Abstract
Urachal adenocarcinoma is a rare neoplasm of the bladder. Most of these tumors arise from urachal remnants in the dome of the bladder and extend into the umbilicus. These tumors can recur and most commonly metastasize to lymph nodes, retroperitoneum, lungs, liver and bone. Here we report a case of an urachal adenocarcinoma followed for seven years with lung metastasis for three years.
Introduction
Urachal adenocarcinoma is a rare neoplasm of the bladder. Most of these tumors arise from urachal remnants in the dome of the bladder and extend into the umbilicus. These tumors can recur and most commonly metastasize to lymph nodes, retroperitoneum, lungs, liver and bone. Here we report a case of an urachal adenocarcinoma followed for seven years with lung metastasis for three years.
Case Report
A 37-year old man, non-smoker, underwent a partial cystectomy with resection of urachus and umbilicus in 2002 for a mucinous urachal adenocarcinoma. Thoracic and abdominal computed tomography (CT) scan showed no abnormalities. Four years later, he was referred for multiple lung nodules (in the middle lobe, the right upper lobe, and left lower lobe) without any other visceral lesion (Figure 1A). There was no deterioration in performance status, and no symptom. Bronchoscopy showed no anomaly and no histological diagnosis was obtained. There was no evidence of infectious disease. A surgical removal of the right upper lobe nodule was performed by axillary thoracotomy. Pathological findings confirmed metastasis of the urachal adenocarcinoma. The patient was treated with chemotherapy consisting of three courses on Day 1 and Day 14 of 5-fluorouracil 400 mg/m2 as intravenous (i.v.) bolus followed by 2400 mg as i.v. continuous injection over 46 h and irinotecan 90 mg/m2. Tolerance was acceptable, despite a grade 2 neutropenia. CT scan showed lung nodules had almost disappeared. Four months later, the disease progressed with increasing size of the previously described lung nodules. Three cycles of chemotherapy with the same drugs were administered, with an acceptable clinical tolerance; due to a persistant neutropenia, chemotherapy was administered on Day 1 and Day 21 instead of Day 1 and Day 14 and the patient had to receive granulocyte colony-stimulating factor (G-CSF). There was then a small regression of the nodules, with persistent activity on PET scan on the right lower lobe nodule. A surgical approach was decided, and a metastasectomy (in the right upper and lower lobes) was performed. Two months later, a CT scan showed two new nodules, one on the left upper lobe and one on the left lower lobe (Figure 1B) with a high metabolic activity on PET scan. The same chemotherapy was administered to the patient. After four cycles, the disease was stable, with decreased activity on PET scan. A surgical removal of the metastases was performed, with a wedge resection of the anterior part of the left basal pyramid and metastasectomy in the anterior part of the left upper lobe. During follow up, six months later, another PET scan showed a new nodule in the right lower lobe and a new mass on the right upper lobe, with parietal involvement (Figure 1C); a CT-guided transparietal biopsy of this mass confirmed the recurrence of the urachal carcinoma. Chemotherapy was administered, consisting of three courses on Day 1 and Day 14 of 5-fluorouracil 400 mg/m2 as i.v. bolus followed by 1200 mg as i.v. continuous injection over 46 h, irinotecan 90 mg/m2 and bevacizumab 5 mg/kg. Tolerance was fair with mild vomiting and no changes in hematologic parameters. The disease was stable. Three courses of chemotherapy with an association of capecitabine 2000 mg/m2/day from Day 1 to Day 14 and oxaliplatin 130 mg/m2 on Day 1and Day 21, with grade 3 nausea and vomiting, were administrated. There was a slight decrease in the metabolism intensity of the metastases on PET scan. Surgery (right pneumonectomy with rib resection) was proposed to the patient. The patient died during surgery because of hemorrhagic shock due to a pulmonary artery wound, in June 2009, seven years after the diagnosis of urachal carcinoma, and three years after appearance of metastases.
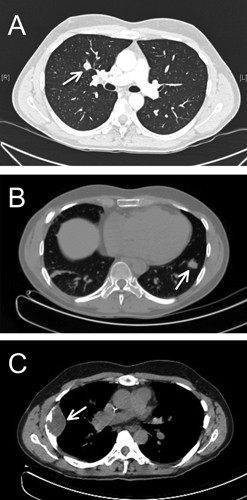
Computed tomography scan (A) upon diagnosis of pulmonary metastasis showing the right upper lobe nodule (arrow), (B) upon first relapse showing the left lower lobe nodule (arrow), (C) upon diagnosis of the last relapse showing the right upper lobe mass (arrow).
