Abstract
Many patients with sulfur mustard (SM) exposure present dyspnea in exertion while they have a normal pulmonary function test (PFT) and imaging. The cardiopulmonary exercise test (CPET) has been used for evaluation of dyspnea in exertion among patients with different pulmonary disorders focusing on assessing gas exchange. We evaluated subjects who were exposed to SM with normal imaging compared to the controls with CPET. A case-control study was carried out on two groups in Tehran, Iran during 2010 to compare the CPET findings. The cases with a history of SM exposure and complaint of exertional dyspnea while they had normal physical examination, chest X-ray, PFT, and nonsignificant air trapping in lung high resolution computed tomography (HRCT) were included. A group of sex- and age-matched healthy people were considered as controls. One hundred fifty-nine male patients (aged 37 ± 4.3 years) were enrolled as a case group and ten healthy subjects (aged 35 ± 5.9 years) as the control group. There was no significant difference in the demographic and baseline PFT characters between the two groups (
Inhalation of sulfur mustard (SM) causes injury of the respiratory system and long-term pulmonary complications. High-dose exposure results in death due to respiratory failure, secondary pneumonia, and occasional hemorrhagic pulmonary edema, especially in the acute phase;[1] however, low-dose exposure could cause insidious onset of exertional dyspnea after many years.[2] Imaging and pathological studies indicate that individuals with SM exposure have suffered by either bronchiolitis obliterans (BO) or bronchiolitis obliterans organizing pneumonia (BOOP).[2,3] Ghanei et al.[2] demonstrated that “exposure to SM was responsible for the occurrence of the BO syndrome observed in exposed patients.”
While BO was indicated as one of the most frequent pulmonary sequels of SM exposure, a chronic lung disorder named mustard lung (ML) was too abstruse to understand clearly. Many clinical signs and symptoms were not consistent with the extent of the injury in ML. Therefore, various mechanisms such as oxidant-antioxidant imbalance, chronic inflammatory process, and genomic mutations were recommended to justify the pathophysiology of the disorder, but none of them were definite.[2,3]
Many clinical manifestations cannot be explained using the previously-mentioned mechanisms. Due to involvement of small bronchioles, it is expected that these patients may be affected by a restricted cardiopulmonary function. While low-dose exposure to SM often presents with mild dyspnea, the constantly-exposed patients did not, in fact, express significant clinical symptoms.
On the other hand, the cardiopulmonary exercise test (CPET) with measuring gas exchange has been used for evaluation of dyspnea on exertion in patients with different pulmonary disorders.[4] There are many disorders that mimic ML and a diagnostic test can be helpful for them;[5] furthermore, understanding the diagnostic protocols helps the entire field in proper and rapid diagnosed.
We have seen many cases with low-dose exposure to SM who have complained of exertional dyspnea while completing normal pulmonary function tests (PFT) and normal full expiratory chest high resolution computed tomography (HRCT). To our knowledge, this is the first study about the role CPET plays on evaluation of low-dose SM exposed cases.
MATERIALS AND METHODS
Study design and participants
During 2010, a case-control study was carried out in Tehran, Iran (barometric pressure is 670 mmHg) to compare the CPET results between two groups: (1) Cases who were exposed to SM 25 years ago and complained of exertional dyspnea with normal general physical examination, normal chest X-ray, PFT, and nonsignificant air trapping in pulmonary HRCT (regarding acute symptoms, all cases have typical symptoms of exposure such as dermatitis, dyspnea, acute bronchitis, and conjunctivitis after exposure that was healed with treatment); and (2) The control group included sex- and age-matched healthy people which were asymptomatic and had a normal physical examination, chest X-ray, and PFT. Written informed consent was taken from subjects (case and control) who had enrolled for this study. This study was approved by the Ethics Committee of Chemical Injuries Research Center at Baqiyatallah University of Medical Sciences.
