Abstract
The aberrant generation or activation of T follicular helper (Tfh) cells contributes to the pathogenesis of systemic lupus erythematosus (SLE), yet little is known about how these cells are regulated. In this study, we demonstrated that the frequency of Tfh cells was increased in lupus-prone B6.MRL-
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a prototypic autoimmune disease characterized by the production of autoantibodies and multiorgan damage1,2. Although the pathogenesis is still unclear, much attention has recently focused on the role of T follicular helper (Tfh) cells in this disease3,4.
With the discovery of B-cell lymphoma-6 (Bcl-6) as the essential transcription factor in 2009, Tfh cells are considered to be a novel subtype of CD4+ T helper cells 5 . They are mainly localized to B-cell follicles in secondary lymphoid tissues and express phenotypic molecules, such as C-X-C chemokine receptor 5 (CXCR5), programmed cell death-1 (PD-1), inducible costimulatory molecule (ICOS), CD40 ligand, and interleukin-21 (IL-21), to mediate their function in assisting B cells and controlling their differentiation6–8. Overactivation of Tfh cells is likely to result in autoimmunity3,9. As suggested by our study and others, circulating Tfh cell proportions are elevated in SLE patients and are correlated with disease symptoms and antinuclear antibody levels10,11. Interestingly, therapies targeting Tfh cells have been shown to dramatically ameliorate lupus syndrome in a lot of autoimmune murine models12–16, making Tfh cells a promising treatment target for SLE patients.
Mesenchymal stem cells (MSCs) are pluripotent stem cells with a wide array of immunomodulatory effects on T and B lymphocytes, natural killer (NK) cells, and antigen-presenting cells, which can be isolated from bone marrow (BM) and a variety of other tissues, such as umbilical cord or adipose tissue17,18. Previously, human umbilical cord-derived MSCs (hUC-MSCs) have been applied to treat lupus mice and refractory SLE patients, with significant improvement in both disease activity and autoantibody levels19,20. However, despite the remarkable therapeutic effect of MSCs in SLE, the mechanisms by which MSCs exert their immunomodulatory and reparative effects are still not completely understood.
Recently, we demonstrated that MSCs suppressed the differentiation of circulating Tfh cells in patients with primary Sjogren's syndrome (pSS), which was mediated by indoleamine 2,3-dioxygenase (IDO)
21
. To find out whether MSCs could also modulate Tfh cells in lupus through the same pathway, in this study the frequency of Tfh cells in lupus-prone B6.MRL-
Materials and Methods
Mice
Female B6.
Isolation, Culture, and Identification of hUC-MSCs and Synovial Fibroblasts (FLSs)
The study on human subjects was approved by the ethics committee of the Affiliated Drum Tower Hospital of Nanjing University Medical School, and written informed consent was obtained from all subjects. hUC-MSCs and FLSs were prepared as previously described22,23 and cultured in Dulbecco's modified Eagle's medium (DMEM)/F12 supplemented with 10% fetal bovine serum (FBS) and 100 U/ml penicillin/streptomycin (all from Gibco, Life Technologies, Grand Island, NY, USA) until confluent. The adherent cells were detached by 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA; Gibco) and reseeded into new flasks for further expansion. All cell cultures were maintained at 37°C in a 5% CO2 humidified atmosphere. Flow cytometry analysis showed hUC-MSCs used in this study were positive for the surface staining of CD73, CD90, and CD105, but lacked CD34, CD45, CD14, CD19, and human leukocyte antigen D-related (HLA-DR) expression. The cells possessed the capacity of osteogenic and adipogenic differentiation.
Isolation, Culture, and Identification of Mouse BM-MSCs
BM cells were flushed out of the tibia and femoral marrow compartments of 6- to 8-week-old B6 mice, and then cultivated in plastic dishes according to the protocol for isolation and culture of MSCs from mouse BM developed by Soleimani and Nadri 24 . Briefly, BM cells were cultured in DMEM/F12 supplemented with 15% FBS for 3 h at 37°C in a 5% CO2 humidified atmosphere. Then nonadherent cells were removed carefully, and fresh medium was replaced. When primary cultures became confluent, the cells were treated with 0.5 ml of 0.25% trypsin-EDTA and reseeded into new dishes for further expansion. Flow cytometry analysis showed mouse BM-MSCs expanded in culture were with positive surface staining for stem cell antigen-1 (Sca-1), CD29, CD44, and CD73, but negative for major histocompatibility complex (MHC) class II (I-A), CD11b, CD19, and CD45. The cells preserved the capacity of osteogenic and adipogenic differentiation.
hUC-MSC Transplantation
Female B6.
