HCs), cone photoreceptors followed by amacrine cells (ACs), and subsequently bipolar cells (BCs), rod photoreceptors and Müller glial cells (MCs) (Fig. 2). Based on this general progression of birth order, retinal cell types can be divided into cohorts of early-born cells which include ganglion cells and cone photoreceptors, and late-born cells which include rod photoreceptors, bipolar cells and Müller glia. 8

The retinal cell types in the adult mouse retina. The adult mouse retina is comprised of three cellular layers separated by two synaptic layers. Rod and cone photoreceptors reside in the outer nuclear layer (ONL), and form synaptic contacts in the outer plexiform layer (OPL) with horizontal cells and bipolar cells, both of which reside in the inner nuclear layer (INL). In addition, amacrine cells and the cell bodies of Müller glia are found in the INL. Synaptic contacts between bipolar cells, amacrine cells and ganglion cells are present in the inner plexiform layer (IPL) and ganglion cells reside in the innermost cellular layer, the ganglion cell layer.
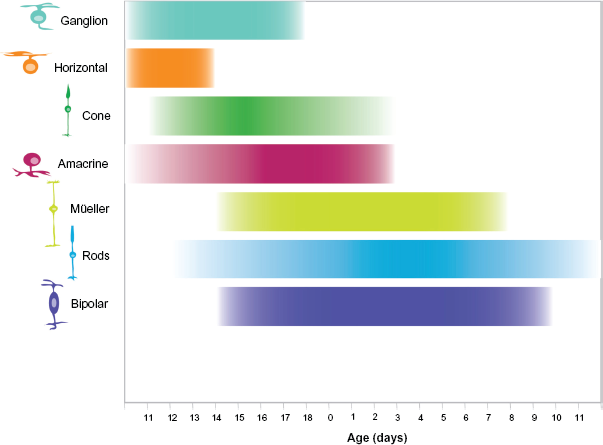
Time course of cell genesis in the developing mouse retina. Retinal cell types are listed on the Y-axis, developmental time on the X-axis. Birth of the animal is indicated as 0, embryonic development is left of 0, postnatal development to the right. The approximate time course of cell genesis is indicated by the bar adjacent each cell type. This figure is based on the work reported by Young. 8
Gene Families that Underlie the Specification of Retinal Cell Types
There are a number of genes that are well known to act in the specification of and/or largely influence the differentiation of retinal cells. They compose a regulatory network that can integrate extrinsic information through signaling pathways like Notch, as well as implement intrinsic programming via transcription factors, many of which can be grouped into the basic helix loop helix (bHLH) gene family and the homeobox gene family.
The family of basic helix loop helix (bHLH) genes is characterized by an α helix-loop-α helix structural motif. The bHLH genes Mash1, Math3, NeuroD, Math5 and Ngn2 cross-regulate each other to specify neuronal types in developing retina,19,40 while Ptf1a,41,42 Bhlhb4 43 and Bhlhb5 44 have roles in the development of more specific retinal cell types or subtypes. Other family members such as Hes1 and Hes5 work as effectors of Notch signaling. These bHLH genes interact with members of homeobox gene subfamilies including Pax, CVC, POU, Lim and Sox. The Pax gene subfamily has critical roles in embryogenesis 45 and Pax6 functions as an early regulatory gene in the development of eye. 46 In contrast, the CVC homeodomain subfamily members Vsx1 and Chx10 have more specific roles in retinogenesis across vertebrate species like mouse,47,48 chicken 49 and fish.50,51 The POU homeodomain subfamily members have a variety of functions related to neural development, 52 and genes Brn3b, Brn3c 53 and Brn3a 54 are all involved in mouse retinal development. The LIM homeodomain gene subfamily are involved in neural patterning 55 with Isll and Lim1 playing crucial roles in retinal development. The Sox subfamily genes 56 are indispensible in many aspects of development including neurogenesis, and Sox8, Sox9 are implicated in retinogenesis. Like the bHLH genes, some homeobox genes like Vsx1,57,58 Barhl2 59 and Irx5 60 appear to specify retinal cell subtypes. Together these genes (see Supplementary Information) work in concert to specify cell fate in the developing retina.