One hundred fifty-nine patients were recruited in our case group with the following inclusion criteria: (1) A well-documented SM exposure by military health services; (2) no history of medical disorders which can affect the CPET results; (3) a complaint of dyspnea on exertion; (4) normal chest X-ray and PFT; and (5) near normal or nonsignificant findings in full expiratory chest HRCT. Low-dose exposure in this study was defined as “exposure to SM that did not cause acute respiratory symptoms at the time of exposure.” Our subjects were selected among all those who were in chemically contaminated areas with SM and had been registered for an annual checkup.
Ten male individuals in the control group were selected with no history of SM exposure or other confounding medical disorders. Volunteers in the control group have not had dust exposure, history of smoking, history of recent respiratory infection or disease, drug uses, or other chronic diseases. Both groups were similarly active.
CPET
The incremental exercise test was performed according to the protocol of Wasserman on an electrically-braked cycle ergometer (SensorMedics 2900). After a three-minute rest and a three-minute period of unloaded pedaling at a cycling speed of 60 rpm, the load was progressively increased by 15 W/min. Cardiopulmonary data was collected breath by breath, metabolic unit (CPX) over the entire period allowing for the measurement of heart rate (HR), ventilation (VE), VO2, VCO2, and determination of maximal heart rate (HR max), peak VE, peak VO2, peak VCO2, and peak oxygen pulse (VO2/HR). Arterial oxygen saturation for hemoglobin was measured by pulse oximeter. Subjects performed exercise test as safely as possible and did not stop as long as they felt breathless.
Predicted HR max was calculated as 220 – age in years × 0.65. Maximal ventilatory capacity (MVC) was estimated as forced expiratory volume in the first second (FEV1) × 40.[6] The dyspnea index, expressed in percent, was calculated by dividing the minute ventilation by maximal voluntary ventilation (VE/MVV). Ventilatory reserve was defined as the difference in liters between MVV and VE. The anaerobic threshold (AT) was determined by the V-slope method as described by Beaver et al.[7]
Calibration of gas analyzers before each test was achieved by the use of a tank of standard gas provided by the manufacturer (SensorMedics). VE was expressed in BTPS, while VO2 and VCO2 were expressed in STPD. All airflow and gas measurements were corrected for ambient temperature, barometric pressure, and water vapor, and are expressed in BTPS units.
HRCT evaluation
HRCT examinations were obtained on one scanner (HiSpeed Advantage, General Electric Medical Systems, Milwaukee, Wisc.). Each HRCT examination consisted of five 1.0-mm collimation images obtained during both deep inspiration and full expiration, respectively, with the patient lying in a supine position. Noncontrast images were obtained at the levels of the aortic arch, midway between the aortic arch and tracheal carina, at the tracheal carina, midway between the tracheal carina and the right hemidiaphragm, and 1 cm above the right hemidiaphragm. All images were reconstructed using a high spatial resolution algorithm and displayed at standard (level 700; width 1,500) and narrow (level 700; width 1,000) lung window settings. The HRCT scans were reviewed by two radiologists and two pulmonologists. A quick read form was developed by the group's consensus for ease and uniformity of reporting. The only data available to the HRCT reviewers were patient's age, sex, and history of exposure to SM. The interobserver agreement (kappa coefficients) for air trapping, bronchiectasis, and mosaic parenchymal attenuation were 0.76 (95% CI: 0.58–0.94), 0.84 (95% CI: 0.69–0.99), and 0.68 (95% CI: 0.48–0.88), respectively. Inspiratory images were read before expiratory ones and images displayed at standard windows before narrow window settings.