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum levels of IL-21, immunoglobulin G (IgG), and anti-double stranded (ds)DNA were measured using mouse IL-21 and IgG ELISA Ready-SET-Go!® kits (eBioscience, San Diego, CA, USA) and mouse anti-dsDNA ELISA kit (Shibayagi, Gunma, Japan), respectively, according to the manufacturer's instructions.
Splenomegaly Assessment and Renal Histopathologic Analysis
When mice were sacrificed, spleens and kidneys were collected. The spleen index (ratio of spleen weight to body weight) was calculated. One kidney was fixed in 4% paraformaldehyde (PFA), embedded in paraffin, sectioned at 3 μm, and stained with hematoxylin and eosin (H&E; Sinopharm Chemical Reagent, Shanghai, P.R. China). The other one was snap frozen in liquid nitrogen and placed in optimal cutting temperature (OCT) embedding matrix (Leica Biosystems, Nussloch, Germany). Frozen sections (3 μm) were stained with fluorescein isothiocyanate (FITC)–anti-mouse IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Histological scores of renal lesions and the intensities of IgG deposits were determined as described previously 25 . Briefly, the severity of glomerulonephritis was graded on a 1–4 scale as follows: 1, focal, mild, or early proliferative; 2, multifocal proliferative with increased matrix; 3, diffuse proliferative; 4, extensive sclerosis/crescents. Interstitial and vascular lesions were also graded on a 1–4 scale according to the number/foci of mononuclear cells around tubules and vessels. Glomerular IgG staining was graded according to intensity on a scale of 0 to 3 as follows: 0, absent; 1, faint; 2, intense; 3, very intense.
Flow Cytometry
For surface staining, human-derived cells were labeled with FITC-anti-CD19, anti-CD34, and anti-CD73, or phycoerythrin (PE)-anti-CD14, anti-CD45, anti-CD90, anti-CD105, and anti-HLA-DR. Mouse-derived cells were labeled with the following antibodies: FITC-anti-CD11b, anti-CD19, anti-CD45, anti-CD34 anti-CD4, and anti-B220, or PE-anti-CD73, anti-CD29, anti-MHC II (I-A), anti-CD138, and anti-PD-1, or allophycocyanin (APC)-anti-Sca-1 and anti-PD-1. CXCR5 was stained with either APC-anti-CXCR5 directly or biotin-anti-CXCR5 for 1 h on ice, followed by PE-streptavidin. Secondary controls (PE-streptavidin only) without primary antibodies were applied to confirm staining specificity. For intracellular cytokine staining, cells were stimulated with 20 ng/ml phorbol-12-myristate-13-acetate (PMA) plus 1 μg/ml ionomycin at 37°C for 4–5 h in the presence of 5 mg/ml brefeldin A (all from Enzo Life Science, Farmingdale, NY, USA). Then cells were fixed and permeabilized with a fixation/permeabilization kit (Nordic-MUbio, Maastricht, Limburg, the Netherlands), followed by staining with APC-anti-interferon-γ (anti-IFN-γ), PE-anti-IL-4, or APC-anti-IL-17A. All antibodies were purchased from Miltenyi Biotec (Bergisch Gladbach, Germany) or eBioscience. Data were acquired by a fluorescence-activated cell sorting (FACS) Calibur flow cytometer (BD Biosciences, San Jose, CA, USA) and analyzed with FlowJo software (Tree Star, Ashland, OR, USA).
Coculture of Mouse B Cells and Tfh Cells
B cells were purified from spleens of 6- to 8-week-old female B6 mice by a B-cell isolation kit (Miltenyi Biotec). To obtain CD4+CXCR5+ T cells, purified CD4+ T cells were labeled with PE-anti-CXCR5 and anti-PE microbeads, and then positively selected by magnetic separation (Miltenyi Biotec).