Discussion and Conclusions
Urachal adenocarcinoma is a rare tumor, accounting for less than 1% of bladder tumors. 1 It is derived from the urachus, a remnant of the allantoid canal. There is no typical symptom leading to the diagnosis, but the most reported are hematuria, pelvic pain, dysuria, pelvic mass, mucosuria and urinary infection. 2 Age at diagnosis is in the late fifties with a little over 1:1 male:female ratio (Table 1).2–8 Diagnosis usually requires cystoscopic biopsy. Diagnostic criteria include: i) tumor located in the dome or elsewhere in the midline of the bladder; ii) sharp demarcation between tumor and normal surface epithelium; iii) and then supportive criteria as an enteric type histology, the absence of urothelial dysplasia, the absence of cystitis cystica or cystitis glandularis, the absence of primary adenocarcinoma of another organ. 9 Local recurrence is frequent after surgery. Distant metastases usually occur late in the natural history of the disease. Urachal carcinoma is associated with a poor prognosis (Table 1).2–8 Identified prognostic factors are tumor stage, lymph node involvement, and positive surgical margins.4,6,7 As far as stage is concerned, the extension to adjacent visceral organs and the presence of distant metastases are correlated to poor survival. We report a prolonged survival after the diagnosis of lung metastases, which are the most frequent visceral metastases of urachal carcinoma, involving 40-50% of metastatic patients (Table 1).2–8 Molina et al. 6 reported a 10.8 year median survival for stage 1/2 disease, and 1.3 year for stage 3/4; 10 patients received chemotherapy with a median survival of 20.4 months. Siefker-Radtke reported a median survival of 24 months at the time metastasis was diagnosed. 4
Summary of large series of urachal adenocarcinoma.

There is no standard treatment for urachal carcinoma. Partial cystectomy with en bloc resection of the urachus and umbilicus seems to be the treatment of choice for localized disease; 4 there is no difference on local recurrence and survival between partial and total cystectomy. 4 The role of neo-adjuvant chemotherapy has not been defined nor is there a standard chemotherapy regimen for metastatic disease. Most of the reports of chemotherapy treatments are single case reports and have been recently summarized. 10 Chemotherapy, such as used in the treatment of urological cancers, have been largely unsuccessfully. There was no response after cisplatinum plus gemcitabine in one case, 10 and after MVAC (methotrexate, vinblastine, adriamycin, ciplatin) in 5 cases. 4 Chemotherapies have been used for digestive metastatic adenocarcinoma. Combination using fluorouracil ± ciplatinum seems to obtain best response: 3 partial response and 6 stable disease were described in 9 patients with a combination including 5FU+cisplatin, 4 and 3 responses were obtained in 4 patients treated with ifosfamide, 5-fluorouracil, VP16 and cisplatin. 11 The Germ-FLP trial is the only ongoing phase II trial for metastatic urachal carcinoma, evaluating gemcitabine, fluorouracil, leucovorin and cisplatin association (NCT00082706).
We report on a case of an urachal carcinoma with lung metastases treated with a combination of chemotherapy and surgical resection leading to a prolonged survival. Surgery should be considered after a good response to chemotherapy. Kawakami et al. described 2 cases of metastatic adenocarcinoma with more than 10-year survival, using repeated resection and chemotherapy for one patient, and chemo-radiotherapy for lung metastases for the other. 12 Krane et al. reported interesting results with cytoreductive surgery with hyperthermic intraperitoneal chemotherapy in patients with peritoneal carcinomatosis secondary to an urachal carcinoma. 13
The death of the patient after a peroperative vascular wound may reflect the controversial increase in surgical complications after treatment with bevacizumab. 14 Currently, the most important aspect of treatment is the timing of surgery. 15 Despite this, aggressive treatment of metastatic urachal adenocarcinoma, as recently reported, should be advocated. 16
Footnotes
Acknowledgment
Jean-Paul Gamondes and Xavier Martin provided and cared for the patient.