Expiratory images were displayed at standard and narrow window settings. These were directly compared to determine differences in the conspicuity of air trapping. The criteria used to diagnose the presence of air trapping were alteration of normal anterioposterior lobar attenuation gradients and/or lack of homogeneous increase in lung attenuation resulting in persistent areas of decreased attenuation.[8]
Statistical analysis
SPSS version 16 was used for the data analysis. PFT and CPET data were expressed as mean values with one standard deviation (±SD) above and below the mean. The continuous variables for both tests were compared using student's
RESULTS
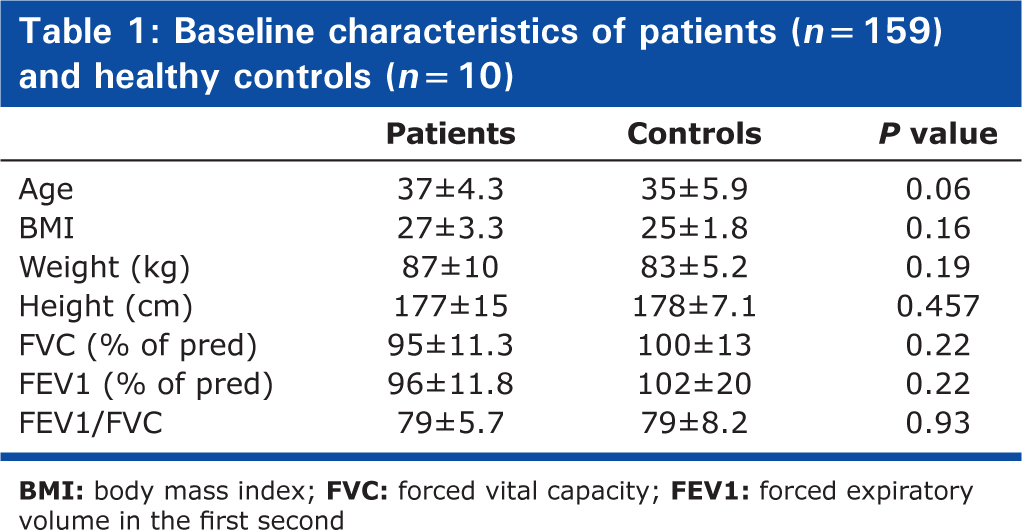
One hundred fifty-nine patients (aged 37 ± 4.3 years) in the case group and ten participants (aged 35 ± 5.9 years) in the control group were evaluated. All cases and controls were male. This study was conducted at the Baqiyatallah University of Medical Sciences, Tehran, a referral center for chemical warfare injuries. In all cases, at least 20 years had passed after dealing with mustard gas, and all were exposed to SM once without any protective devices. All cases had initial symptoms of exposure such as dermatitis, dyspnea, acute bronchitis, and conjunctivitis, which were healed with treatment. All 159 cases were included among more than 530 cases that have been referred to this medical center. Demographic and PFT data of cases and healthy controls are presented in Table 1. There is no significant difference in the demographic and baseline PFT characters between the two groups.
Baseline characteristics of patients (
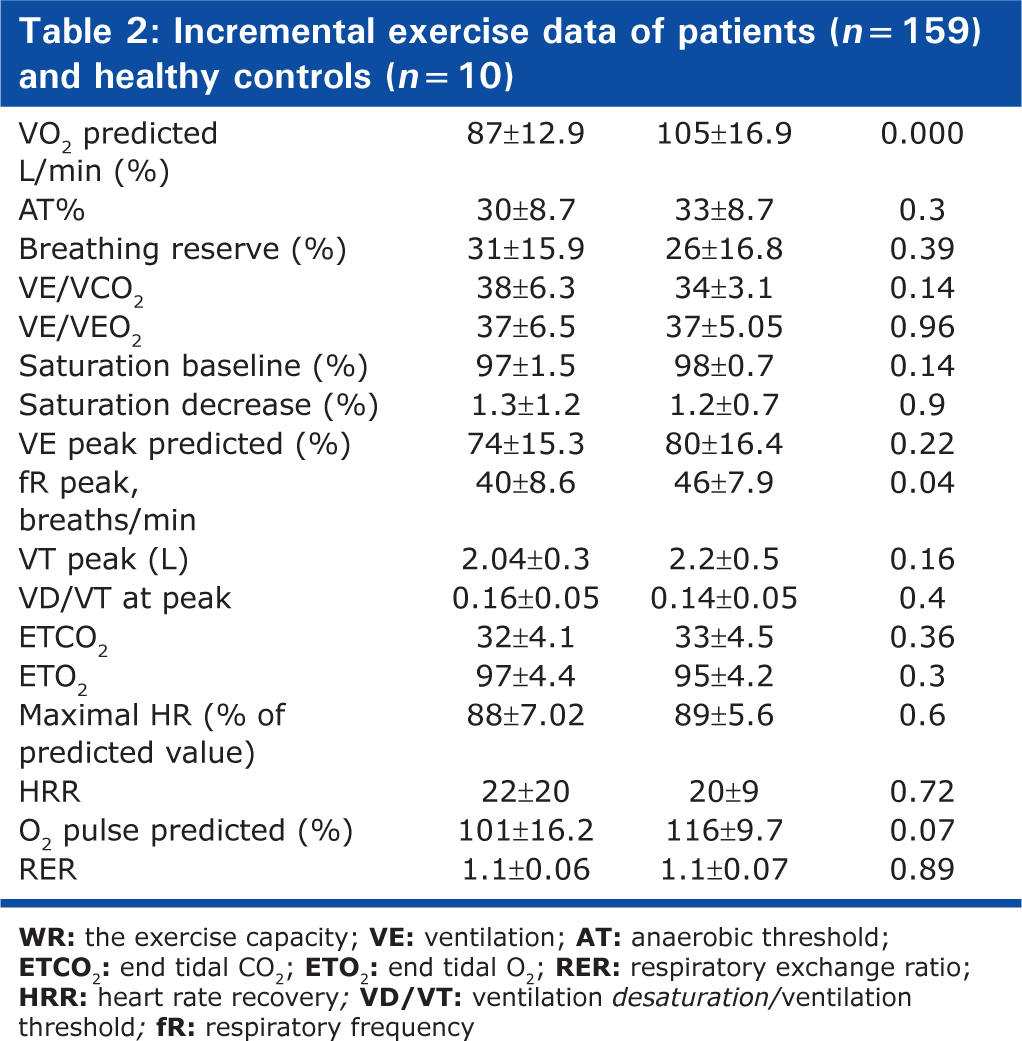
Regarding the CPET findings expressed in Table 2, only WR max, Peak VO2/kg, VO2 predicted, and RR peak were statistically different between cases and controls (