B cells (3 × 105 cells/well) were cocultured with CD4+ CXCR5+ T cells (1×105 cells/well) from either B6.
Coculture of Mouse CD4+ T Cells and MSCs
Total CD4+ T cells were isolated from spleens of 6- to 8-month-old female B6.
Measurement of Tfh Cell Differentiation
Naive CD4+ T cells were isolated from the spleens of 6- to 8-month-old female B6.
Proliferation and Apoptosis Assays
CD4+CXCR5+ T cells (2×105/well) were cultured alone or cocultured with hUC-MSCs at a ratio of 10:1 in the presence of 2.5 μg/ml anti-CD3 and anti-CD28. Four days later, 60 μM 5-bromo-2-deoxyuridine (BrdU; Sigma-Aldrich) was added into the cultures and incubated for 3 h. Subsequently, suspension cells were collected and fixed overnight with 1% PFA in PBS containing 0.1% Tween 20 (Biosharp, Hefei, Anhui, P.R. China). After fixation, cells were treated with 100 Kunitz U/ml DNase I (Invitrogen) and stained with FITC-anti-BrdU (eBioscience). The incorporation of BrdU in CD4+CXCR5+ T cells was detected by flow cytometry.
For the detection of apoptosis, CD4+CXCR5+ T cells (2×105/well) were cultured alone or cocultured with hUC-MSCs at a ratio of 10:1 in the presence of 2.5 μg/ml anti-CD3 and anti-CD28. Three days later, CD4+ CXCR5+ T cells were collected, resuspended in 1× annexin V binding buffer, and stained with FITC-annexin V (BD Biosciences). Annexin V+ cells were detected by flow cytometry.
Real-Time Polymerase Chain Reaction (PCR) and Western Blot
Total RNA was extracted from hUC-MSCs using TRIzol (Takara, Dalian, Liaoning, PR. China), and cDNA was synthesized using reverse transcription by PrimeScript RT Master Mix (Takara). Quantitative real-time PCRs containing the SYBR Premix EX Taq™, ROX Reference Dye, cDNA, and specific gene primers (all from Takara) were run on the StepOnePlus Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The relative expressions of each gene were determined and normalized to the expression of housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and calculated using the 2–ΔΔCT method. Gene primers (Takara) are listed in Table 1.
Gene Primers Used for Real-Time PCR

GAPDH, glyceraldehyde 3-phosphate dehydrogenase; MMP-1, matrix metalloproteinase-1; iNOS, inducible nitric oxide synthase; HGF, hepatocyte growth factor; CCL2, chemokine (C-C motif) ligand 2; TGF-β1, transforming growth factor-β1; sHLA-G, soluble human leukocyte antigen-G; IDO, indoleamine 2,3-dioxygenase; IL-10, interleukin-10; COX2, cyclooxygenase 2.
For Western blot, a mouse antibody to iNOS (Novus Biologicals, Littleton, CO, USA), rabbit antibodies to protein kinase B (Akt), p65, IκBα, signal transducer and activator of transcription (Stat)1, Stat3, Stat5 and their phosphorylated forms [Cell Signaling Technology (CST), Danvers, MA, USA], and rabbit antibody to GAPDH (CST) were used as primary antibodies. Horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (H + L) and anti-mouse IgG (H + L) (both from Proteintech, Wuhan, Hubei, PR. China) were used as secondary antibodies, respectively. hUC-MSCs were washed twice with PBS and lysed on ice for 30 min with 1× radioimmunopre-cipitation assay (RIPA) buffer (CST) containing 1% 100× protease/phosphatase inhibitor cocktail (CST). Lysates were centrifuged at 12,000×
Nitric Oxide (NO) Production Assay
NO is quickly converted to NO2– and NO3– in the culture medium. Since there is a large amount of NO3– in the RPMI-1640 media, NO2– concentration was measured to represent NO production using a Griess reagent kit (Beyotime Biotechnology, Shanghai, P.R. China).
Statistical Analysis
Data are shown as mean ± standard deviation (SD). Student's
Results
Increased Tfh Cell Proportion Contributed to Abnormal B-Cell Activation in B6.lpr Mice
Similar to a previous report
26
, lupus nephritis began to develop in 6- to 8-month-old female B6.