Using a scaffold of bHLH and homeobox gene family members, we developed a seed network to summarize key gene relationships that govern the development of each of the retinal cell types in mouse retina. These seed networks are based on published studies that have demonstrated a role for the seed genes in the determination and differentiation of retinal cell types via either loss of function experiments41,61––68 gain of function experiments69,70 or transcriptional regulation experiments.71,72 Genes involved in the specification of multiple retinal cell types (see Supplementary Information) are not always included, in a given seed network due to the lack of strong evidence they interact with other essential genes in the seed network specifying a particular cell type. These seed networks can be used in two complimentary ways: 1) to design database queries to identify additional key molecules for cell-specific development, 2) to assemble a comprehensive summary of known gene relationships and identify key decision points in cell-specific specification that may be important regulatory targets for future application.
Müller Glial cells
The gene relationships that underlie Müller glia determination and differentiation are summarized in the seed network in Figure 3. Müller glia are the only glial cells to arise from the retinal progenitor cell population. Thus, the factors which influence the progenitor cell choice between gliogenesis and neurogensis are critical for the creation of these cells. Previous work has demonstrated that Notch signaling plays a major role in the choice between neural and glial cell fate. 73 Notch is a transmembrane receptor that functions at the cell surface to both receive extracellular signals and to regulate gene expression in the nucleus. Notch signaling is widely used to control developmental processes in many animal species. 74 In the developing retina, the Notch pathway is implicated in the control of progenitor cell proliferation and apoptosis, as well as the multipotency of progenitor cells. 75 In addition to its role in maintaining the undifferentiated and proliferative state of retinal progenitor cells (RPCs), Notch also seems to regulate the neuronal versus glial cell fate choice by inhibiting the photoreceptor cell fate in mouse retina.76,77

A network of genes essential for Müller glia development. Edges in this graph are based on evidence that Rax promotes notch1 and Hes1 transcription, 73 Notch signaling positively regulates expression of hes1, hes5, 78 sox8 and sox9,71,80 and Hes1 suppresses the proneuronal gene Mash1.79,162 Blue edges between genes indicate activation, while red edges indicate repression.
As essential effectors of Notch signaling, 78 bHLH genes Hes1 and Hes5 have partly overlapping but distinct roles in Müller cell determination and differentiation. Both Hes1 and Hes5 are thought to repress expression of neuronal bHLH genes. 19 However, their specific target genes appear to be different since Hes1 maintains the progenitors and inhibits both neuronal and glial differentiation, whereas Hes5 cooperatively regulates maintenance of progenitors but promotes the glial cell fate.61,62 Specifically, Hes1 is known to inhibit the proneuronal gene Mash1 79 and thus promotes glial cell determination. Consistent with their different effects, both Hes1 and Hes5 are expressed in undifferentiated cells while Hes5 is also expressed in differentiating Müller glial cells.