Incremental exercise data of patients (
DISCUSSION
The main findings of this study are as follows: (1) The VO2 (peak and predicted) reduced in SM exposed groups in comparison with control groups; and (2) Respiratory and cardiac response to exercise did not differ significantly in both groups. The measurement of VO2 max or VO2 peak remains the best available index for the assessment of exercise capacity. Peak oxygen consumption (VO2 peak) should be measured directly because its estimation from resting indices, work rate, or exercise protocols is unreliable.[9] VO2 peak could be expressed in absolute values (L/minute) and as predicted percent, or ideally for fat-free mass (mL/kg/minute).[10]
Peak VO2 was reduced in these cases, in both value expressions: absolute and fat free mass. A reduced VO2 peak is the starting point in the evaluation of reduced exercise capacity. Also, a reduced O2 peak response to exercise reflects problems with oxygen delivery (heart, lung, systemic and pulmonary circulation, and blood) and/or peripheral abnormalities (i.e., reduced oxygen use or muscle dysfunction).[11–14] A reduced O2 peak also may reflect poor effort.
The essential step for interpretation of CPET is evaluation of patient effort. Heart rate reserve (HRR), breathing reserve (BR), and respiratory exchange ratio (RER) were normal and indicated that poor effort was not a reason for low VO2.
Cardiac response to exercise in these cases was normally observed. These findings were attained by normal maximum heart rate, heart rate reserve and O2 pulse, and they showed that cardiac problem did not lead to low VO2. The mean age of our cases (37 years old) and lack of atherosclerosis risk factors corroborate this result of CPET.