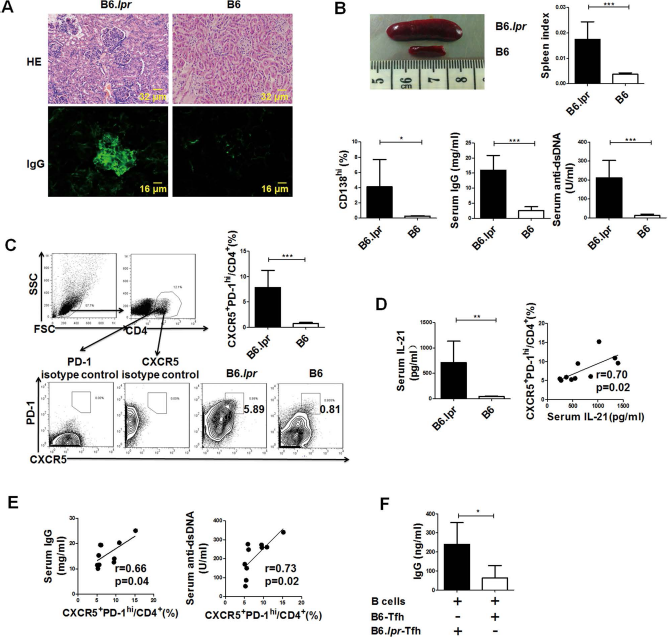
Increased T follicular helper (Tfh) cell proportion contributed to the abnormal B-cell response in B6.
Next, we compared the percentages of CD4+CXCR5+ PD-1hi Tfh cells, the main T helper cell subset that provides help to B cells, between B6.
Our data showed that the proportions of Tfh cells were positively correlated with both IgG and anti-dsDNA levels (Fig. 1E), suggesting that Tfh cells were related to aberrant B-cell response in B6.
hUC-MSC Transplantation Decreased Tfh Cell Proportions in B6.lpr Mice
To find out whether hUC-MSCs had an impact on Tfh cells in lupus-prone mice, 1 × 106 hUC-MSCs were transfused into B6.

Human umbilical cord mesenchymal stem cell (hUC-MSC) transplantation decreased Tfh-cell proportion in B6.
One month after treatment, mice were sacrificed, and histopathology examinations revealed that renal interstitial and perivascular lesions were greatly improved in hUC-MSC-treated mice, as characterized by diminished mononuclear cell infiltration around tubules and vessels. Meanwhile, IgG depositions in glomeruli were greatly reduced in hUC-MSC-treated mice (Fig. 2B), indicating that hUC-MSC transplantation effectively ameliorated disease progression in B6.
Next, we measured the frequencies of four main CD4+ T helper cell subsets, including Th1, Th2, Th17, and Tfh cells, after the treatment. As shown in Figure 2C, only Tfh cell proportions were significantly decreased in hUC-MSC-treated mice, while Th1, Th2, and Th17 cells remained unchanged. Decreased CD138hi splenic plasma cell proportions and serum IgG levels were observed after hUC-MSC transplantation (Fig. 2D). The level of anti-dsDNA antibody also showed a declining trend after MSC transplantation (180.2 ± 53.8 U/ml) compared with either PBS (260.5 ± 60.0 U/ml) or FLS (245.7 ± 60.0 U/ml) treatment (Fig. 2D). Additionally, there was a strong correlation between Tfh cells and plasma cells among the groups (Fig. 2E), suggesting that hUC-MSCs could act through the regulation of Tfh cells to restore balance to B cells.
hUC-MSCs Suppressed Tfh Cell Expansion Through the Inhibition of Cell Differentiation and Proliferation
To confirm the effect of hUC-MSCs on Tfh cells, splenic CD4+ T cells isolated from B6.

hUC-MSCs suppressed mouse Tfh cell expansion in vitro. (A) Splenic CD4+ T cells from of B6.
To explore how hUC-MSCs act on Tfh cells, experiments of cell differentiation, proliferation, and apoptosis were performed. Naive CD4+ T cells from B6.