The homeobox gene, Rax, promotes the glial cell fate choice, potentially via activation of promoters of Notch1 and Hes1. 73 The homeobox genes Sox8 and Sox9 have also been implicated in the specification of Müller glial cells,71,80 though neither of them alone is sufficient to induce Müller glial cell differentiation. Notch signaling regulates Sox8 and Sox9 transcription, though it does not appear to be through its activation of Hes1 and Hes5. 71
Retinal Ganglion Cells
The gene relationships that underlie retinal ganglion cell determination and differentiation are summarized in the seed network in Figure 4. The bHLH gene Math5 plays a critical role in retinal ganglion cell (RGC) development. The targeted deletion of Math5 results in the loss of more than 80% of RGCs, 64 and a cell fate shift to other retinal cell types.64,81,82 It seems that Math5 underlies RGC differentiation in two ways. First, Math5 activates a downstream transcriptional network that controls ganglion cell differentiation and development.83,84 Second, Math5 suppresses other bHLH proneuronal genes such as Math3, NeuroD and Ngn2 that are involved in the adoption of other retinal cell fates.82,84 The available evidence suggests that Math5 is directly regulated by Pax6.85,86 Downstream of Math5, Brn3b and lsl-1 are known to play critical roles in ganglion cell differentiation.64,87,88 Brn3b, a POU subfamily gene, while not required for the initial commitment of RGC fate, is essential for early retinal ganglion cell differentiation.63,89 Homozygous disruption of Brn3b leads to a selective loss of 70% RGCs 63 suggesting not all RGC differentiation is dependent on Brn3b. 90 Consistent with this, it is hypothesized that Brn3b regulates genes important for formation of RGC axons and axon path-finding.89,91 In addition to loss of Brn3b, deletion of the Lim family gene lsl-1 also causes a marked reduction in the number of ganglion cells. 65 Recent studies indicate that both lsl-1 and Brn3b regulate genes such as Eomes and Shh.87,88 Eomes is a T-box transcription factor, now known as a direct target of Brn3b and required for RGCs and optic nerve development. 92 Other Brn3b-related genes are also found to contribute to ganglion cell development. For example, the zinc finger protein Wt1, acts upstream of Brn3b, and plays a role in the development of RGCs.93,94 Barhl2 functions downstream of Brn3b to regulate the maturation and survival of RGCs. 59 Math5 and Brn3b are essential for ganglion cell determination. In addition here are other Brn3b dependent-genes, 72 Math5-dependent genes, 84 and genes identified in RGC single cell expression studies. 95 However, the relationships of these genes to the network described here are not yet understood and were not included in our seed network.
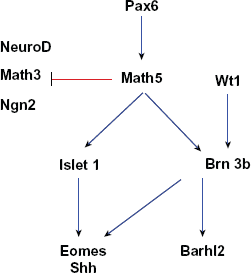
A network of genes essential for ganglion cell development. Edges in this graph are based on evidence that Pax6 actives Math5 expression 163 and Math5 suppresses Math3 and NeuroD to promote ganglion cell fate.82,84 In addition, Math5 promotes Brn3b and Isletl expression,64,87,88 which in turn positively regulate genes like Eomes, 92 Shh 164 and Barhl2. 59 Brn3b is also activated by wt1. 94 Blue edges between genes indicate activation, while red edges indicate repression.
Bipolar Cells
Compared to other retinal cell types, data supporting the relationships among genes essential for bipolar cells specification and differentiation are relatively sparse; however, the genes with key regulatory roles in bipolar cells determination and differentiation are summarized in Figure 5. The bHLH gene Mash 1 plays a pivotal role in bipolar cell differentiation. In both rat and mouse, the onset of Mash1 expression (about E15) most closely correlates with the appearance of bipolar cells and Müller glia.96,97 In Mash1 –/– retinal explants, the differentiation of all late born retinal cells (bipolar cells, rod photoreceptors and Müller glia) was delayed, and the number of the mature bipolar cells was significantly reduced, though the number of vimentin-positive cells (likely Müller glial cells) was increased. 98 Additionally, Mash1 is expressed by a subset (10%–30%, depending on age) of the total proliferating progenitor cells, providing a molecular marker of heterogeneity among retinal progenitor cells (RPCs). 97 Together, this evidence suggests that Mash1 plays a role in the commitment and/or differentiation of late born retinal cells, particularly bipolar cells.

A network of genes essential for bipolar cell development. The edges in this graph are based on evidence that Otx2 may affect the competence of progenitor cells to adopt a bipolar vs. rod photoreceptor cell fate, 103 that Chx10 is hypothesized to work downstream of Otx2 to promote bipolar cell fate, 102 and that Chx10, together with Mash1 and Math3, specify bipolar cell fate.67,100 Dotted edges indicate indirect or poorly characterized gene relationships.
Mash1 and Math3 are co-expressed in various regions of CNS suggesting these genes may have some functional redundancy.