Ventilatory response to exercise was evaluated by ventilatory capacity, breathing reserve, breathing pattern, and ventilatory equivalent for VCO2 and VO2, end tidal CO2 and O2, and pulmonary gas exchange. All of ventilatory indices were normal in these cases. Ventilatory equivalent for VCO2 (VE/VCO2) did not differ significantly in both groups, while absolute value was higher than normal in the exposed cases. VE/VCO2 is usually less than 32–34 at or near the AT and less than 36 (rarely 40) at the peak of exercise in normal subjects.[15] VE/VCO2 at AT was used as a noninvasive method for measuring ventilation perfusion (V/Q) mismatch;[6] furthermore, patients with chronic obstructive lung disease, restrictive lung disease, left ventricular failure, and pulmonary vascular diseases usually have V/Q mismatch.
Regarding our previous studies, we confirmed that many exposed cases are suffering from small airway pathology in spite of nonsignificant findings in laboratory data,[3] but in this study we wanted to assess the pure effect of SM in patients regardless of considerable pulmonary consequences because CPET results can be affected by pulmonary disorders such as bronchiolitis. High VE/VCO2 at AT in our cases could be a marker for V/Q mismatch and abnormal gas exchange. The major limitation of this study is lack of noninvasive assessment of uneven V/Q.
Abnormal changes in P (alveolar-arterial) O2 and P (arterial-end tidal) CO2 are the most sensitive markers of lung diseases. Airway diseases are ruled out with normal values since these measurements are almost always abnormal in these disorders.[4] Calculation of VD/VT according to PETCO2 will underestimate the severity of gas exchange found in patients having elevated VD/VT, and will overestimate VD/VT as seen in patients with normal gas exchange.[17]
Despite the fact that abnormal gas exchange may be present in our cases, it cannot be a good explanation for low VO2 in CPET. Also, impaired cell O2 consumption could be a hypothesis for low VO2 in these cases. Some animal studies investigated the impact of SM exposure on cell NAD+ storage and it was declared that NAD+ depletion and glycolysis inhibition occurred in acute exposure; however, no study has been observed on these abnormalities in late complications of SM poisoning.[18–20] Also, given that there were no apparent differences of the pulmonary parenchyma consequences, SM can affect the peripheral musculature and the ability to utilize O2 (resulting in a decreased VO2 max). Accordingly, Fedeles et al. conducted an investigation on oxygen consumption capacity in tissue exposed with an antitumor agent 11b as mustard derivate. They finally concluded that “11b inhibited oxygen consumption both in intact HeLa cells (a cancerous cell) and in isolated mitochondria.”[21] They additionally stated, “11β blocked uncoupled oxygen consumption when mitochondria were incubated with complex I substrates, but it had no effect on oxygen consumption driven by substrates acting downstream of complex I in the mitochondrial electron transport chain.”[21] This finding supports our mentioned hypothesis about inhibitory effects of SM on cellular O2 metabolism. On the other hand, Fedeles et al. hypothesized that this inhibitory effect of SM on oxygen metabolism may be due to enhanced ROS generation and antioxidant imbalance that is concordant with the previous findings about oxidative-antioxidant imbalance in these patients.[21]
The post-test RR peak was significantly higher than in controls. Increase in RR may be due to increased demand of insufficient supply. It seems that among SM exposed patients who have normal imaging and also normal PFT, the suspected mechanism was increased in demand because there are evidences about sufficient oxygen supply observed in this study (such as proper O2 saturation in CPET in SM exposed cases versus controls).[22] Also, regarding our findings about WR max and VO2 predicted and max, it seems that cell oxygen consumption in exposed cases was lower than in the healthy cases.
In conclusion, although CPET was recommended for symptomatic cases with normal imaging, apparently CPET cannot be considered as a useful method for discrimination of small airway lesion in symptomatic low-dose SM exposed cases.
Footnotes
ACKNOWLEDGMENTS
The authors would like to state their warm gratitude for the participants of this study.