The Inhibitory Effects of hUC-MSCs on Tfh Cells We re Mediated by iNOS
Previous studies have suggested that MSCs inhibited T-cell proliferation mainly through the secretion of soluble factors, including iNOS, HGF, chemokine (C-C motif) ligand 2 (CCL2), TGF-β1, soluble human leukocyte antigen-G (sHLA-G), IDO, cyclooxygenase 2 (COX2)/PGE2, and IL-1028. To find out the specific factors participating in regulating Tfh cells in B6.
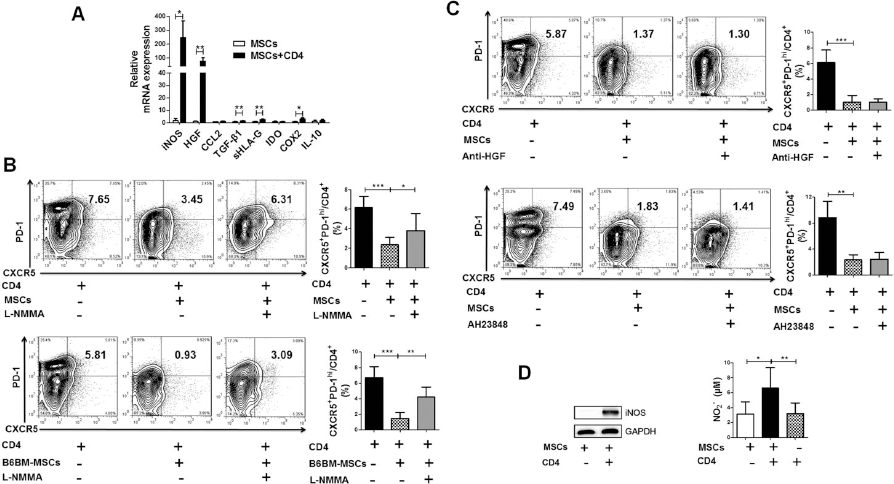
The inhibitor of inducible nitric oxide synthase (iNOS) partly restored the generation of mouse Tfh cells. (A) Real-time polymerase chain reaction (RT-PCR) showed that the expression of iNOS in hUC-MSCs was significantly increased after coculturing with mouse CD4+ T cells. (B) NG-methyl-L-arginine acetate salt (L-NMMA), the inhibitor of iNOS, reversed the suppression of hUC-MSCs (
To verify whether these differentially elevated factors were truly involved in the regulation of MSC function, an iNOS inhibitor, a PGE2 antagonist, and a neutralizing antibody against HGF were added into the hUC-MSCs-CD4+ T-cell coculture system, respectively. Our data showed that only L-NMMA, a specific inhibitor of iNOS, significantly restored the generation of Tfh cells (Fig. 4B), while neither anti-HGF treatment nor PGE2 blockade had an impact on Tfh cell production (Fig. 4C). Consistently, the inhibitory effect of mouse MSCs on Tfh cells was also reversed by L-NMMA (Fig. 4B). When culturing with mouse CD4+ T cells, hUC-MSCs produced more iNOS protein and higher levels of NO production were detected in coculture supernatants (Fig. 4D). Collectively, these data suggest that iNOS is an important mediator in MSC–Tfh cell regulation.
Cell-to-Cell Contacts Were Required for High iNOS Production in hUC-MSCs
Recently, it has been suggested that cell-to-cell contact was important for MSC-mediated inhibition of Tfh-cell differentiation
29
. To verify this phenomenon, CD4+ T cells collected from the spleens of B6.
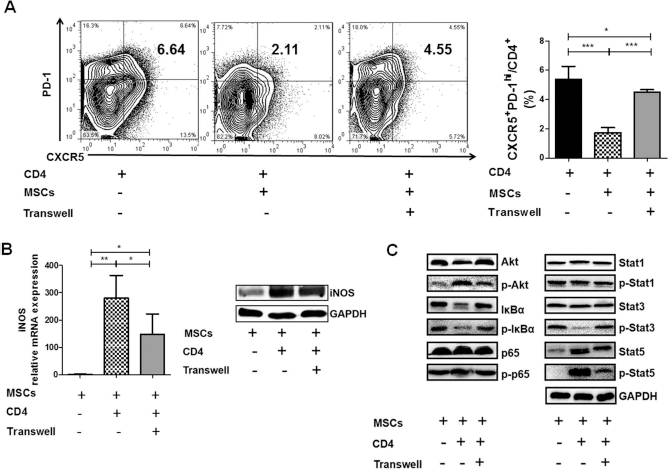
Complete inhibition of Tfh cells and full induction of iNOS in hUC-MSCs were dependent on direct cell-to-cell contact with CD4+ cells. CD4+ T cells from spleens of B6.