67
Interestingly, the
The expression of the homeobox gene Chx10 is also integral to bipolar cell fate. Chx10 is restricted to the inner nuclear layer (INL) in the mature retina, though in the developing mouse eye, the Chx10 transcript is confined to the anterior optic vesicle and all neuroblasts of the optic cup. 101 Loss of Chx10 results in reduced proliferation of retinal progenitors and a specific absence of differentiated bipolar cells. 68 Misexpression of Chx10 induces generation of inner nuclear layer cells, 100 while misexpression of Mash1 or Math3 together with Chx10 increases the number of mature bipolar cells while decreasing the mature Müller glial cell number. 100 Thus, it is proposed that Chx10 confers the specific inner nuclear layer identity to retinal neurons while bHLH genes such as Mash1 and Math3 subsequently specify the bipolar cell fate. 100 In addition, Chx10 promotes bipolar cell fate determination by inhibiting photoreceptor specification, presumably by acting downstream of Otx2 or other Otx genes. 102 Otx2 subcellular localization is hypothesized to play a role in the rod versus bipolar cell fate choice. 103 In the retina of a postnatal, bipolar-cell-specific-Otx2 conditional knockout mouse the expression of mature bipolar cell markers is significantly down-regulated, 104 demonstrating its importance in bipolar cell differentiation.
Amacrine Cells
The gene relationships that underlie amacrine cell determination and differentiation are summarized in the seed network in Figure 6. For amacrine cell specification, the bHLH gene Math3 cooperates with another bHLH gene, NeuroD, and amacrine cells are completely missing in Math3-NeuroD double mutant retinas. The cells in the double knockout retinas that fail to differentiate into amacrine cells adopt both ganglion and Müller glial cell fates. However, while these genes are necessary for amacrine cell fate determination, they are not sufficient; misexpression of either Math3 or NeuroD alone cannot induce amacrine cell genesis. 69
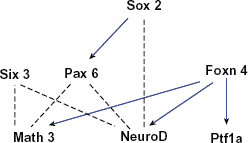
A network of genes essential for amacrine cell development. The edges in this graph are based on evidence that Sox2 activates Pax6 and NeuroD to promote amacrine cell fate,70,85 that Pax6 and Six3, with the cooperation of Math3 and NeuroD, specify amacrine cell fate, 69 and that Foxn4 positively regulates Ptf1a, Math3 and NeuroD expression.41,42,66 Blue edges between genes indicate activation while dotted edges indicate indirect or poorly characterized relationships between genes.
In the Pax6-knockout mouse retina, the retinal progenitor cells become totally restricted to an amacrine cell fate. 85 While misexpression of Pax6, Math3 or NeuroD alone does not induce amacrine genesis, the misexpression of a combination of bHLH genes Math3 or NeuroD with homeobox genes Pax6 or Six3 (the transcription of which is independent of Pax6) 105 does promote amacrine cell genesis. 69 Furthermore, misexpression of Pax6 with only Math3 results in the production of amacrine cells and horizontal cells, while the combination of Pax6 and NeuroD predominantly increases only the number of amacrine cells, suggesting that when expressed with Pax6, NeuroD is more specific for amacrine cell differentiation than Math3. 69 The homeobox gene, Sox2, is expressed in a subset of amacrine cells and misexpression of Sox2 results in a dramatic increase of amacrine cells in INL. Experimental evidence indicates that Sox2 transcriptionally induces Pax6 and may also induce NeuroD. 70 Taking all these data into account, it appears that Sox2 functions upstream of Pax6 and NeuroD to affect/promote amacrine cell fate.