To further explore the mechanisms involved in iNOS production, key proteins of PI3K/Akt, nuclear factor κ-light-chain-enhancer of activated B cells (NF-κB) and Janus kinase (Jak)/Stat signaling pathways were measured by Western blot. As shown in Figure 5C, the levels of phosphorylated Akt and Stat5 were increased, while the levels of phosphorylated IκBα and Stat3 were decreased in hUC-MSCs after coculturing with mouse CD4+ T cells.
Discussion
Tfh cells are essential to the development of germinal center (GC) B-cell responses, and thus may play a critical role in the pathogenesis of lupus3,4,9. Recently, it has been reported that MSCs slowed down the progression of lupus nephritis by suppressing the emergence of the Tfh cells
29
. However, the underlying mechanism remains unclear. Here we confirmed there was a significantly higher frequency of CXCR5+PD-1hi Tfh cells in lupus-prone B6.
In this study, we confirmed that there was an excessive accumulation of Tfh cells and elevated IL-21 expression in lupus-prone B6.
Tfh cells may serve as a new therapeutic target for lupus, as blocking overactivated signals to target Tfh cells has been shown to alleviate Tfh and GC B-cell accumulation and result in the amelioration of lupus symptoms12–16. Previously we have demonstrated that MSC transplantation remarkably ameliorated lupus symptoms both in SLE patients and lupus mice and led to increased survival rates and improvement of renal function20,34. In this study, we demonstrated that hUC-MSCs restrained the expansion of Tfh cells both in vitro and in vivo, while the decline of Tfh cells was closely correlated with the levels of IgG and anti-dsDNA. Although there was evidence showing MSCs could directly regulate the proliferation and differentiation of B cells
35
, it is likely that MSCs could also act on Tfh cells to regulate B-cell response in B6.
As suggested by a recent study in NZB/W F1 mice
29
, the effect of MSCs on Tfh-cell differentiation was dependent on cell-to-cell contacts, yet the underlying mechanism remains unclear. In patients with pSS, we have previously verified that hUC-MSCs inhibited circulating Tfh cell differentiation through the production of IDO
21
. However, in the present study, hUC-MSCs cocultured with mouse CD4+ T cells did not express high levels of IDO; instead, they produced a large amount of iNOS. Inhibiting iNOS reversed the effects of hUC-MSCs on mouse Tfh cells, suggesting that iNOS is a key factor involved in MSC–Tfh cell regulation in B6.
The pathways involved in the induction of iNOS in MSCs are still not clear. Previously we have shown that the Jak/Stats signaling pathway was involved in the suppression of T cells by MSCs in human SLE 23 . In macrophage, lipopolysaccharide (LPS)-induced iNOS production has been reported to be dependent on PI3K/Akt and NF-κB signaling pathway 36 . In this study, we demonstrated that the phosphorylation of Akt and Stat5 was increased and the phosphorylation of IκBα and Stat3 was decreased in hUC-MSCs when they were cocultured with mouse CD4+ T cells, suggesting these three signaling pathways are all involved in the induction of iNOS in hUC-MSCs. Interestingly, the effect was almost diminished when hUC-MSCs were not directly cultured with CD4+ T cells, indicating that direct cell-to-cell contact is critical for the activation of these pathways in hUC-MSCs.
In summary, we show, for the first time, that hUC-MSCs inhibit Tfh cell expansion through the activation of iNOS in lupus-prone B6.
Footnotes
Acknowledgments
This work was funded by grants from the National Natural Science Foundation of China (Nos. 81172846, 81373198, 81302558, and 81601414) and the Jiangsu Provincial Science and Technology Project (No. BE2015602). The authors declare no conflicts of interest.