The expression of the forkhead gene family member Foxn4 in mouse retina correlates closely with the birth date of amacrine cells and misexpression of Foxn4 promotes amacrine cell genesis. 66 Further, Foxn4-null mice exhibit a significant decrease in amacrine cells and a complete loss of horizontal cells. 66 The effect of Foxn4 on amacrine cell differentiation may be via activation upstream of NeuroD and Math3 signaling, since in Foxn4–/– retinas there is a marked down-regulation of NeuroD and Math3 with no observable alteration in Math5, Ngn2, Chx10 or Pax6 expression. 66 Downstream of Foxn4 is Ptf1a. 106 Lineage tracing reveals that Ptf1a expression in the developing mouse retina marks the horizontal and amacrine cell precursors. 42 Loss of Ptf1a affects the differentiation of a small population of amacrine cells and the entire population of horizontal cells. While Foxn4 may influence amacrine cell differentiation via NeuroD and Math3, Ptf1a does not appear to work in this way as in the Ptf1a-null retina expression of the two genes was unaffected.41,42
Horizontal Cells
The gene relationships that underlie horizontal cell determination and differentiation are summarized in the seed network in Figure 7. It appears that amacrine (Fig. 6) and horizontal (Fig. 7) cell fates are linked as they share several key regulatory genes including Foxn4, Ptf1a, Math3, and Pax6 (Fig. 6 and 7). As previously mentioned, misexpression of Pax6 with Math3 results in an increase of both horizontal cells and amacrine cells, though the effect on horizontal cell genesis is greater (14% increase) than the effect on amacrine cell genesis (7% increase). 69 At the same time, deletion of Foxn4 results in complete loss of horizontal cells, presumably via the downregulation of Math3. 66
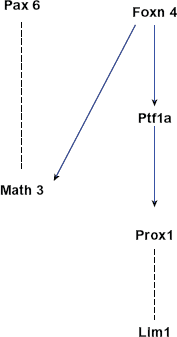
A network of genes essential for horizontal cell development. The edges in this graph are based on evidence that Foxn4 positively regulates Math3 and Ptf1a expression, 66 that coexpression of Pax6 and Math3 promotes horizontal cell fate, 69 and that Ptf1a positively regulates Prox1 expression41,66,107 which in turn affects Lim1 expression. 106
Prox1, the Prospero-related homeobox 1, is also important for horizontal cell differentiation. Prox1 is expressed in, and is required for efficient cell cycle exit for, early RPCs (but not in late RPCs). 107 Prox1-null retinas exhibit a complete loss of horizontal cells and the misexpression of Prox1 results in the production of horizontal cells.107,108 Considering the fact that there is a lack of Prox1 expression in Foxn4-null retina and a downregulation of Prox1 in Ptf1a-null retina,41,66 Prox1 seems to promote horizontal cell fate by acting downstream of the Foxn4-Ptf1a axis. Downstream of Foxn4-Ptf1a-Prox1 is another essential gene, Lim1. 106 Lim1 is required for specific morphogenesis of horizontal cells in chick retina. 109 In mouse retina, Lim1 is essential to instruct the differentiation and migration of horizontal cells to the correct laminar position.110,111
Cone and Rod photoreceptors
Both cones and rods employ phototransduction, a process that captures and converts photons of light to an electrical signal; however, each cell type expresses a particular visual protein (opsin) to absorb a specific portion of the light spectrum. In mice, cones express either a S-opsin (short wavelength sensitive) or a M-opsin (middle wavelength sensitive) while rods express rhodopsin. Interestingly, both rod and cone photoreceptors share several key genes essential for cell fate specification and differentiation. Thus, the relationships of genes underlying the differentiation of cones and rods are shown together in a single network (Fig. 8).
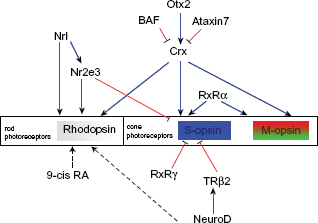
A network of genes essential for rod and cone photoreceptor cell development.
NeuroD is the only bHLH gene known to be essential for photoreceptor differentiation. NeuroD is expressed in developing photoreceptors and is maintained in a subset of mature photoreceptors in the adult mouse retina.112,113 In the NeuroD-null retina, the number of rods is reduced, while the number of the bipolar cells is increased in a dose-dependent fashion 112 Misexpression of NeuroD not only blocks gliogenesis, but also favors rod photoreceptor differentiation while reducing bipolar cell differenation. 112 NeuroD is also necessary for sustained expression of TRβ2, an essential gene for cone photoreceptor development. 113
Photoreceptor cell types are generated by common activity of genes like Crx (Cone rod homeobox), Nrl (neural retina leucine zipper), and Nr2e3. For example, Crx is expressed early in the developing retina, and is predominantly expressed in photoreceptors in mature retina. 114 Crx transactivates the Rhodopsin promoter and acts synergistically with Nrl to drive rhodopsin expression in rods. 114 Crx also activates cone opsins.115,116 Two genes are known to suppress Crx function Ataxin-7117,118 and BAF, 119 both contribute to photoreceptor degenerative disease. Otx2, a member of Otx homeobox gene family, transactivates Crx 120 and misexpression of Otx2 directs retinal progenitor cells towards photoreceptor fate. 120
Nrl is a basic motif-leucine zipper transcription factor preferentially expressed in rod photoreceptors,121,122 which positively regulates rhodopsin.123,124 In the Nrl–/– mouse retina, cone-like photoreceptor cells are clearly different from WT rods and cones, revealing a functional transformation from rods to S-cones.125,126 From these results, it is inferred that Nrl modulates rod-specific genes as well as inhibits S-cone differentiation through the activation of Nr2e3.126,127 Nr2e3 expression is restricted to photoreceptor cells. It is a ligand-dependent transcription factor that requires itself for the repression of its own transcription.128,129 Mutation of Nr2e3 causes enhanced S cone syndrome (ESCS), 130 a retinal degenerative disease in humans that results in an abundance of short-wavelength sensitively cones (S cones) at the expense of rod photoreceptors. 131 It is hypothesized that when photoreceptors are first generated the defective Nr2e3 cannot prevent a ‘default’ shift of rod progenitors to an S-cone fate, producing a large number of S-cones and an absence of rods. 132 This is supported by the fact that Nr2e3 acts as a repressor of cone-specific genes in rods, 133 and directly interacts with Crx to enhance rhodopsin and repress cone opsins. 134
In addition to upstream genes including Otx2, Crx, Nrl and Nr2e3 as well as photoreceptor-specific genes like rhodopsin, S-opsin and M-opsin, retinoid receptors are indispensible for appropriate photoreceptor differentiation. Retinoid receptors belong to a steroid receptor superfamily of proteins that serve as ligand-dependent transcription factors. Retinoic acid (RA) plays its role in transcription through retinoic acid receptors (RARs) and retinoid X receptors (RXRs). 9-cis RA binds to and transactivates both RXRs and RARs. 135 In addition, 9-cis RA directs progenitor cells to the rod cell fate through activation of members of the steroid/thyroid superfamily of receptors. 136 Another effector of this family, thyroid hormone (TH), is found to induce progenitor cells to differentiate into cones in embryonic rat retinal cultures. 137 Many effects of TH are mediated by TH receptors (TRs). 138 The most important TR in retina development is TRPβ. TRPβ is expressed in the outer nuclear layer of the embryonic retina.139,140 The mouse retina has an opposing S-cone (greater expression ventrally) and M-cone (expressed more dorsally) distribution. Deletion of TRβ2 in mice causes the selective loss of M-cones and a concomitant increase in S-opsin immunoreactive cones, disturbing the gradient of an opposing S- (ventral) and M-cone (dorsal) distribution. 141 TH is also required to inhibit S-opsin and activate M-opsin expression. 142 Other studies confirm that thyroid hormone action is required for normal cone opsin expression during mouse retinal development.143,144 RXRγ cooperates with TRβ2 to suppress S-opsin in all immature cones and in dorsal cones of the mature retina though it is not necessary for M-opsin regulation. 145 Finally, RXRα acts in synergy with Crx to activate many cone-specific genes. 146
Identification of Experimentally-Determined Gene Relationships in a High Throughput Gene Expression Dataset
The gene relationships in the seed networks described above are supported by experimental evidence and thus have been validated in the narrow sense by identifying direct or indirect interactions between two genes under particular experimental conditions. The next step to identify the ‘system’ of genes that work together to influence cell-specific determination and differentiation will require the use of large gene expression datasets and potentially additional dataset types such as protein-protein interaction datasets, ChIP-chip datasets, datasets from animals with specific mutations, etc. We have previously demonstrated the successful application of literature-derived seed-networks to query high-throughput gene expression datasets.2,3 One motivation for this review article was to assemble the available experimental evidence in a way that it might be readily applied to future studies of other cell types, and perhaps to even guide the experimental design processes that underlie the generation of new datasets.
An implicit assumption when using large gene expression datasets is that legitimate gene relationships will be discoverable by identifying a correlation of expression between them. An important question, then is, are
Using previously published data collected from developing rod photoreceptors isolated from the retina at E16, P0, P2, P6 and P10, 147 we calculated the correlation coefficients between all pairs of genes (edges) present in the seed network (Table 1). In the photoreceptor seed-network, there were 13 genes and 17 edges (relationships) between them. Two genes (BAF and 9-cis-RA) were not present in the dataset, which left 15 edges to identify. Seven of the 15 edges were recognized as high correlation coefficients (> |0.85|) and an additional three of the 15 edges were supported with weaker correlation coefficients (> |0.45|).
Pairwise correlation coefficients between genes of the photoreceptor-specific seed network. Pearson correlation coefficients were calculated based on the developmental gene expression in rod photoreceptors isolated from retina at ages E16, P2, P6 and P10. 147 Two genes, BAF and 9-cis RA were not present in the expression dataset and therefore no correlation coefficient could be calculated (NO DATA). Seed network is shown in Figure 8.

Thus, two-thirds of the seed-network relationships are present in the dataset and nearly half of the seed-network relationships are strongly correlated. Encouragingly, our result suggests that a significant number of legitimate gene relationships can be discovered using gene expression data. Previously, we have used seed networks to discover new candidate genes by focusing on genes that were correlated with multiple seed-network genes.2,3 Ultimately, it appears that it will require a combination of datasets and approaches to describe the entire gene network that underlies cell fate determination and differentiation.
Summary
The seed networks presented here can be the basis for queries of high throughput datasets to identify larger, more comprehensive networks that participate in cellular fate specification and differentiation in the developing mouse retina. In addition to summarizing prior knowledge of these processes, seed networks can also be the basis for comparative studies between tissue types within a species or between diverged organisms in order to identify genetic pathways that are conserved through development and evolution.3,148–150 while a more generalized gene-by-gene comparative approach has been effective in identifying orthologs that may play a role in a complex process or a disease state in different organisms,151–154 it is the conservation of not only the gene, but of its relationships to other genes in a network, that dramatically increases the likelihood that the gene, in fact, functions in similar way. Being able to include relational data is one advantage of the seed network approach over more generalized comparative studies. The effectiveness of a cross-species seed network approach has been demonstrated elsewhere.3,149
These seed networks were constructed to help demonstrate the potential of the developing vertebrate retina as a model system for the development and evaluation of systems based approaches. In addition to its characteristic organization and developmental time course, there is a significant amount of high throughput data that has been collected from the developing retina,155–159 and single cells from the developing retina.95,147,160,161 Because of its characteristic organization during development, candidate molecules that are generated using systems based approaches can be rapidly, albeit cursorily, evaluated based on in situ spatial and temporal expression.3,160 Finally, due to its accessibility, candidates can be functionally evaluated in developing retinas using in vivo electroporation to either drive overexpression or knockdown expression of candidate molecules. 161
Networks and network representation of processes have an important role in the implementation of systems based approaches and the analysis of large datasets and complex processes. Demonstrating the ability of these seed networks to effectively focus the generation of hypotheses from high throughput data sets would significantly advance the discoveries that depend upon this type of data. In addition, we have also demonstrated that seed networks are an effective way to do comparative analysis of retinal development and use knowledge of one model system to drive discovery in another. 3 The use of seed networks to identify conserved networks that act in similar ways (as opposed to conserved genes) will be tremendously useful in the extrapolation of discovery in one model system to another. Thus, development of systems based approaches to investigate cell fate determination in the developing mouse retina will not only lead to important discoveries in the developing retina, but strategies that can be broadly generalized to address many biological questions.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material
